辉钼精矿的氧化焙烧
来源期刊:中国有色金属学报(英文版)2015年第12期
论文作者:王璐 张国华 党杰 周国治
文章页码:4167 - 4174
关键词:氧化焙烧;辉钼精矿;形貌;烧结
Key words:oxidation roasting; molybdenite concentrate; morphology; sintering
摘 要:为研究辉钼精矿的氧化焙烧机理分别在673、723、773、873、973 K下进行氧化焙烧实验。分别采用X射线衍射(XRD)和扫描电子显微镜(SEM)对氧化过程中产物的物相和形貌进行分析。结果表明,在纯氧条件下,MoS2直接氧化成MoO3,并且随着焙烧温度的升高,产物的形貌发生了明显变化。在纯氧气氛下,当温度高于873 K时有烧结现象发生,烧结物大部分是由氧化产物MoO3组成,同时含有少量未氧化完全的MoS2。
Abstract: In order to investigate the oxidation roasting of molybdenite concentrate in pure oxygen atmosphere, experiments at 673, 723, 773, 873 and 973 K were carried out. The phase transitions and morphology evolutions of the samples obtained at different temperatures after reacting for different time were analyzed by X-ray diffraction (XRD) and scanning electron microscopy (SEM). The results showed that molybdenite concentrate was oxidized directly to MoO3 in pure oxygen atmosphere. There were remarkable changes of the morphologies of products with the increase of the roasting temperature. It was also found that sintering phenomenon occurred during the roasting process in pure oxygen when the temperature was above 873 K. The composition of sintered sample was mainly comprised of MoO3 and some unreacted MoS2.
Trans. Nonferrous Met. Soc. China 25(2015) 4167-4174
Lu WANG, Guo-hua ZHANG, Jie DANG, Kuo-chih CHOU
State Key Laboratory of Advanced Metallurgy, University of Science and Technology Beijing, Beijing 100083, China
Received 26 January 2015; accepted 20 May 2015
Abstract: In order to investigate the oxidation roasting of molybdenite concentrate in pure oxygen atmosphere, experiments at 673, 723, 773, 873 and 973 K were carried out. The phase transitions and morphology evolutions of the samples obtained at different temperatures after reacting for different time were analyzed by X-ray diffraction (XRD) and scanning electron microscopy (SEM). The results showed that molybdenite concentrate was oxidized directly to MoO3 in pure oxygen atmosphere. There were remarkable changes of the morphologies of products with the increase of the roasting temperature. It was also found that sintering phenomenon occurred during the roasting process in pure oxygen when the temperature was above 873 K. The composition of sintered sample was mainly comprised of MoO3 and some unreacted MoS2.
Key words: oxidation roasting; molybdenite concentrate; morphology; sintering
1 Introduction
Molybdenite concentrate has excellent properties in catalysis and lubrication [1,2]. Also, molybdenite concentrate (about 90% MoS2) is the essential ore mineral of the molybdenum industry for the production of technical grade molybdenum trioxide, which is further used to produce molybdenum dioxide [3], molybdenum, ferromolybdenum alloy and other pure molybdenum compounds, such as ammonium paramolybdate, sodium and calcium molybdate. The oxidation roasting of molybdenite concentrate is necessary to generate molybdenum trioxide. At present, the oxidation roasting of molybdenite concentrate has been commercially carried out in multiple heart furnace and fluidized bed furnace. The main reaction during the oxidation process is
MoS2+3.5O2=MoO3+2SO2 (1)
The change of the standard free energy of Reaction (1) is given by the following equation [4]:
ΔG/(J・mol-1)=-1074744.08+61.38TlgT-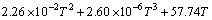 (2)
(2)
The equilibrium constant K of Reaction (1) at temperature T can be calculated by the following equation:
 (3)
(3)
Owing to the large equilibrium constant of the above reaction, for instant, up to 1052 when the temperature is 873 K, the oxidation reaction of molybdenite concentrate in air or pure oxygen could be considered to be an irreversible one.
