���ּ������ȹž��Ի�ͭ��������ͽ�����Ϊ
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2018���12��
�������ߣ������� ����ΰ ������ ������
����ҳ�룺2538 - 2544
�ؼ��ʣ��������ȹž���A. brierleyi��S. metallicus����ͭ������������
Key words��thermophilic archaea; A. brierleyi; S. metallicus; chalcopyrite; adsorption; leaching
ժ Ҫ���о�������д����Եļ������ȹž�A. brierleyi��S. metallicus��������Ի�ͭ��������ͽ�����Ϊ�����������S. metallicus��ͭ�������Ը���A. brierleyi�ģ����л�Ͼ���ϵ��ͭ��������ߡ����������е�Ⱥ��ṹ����������S. metallicus�ھ�Ⱥ���������ƾ�Ⱥ��A. brierleyi��ռ���������������ƣ�����һ�����������ͭ���ڻ����ϵ�������ڵ�ͭŨ�ȱ��ڵ�����ϵ���������йء�Langmuir���������ó����ֹž�֮�䲻���ھ�����������ϵ����qPCR�����ʾ���ڻ�Ͻ����Ĺ����У�S. metallicus��A. brierleyi֮�������ٽ������������о���һ��������ϼ������Ⱦ��ڿ����������������������Ϊ��
Abstract: The adsorption and leaching of chalcopyrite by two extreme thermophilic archaea (A. brierleyi and S. metallicus) and their mixture were studied. The results revealed that the chalcopyrite leaching rate of S. metallicus was slightly higher than that of A. brierleyi; the mixed system showed the highest rate. Community structure analysis during the leaching process showed that S. metallicus was maintained in a predominant state. However, the proportion of A. brierleyi in the community increased during leaching. Copper concentrations, which increased faster in the mixed system than in the single-organism systems during later stages, was related to the change of A. brierleyi in the community. Langmuir parameter analysis revealed no competitive adsorption between these two thermophilic archaea. Furthermore, qPCR (quantitative polymerase chain reaction) confirmed that adsorption was promoted between A. brierleyi and S. metallicus during mixed leaching. These findings can improve our understanding of the adsorption behaviors of mixed extreme microbial populations on mineral surfaces.
Trans. Nonferrous Met. Soc. China 28(2018) 2538-2544
Yu-ting LIANG, Jun-wei HAN, Chen-bing AI, Wen-qing QIN
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 23 May 2018; accepted 27 November 2018
Abstract: The adsorption and leaching of chalcopyrite by two extreme thermophilic archaea (A. brierleyi and S. metallicus) and their mixture were studied. The results revealed that the chalcopyrite leaching rate of S. metallicus was slightly higher than that of A. brierleyi; the mixed system showed the highest rate. Community structure analysis during the leaching process showed that S. metallicus was maintained in a predominant state. However, the proportion of A. brierleyi in the community increased during leaching. Copper concentrations, which increased faster in the mixed system than in the single-organism systems during later stages, was related to the change of A. brierleyi in the community. Langmuir parameter analysis revealed no competitive adsorption between these two thermophilic archaea. Furthermore, qPCR (quantitative polymerase chain reaction) confirmed that adsorption was promoted between A. brierleyi and S. metallicus during mixed leaching. These findings can improve our understanding of the adsorption behaviors of mixed extreme microbial populations on mineral surfaces.
Key words: thermophilic archaea; A. brierleyi; S. metallicus; chalcopyrite; adsorption; leaching
1 Introduction
Rich, global ore resources are becoming increasingly scarce because of their continuous development and utilization. Researchers in China have efficiently and rationally developed and utilized metal mineral resources classified as low-grade and difficult to process [1]. Recently, bioleaching has emerged as an effective method for processing low-grade minerals because of its low cost, energy consumption, and environmental impact [2-4]. According to their optimum growth temperatures, common metallurgical micro- organisms have been classified into mesophilic, moderately thermophilic, and extremely thermophilic ones. Mesophilic and moderately thermophilic organisms are currently used in industrial applications [5]. However, the application of extreme thermophilic microorganisms is rare. Extreme thermophilic archaea show high temperature resistance, high extraction speed, and high leaching rates compared to mesophilic bacteria; thus, such organisms have great potential in industrial applications [6] and have become a focal point of research.
