P含量对CaO-Al2O3-SiO2体系在高温烧结过程中矿相转变的影响
来源期刊:中国有色金属学报(英文版)2019年第3期
论文作者:于海燕 潘晓林 董凯伟 吴艳
文章页码:650 - 656
关键词:石灰烧结法;铝酸钙;硅酸二钙;晶体结构;自粉性能;浸出
Key words:lime-sinter process; calcium aluminate; dicalcium silicate; crystal structure; self-pulverization performance; leaching
摘 要:采用XRF、XRD、SEM-EDS和DSC-TG等技术手段研究不同P含量下铝酸钙熟料在高温烧结过程中的矿相转变、显微组织、自粉性能及其氧化铝浸出性能。1350 °C下烧结熟料主要由12CaO・7Al2O3、 CaO・Al2O3、β-2CaO・SiO2和γ-2CaO・SiO2等矿相组成;随着P含量的增加,β-2CaO・SiO2含量逐渐升高,而γ-2CaO・SiO2含量逐渐降低。熟料的自粉性能随着P含量的增加逐渐恶化。P在熟料中均匀分布,其存在抑制β-2CaO・SiO2向γ-2CaO・SiO2的转变。12CaO・7Al2O3和CaO・Al2O3的晶胞体积随着P含量的增加分别降低和提高,且P能降低铝酸钙化合物的形成温度。铝酸钙熟料的氧化铝浸出率随着P含量的增加明显降低,当P含量从0提高到0.85%时,氧化铝浸出率从95.01%降低到83.84%。
Abstract: The mineral transition, microstructure and self-pulverization as well as the Al2O3 leaching performance of calcium aluminate clinkers with different P additions in CaO-Al2O3-SiO2 system during high-temperature sintering were systematically studied by XRF, XRD, SEM-EDS and DSC-TG technologies. The clinkers sintered at 1350 °C mainly contain 12CaO・7Al2O3, CaO・Al2O3, β-2CaO・SiO2 and γ-2CaO・SiO2. The content of β-2CaO・SiO2 increases while the content of γ-2CaO・SiO2 decreases as the P addition increases. The self-pulverization property of clinkers deteriorates with increasing P addition. P distributes evenly in the clinkers, and inhibits the transformation of β-2CaO・SiO2 to γ-2CaO・SiO2. The cell volumes of 12CaO・7Al2O3 and CaO・Al2O3 decrease and increase respectively as the P addition increases, and P decreases their formation temperatures. The Al2O3 leaching rate of calcium aluminate clinkers decreases obviously with increasing P addition, which decreases form 95.01% to 83.84% as the P addition increases from 0 to 0.85%.
Trans. Nonferrous Met. Soc. China 29(2019) 650-656
Hai-yan YU, Xiao-lin PAN, Kai-wei DONG, Yan WU
School of Metallurgy, Northeastern University, Shenyang 110819, China
Received 8 March 2018; accepted 12 June 2018
Abstract: The mineral transition, microstructure and self-pulverization as well as the Al2O3 leaching performance of calcium aluminate clinkers with different P additions in CaO-Al2O3-SiO2 system during high-temperature sintering were systematically studied by XRF, XRD, SEM-EDS and DSC-TG technologies. The clinkers sintered at 1350 °C mainly contain 12CaO・7Al2O3, CaO・Al2O3, β-2CaO・SiO2 and γ-2CaO・SiO2. The content of β-2CaO・SiO2 increases while the content of γ-2CaO・SiO2 decreases as the P addition increases. The self-pulverization property of clinkers deteriorates with increasing P addition. P distributes evenly in the clinkers, and inhibits the transformation of β-2CaO・SiO2 to γ-2CaO・SiO2. The cell volumes of 12CaO・7Al2O3 and CaO・Al2O3 decrease and increase respectively as the P addition increases, and P decreases their formation temperatures. The Al2O3 leaching rate of calcium aluminate clinkers decreases obviously with increasing P addition, which decreases form 95.01% to 83.84% as the P addition increases from 0 to 0.85%.
