
Effect of EPS on adhesion of Acidithiobacillus ferrooxidans on chalcopyrite and pyrite mineral surfaces
YU Run-lan, OU Yang, TAN Jian-xi, WU Fa-deng, SUN Jing, MIAO Lei, ZHONG Dai-li
Key Laboratory of Biometallurgy of Ministry of Education, School of Minerals Processing and Bioengineering,
Central South University, Changsha 410083, China
Received 18 March 2010; accepted 2 July 2010
Abstract: Extracellular polymeric substances (EPS) were extracted from Acidithiobacillus ferrooxidans through sonication method associated with centrifugation, which was evaluated tentatively with 2-keto-3-deoxyoctonic acid (KDO) as the indicator of EPS by spectrophotometry. Then the effect of EPS of A. ferrooxidans on the adhesion on chalcopyrite and pyrite surfaces was studied through a series of comparative experiments. The untreated cells and EPS-free cells of A. ferrooxidans were mixed with EPS suspension, Fe2+ or Fe3+, respectively. The planktonic cells were monitored in 2 h during bioleaching. The results indicate that the presence of EPS on the cell is an important factor for the adhesion to chalcopyrite and pyrite. A decrease of attachment of A. ferrooxidans to minerals was produced by the deficiency of EPS, which can recover mostly when the EPS was re-added into the EPS-free cells. The restoring extent is more obvious in pyrite than in chalcopyrite. The extent of cell adhesion to chalcopyrite increased when EPS and Fe3+ added, and decreased when Fe2+ added, which imply the electrostatic interaction plays a main role in initial adhesion between bacteria and minerals and it is a driving force for bacteria to produce EPS probably as a result of regaining their attachment ability to copper sulphides.
Key words: attachment; Acidithiobacillus ferrooxidan; extracellular polymeric substances; chalcopyrite; pyrite
1 Introduction
Acidithiobacillus ferrooxidans as an acidophilic leaching bacterium play a pivotal role in the extraction of metals from sulfidic minerals by bioleaching[1-2]. Bioleaching of chalcopyrite, pyrite and other minerals is enhanced by the attachment of these bacteria[3-4]. It is increasingly accepted that extracellular polymeric substances (EPS) mediate the attachment of microorganisms to material surfaces[5], but the interface-action mechanism is sophisticated and not fully understood[6]. GEHRKE et al[7] demonstrated that A. ferrooxidans possessed an extracellular layer of polysaccharides, proteins and lipids and also showed that EPS was necessary for the attachment and subsequent leaching of sulfidic minerals. Because of the insolubility of metal sulphides, direct bacterial attack must be initiated by adhesion of cells to the substrate surface. Physical evidence of the adhesion of A. ferrooxidans onto mineral surfaces was described in several studies[8-10]. A prerequisite for initial attachment of bacterial cells is that EPS mediates this contact[3, 11-13].
The EPS consists mainly of extracellular lipopolysaccharide (LPS) and outer membrane proteins (OMP)[14]. The polymers’ composition depends on the extraction protocol used. Numerous EPS extraction methods have already been developed, but no universal method was adopted as a compromise between high yields of extraction and minimum cell lysis during the extraction process[15]. Some of the methods, especially the chemical protocols, may actually be in error, depending on the approach used. Some work[16-17] showed that the EPS extracted by chemical methods (EDTA, glutaraldehyde or formaldehyde + NaOH) are contaminated by the extracting reagents, which sometimes affects the EPS composition and also affects complexation properties. According to other work[18] by determining the pKas (acidic constant) of each EPS suspension, the physical protocols, except heating, are thus preferable for the extraction of EPS.
In contrast to cells of A. ferrooxidans grown on sulfur, cells of A. ferrooxidans grown on pyrite or Fe2+ incorporate uronic acid and Fe3+ in their EPS[19]. However, the EPS of A. ferrooxidans grown on chalcopyrite, which reflects the condition in bioleaching, was investigated rarely. In the present study, the EPS of A. ferrooxidans grown on chalcopyrite or pyrite as the energy source, were extracted by sonication method associated with centrifugation, and their efficiency was evaluated. The role of EPS during bioleaching chalcopyrite and pyrite concentrates was studied. Especially, the attachment capacity and recovery ability were investigated by addition of EPS suspension to the untreated cells and EPS-free cells as a comparison. Furthermore, the effect of ferric ions and ferrous ions on the attachment was also studied.