The most important factors influencing bioleaching are microorganisms which play very important roles in mineral leaching. The roles of various microorganisms in the leachate are highly complex and may be both positive and negative. The microorganisms are considered to coexist in synergistic, competitive, or mutually beneficial symbiotic relationships [7]. These different relationships are based on different physiological and biochemical characteristics of the various microorganisms, such as carbon fixation [8], sulfur oxidation and reduction [9], nitrification fixation, oxidation of organic matter [10,11], and ferrous iron oxidation and reduction capability [12]. Competitive relationships in bio-metallurgy mainly refer to substrate competition between organic and inorganic electron donors [13-15]. Environmental factors, such as temperature, pH, metal ion concentrations, and substrate concentrations, also strongly influence the competitive relationships in bio-metallurgy [16]. In contrast, in the physiological complementation, microorganisms synergistically oxidize and decompose minerals in mixed culture and display significant promoting effects. For example, chalcopyrite leaching efficiency in mixed leaching systems of Leptospirillum ferrooxidans (L. ferrooxidans) and Acidithiobacillus thiooxidans (A. thiooxidans) was significantly higher than that in any single-organism system [17-19]. This was primarily because despite their inability to directly oxidize and decompose minerals, sulfur-oxidizing bacteria produce acid, remove sulfur passivation film, and prevent the formation of jarosite. Additionally, related studies showed that minerals can only be oxidized and decomposed when Ferroplasma and A. thiooxidans coexist [17]. Regardless of the relationship between microorganisms, during the leaching process, the surface of the mineral is adsorbed first [20,21]. This step can also determine the relationship between populations during the leaching process to some extent. Microbial adsorption on the mineral surface is a prerequisite for effective interfacial interactions and plays an important role in the mineral dissolution process. It has been reported that the surface properties of minerals and different energy metabolism strategies of microorganisms affect the adsorption behaviors, thereby affecting microbial-mineral interactions.
Few studies have examined the interactions between extreme thermophiles in bioleaching. However, the use of thermophilic archaea in leaching processes has broad application potential. Therefore, Acidianus brierleyi and Sulfolobus metallicus were selected as representative thermophilic leaching microorganisms, to study mutual relationships in the adsorption of chalcopyrite surfaces and their influence on leaching processes. Both archaea are thermophilic autotrophic paleontological archaea that can grow in the presence of ferrous, sulfur, and reducing inorganic sulfur compounds. According to previous studies, A. brierleyi has strong adaptability and sulfur oxidation capacity and S. metallicus has a very strong ferrous oxidation ability, but it is sensitive to environmental conditions [22-24]. Thus, both strains are representative of extremely thermophilic archaea.
2 Experimental
2.1 Organisms and minerals
Chalcopyrite was supplied by the Key Lab of Biohydrometallurgy of the Ministry of Education, Central South University, Changsha, China. Pure chalcopyrite minerals were manually selected and ground to 0.045-0.074 mm in diameter. For adsorption and zeta potential measurements, chalcopyrite was ground with a diameter <5 ��m using agate. The mass fractions of elemental copper, iron, and sulfur were 31.45%, 26.74%, and 31. 87%, respectively.
A. brierleyi and S. metallicus were obtained from the Key Lab of Biohydrometallurgy of the Ministry of Education, Central South University, Changsha, China. They were inoculated in basal salt medium and supplemented with 1% elemental sulfur and 0.02% yeast extract [25]. The pH of the medium was adjusted to be 2.0 with 2.5 mol/L H2SO4. The basal salt medium consisted of 3.09 g/L (NH4)2SO4, 0.1 g/L KCl, 0.5 g/L K2HPO4, 0.5 g/L MgSO4��7H2O, and 0.01 g/L Ca(NO3)2.