Key words: lime-sinter process; calcium aluminate; dicalcium silicate; crystal structure; self-pulverization performance; leaching
1 Introduction
Calcium aluminate compounds in CaO-Al2O3-SiO2 ternary system are the research focus in basic theories and applications, which have played an important role in the areas of refractories, oxide ceramics, cement chemistry, metallurgical slags and geochemistry [1-4]. The calcium aluminates are mainly comprised of 3CaO・Al2O3 (C3A), 12CaO・7Al2O3 (C12A7), CaO・Al2O3 (CA), CaO・2Al2O3 (CA2), CaO・6Al2O3 (CA6) [5]. Combustion synthesis and mechanical activation were used to synthesize calcium aluminates by RODRIGUEZ et al [6] and CHEN [7]; MOHAMED and SHARP [8,9] obtained the formation mechanism and microstructure characteristics of CA and C3A; OSTROWSKI and ELAZNY [10] determined the solid solutions of calcium aluminates; STOBER et al [11] and VERMA et al [12] studied the crystal structure and physical-chemical properties in Mn4+, Mn2+, Fe3+ and Tb3+ doped C12A7 and other aluminates, respectively.
Calcium aluminates are also the main constitutes of sintering clinker in the lime-sinter process from alumina industry [13]. TIAN et al [14] and ZHANG et al [15] studied the formation mechanisms of different calcium aluminate compounds with different molar ratios of CaO to Al2O3 in CaO-Al2O3 system and CaO-Al2O3-SiO2 system respectively, and found that the calcium aluminates present as a layered distribution and all the calcium aluminates play role as the intermediate phases to the final equilibrium phases. YU et al [16,17] discussed the effects of Na2O and iron oxides on the formation activity of calcium aluminate clinker, and found that Na2O and FeO can form solid solutions in C12A7 and inhibit the transformation of β-2CaO・SiO2 to γ-2CaO・SiO2. WANG et al [18,19] investigated the effects of MgO and S on the mineral transition and leaching properties of calcium aluminate clinkers, and found that they can form 20CaO・13Al2O3・3MgO・3SiO2 and 3CaO・3Al2O3・CaSO4, respectively. However, as an important impurity in bauxite ores and other raw materials, the effects of P on the formation process and leaching properties of calcium aluminate clinkers are unknown. The content of P in bauxites usually ranges from 0.05% to 0.5% in the mineral form of apatite. In this work, the self-pulverization, sintering characteristics and leaching properties of calcium aluminate clinkers with different P additions were systematically investigated in the CaO-Al2O3-SiO2 system based on the lime-sinter process.
2 Experimental
Analytically pure reagents of CaCO3, Al2O3, SiO2 and Ca3(PO4)2 were used in this study, and the calculated mass fractions of oxides for the sintering process are 49.70% CaO, 10.06% SiO2, 40.24% Al2O3 and 0-0.85% P. Then, the mixtures were milled in a ball mill for 3 h. A programmable MoSi2 heating furnace was used for sintering in a graphite crucible at 1350 °C for 1 h, and then the clinkers were cooled to room temperature in the furnace after sintering.
The calcium aluminate clinkers were ground to below 74 mm in particle size and then leached at 80 °C for 30 min in sodium carbonate solution. The concentration of sodium carbonate solution (in form of Na2O) is 80 g/L. The liquid-to-solid ratio of sodium carbonate solution to clinker for leaching is 10:1. The leached slurry was filtrated using a Buchner funnel. The concentrations of caustic alkali (NK, in form of Na2O), total alkali (NT, in form of Na2O) and Al2O3 (AO) in the filtrate were determined by the volumetric method, and the filter residues were washed with hot water and dried for chemical analysis. The Al2O3 leaching rate ( ) is calculated according to the following formula:
) is calculated according to the following formula:
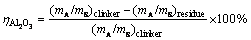 (1)
(1)
where (mA/mS)clinker and (mA/mS)residue are the mass ratios of Al2O3 to SiO2 in the sintered clinkers and leached residues, respectively.
The contents of Al2O3 and SiO2 in the sintered clinkers and leached residues were determined using X-ray fluorescence (XRF, ZSX100e) [20]. The mineral compositions of clinkers and residues were estimated by X-ray diffraction (PANalytical PW3040/60). Quantitative analyses were performed by the Rietveld refinement method. The differential scanning calorimetry (DSC) and thermogravimetric (TG) analysis were performed using NETZSCH STA409C/CD simultaneous thermal analyzer in dynamic Ar atmosphere with a heating rate of 10 °C/min. The morphology and micro-composition of minerals in clinker were obtained by SEM (SHIMADZU SSX-550) coupled with EDS (DX-4).