2 Experimental
2.1 Preparation of chalcopyrite and pyrite sample
The flotation chalcopyrite concentrate with a mean particle size of about 75 μm was obtained from Dexing Copper Mine (Jiangxi Province, China). The chalcopyrite sample contained 33.15% Cu, 29.86% Fe, 33.22% S, and minor amounts of quartz, pyrite and covellite as accessory minerals. The pyrite was bought in Changsha mineral market. The particles were crushed and ground to average size of 75 μm. The pyrite sample contained 40.11% Fe and 49.02% S.
The samples were washed with 1 mol/L HCl and 2 mol/L H2SO4 in turn three times, then washed with acetone to remove the flotation reagent. Then the chalcopyrite and pyrite concentrates were placed in air for 4 h, then dried at 100 °C for 24 h. Finally, the minerals were sterilized by UV for 24 h in asepsis room[20].
2.2 Culture and harvest of cells of A. ferrooxidans
A. ferrooxidans used in the experiments were obtained from the Key Laboratory of Biometallurgy of Ministry of Education, Central South University. They were initially adapted to grow using several subcultures in liquid medium through successive replacement of Fe2+ with chalcopyrite and pyrite, respectively. The supernatant from the culture was used as inoculum. Then they were grown in iron-free 9K medium supplemented with 1% chalcopyrite concentrate and 1% pyrite concentrate, respectively. The initial pH value was 2.0, the temperature of the shaking table was 30 °C, and the rotating speed was 180 r/min. The iron-free 9K medium contained 3.00 g/L (NH4)2SO4, 0.10 g/L KCl, 0.50 g/L K2HPO4, 0.50 g/L MgSO4・7H2O and 0.01 g/L Ca(NO3)2.
Cells were harvested by centrifugation and then suspended in iron-free 9K medium without energy sources. The concentration of the soliquid was over 1×108 cell/mL.
2.3 Extraction of EPS
The extraction conditions were evaluated through orthogonal experiments with two factors of ultrasonic time and ultrasonic power. According to a series of preliminary experiments, 6, 12, 18 and 24 min were selected as ultrasonic time and 40, 80, 120 and 160 W as ultrasonic power.
After the treatment of sonication and centrifugation(12 000g for 30 min)[21], the supernatant containing EPS and sediment was defined as EPS-free cells. According to the constituents of EPS of the A. ferrooxidans[22], lipopolysaccharide (LPS), of which 2-keto-3-deoxyoctonic acid (KDO) is a unique constituent, can be detected tentatively as an indicator of EPS. The supernatant solution was treated with 0.2 mol/L H2SO4 at 100 °C for 30 min to release KDO followed by reaction with 0.04 mol/L HIO4, 2.6% NaAsO2 and 0.6% thiobarbituric acid (TBA). The red chromophore thus formed, which has been stable for days at room temperature by adding dimethylsulfoxide (DMSO) to the reaction mixture[23]. It has an absorption maxima at 548 nm. Therefore, the best ultrasonic condition, which was adopted to extract EPS used in adhesion experiments, can be obtained from the 16 results of the above orthogonal experiments by spectrophotometry.
2.4 Attachment experiments
The EPS suspension and EPS-free suspension of A. ferrooxidans, which were re-suspended in iron-free 9K medium, were obtained after the extraction step. Iron-free 9K medium (10 mL) or EPS suspension (10 mL) were mixed with bacterial suspension (10 mL) containing either untreated cells or EPS-free cells of A. ferrooxidans. After 20 min, 0.75 g of chalcopyrite or pyrite was added into the mixture to investigate the attachment comparatively. In addition, on the basis of attachment experiments, similar experiments were carried out through adding 40 mg Fe2+ or Fe3+ to investigate the effect of Fe2+ or Fe3+ on the attachment.
Each adhesion experiment was carried out in test tubes, which were agitated at 180 r/min, 30 °C for 2 h. During the experiment the number of planktonic cells was monitored. The number of planktonic cells was determined by direct counting using a Petroff Hausser counting chamber with an optical microscope. All experiments were carried out at least twice and the standard deviations were equal to or less than 7.5%. The amount of attached cells was calculated by subtracting the planktonic from the initial cell number.