2.2 Bioleaching experiment
Wide-mouth conical flasks (250 mL) and basal salt medium with pH 2.0 were utilized in the leaching experiment. The pH required no adjustment. The leaching test was performed in a high-temperature water bath oscillator at 68 ��C and a rotation speed of 170 r/min. Chalcopyrite powders were placed into the flask to a mineral concentration of 0.2% (w/v). In the single- archaea leaching test, the leaching microorganisms were present at a concentration of 1��107 cell/mL. The mixed sample consisted of equal amounts of the two archaea in a total concentration of 1��107 cell/mL. A blank control group without archaea was also used. For the initial mixed-system experiment, 18 parallel samples with the same initial leaching conditions were used, with three parallel samples in each group. Thus, six flasks were used at each stage and were divided into two groups. One group was used to extract DNA and slag from the solutions, while the other group was used to construct adsorption curves. The microorganisms were collected and centrifuged at 3000 r/min for 2 min. The supernatant was separated, and then the slag was mixed with sterile PBS buffer. After magnetic stirring at high speed for 10 min, the washing solution was collected under aseptic conditions for DNA extraction. The process was repeated until no further organisms were microscopically identified in the slag. The supernatant was collected, and its slag washing solution was centrifuged to collect microorganisms for later use.
2.3 Experimental sampling and analysis of leaching process
The pH levels and redox potential were measured every two days. Additionally, 2 mL solution was extracted and used to measure the Cu2+, Fe2+, and Fe3+ contents. Collected samples were replaced with the same volumes of medium. Evaporation losses from the solution during testing were replaced with the same amount of sterile water. pH values were measured using a pH meter (PHS-3C), and the oxidation-reduction potential (��) was measured by an inductively coupled plasma atomic emission spectrometer (ICP-AES) combined with Ag/AgCl platinum (3 mol/L KCl) (bpp-922). The concentrations of Cu2+ and total iron in the solution were determined by atomic absorption spectrophotometry. Additionally, Fe2+ levels were determined by potassium dichromate titration, and the Fe3+ concentration was calculated by subtracting the Fe2+ concentrations from the total iron concentrations.
2.4 Analysis of microbiological communities
DNA extraction was conducted as described previously [26]. Free and attached microorganisms were harvested by filtration and centrifugation (12000 r/min, 20 min). The attached cells were collected from the ore in advance by repeated vortexing and resuspension. DNA was extracted immediately using the TIANamp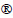 Bacterial DNA Kit (Tiangen, Beijing, China) according to the manufacturer��s instructions. All tests were performed in triplicate. The real-time quantitative polymerase chain reaction (qPCR) was used to analyze the population dynamics during bioleaching. The quality of the amplified DNA fragment stained with ethidium bromide was examined with 1.5% agarose gel electrophoresis, followed by purification of the DNA in the bands using the QIAquick-spin PCR Purification Kit was used (Qiagen, Hilden, Germany). Next, DNA sequencing was performed by Shanghai Biotech, China, and BLAST analysis was performed in GenBank (http://www.ncbi.nlm.nih.gov/BLAST/). Each sample leaching system was evaluated as follows: pre-denaturation at 95 ��C for 3 min, 95 ��C for 30 s, 60 ��C for 30 s, and 72 ��C for 30 s for a total of 40 cycles. qPCR data were analyzed using iCycler MyiQ software v1.0 (Bio-Rad, Hercules, CA, USA). The primers were arranged as described previously [27]. The six standard curves generated from known concentrations of PCR products were used to quantify the copy number of each conserved gene. The correlation coefficient of the standard curve of each gene was greater than 0.99, and the PCR amplification efficiency was between 90% and 110%. The number of cells for each strain was calculated from the total number of cells and percentage of each strain.
Bacterial DNA Kit (Tiangen, Beijing, China) according to the manufacturer��s instructions. All tests were performed in triplicate. The real-time quantitative polymerase chain reaction (qPCR) was used to analyze the population dynamics during bioleaching. The quality of the amplified DNA fragment stained with ethidium bromide was examined with 1.5% agarose gel electrophoresis, followed by purification of the DNA in the bands using the QIAquick-spin PCR Purification Kit was used (Qiagen, Hilden, Germany). Next, DNA sequencing was performed by Shanghai Biotech, China, and BLAST analysis was performed in GenBank (http://www.ncbi.nlm.nih.gov/BLAST/). Each sample leaching system was evaluated as follows: pre-denaturation at 95 ��C for 3 min, 95 ��C for 30 s, 60 ��C for 30 s, and 72 ��C for 30 s for a total of 40 cycles. qPCR data were analyzed using iCycler MyiQ software v1.0 (Bio-Rad, Hercules, CA, USA). The primers were arranged as described previously [27]. The six standard curves generated from known concentrations of PCR products were used to quantify the copy number of each conserved gene. The correlation coefficient of the standard curve of each gene was greater than 0.99, and the PCR amplification efficiency was between 90% and 110%. The number of cells for each strain was calculated from the total number of cells and percentage of each strain.