Many investigations have been done to study the oxidation roasting of molybdenite concentrate.  and
and  [5] studied the roasting process of MoS2 synthesized in the laboratory and determined the kinetic parameters using the Kissinger and the Ozawa methods. SHIGEGAKI et al [6] investigated the oxidation mechanism of pure MoS2 using the TG-DTA method, and reported that oxidation process of MoS2 powder is controlled by a three- dimensional boundary reaction. MARIN et al [7] carried out oxidation roasting using a thin layer of sample in a muffle furnace in air in order to simulate the reactions taking place in the multiple heart furnace. The results showed that the rate of oxidation was very slow below 713 K, and then it increased and remained constant from about 813 K to 913 K. A kinetic model involving the intermediate phase of MoO2 was proposed (MoS2→ MoO2→MoO3). UTIGARD [8] conducted a large number of experiments about the oxidation roasting of molybdenite concentrate using a thermal gravimetric unit on a small scale both in air and oxygen atmosphere. The solid-state reaction between MoS2 and MoO3 to form MoO2 was found to take place. WILKOMIRAKY et al [9] studied the reaction of molybdenite concentrate with oxygen and air in a hot-stage microscope and reported that acicular crystals of MoO3 grown rapidly outward from the molybdenite. KIM et al [10] reported that the rate of mass loss decreases as the particle size increases when roasting the low grade Mongolian molybdenite concentrate in dried air. However, the particle size effect was slight when the molybdenite particle size was less than 67 μm. Very recently, a looping oxidation process with the aim to decrease energy consumption has been proposed by MCHUGH et al [11].
[5] studied the roasting process of MoS2 synthesized in the laboratory and determined the kinetic parameters using the Kissinger and the Ozawa methods. SHIGEGAKI et al [6] investigated the oxidation mechanism of pure MoS2 using the TG-DTA method, and reported that oxidation process of MoS2 powder is controlled by a three- dimensional boundary reaction. MARIN et al [7] carried out oxidation roasting using a thin layer of sample in a muffle furnace in air in order to simulate the reactions taking place in the multiple heart furnace. The results showed that the rate of oxidation was very slow below 713 K, and then it increased and remained constant from about 813 K to 913 K. A kinetic model involving the intermediate phase of MoO2 was proposed (MoS2→ MoO2→MoO3). UTIGARD [8] conducted a large number of experiments about the oxidation roasting of molybdenite concentrate using a thermal gravimetric unit on a small scale both in air and oxygen atmosphere. The solid-state reaction between MoS2 and MoO3 to form MoO2 was found to take place. WILKOMIRAKY et al [9] studied the reaction of molybdenite concentrate with oxygen and air in a hot-stage microscope and reported that acicular crystals of MoO3 grown rapidly outward from the molybdenite. KIM et al [10] reported that the rate of mass loss decreases as the particle size increases when roasting the low grade Mongolian molybdenite concentrate in dried air. However, the particle size effect was slight when the molybdenite particle size was less than 67 μm. Very recently, a looping oxidation process with the aim to decrease energy consumption has been proposed by MCHUGH et al [11].
Although many investigations on the oxidation roasting of molybdenite concentrate have been done, most of them were preceded in air atmosphere. Nowadays, oxygen-enriched air technology has been widely used to the iron-making process in blast furnace and it greatly improves the smelting efficiency. Similarly, the present study aims to investigate the oxidation roasting of molybdenite concentrate in pure oxygen in order to explore a new way of treating molybdenite. The phase transitions and morphology evolutions during the oxidation process will be analyzed.
2 Experimental
2.1 Sample preparation
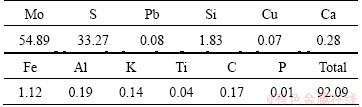
The molybdenite concentrate from Jinduicheng Molybdenum Industry Co., Ltd., Xi’an, China, was used as the raw material. The mass fraction of Mo content is about 54.89%. The main impurities in the molybdenite concentrate are sulfides or oxides of lead, copper, calcium, iron and silicon, etc. The chemical composition of the studied molybdenite concentrate is given in Table 1.