3 Results and discussion
3.1 Cell concentration of A. ferrooxidans with pyrite or chalcopyrite
Figure 1 shows the change of the bacterial concentration of A. ferrooxidans with pyrite or chalcopyrite as the only energy sources, respectively. An adaptive phase occurred in 3 d and the logarithmic growth phase was 4-8 d with pyrite as energy sources. While with chalcopyrite, the adaptive phase occurred in 4 d, and the logarithmic growth phase was 5-15 d. Then, the bacteria concentration began to decrease and a secondary growth peak appeared again, which was 16-17 d with pyrite and 18-19 d with chalcopyrite. According the change of EPS content with the growth time in some studies[24], the secondary growth phase is the best period for the development of EPS. Therefore, the bacterial cells were harvested after the secondary growth immediately in order to obtain more abundant EPS. The interference from impurities like autolysate and secondary metabolites, which were produced during the secondary growth, can be eliminated effectively by re-suspending the bacterial cells by iron-free 9K medium after the harvest.
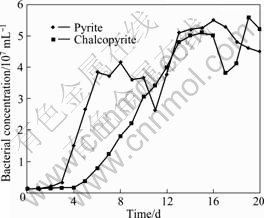
Fig.1 Bacterial growth curves of A. ferrooxidans with pyrite or chalcopyrite as energy source
3.2 Evaluation of sonication in extraction of EPS
As can be seen from Fig.2, the optical density (OD) value of KDO changed acutely with ultrasonic power. However, the change trends of OD value seemed to be irregular with the ultrasonic time. According to the result, we can conclude that the ultrasonic power is the significant factor. Obviously, for the 4 kinds of different ultrasonic power, the maximum of OD value appeared ultrasonic power of 80 W, no matter how long the sonication was operated. Therefore, 80 W was the most appropriate ultrasonic power for extraction of EPS of A. ferrooxidans by sonication. With ultrasonic power of 80 W, the OD value increased firstly because more and more EPS were shaken off from bacterial cells with increasing the time. The largest efficiency of extraction was obtained at 12 min. Then the OD value began to decrease gradually. The reason may be concluded that the component of KDO was oxidized and decomposed by the radical which was produced during the sonication[25]. Moreover, the thermal effect was more remarkable with increasing the ultrasonic time, which can be attributed to the degradation of EPS. As shown in Fig.2, as for the other 3 kinds of power, the OD value changed little with the time. In conclusion, the best ultrasonic conditions to get the highest content of EPS is under 80 W for 12 min. The results indicated that the EPS extraction method using sonication associated with centrifugation is feasible. However, the method of using KDO as an indicator of EPS is tentative, which needs further research on its feasibility and veracity.

Fig.2 Change of OD value of KDO from EPS of A. ferrooxidans
3.3 Attachment
3.3.1 Effect of EPS on cell adhesion to chalcopyrite and pyrite
The adhesion amount of cell to chalcopyrite and pyrite is shown in Fig.3 and Fig.4, respectively. In a comparison between the parallel experimental groups of chalcopyrite and pyrite, the attachment ability of A. ferrooxidans to pyrite was superior to that to chalcopyrite. This validates that the attachment of A. ferrooxidans is mineral-selective[19]. The decrease of attachment of A. ferrooxidans to minerals was produced by the deficiency of EPS, as indicated both in Fig.3 and Fig.4. This could be partially recovered by the addition of EPS suspension to EPS-free cells, but the extent was different. The speed and capacity of attachment of EPS-free cells were much smaller than those of the original, which was recovered over 80% in the initial 90 min as soon as the EPS was added. As for pyrite, it was recovered mostly in 20 min. The difference can be attributed to the component of EPS, which varies with the culture. The concentration of EPS with pyrite as the substrate was over twice that with chalcopyrite as the substrate[26]. Therefore, the concentration of EPS suspension extracted from A. ferrooxidans with pyrite as the energy source is higher than that with chalcopyrite, which probably results in the difference of their recovery extent. Nevertheless, it cannot be neglected that the adhesion extent of EPS-free cells increased spontaneously with the time slightly. That may be suggested that EPS-free cells regained their ability to attach to minerals. These results are in agreement with the previous work[27], which indicates that EPS-free cells regained their adhesion ability to the covellite after about 24 h.