2.5 Strain adsorption experiments
The pH of NaCl solution with an ionic strength of 1��10-3 mol/L was adjusted to 2.0. The volume of the solution was 100 mL, and then 0.2 g chalcopyrite was added and shaken at 170 r/min and 68 ��C for 1 h. In separate adsorption experiments, the concentration of archaea in the solution was adjusted to be 1.0��106 cell/mL. In the mixed archaea adsorption experiment, the same number of archaea was used to adjust the archaea concentration in the solution to be 1.0��106 cell/mL. The number of archaea in the separate adsorption experiments was directly counted using a microscope. In the mixed archaea adsorption experiment, the number of each archaea was determined by qPCR. To eliminate the measurement error, sample determination and quantitative standard curve establishment were performed simultaneously in the same operation, and all experiments were performed three times. Both the initial archaeal amount (1.0��108 cells) and mineral mass (0.2 g) in the solution, and the number of archaea in the liquid at the time of adsorption equilibrium (CL) were all used to calculate the number of adsorbed archaea (CA) [28].
3 Results and discussion
3.1 Bioleaching experiment
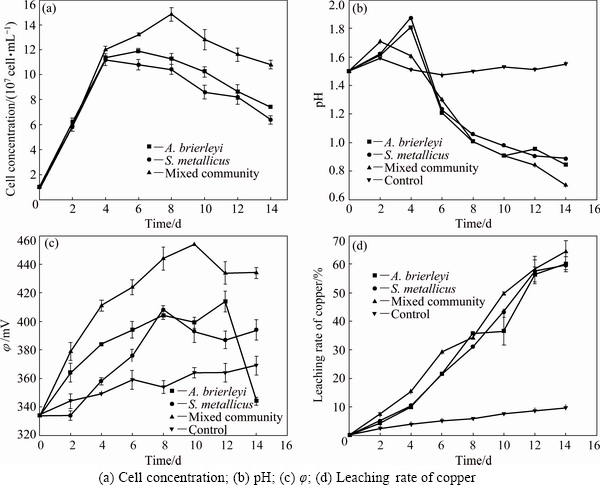
Fig. 1 Differences in chalcopyrite leaching of single- and mixed-archaea systems
Fig. 2 Concentrations of iron ion of A. brierleyi (a), S. metallicus (b), mixed community (c) and control group (d)
Changes in planktonic conditions of the leaching- solution from the three experimental groups are shown in Fig. 1(a). During the early period, planktonic levels rose sharply, and then began to decrease during the middle and late periods. Compared with that of the single-archaea system, the decrease of the mixed system was slower. Figure 1(b) shows the changes in pH levels during the leaching process for the three experimental groups. While the pH of the blank control slowly decreased, the pH levels in the three experimental groups increased during the early stage and declined rapidly at the middle and later stages. As shown in Fig. 1(c), the �� of the blank control showed a steady and slow-rising trend. The experimental groups with the two single strains showed obviously upward trends, and only showed significant decrease near the end of the leaching process. The �� of the mixed archaea system changed similarly to those of the single-archaea systems at the early and middle stages of the leaching process. However, no significant decrease was observed at the end of the leaching process. This difference was similar to that between copper ion concentrations in the leachate of the mixed and single-archaea systems, as detailed in Fig. 1(d). The leaching rates of copper ions after leaching with A. brierleyi and S. metallicus were 60.01% and 59.61%, respectively, while the copper leaching rate of mixed archaea was 64.43%. Therefore, the leaching rate of the mixed thermophilic archaea system was significantly greater than that of the single-archaea systems, with the copper leaching rate increased by approximately 5%. As shown in Fig. 2, the concentration of ferrous ions in the archaeal system was low during the leaching process and showed minimal changes, because archaea oxidized ferrous ions released by the dissolution of chalcopyrite into ferric ions, and thus the iron in the leaching system is mainly in the form of ferric ion. The ferric ion concentration in the two groups of single archaea solution began changing in the later stage. The plateau concentration of the mixed archaea still shows a rapidly increasing trend. Compared to the copper ion concentration change of leaching in Fig. 1(d), according to the previous study, Fe3+ combines with  in solution to form jarosite. The formation of jarosite during leaching of chalcopyrite by the mixed archaea system was slowed, and thus the leaching rate of copper ions of the mixed archaea system was higher than that of the single-archaea system.