Table 1 Chemical composition of molybdenite concentrate (mass fraction, %)
The X-ray diffraction (XRD) pattern of molybdenite concentrate sample is shown in Fig. 1. The main peaks are well-defined to be MoS2. From Fig. 2, it can be seen that molybdenite concentrate particle has a layer structure and a wide particle size distribution ranged from several microns to 100 μm.
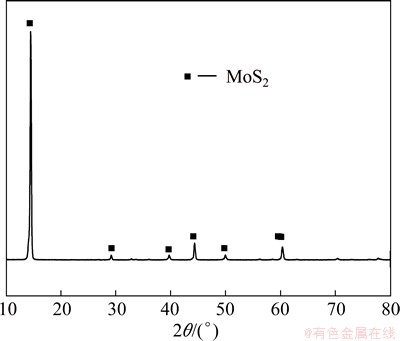
Fig. 1 XRD pattern of studied molybdenite concentrate

Fig. 2 SEM images of molybdenite concentrate sample
2.2 Apparatus and procedure
Figure 3 shows the schematic diagram of the apparatus. Alumina crucibles with 50 mm in length, 25 mm in width and 20 mm in height were used. At first, molybdenite concentrate was dried at 383 K in the oven for 12 h. In each experimental run, a sample of about 3 g (about 5 mm in thickness) was used and filled into the alumina crucible. After the crucible with the sample was placed into the quartz tube, argon was purged to remove air from the quartz tube. Then the quartz tube was put into the electrical furnace with the heating element of Si-C rods, the temperature of which was already raised to the desired value. When the temperature was stabilized, the argon was switched to pure oxygen to start the oxidation reaction. After reacting for a certain time, gas inlet was switched to argon again, and the quartz tube was taken out quickly to cool the sample to the room temperature. The mass loss after the oxidation process was measured. In the present study, oxidation experiments at five temperatures (673, 723, 773, 873 and 973 K) for different reacted time were conducted.
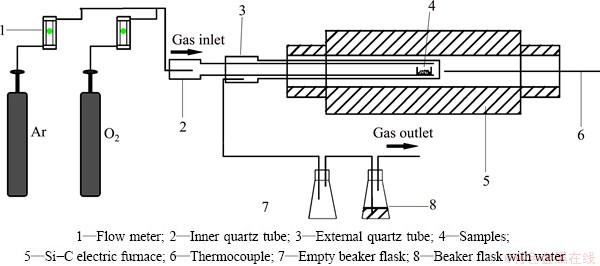
Fig. 3 Schematic diagram of experimental apparatus
In all the experimental runs, a constant oxygen flow rate of 400 mL/min was kept during the oxidation process. XRD (Model, TTR III, Japan) and SEM (Model S250MK3, CAMBRIDGE) were used to analyze the samples.
3 Results
3.1 Mass loss
The mass loss (η) was calculated with the following equation:
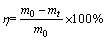 (4)
(4)
where m0 and mt are the masses of the initial sample and that reacted for a period time of t, respectively. The experimental data are shown in Fig. 4. It is clearly seen that the rate of reaction is very slow at 673 K. Even the roasting time was up to 100 min, there was a very small mass loss. Meanwhile, the maximum mass loss is different at different roasting temperatures and the value increases with the increase of temperature. The plateaus were reached after reacting for 20 min in all cases except the case when the temperature was 673 K. One surprising aspect of the roasting tests was that the sample was partially sintered and appeared to stick to the bottom of alumina crucible when reacting at 873 K for 20 min or longer. At 973 K, the sintering phenomenon occurred when reacting for 15 min. However, when the temperature was lower than 873 K, there was no sintering occurred even roasting for 1 h. It was also found that the color of the product continuously changed as the reaction proceeded. For example, when samples were roasted at 873 K, the color changed from black or grey to white with a light-greenish tint, as shown in Fig. 5. Furthermore, on the surface of product, many acicular, elongated and thin products were produced.