Fig.3 Adhesion of A. ferrooxidans to chalcopyrite

Fig.4 Adhesion of A. ferrooxidans to pyrite
Our results suggested that the presence of EPS on the cell surface of A. ferrooxidans is an important factor for the attachment to chalcopyrite and pyrite. These results were in agreement with those obtained by GEHRKE et al[7] with pyrite. When EPS-free cells were mixed with EPS-suspension, their capacity of attachment to chalcopyrite was restored. In contrast, the adhesion capacity of EPS-free cells to chalcopyrite was low. These facts are similar to the results of previous studies about LPS[28].
3.3.2 Effect of EPS with iron ions on cell adhesion to chalcopyrite and pyrite
In order to investigate the role of EPS and exopolymer-complexed Fe3+ in the interaction extraction, experiments using untreated A. ferrooxidans, EPS and Fe2+ or Fe3+ were performed. As can be seen from Fig.5, the extent of cell adhesion to chalcopyrite increased in the presence of EPS or EPS and Fe3+, and it became weak when Fe2+ was added. Nevertheless, the extent of cell adhesion to chalcopyrite increased with time more or less in all the experiments.
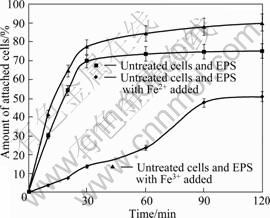
Fig.5 Adhesion of A. ferrooxidans to chalcopyrite
GEHRKE et al[7] proposed that ferric ions were entrapped in the EPS layer. In addition, FU et al[6] observed iron ion in the EPS of A. ferrooxidans by electronic microscope and cellular chemistry. And also, some researches[12, 29] found that the isoelectric points of iron-grown cells were at pH 2.0. These implied that the cells were positively charged in our attachment experiments. According to previous study [30], copper sulphides have a negative surface charge. Therefore, these facts implied that the electrostatic interactions may play a main role in initial adhesion. And the electrostatic interaction may be also the driving force for bacteria to produce EPS as a result of regaining their ability to attach to copper sulphides.
As can be expected, the adhesion extent of cells reached the maximum value when the EPS and Fe3+ were added simultaneously. The added EPS increased the positive charges by chelating Fe3+, and made bacteria attach to chalcopyrite surface easily due to sulphides having a negative surface charge. Therefore, the electrostatic interaction may play an important role in adhesion between bacteria and chalcopyrite. However, in the similar experiments with Fe2+ added, the adhesion of bacteria became weak, suggesting that bacteria use Fe2+ as energy source more easily in the condition of chalcopyrite. The bacteria don’t have to congregate on the surfaces of minerals to obtain energy, which go against the production of EPS. In contrast, the role of Fe3+ added is to stimulate bacteria to attach to the mineral surface and produce EPS.
4 Conclusions
1) Extraction of EPS from A. ferrooxidans using sonication associated with centrifugation is feasible. The best ultrasonic condition is under 80 W for 12 min.
2) The attachment of A. ferrooxidans to minerals decreases by the deficiency of EPS. The capacity for attachment can restore when EPS-free cells are mixed with EPS suspension. The extent of restoring in pyrite is more obvious than that in chalcopyrite.
3) The extent of cell adhesion to chalcopyrite increases when EPS and Fe3+ are added, and becomes weak when Fe2+ is added, which imply that the electrostatic interaction plays a main role in initial adhesion between bacteria and minerals and it is a driving force for bacteria to produce EPS as a result of regaining their ability to attach to copper sulphides.
References
[1] RAWLINGS D E, DEW D, DU PLESSIS C. Biomineralization of metal-containing ores and concentrates [J]. Trends Biotechnol, 2003, 21(1): 38-44.
[2] OLSON G J, BRIERLEY J A, BRIERLEY C L. Bioleaching review (part B): Progress in bioleaching: applications of microbial processes by the minerals industries [J]. Appl Microbiol Biotechnol, 2003, 63: 249-257.
[3] SAND W, GEHRKE T, HALLMANN R, SCHIPPERS A. Sulfur chemistry, biofilm and the (in)direct attack mechanism: A critical evaluation of bacterial leaching [J]. Appl Microbiol Biotechnol, 1995, 43(3-4): 961-968.
[4] ROHWERDER T, GEHRKE T, KINZLER K, SAND W. Bioleaching review (part A): Progress in bioleaching: Fundamentals and mechanisms of bacterialmetal sulfide oxidation [J]. Appl Microbiol Biotechnol, 2003, 63: 239-248.
[5] SAND W, GEHRKE H. Extracellular polymeric substances mediate bioleaching/biocorrosion via interfacial processes involving iron(Ⅲ) ions and acidophilic bacteria [J]. Research in Microbiology, 2006, 157: 49-56.