in solution to form jarosite. The formation of jarosite during leaching of chalcopyrite by the mixed archaea system was slowed, and thus the leaching rate of copper ions of the mixed archaea system was higher than that of the single-archaea system.
3.2 Analysis of microbial communities during mixed-archaea leaching process
Figure 3(a) shows the strains in the leaching system on the second day of the leaching process. As for the planktonic archaea in solution and those adsorbed on the surface of the chalcopyrite, the predominant strain was S. metallicus, while A. brierleyi was less abundant. This is because S. metallicus has a comparatively high ability to oxidize ferrous ion in low-Fe2+-concentration systems. Comparison of planktonic and adsorbed populations revealed that the proportion of S. metallicus in the planktonic archaea population was approximately 5.28% higher than that in the adsorbed microbial population. As shown in Fig. 3(b), in the middle of the leaching process (day 8), changes were observed in the microbial communities. Although S. metallicus was still a dominant species, the proportions of A. brierleyi were increased by 2.6% and 0.53% of the planktonic and adsorbed populations, respectively, while the proportion of S. metallicus was decreased by the same percentage. Figure 3(c) shows the microbial community structure at the end stage of the leaching process (day 14). Although the predominant microbial population was still S. metallicus (56.74% of planktonic and 66% of adsorbed), the percentage of A. brierleyi increased significantly by 15.39% (planktonic) and 2.87% (adsorbed). Throughout the leaching process, A. brierleyi increased, likely because of its strong sulfur oxidation activity. Additionally, instability of S. metallicus in the leaching environment may have caused its proportion in the leaching system to decrease.
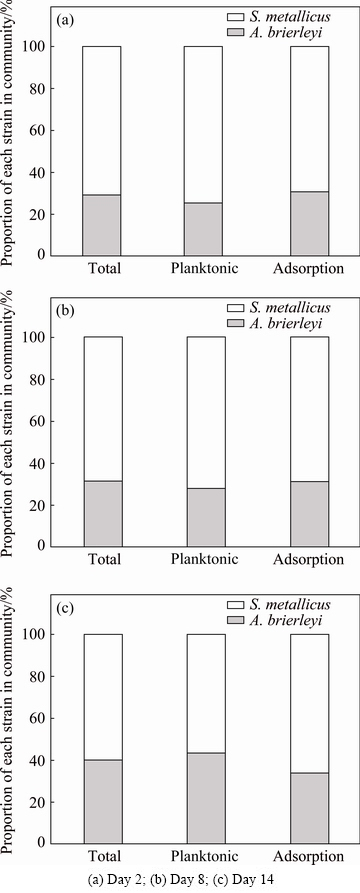
Fig. 3 Population structure of chalcopyrite leachate from mixed-archaea system including total community, planktonic community, and community adsorbed on surface of minerals at different time
3.3 Adsorption of chalcopyrite by microorganisms
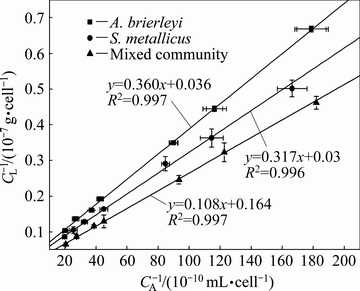
Fig. 4 Langmuir curves of A. brierleyi, S. metallicus, and mixed community adsorbed on chalcopyrite surface
Langmuir curves of each separate strain and the mixed community on chalcopyrite are shown in Fig. 4. Based on the adsorption data, the reference parameters of the isothermal adsorption curve model were obtained by linear regression using Eq. (1) [28] as follows:
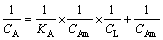 (1)
(1)
where CA denotes the amount of microorganisms adsorbed per unit mass of chalcopyrite when the absorption balance is reached, KA represents the adsorption equilibrium constant, CL is the amount of planktonic microbial mass in the solution, and CAm represents the ability of unit chalcopyrite to adsorb the maximum cell number. Next, KA and CAm were obtained by plotting CL/CA against CA.