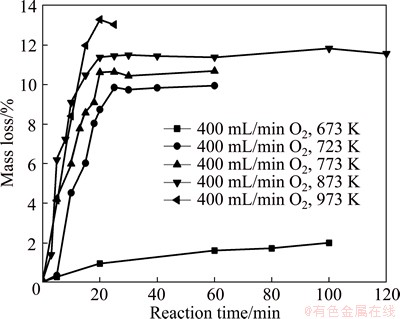
Fig. 4 Mass loss versus reaction time curves for molybdenite concentrate
3.2 XRD analysis
The X-ray diffraction patterns of products roasted at 723, 773 and 873 K are shown in Figs. 6(a)-(c), respectively. Figure 6(a) shows that after 15 min, there are some peaks for MoO3, but the main phase is still MoS2 in the molybdenite concentrate. The X-ray diffraction pattern for sample reacted for 20 min is very similar as that reacted for 15 min. Even if the reacting time is as long as 60 min when mass loss is almost stopped as shown in Fig. 4, there are still many unreacted MoS2. Therefore, the completed oxidation of MoS2 is very hard at 723 K. The X-ray diffraction patterns for sample reacting at 773 K as shown in Fig. 6(b) are somewhat similar to those of Fig. 6(a). The obvious difference is that when the roasting time is longer than 20 min, most of the MoS2 could be oxidized to MoO3. However, when the temperature is up to 873 K, as shown in Fig. 6(c), no peaks of MoS2 are detected in the final products, and the main constituent is MoO3 after reaction for 20 min.

Fig. 5 Color change after blending associated with oxidation in pure oxygen at 873 K
3.3 Microscopic examination
Scanning electron micrographs of molybdenite concentrate sample oxidized in pure oxygen at 873 K for various time are shown in Fig. 7. After reacting for 5 min, as shown in Fig. 7(a), the morphology of product is similar to the raw molybdenite concentrate (Fig. 2 (b)). Figures 7(b) and (c) show that the morphologies have significant changes after reaction for a long time when compared with Fig. 7(a). It can be found that as the reaction proceeds, large MoS2 particles disappeared with the formation of a large amount of plate-shaped MoO3. When sampling the surface product for observation, as shown in Fig. 7(d), almost all the MoO3 were plate-shaped and acicular or elongated.
Comparisons of morphologies of the samples oxidized in pure oxygen at different temperatures for various time are shown in Fig. 8. At 723 K, as shown in Fig. 8(a), the compact MoO3 is evident. Whereas, at 773 K, as shown in Fig. 8(b), the platelet-shaped crystals can be seen and some small crystals appear to be broken from the oxidized particles and a lot of fragmentations can be seen easily, which may be the result of the release of gaseous SO2. The particles have a tendency to agglomerate together. At 873 K, as shown in Fig. 8(c), the transformation to MoO3 is completed with crystals growing away from the original MoS2. Since these crystals grow out away from the surface, it appears that a vapor phase mechanism is involved, i.e., gaseous MoO3 forms at the nucleation site and then condenses as it leaves the hot reaction region.