3 Results and discussion
3.1 Pulverization property of calcium aluminate clinkers
The macrostructures of calcium aluminate clinkers with different P additions are shown in Fig. 1. The clinker without P is pulverized very well; as the P addition increases to 0.50%, about 40% of the clinker is not pulverized as vitreous particles, which is over 74 mm in particle size, and the other is shown in Fig. 1(b); when the P addition is 0.85%, the clinker is absolutely not pulverized.

Fig. 1 Macrostructures of calcium aluminate clinkers with different P additions
The main sintering reactions of CaO-Al2O3-SiO2 ternary system at 1350 °C are the formation of calcium aluminates and dicalcium silicate, as shown in Formulae (2) and (3). Dicalcium silicate (2CaO・SiO2, C2S) has a variety of crystal structures [21], e.g. α, α′H, α′L, β and γ, which transform to each other under different conditions. The crystal of C2S during the lime-sinter process transforms from β to γ at (700±20) °C during the cooling process of calcium aluminate clinkers. The volume of clinkers will expand about 10% during the transformation process because of different relative densities of the two types of C2S (the densities of β-C2S and γ-C2S are 3.28 and 2.97 g/cm3, respectively). Then, the internal stress in the inner crystals of C2S will make the clinker pulverized itself. The pulverization properties of calcium aluminate clinkers get much worse with increasing P addition, as shown in Fig. 1, indicating that P inhibits the transformation of β-2C2S to γ-2C2S. The reason may be that P can enter into the crystal lattice of C2S and forms solid solutions, and then the glassy structure will form in the cooling process, as seen in Fig. 1(c), which inhibits the β to γ transformation. In fact, B2O3, Na2O, K2O, MgO, BaO, MnO2 and Cr2O3 were also reported to stabilize β-C2S [22].
mCaO(s)+nAl2O3(s)→mCaO・nAl2O3(s) (2)
2CaO(s)+SiO2(s)→2CaO・SiO2(s) (3)
3.2 Mineral transition of calcium aluminate clinkers
The XRD patterns of calcium aluminate clinkers with different P additions are shown in Fig. 2. All the clinkers contain C12A7, CA, β-C2S and γ-C2S, and the P impurity does not affect the phase types of calcium aluminate clinkers. However, the mineral contents in clinkers are different. The peak intensity of γ-C2S decreases and the peak intensity of β-C2S increases as the P addition increases, as seen in Fig. 2.
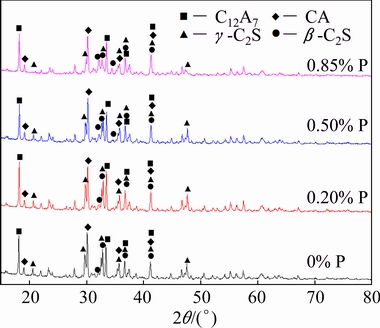
Fig. 2 XRD patterns of calcium aluminate clinkers with different P additions
The mineral compositions in calcium aluminate clinkers with different P additions were calculated by the Rietveld method as listed in Table 1. The contents of C12A7 and CA do not change obviously, but the contents of β-C2S and γ-C2S change accordingly with increasing P addition. When there is no P addition, C2S is mainly in the form of γ-C2S. However, as the P addition increases, the content of γ-C2S decreases while the content of β-C2S increases. When the P addition is 0.85%, β-C2S is the dominant existence of C2S. The above results are consistent with the self-pulverization properties of calcium aluminate clinkers, which demonstrate that the P impurity restrains the transformation of β-C2S to γ-C2S.
Table 1 Mineral compositions in calcium aluminate clinkers with different P additions (wt.%)

The crystal parameters of calcium aluminate compounds in clinkers with different P additions were calculated and listed in Table 2. C12A7 belongs to the cubic system with 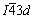 space group, and CA belongs to the monoclinic system with P21/n space group. As the P addition increases, the lattice constant (a) of C12A7 decreases gradually, and the corresponding cell volume (V) decreases. Meanwhile, the lattice constant a of CA decreases while the b and c increase with increasing P addition, and the corresponding cell volume increases gradually. Because no P-containing phase was found in the XRD patterns, the changes in the lattice constants and cell volumes of C12A7 and CA indicate that P can form solid solutions in calcium aluminates, which is also proved by the following P distribution in the calcium aluminate clinker according to the map scan analysis of SEM.
space group, and CA belongs to the monoclinic system with P21/n space group. As the P addition increases, the lattice constant (a) of C12A7 decreases gradually, and the corresponding cell volume (V) decreases. Meanwhile, the lattice constant a of CA decreases while the b and c increase with increasing P addition, and the corresponding cell volume increases gradually. Because no P-containing phase was found in the XRD patterns, the changes in the lattice constants and cell volumes of C12A7 and CA indicate that P can form solid solutions in calcium aluminates, which is also proved by the following P distribution in the calcium aluminate clinker according to the map scan analysis of SEM.