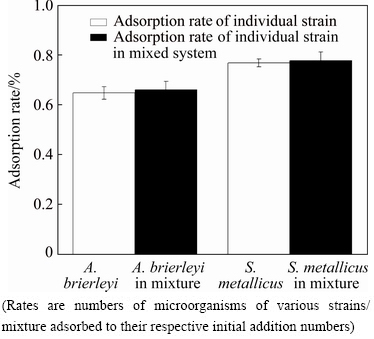
Fig. 5 Adsorption rates of individual strain and mixed system
The CAm (6.113��1012 cell/g) of the mixed system was 2.18- and 1.86-fold CAm of the two single-strain adsorption models, respectively, and the CAm values of A. brierleyi and S. metallicus were 2.807��1012, 3.287��1012 cell/g, respectively. The CAm value of the mixed system approached the sum of the CAm values of the two individual archaea (6.094��1012 cell/g). These findings indicated that the two strains had their own unique adsorption sites on the chalcopyrite surface, and nearly no common adsorption sites were observed. Therefore, the two strains produced no competitive adsorption in the mixed system. The KA value of the same mixed system (1.97��10-9 mL/cell) was greater than that of the two strains when adsorbed separately (A. brierleyi: 0.98��10-9 mL/cell and S. metallicus: 0.96��10-9 mL/cell). According to previous studies, when the CAm and KA values of the mixed system are greater than the sum of the two single archaea, the promotion of adsorption occurs before the mixed archaea group.
The two strains in the mixed system influenced one another. Therefore, qPCR was conducted to measure the number of microorganisms that were adsorbed by each strain in the mixed system. As shown in Fig. 5, the adsorption rates of A. brierleyi and S. metallicus were 64% and 76.7%, respectively, when they were individually adsorbed to equilibrium. However, when the two strains in the mixed system were adsorbed in solution until equilibrium was reached, their initial adsorption rates were 66.01% and 77.59%, respectively. Therefore, the adsorption rates of both strains in the mixed system increased. This result indicated that when the mixed system was adsorbed on the surface of the chalcopyrite, adsorption occurred between A. brierleyi and S. metallicus, and both showed increased adsorption rates on the mineral surface.
4 Conclusions
The results of this study showed that the leaching rate of chalcopyrite by S. metallicus was slightly higher than that of A. brierleyi. Additionally, the chalcopyrite leaching rate of a mixed system of both strains was much higher than that of the single strain. Community structure analysis during the leaching process demonstrated that S. metallicus was consistent in a predominant state within the community. However, the proportion of A. brierleyi in the community exhibited an increasing trend. Changes in the concentrations of copper in the leachate were due to the fast increase of the leaching rate of the mixed archaea than that of two single-archaea systems in the later stage of leaching. Langmuir parameter analysis revealed no competitive adsorption between the two thermophilic archaea. Moreover, qPCR confirmed that adsorption promotion occurred between S. metallicus and A. brierleyi in the mixed system.
References
[1] LI Y, KAWASHIMA N, LI J, CHANDRA A, GERSON A. A review of the structure, and fundamental mechanisms and kinetics of the leaching of chalcopyrite [J]. Advances in Colloid and Interface Science, 2013, 197: 1-32.
[2] PATHAK A, MORRISON L, HEALY M G. Catalytic potential of selected metal ions for bioleaching, and potential techno-economic and environmental issues: A critical review [J]. Bioresource technology, 2017, 229: 211-221.