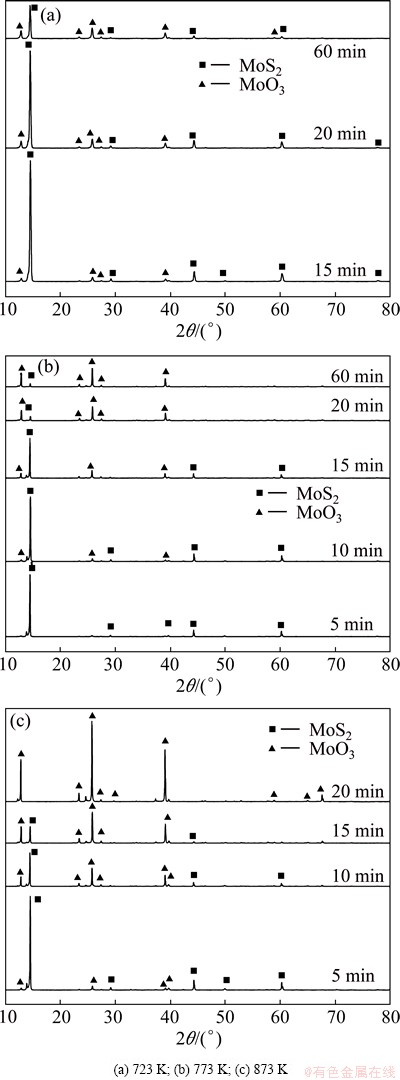
Fig. 6 XRD patterns of products after oxidation roasting of molybdenite concentrate at different temperatures for various time

Fig. 7 SEM images of molybdenite concentrate samples oxidized in pure oxygen at 873 K for various time
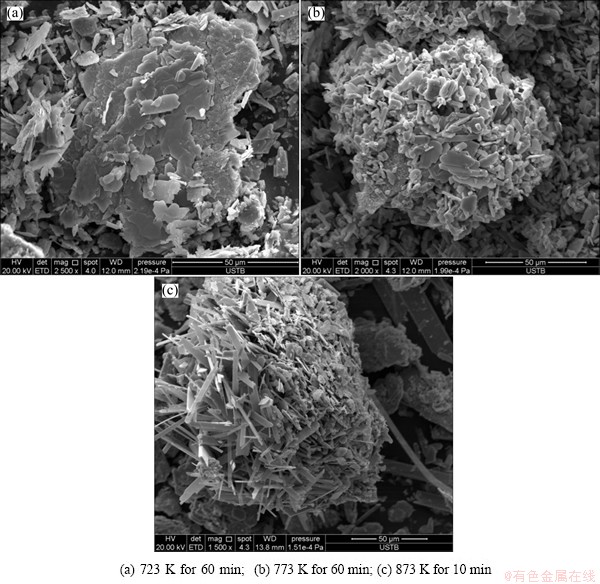
Fig. 8 SEM images of molybdenite concentrate samples oxidized in pure oxygen at different temperatures
4 Discussion
4.1 Formation mechanism of sintering
Previously, many researchers [7,8] have found that there were sintering or agglomerating phenomena at the temperatures above 873 K in air when oxidizing molybdenite concentrate. In the present study, the sintering took place from 20 min at 873 K, when MoS2 disappeared as shown in Fig. 6(c), and from 15 min at 973 K, while at other temperatures, there was no sintering occurred.
Figure 9 shows the XRD pattern for sintering at 873 K for 120 min. It is obviously seen that its main component was MoO3, but it also had some unreacted MoS2. This means that the molybdenite concentrate had not been oxidized completely at the bottom due to the sintering. Obviously, when the temperature was increased up to 973 K, the sintering phenomenon is more serious, which may be resulted from the high local reaction rate which released lots of heat leading to a much higher local temperature. Since the melting point of MoO3 is 1068 K [12,13], when the local temperature is higher than it, the liquid phase will present which results in the occurrence of sintering. However, since the vapor pressure of MoO3 increases with the increasing temperature [14,15], the sublimation becomes more significant at 973 K than that at 873 K, thus the mass loss is larger than that at 873 K, as shown in Fig. 4.
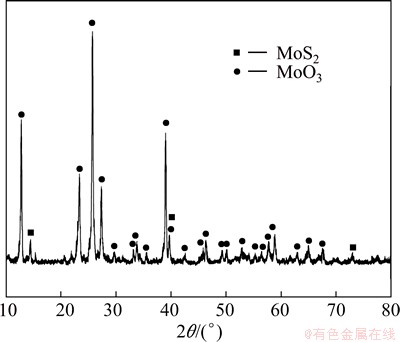
Fig. 9 XRD pattern for sintered sample oxidized at 873 K, 400 mL/min O2 for 120 min
The morphologies of the sintered products are shown in Fig. 10. It is observed that smooth surface was produced due to the fusion of particles which change the surface morphology greatly. EDS analysis for the smooth surface shows that the main component is MoO3, but with low content of other metal oxides, such as calcium oxide. Therefore, the glazing of molybdenite concentrate may be caused by the formation of low melting of eutectic metal oxides and molybdenum trioxide.