[6] FU Jian-hua, QIU Guan-zhou, HU Yue-hua, XU Jing. The role of EPS of Thiobacillus ferrooxidans during bioleaching [J]. Acta Laser Biology Sinica, 2004, 13(1): 62-66. (in Chinese)
[7] GEHRKE T, HALLMANN R, SAND W. Importance of exopolymers from Thiobacillus ferrooxidans and Leptospirillum ferrooxidans for bioleaching [J]. Biohydrometallurgical Processing, 1995, 1: 1-11.
[8] BLAKE R, LYLES M M, SIMMONS R. Morphological and physical aspects of attachment of Thiobacillus ferrooxidans to pyrite and sulphur [J]. Biohydrometallurgical Processing, 1995, 1: 13-22.
[9] POGLIANI C, CURUTCHET G, DONATI E, TEDESCO P. A need for direct contact with particle surfaces in the bacterial oxidation of covellite in the absence of a chemical lixiviant [J]. Biotechnol Lett, 1990, 12: 515-518.
[10] SOLARI J A, HUERTA G, ESCOBAR B, VAGRAS T, BADILLA-OHLBAUM R, RUBIO J. Interfacial phenomena affecting the adhesion of Thiobacillus ferrooxidans to sulphide mineral surfaces [J]. Colloids Surfaces, 1993, 69: 159-166.
[11] SAND W, GEHRKE T, JOZSA P G, SCHIPPERS A. Direct versus indirect bioleaching [C]//AMILS R. Biohydrometallurgy and the Environment Toward the Mining of the 21st Century (Part A). Amsterdam: Elsevier, 1999: 27-49.
[12] KAR N, DASGUPTA A. The possible role of surface charge in membrane organization in an acidophile[J]. Biochem Biophys,1996, 33: 398-402.
[13] NORRIS P R. Acidophilic bacteria and their activity in mineral sulphide oxidation [C]//EHRLICH H L, BRIERLEY C L. Microbial Mineral Recovery. New York: McGraw-Hill, 1990: 3-28.
[14] AGATE A D, KOREZYNSKI M S, LUNDGREN D G. Extracellular complex from the culture filtrate of Thiobacillus ferrooxidans [J]. Microbiol, 1969, 15: 59-264.
[15] LIU Y, FANG H H P. Influences of extracellular polymeric substances (EPS) on flocculation, settling, and dewatering of activated sludge [J]. Crit Rev Environ Sci Technol, 2003, 33: 237-273.
[16] COMTE S, GUIBAUD G, BAUDU M. Relations between extraction protocols of the activated sludge extracellular polymeric substances (EPS) and EPS complexation properties: part I. Comparison of the efficiency of eight EPS extraction properties [J]. Enzyme Microbial Technol, 2006, 38: 237-245.
[17] CHENG M H, PATTERSON J W, MINEAR R A. Heavy metals uptake by activated sludge [J]. Journal of the Water Pollution Control Federation, 1975, 47: 362-376.
[18] COMTE S, GUIBAUD G, BAUDU M. Relations between extraction protocols of the activated sludge extracellular polymeric substances (EPS) and EPS complexation properties: part II. Consequences of EPS extraction methods on Pb2+ and Cd2+ complexation [J]. Enzyme Microbial Technol, 2006, 38: 246-252.
[19] HARNEIT K, G?KSEL A, KOCK D, KLOCK J H, GEHRKE T, SAND W. Adhesion to metal sulfide surfaces by cells of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans and Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2006, 83: 245-254.
[20] YU Run-lan, TAN Jian-xi, YANG Peng, SUN Jing, OU Yang, XIONG Jing, DAI Yun-jie. EPS-contact-leaching mechanism of chalcopyrite concentrates by A.ferrooxidans [J]. Transactions of Nonferrous Metals Society of China, 2008, 18: 1427-1432.
[21] GEHRKE T, TELEGDI J, THIERRY D, SAND W. Importance of extracellular polymeric substances from Thiobacillus ferrooxidans for bioleaching [J]. Appl Environ Microbiol, 1998, 64(7): 2743-2747.