Table 2 Crystal parameters of calcium aluminates in clinkers with different P additions

3.3 Morphology of calcium aluminate clinker
The SEM image of clinker sintered with 0.85% P is shown in Fig. 3. The morphology of clinker is divided to three regions, i.e. light grey, medium grey and dark gray, which are adjacent to each other but the borderlines are clearly discerned. The micro-compositions of the three regions determined by EDS are listed in Table 3, and the corresponding C/A ratio (the molar ratio of CaO to Al2O3) and C/S ratio (the molar ratio of CaO to SiO2) were calculated.

Fig. 3 SEM image of calcium aluminate clinker containing 0.85% P
Table 3 EDS results of clinker corresponding to Fig. 3 (at.%)
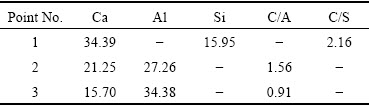
The light grey region (Point 1 in Fig. 3) is mainly composed of Ca, Si and O, and the C/S ratio is about 2, which corresponds to C2S based on the XRD results. The crystal size of C2S is 2-8 μm. Both the medium grey region (Point 2) and the dark gray region (Point 3) are composed of Ca, Al and O, but the C/A ratios are quite different. According to the C/A ratios and XRD results, the medium grey region corresponds to C12A7, while the the dark gray region corresponds to CA.
The elemental distribution of calcium aluminate clinker when sintered with 0.85% P was obtained by map scan analysis of SEM, as shown in Fig. 4. The elements of Ca, O and P distribute evenly, while Al mostly distributes in the medium grey and dark gray regions where the calcium aluminate compounds distribute. Si mainly distributes in the light gray region of C2S.
3.4 Thermal analysis
The DSC and TG curves of the mixtures of CaCO3, Al2O3 and SiO2 without P and with 0.85% P during heating process are shown in Fig. 5. The mass of both TG curves has little change over 800 °C after the decomposition of CaCO3, but a series of endothermic peaks appear in the DSC curves. According to the related references [8,9], the endothermic peaks at 1100-1300 °C represent the reactions of C2S formation. As CA is the main calcium aluminate compound in the clinkers, the endothermic peaks at 1334.8 °C in Fig. 5(a) and at 1331.0 °C in Fig. 5(b) are related to the formation of CA, while the endothermic peaks at 1382.9 °C in Fig. 5(a) and at 1374.3 °C in Fig. 5(b) are related to the formation of C12A7. The P addition decreases the formation temperatures of calcium aluminate compounds in clinkers, which has the same effect with Na2O [16].
Combining the macrostructure and the XRD analyses of the clinkers with the DSC results, the following conclusions can be drawn: P can decrease the mineral formation temperature and the melting temperature of clinkers, and distributes evenly in the liquid which forms solid solutions in both calcium aluminates and dicalcium silicate; then P inhibits the transformation of β-C2S to γ-C2S, which makes the clinkers glassy and unpulverized.

Fig. 4 SEM image (a) and elemental distributions (b-f) of calcium aluminate clinker containing 0.85% P
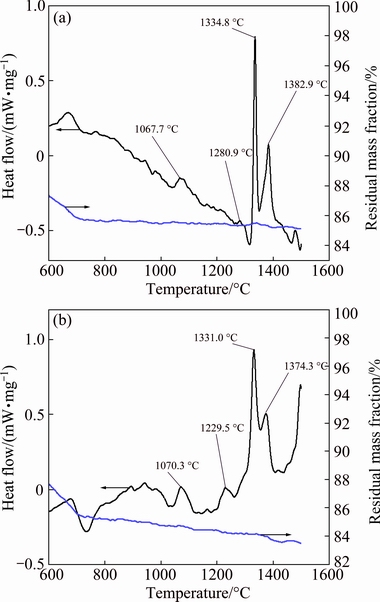
Fig. 5 DSC-TG curves of mixtures without P (a) and with 0.85% P (b) during heating process
3.5 Leaching property of calcium aluminate clinkers
The leaching reactions of calcium aluminate compounds in sodium carbonate solution are shown in Formulae (4) and (5), respectively. Meanwhile, C2S can also react with the alkali solution as listed in Formulae (6) and (7), and then transform to the corresponding desilication products (DSPs) as listed in Formulae (8) and (9) known as the secondary reaction in the sinter process [23]. The leaching results of clinkers with different P additions in sodium carbonate solution are shown in Table 4. The concentrations of caustic alkali and alumina in sodium aluminate solution after leaching decrease with increasing P addition, especially an obvious decrease when the P addition reaches 0.85%.