[3] DONATI E R, CASTRO C, URBIETA M S. Thermophilic microorganisms in biomining [J]. World Journal of Microbiology and Biotechnology, 2016, 32(11): 1-8.
[4] CLARK M E, BATTY J D, VAN BUUREN C B, DEW D W, EAMON M A. Biotechnology in minerals processing: Technological breakthroughs creating value [J]. Hydrometallurgy, 2006, 83(1-4): 3-9.
[5] WATLING H R. The bioleaching of sulphide minerals with emphasis on copper sulphides��A review [J]. Hydrometallurgy, 2006, 84(1-2): 81-108.
[6] COUNTS J A, ZELDES B M, LEE L L, STRAUB C T, ADAMS M W W, KELLY R M. Physiological, metabolic and biotechnological features of extremely thermophilic microorganisms [J]. Wiley Interdisciplinary Reviews: Systems Biology and Medicine, 2017, 9(3): 1-23.
[7] RUIZ L M, VALENZUELA S, CASTRO M, GONZALEZ A, FREZZA M, SOULERE L, ROHWERDER T, QUENEAU Y, DOUTHEAU A, SAND W, JEREZ C A, GUILIANI N. AHL communication is a widespread phenomenon in biomining bacteria and seems to be involved in mineral-adhesion efficiency [J]. Hydrometallurgy, 2008, 94(1-4): 133-137.
[8] BERG I A, KOCKELKORN D, RAMOS-VERA W H, SAY R F, ZARZYCKI J, HUGLER M, ALBER B, FUCHS G. Autotrophic carbon fixation in archaea [J]. Nature Reviews: Microbiology, 2010, 8(6): 447-460.
[9] LING Wei-bo, WANG Lei, LIU Hong-chang, NIE Zhen-yuan, YANG Yun, YANG Yi, MA Chen-yan, ZHENG Lei, ZHAO Yi-dong, XIA Jin-lan. The evidence of decisive effect of both surface microstructure and speciation of chalcopyrite on attachment behaviors of extreme thermoacidophile Sulfolobus metallicus [J]. Minerals, 2018, 8(4): 159-173.
[10] CARDENAS J P, QUATRINI R, HOLMES D S. Genomic and metagenomic challenges and opportunities for bioleaching: A mini-review [J]. Research in Microbiology, 2016, 167(7): 529-538.
[11] KADNIKOV V V, IVASENKO D A, BELETSKY A V, MARDANOV A V, DANILOVA E V, PIMENOV N V, KARNACHUK O V, RAVIN N V. Effect of metal concentration on the microbial community in acid mine drainage of a polysulfide ore deposit [J]. Microbiology, 2016, 85(6): 745-751.
[12] ZHAO Hong-bo, WANG Jun, GAN Xiao-wen, ZHENG Xi-hua, TAO Lang, HU Ming-hao, LI Yi-ni, QIN Wen-qing, QIU Guan-zhou. Effects of pyrite and bornite on bioleaching of two different types of chalcopyrite in the presence of Leptospirillum ferriphilum [J]. Bioresource Technology, 2015, 194: 28-35.
[13] NI G, CHRISTEL S, ROMAN P, WONG Z L, BIJMANS M F M, DOPSON M. Electricity generation from an inorganic sulfur compound containing mining wastewater by acidophilic microorganisms [J]. Research in Microbiology, 2016, 167(7): 568-575.
[14] WHEATON G, COUNTS J, MUKHERJEE A, KRUH J, KELLY R. The confluence of heavy metal biooxidation and heavy metal resistance: Implications for bioleaching by extreme thermoacidophiles [J]. Minerals, 2015, 5(3): 397-451.
[15] GUMULYA Y, BOXALL N, KHALEQUE H, SANTALA V, CARLSON R P, KAKSONEN A H. In a quest for engineering acidophiles for biomining applications: Challenges and opportunities [J]. Genes, 2018, 9(2): 116-144.
[16] XIAO Yun-hua, XU Yong-Dong, DONG Wei-ling, LIANG Yi-li, FAN Fen-liang, ZHANG Xiao-xia, ZHANG Xian, NIU Jiao-jiao, MA Li-yuan, SHE Si-yuan. The complicated substrates enhance the microbial diversity and zinc leaching efficiency in sphalerite bioleaching system [J]. Applied Microbiology and Biotechnology, 2015, 99(23): 10311-10322.