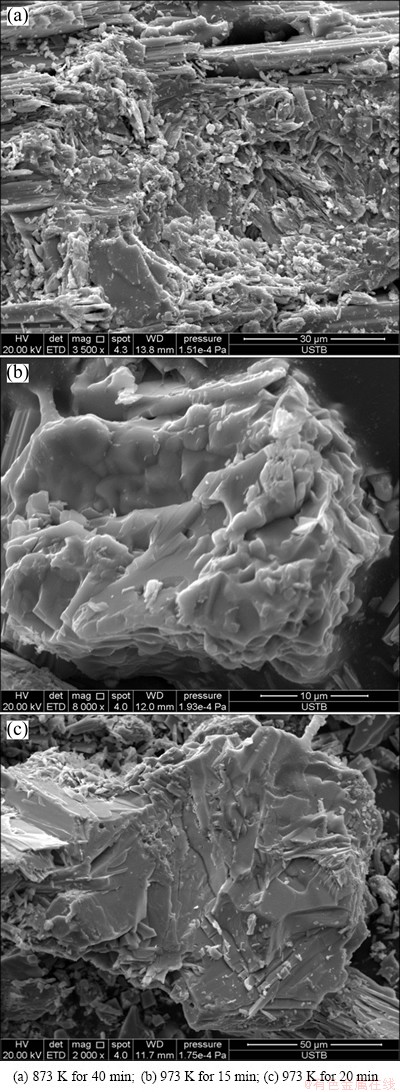
Fig. 10 SEM images of sintered sample
4.2 Oxidation mechanism
WILKOMIRSKY et al [9] carried out the oxidation of molybdenite concentrate in a hot-stage microscope and visually observed the rapid growth of MoO3 crystals outward other than inward. The growth of these crystals was interpreted as a vaporization-condensation mechanism, in which the product MoO3 is heated to a relatively high temperature by the exothermic reaction, and then it vaporizes from the surface, at last condenses on the protruding oxide crystals.
As seen from Fig. 8, as the temperature increases from 723 K to 873 K, molybdenum oxide crystallizes to polyhedral and plate-shaped particles that grow directionally. This phenomenon changes the shape of the particles dramatically. Directional crystallization of MoO3 could remove oxide layer from the surface of MoS2 particles, which is in agreement with the work of WILKOMIRSKY et al [9].
4.3 Formation of MoO2
In the present study, MoS2 powder was oxidized to MoO3 in a single step in pure oxygen without any intermediate oxide. These results are in agreement with those of WILKOMIRSK et al [9,16], DOHEIM et al [17], ROSS and SUSSMAN [18] and SHIGEGAKI et al [6], but differ from the results of MARIN et al [7], UTIGARD [8], ONG [19], ZELIKMAN and BELYAEVSKAYA [20], and COUDURIER et al [21] in which MoO2 as an intermediate oxidative product was produced before the formation of MoO3.
In order to explain the discrepancies between the results, additional experiments in air atmosphere were carried out. Figure 11 shows the differences of the products produced under pure oxygen and air atmosphere under identical conditions. It is clearly seen that the rate of oxidation in oxygen is much larger than that in air, and it took 60 min to reach plateau in air, while in pure oxygen, it only needed 20 min. Figure 12 shows the SEM images of products after roasting molybdenite concentrate in air and oxygen. It is clearly seen that the micrographs of products are similar to each other (the black part is SiO2). In addition, when compared with the raw material (Fig. 2), it can be seen that there are no obvious changes on morphologies of the cross sections of different samples and it is hard to identify MoO2, MoO3 and MoS2. Figure 13 shows the XRD patterns of products after roasting molybdenite concentrate in air for different time. Unexpectedly, it can be seen that MoO2 could be formed when roasting in air. Thus, the high oxygen potential in pure oxygen atmosphere may be the reason for the disappearance of intermediate phase MoO2.