The Al2O3 leaching rate of clinkers and the molar ratio of caustic alkali to alumina (αK) in sodium aluminate solution after leaching were calculated and shown in Fig. 6. Both the Al2O3 leaching rate and αK decrease with increasing P addition. The Al2O3 leaching rate decreases from 95.01% to 90.26% when the P addition increases from 0 to 0.50%; as the P addition increases to 0.85%, the Al2O3 leaching rate is only 83.34%, demonstrating that P deteriorates the leaching properties of clinkers.
12CaO・7Al2O3(s)+12Na2CO3(l)+33H2O=12CaCO3(s)+14NaAl(OH)4(l)+10NaOH(l) (4)
CaO・Al2O3(s)+Na2CO3(l)+4H2O=CaCO3(s)+2NaAl(OH)4(l) (5)
2CaO・SiO2(s)+2NaOH(l) +H2O=2Ca(OH)2(l)+Na2SiO3(l) (6)
2CaO・SiO2(s)+2Na2CO3(l)+H2O=2CaCO3(s)+Na2SiO3(l)+2NaOH(l) (7)
1.7Na2SiO3(l)+2NaAl(OH)4(l)=Na2O・Al2O3・1.7SiO2・nH2O(s)+3.4NaOH(l)+(2.3-n)H2O (8)
3Ca(OH)2(l)+xNa2SiO3(l)+2NaAl(OH)4(l)=3CaO・Al2O3・xSiO2・(6-2x)H2O(s)+2(x+1)NaOH(l)+xH2O (9)
Table 4 Leaching results of calcium aluminate clinkers with different P additions
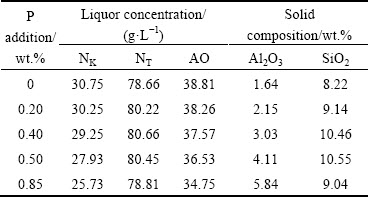

Fig. 6 Al2O3 leaching rate and αK of calcium aluminate clinkers with different P additions after leaching
The XRD patterns of the leached residues without P and with 0.85% P are shown in Fig. 7. The main minerals in the leached residues are CaCO3, β-C2S and γ-C2S because of the decomposition of calcium aluminate compounds. However, some C12A7 still exists in the residue when the clinker contains P impurity, demonstrating that P deteriorates the leaching activity of C12A7 of clinkers in sodium carbonate solution. In addition, CA is not found in both residues with and without P addition, indicating that the leaching property of CA is good and it is not affected by the P impurity.
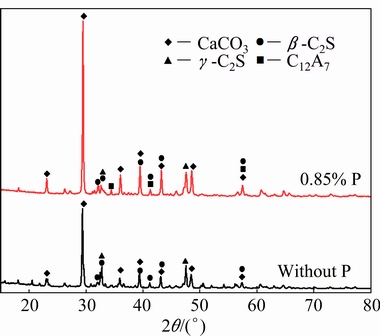
Fig. 7 XRD patterns of leached residues of calcium aluminate clinkers without P and with 0.85% P
No DSPs were found in the leached residues, as seen in Fig. 7, indicating that the secondary reaction of C2S does not happen because of the high liquid-to-solid ratio during leaching [23,24]. As listed in Formulae (4) and (5), the decomposition of C12A7 in sodium carbonate solution leads to the increase of NK because of the extra formation of NaOH as compared to CA. On the other hand, as shown in Table 1, the addition of P during sintering can increase the content of C12A7 but decrease the content of CA. However, the NK in sodium aluminate solution after leaching decreases with the increase of P addition as seen in Fig. 6, which also demonstrates that P increases the stability of C12A7 during the sintering process and then results in more un-decomposed C12A7 in clinker during leaching.