[22] LIU Jian-she, YAN Ying, WANG Hua-tai, WANG Xiu-mei. Progress in research on extracellular polymeric substance of Thiobacillus ferrooxidans[J]. Metal Mine, 2007(12): 14-16. (in Chinese)
[23] KARKHANIS Y D, ZELTNER J Y, JACKSON J J, CARLO D J. A new and improved microassay to determine 2-keto-3-deoxyoctonate in lipopolysaccharide of gram-negative bacteria[J]. Analytical Biochemistry, 1978, 85: 595-601.
[24] ZENG Wei-min, QIU Guan-zhou, ZHOU Hong-bo, LIU Xue-duan, CHEN Miao, CHAO Wei-liang, ZHANG Cheng-gui, PENG Juan-hua. Characterization of extracellular polymeric substances extracted during the bioleaching of chalcopyrite concentrate [J]. Hydrometallurgy, 2010, 100: 177-180.
[25] LONG Teng-rui, LI Jin-yin, LONG Xiang-yu, LUO Tai-zhong, TANG Ran. Extracting extracelluar polymer subtance from the actived sludge using ultrasonic[J]. Environmental Chemistry, 2008, 27(3): 310-313. (in Chinese)
[26] KINZLER K, GEHRKE T, TELEGDI J, SAND W. Bioleaching―A result of interfacial processes caused by extracellular polymeric substances(EPS) [J]. Hydrometallurgy, 2003, 71: 83-88.
[27] POGLIANI C, DONATI E. The role of exopolymers in the bioleaching of a non-ferrous metal sulphide [J]. Journal of Industrial Microbiology & Biotechnology, 1999, 22: 88-92.
[28] ESCOBAR B, HUERTA G, RUBIO J. Influence of lipopolysaccharides on the attachment of Thiobacillus ferrooxidans to minerals[J]. World J Microbiol Biotechnol, 1997, 13: 593-594.
[29] DEVASIA D, NATARAJA K A, SATHYANARYANA D N, RAO G R. Surface chemistry of Thiobacillus ferrooxidans relevant to adhesion on mineral surfaces [J]. Appl Environ Microbiol, 1993, 59: 4051-4055.
[30] SKLODOWSKA A, MATLAKOWSKA R. Influence of exopolymers produced by bacterial cells on hydrophobicity of substrate surface [J]. Biotechnol Technol, 1997, 11: 837-840.
EPS对Acidithiobacillus. ferrooxidans 在黄铜矿与黄铁矿表面吸附的影响
余润兰, 欧 阳, 谭建锡, 吴发登, 孙 静, 苗 雷, 钟代立
中南大学 资源加工与生物工程学院 生物冶金教育部重点实验室,长沙 410083
摘 要:运用超声波结合离心方法提取Acidithiobacillus. ferrooxidans 的胞外多聚物(EPS),用2-酮基-3-脱氧辛酸(KDO)作为表征EPS含量的指标,采用分光光度法对该提取方法进行评估。通过一系列对比性实验研究EPS对Acidithiobacillus ferrooxidans 在黄铜矿与黄铁矿表面吸附的影响。将未处理的Acidithiobacillus ferrooxidans与经处理脱去EPS层的Acidithiobacillus ferrooxidans 分别与EPS悬液、Fe2+ 和 Fe3+重新混合,在2 h的反应过程中,实时检测混合液中游离的细菌含量。结果表明:细菌表面EPS的存在是其吸附于黄铁矿和黄铜矿表面的一个重要因素。当缺失EPS层时,Acidithiobacillus ferrooxidans 吸附于矿物表面的能力有所下降,但当重新加入EPS混合液时这种能力能大部分恢复,这种恢复程度在黄铁矿中较黄铜矿中更加明显。当加入EPS 和 Fe3+时其细菌吸附于黄铜矿表面的程度有所升高,而加入Fe2+ 时吸附程度明显降低,这个结果表明静电的相互作用也许是细菌最初吸附于矿物表面的一个主要原因,并且这也许是细菌生产EPS的一种驱动力以使细菌吸附于硫化铜矿物后重新获得其吸附能力。
关键词:吸附;A. ferrooxidan;胞外多聚物(EPS);黄铜矿;黄铁矿
(Edited by YUAN Sai-qian)
Foudation item: Project (2010CB630901) supported by the National Basic Research Program of China; Project (50621063) supported by the National Natural Science Foundation of China
Corresponding author: YU Run-lan; Tel: +86-731-88716592; E-mail: YRL715@sina.com.cn
DOI: 10.1016/S1003-6326(11)60729-2