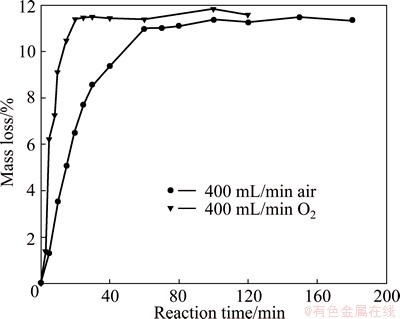
Fig. 11 Mass loss versus time for molybdenite concentrate at 873 K in oxygen and air

Fig. 12 SEM images of products obtained at 873 K for various time
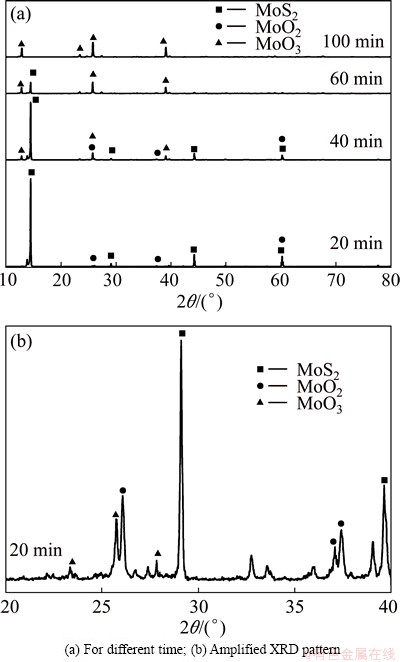
Fig. 13 XRD patterns of products after oxidation roasting of molybdenite concentrate at 873 K in air
5 Conclusions
1) The roasted sample begins to sinter at 873 K in pure oxygen. The compositions of the sintered products include MoO3 and unreacted MoS2.
2) The reaction rate is very slow when roasting at 673 K and the sublimation of MoO3 is significant at 973 K.
3) The intermediate oxide, MoO2, could be detected when roasting molybdenite concentrates in air atmosphere.
References
[1] HU Kun-hong, ZHAO Di-fang, LIU Jun-sheng. Synthesis of nano-MoS2/bentonite composite and its application for removal of organic dye [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(10): 2484-2490.
[2] CHEN Xing-yun, ZHAO Zhong-wei, HAO Ming-ming, LIU Xu-heng. Measurement of binary phase diagram of Cu2S-MoS2 system [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(1): 271-275.
[3] DANG J, ZHANG G H, CHOU K C. REDDY R G, HE Y, SUN Y J. Kinetics and mechanism of hydrogen reduction of MoO3 to MoO2 [J]. International Journal of Refractory Metals and Hard Materials, 2013, 41: 216-223.
[4] MIRSON G, ZELIKMAN A. Metallurgy of rare metals [M]. Moscow: Metallurgia Publication, 1965.
[5] 
 ,
,  J. Kinetics and mechanism of the oxidation of molybdenum sulphide [J]. Journal of Thermal Analysis and Calorimetry, 1998, 53: 263-267.
J. Kinetics and mechanism of the oxidation of molybdenum sulphide [J]. Journal of Thermal Analysis and Calorimetry, 1998, 53: 263-267.
[6] SHIGEGAKI Y, BASU S, WAKIHARA M, TANIGUCHI M. Thermal analysis and kinetics of oxidation of molybdenum sulfides [J]. Journal of Thermal Analysis and Calorimetry, 1988, 34: 1427-1440.
[7] MARIN T, UTIGARD T, HERNANDEZ C. Roasting kinetics of molybdenite concentrates [J]. Canadian Metallurgical Quarterly, 2009, 48: 73-80.