Therefore, P has a negative impact on the sintering and leaching properties of calcium aluminate clinkers in CaO-Al2O3-SiO2 system. P cannot only inhibit the transformation of β-C2S to γ-C2S to deteriorate the self-pulverization property of clinker, but also form solid solutions in calcium aluminates to decrease the alumina leaching rate of clinker. The content of P should be controlled to be less than 0.50% in the final calcium aluminate clinker.
4 Conclusions
(1) The self-pulverization properties of calcium aluminate clinkers are increasingly deteriorated by increasing P addition because P hinders the transformation of β-C2S to γ-C2S.
(2) The clinkers mainly contains C12A7, CA, β-C2S and γ-C2S, but the content of β-C2S increases while the content of γ-C2S decreases as the P addition increases.
(3) The cell volumes of C12A7 and CA decrease and increase respectively as the P addition increases, and P decreases the formation temperatures of calcium aluminates and increases the stability of C12A7 during sintering.
(4) The Al2O3 leaching rate of calcium aluminate clinkers decreases from 95.01% to 83.84% as the P addition increases from 0 to 0.85%. The content of P should be controlled to be less than 0.50% in the calcium aluminate clinker.
References
[1] AKONO A, CUI Y, KATARUKA A, ANDERSON K, KABIR P. Intrinsic mechanical properties of calcium aluminate crystals via the linear comparison composite method coupled with nano-indentation [J]. Mechanics of Materials, 2018, 118: 74-84.
[2] LEE N K, KOH K T, PARK S H, RYU G S. Microstructural investigation of calcium aluminate cement-based ultra-high performance concrete (UHPC) exposed to high temperatures [J]. Cement and Concrete Research, 2017, 102: 109-118.
[3] YANG Shou-lei, XIAO Guo-qing, DING Dong-hai, REN Yun, LV Li-hua, YANG Pan. Dissolution-precipitation mechanism of combustion synthesis of calcium aluminate [J]. Ceramics International, 2017, 43(17): 15918-15926.
[4] KANG Shuai, WANG Xue, XU Wen-bin, WANG Xin, HE Dong-bing, HU Li-li. Effect of B2O3 content on structure and spectroscopic properties of neodymium-doped calcium aluminate glasses [J]. Optical Materials, 2017, 66: 287-292.
[5] FABRICHNAYA O B, NERAD I. Thermodynamic properties of liquid phase in the CaO・SiO2-CaO・Al2O3・2SiO2-2CaO・Al2O3・SiO2 system [J]. Journal of the European Ceramic Society, 2000, 20(4): 505-515.
[6] RODRIGUEZ M A, AGUILAR C L, AGHAYAN M A. Solution combustion synthesis and sintering behavior of CaAl2O4 [J]. Ceramics International, 2012, 38(1): 395-399.
[7] CHEN Guo-hua. Mechanical activation of calcium aluminate formation from CaCO3-Al2O3 mixtures [J]. Journal of Alloys and Compounds, 2006, 416(1-2): 279-283.
[8] MOHAMED B M, SHARP J H. Kinetics and mechanism of formation of monocalcium aluminate, CaAl2O4 [J]. Journal of Materials Chemistry, 1997, 7(8): 1595-1599.
[9] MOHAMED B M, SHARP J H. Kinetics and mechanism of formation of tricalcium aluminate, Ca3Al2O6 [J]. Thermochimica Acta, 2002, 388(1-2): 105-114.
[10] OSTROWSKI C, ELAZNY J. Solid solutions of calcium aluminates C3A, C12A7 and CA with sodium oxide [J]. Journal of Thermal Analysis and Calorimetry, 2004, 75(3): 867-885.
[11] STOBER R, NOFZ M, GEBNER W, SCHROTER C, KRANZ G. Paramagnetic monitors (Mn2+, Mn4+, Fe3+, and O2-) in the solid-state reaction yielding 12CaO・7Al2O3 and other aluminates [J]. Journal of Solid State Chemistry, 1989, 81(2): 152-164.
[12] VERMA R K, KAUR G, RAI A, RAI S B. Dual mode green fluorescence from Tb3+: Ca12Al14O33 and its applicability as delayed fluorescence [J]. Materials Research Bulletin, 2012, 47(11): 3726-3731.
[13] CHOU K S, BURNET G. Formation of calcium aluminates in the lime-sinter process [J]. Cement and Concrete Research, 1981, 11(2): 167-174.
[14] TIAN Yong-pan, PAN Xiao-lin, YU Hai-yan, TU Gan-feng. Formation mechanism of calcium aluminate compounds based on high-temperature solid-state reaction [J]. Journal of Alloys and Compounds, 2016, 670: 96-104.
[15] ZHANG Di, PAN Xiao-lin, YU Hai-yan, ZHAI Yu-chun. Mineral transition of calcium aluminate clinker during high-temperature sintering with low lime dosage [J]. Journal of Materials Science and Technology, 2015, 31(12): 1244-1250.
[16] YU Hai-yan, PAN Xiao-lin, WANG Bo, ZHANG Wu, SUN Hui-lan, BI Shi-wen. Effect of Na2O on the formation of calcium aluminates in the CaO-Al2O3-SiO2 system [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(12): 3108-3112.
[17] YU Hai-yan, PAN Xiao-lin, LIU Bao-wei, WANG Bo, BI Shi-wen. Effect of iron oxides on the activity of calcium aluminate clinker in CaO-Al2O3-SiO2 system [J]. Journal of Iron and Steel Research, International, 2014, 21(11): 990-994.
[18] WANG Bo, SUN Hui-lan, GUO Dong, ZHANG Xue-zheng. Effect of Na2O on alumina leaching property and phase transformation of MgO-containing calcium aluminate slags [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(12): 2752-2757.
[19] WANG Bo, CHU Wei-qin, HAO Yuan-liang, RONG Shuo, SUN Hui-lan. Synthesis and alumina leaching mechanism of calcium sulphoaluminate [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(9): 2090-2095.
[20] PAN Xiao-lin, YU Hai-yan, TU Gan-feng. Reduction of alkalinity in bauxite residue during Bayer digestion in high-ferrite diasporic bauxite [J]. Hydrometallurgy, 2015, 151: 98-106.
[21] GHOSH S N, RAO P B, PAUL A K, RAINA K. The chemistry of dicalcium silicate mineral [J]. Journal of Materials Science, 1979, 14(7): 1554-1566.
[22] ANGELES G, KHADIJA M, MOHAMMED Z, ARANDA A G. In situ synchrotron powder diffraction study of active belite clinkers [J]. Journal of Applied Crystallography, 2007, 40(6): 999-1007.
[23] YU Hai-yan, WANG Bo, PAN Xiao-lin, Ding Ting-ting, BI Shi-wen. Electrochemical study on adsorption behavior of surfactants at β-2CaO・SiO2/NaAlO2 interface [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(8): 2416-2421.
[24] SUN Hui-lan, WANG Bo, ZHANG Jian-xin, ZONG Shu-feng, LIU Jia-jia. Secondary reaction mechanism of leaching process of calcium aluminate slag [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(4): 1334-1340.
于海燕,潘晓林,董凯伟,吴 艳
东北大学 冶金学院,沈阳 110819
摘 要:采用XRF、XRD、SEM-EDS和DSC-TG等技术手段研究不同P含量下铝酸钙熟料在高温烧结过程中的矿相转变、显微组织、自粉性能及其氧化铝浸出性能。1350 °C下烧结熟料主要由12CaO・7Al2O3、 CaO・Al2O3、β-2CaO・SiO2和γ-2CaO・SiO2等矿相组成;随着P含量的增加,β-2CaO・SiO2含量逐渐升高,而γ-2CaO・SiO2含量逐渐降低。熟料的自粉性能随着P含量的增加逐渐恶化。P在熟料中均匀分布,其存在抑制β-2CaO・SiO2向γ-2CaO・SiO2的转变。12CaO・7Al2O3和CaO・Al2O3的晶胞体积随着P含量的增加分别降低和提高,且P能降低铝酸钙化合物的形成温度。铝酸钙熟料的氧化铝浸出率随着P含量的增加明显降低,当P含量从0提高到0.85%时,氧化铝浸出率从95.01%降低到83.84%。
关键词:石灰烧结法;铝酸钙;硅酸二钙;晶体结构;自粉性能;浸出
(Edited by Wei-ping CHEN)
Foundation item: Projects (51674075, 51774079) supported by the National Natural Science Foundation of China
Corresponding author: Xiao-lin PAN; Tel: +86-24-83686460; E-mail: panxl@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(19)64975-7