[8] UTIGARD T. Oxidation mechanism of molybdenite concentrate [J]. Metallurgical and Materials Transactions B, 2009, 40: 490-496.
[9] WILKOMIRSKY I. WATKINSON A, BRIMACOMBE J. Kinetics of oxidation of molybdenite [J]. Transactions of the Institution of Mining andMetallurgySectionC, 1997, 87: C16-C22.
[10] KIM B S, LEE H I, CHOI Y Y, KIM S. Kinetics of the oxidative roasting of low grade mongolian molybdenite concentrate [J]. Materials Transactions, 2009, 50: 2669-2674.
[11] MCHUGH L F, BALLIETT R, MOZOLIC J A. The sulfide ore looping oxidation process: An alternative to current roasting and smelting practice [J]. JOM, 2008, 60: 84-87.
[12] SHIBATA K, TSUCHIDA K, KATO A. Preparation of ultrafine molybdenum powder by vapour phase reaction of the MoO3-H2 system [J]. Journal of the Less Common Metals, 1990, 157: L5-L10.
[13] SAGHAFI M, ATAIE A, KHODADADI A. Synthesis of nanocrystalline molybdenum by hydrogen reduction of mechanically activated MoO3 [J]. International Journal of Refractory Metals and Hard Materials, 2012, 30: 128-132.
[14] GULBRANSEN E, ANDREW K, BRASSART F. Vapor pressure of molybdenum trioxide [J]. Journal of the Electrochemical Society, 1963, 110: 242-243.
[15] PAUL E B, MICHAEL H, HERRICK L J. The vaporization of molybdenum and tungsten oxides [J]. The Journal of Physical Chemistry, 1958, 62: 769-773.
[16] WILKOMIRSKY I, OTERO A, BALLADARES E. Kinetics and reaction mechanisms of high-temperature flash oxidation of molybdenite [J]. Metallurgical and Materials Transactions B, 2010, 41: 63-73.
[17] DOHEIM M, ABDEL M, RASSOUL S. Fluidized bed roasting of molybdenite―Effect of operating variables [J]. Metallurgical Transactions B, 1976, 7: 477-483.
[18] ROSS S, SUSSMAN A. Surface oxidation of molybdenum disulfide [J]. The Journal of Physical Chemistry, 1955, 59: 889-892.
[19] ONG J. Oxidatiion of sulfied [D]. Utah: University of Utah, 1955.
[20] ZELIKMAN A, BELYAEVSKAYA L. The melting point of molybdenite [J]. Journal of Inorganic Chemistry, 1956, 1: 65-72.
[21] COUDURIER L, WILKOMIRSK Y I, MORIZOT G. Molybdenite roasting and rhenium volatilization in a multiple-hearth furnace [J]. Transactions of the Institution of Mining and Metallurgy Section C, 1970, 79: C34-C40.
王 璐,张国华,党 杰,周国治
北京科技大学 钢铁冶金新技术国家重点实验室,北京 100083
摘 要:为研究辉钼精矿的氧化焙烧机理分别在673、723、773、873、973 K下进行氧化焙烧实验。分别采用X射线衍射(XRD)和扫描电子显微镜(SEM)对氧化过程中产物的物相和形貌进行分析。结果表明,在纯氧条件下,MoS2直接氧化成MoO3,并且随着焙烧温度的升高,产物的形貌发生了明显变化。在纯氧气氛下,当温度高于873 K时有烧结现象发生,烧结物大部分是由氧化产物MoO3组成,同时含有少量未氧化完全的MoS2。
关键词:氧化焙烧;辉钼精矿;形貌; 烧结
(Edited by Yun-bin HE)
Foundation item: Projects (51474141, 51174022) supported by the National Natural Science Foundation of China
Corresponding author: Guo-hua ZHANG; Tel: +86-10-62333703; E-mail: ghzhang_ustb@163.com
DOI: 10.1016/S1003-6326(15)64067-5