[17] JOHNSON D B. Biodiversity and ecology of acidophilic microorganisms [J]. FEMS Microbiology Ecology, 1998, 27(4): 307-317.
[18] JOHNSON D B. Biodiversity and interactions of acidophiles: Key to understanding and optimizing microbial processing of ores and concentrates [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1367-1373.
[19] QIN Wen-qing, YANG Cong-ren, LAI Shao-shi, WANG Jun, LIU Kai, ZHANG Bo. Bioleaching of chalcopyrite by moderately thermophilic microorganisms [J]. Bioresource Technology, 2013, 129: 200-208.
[20] FENG Shou-shuai, YANG Hai-lin, WANG Wu. Microbial community succession mechanism coupling with adaptive evolution of adsorption performance in chalcopyrite bioleaching [J]. Bioresource Technology, 2015, 191: 37-44.
[21] LIANG Yu-ting, ZHU Shan, WANG Jun, AI Chen-bing, QIN Wen-qing. Adsorption and leaching of chalcopyrite by Sulfolobus metallicus YN24 cultured in the distinct energy sources [J]. International Journal of Minerals, Metallurgy, and Materials, 2015, 22(6): 549-552.
[22] VILCAEZ J, YAMADA R, INOUE C. Effect of pH reduction and ferric ion addition on the leaching of chalcopyrite at thermophilic temperatures [J]. Hydrometallurgy, 2009, 96(1-2): 62-71.
[23] FRANZMANN P D, HADDAD C M, HAWKES R B, ROBERTSON W J, PLUMB J J. Effects of temperature on the rates of iron and sulfur oxidation by selected bioleaching Bacteria and archaea: Application of the Ratkowsky equation [J]. Minerals Engineering, 2005, 18(13-14): 1304-1314.
[24] VILCAEZ J, SUTO K, INOUE C. Bioleaching of chalcopyrite with thermophiles: Temperature�CpH-ORP dependence [J]. International Journal of Mineral Processing, 2008, 88(1-2): 37-44.
[25] ZHU Wei, XIA Jin-lan, PENG An-an, NIE Zhen-yuan, QIU Guan-zhou. Characterization of apparent sulfur oxidation activity of thermophilic archaea in bioleaching of chalcopyrite [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(8): 2383-2388.
[26] ZENG Wei-min, QIU Guan-zhou, ZHOU Hong-bo, PENG Juan-hua, CHEN Miao, TAN Su Nee, CHAO Wei-liang, LIU Xue-duan, ZHANG Yan-sheng. Community structure and dynamics of the free and attached microorganisms during moderately thermophilic bioleaching of chalcopyrite concentrate [J]. Bioresource Technology, 2010, 101(18): 7068-7075.
[27] ZHU Wei, XIA Jin-lan, YANG Yi, NIE Zhen-yuan, PENG An-an, LIU Hong-chang, QIU Guan-zhou. Thermophilic archaeal community succession and function change associated with the leaching rate in bioleaching of chalcopyrite [J]. Bioresource Technology, 2013, 133: 405-413.
[28] SONG Jian, LIN Jian-qun, REN Yi-lin, LIN Jian-qiang. Competitive adsorption of binary mixture of Leptospirillum ferriphilum and Acidithiobacillus caldus onto pyrite [J]. Biotechnology and Bioprocess Engineering, 2010, 15(6): 923-930.
�����ã�����ΰ����������������
���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083
ժ Ҫ���о�������д����Եļ������ȹž�A. brierleyi��S. metallicus��������Ի�ͭ��������ͽ�����Ϊ�����������S. metallicus��ͭ�������Ը���A. brierleyi�ģ����л�Ͼ���ϵ��ͭ��������ߡ����������е�Ⱥ��ṹ����������S. metallicus�ھ�Ⱥ���������ƾ�Ⱥ��A. brierleyi��ռ���������������ƣ�����һ�����������ͭ���ڻ����ϵ�������ڵ�ͭŨ�ȱ��ڵ�����ϵ���������йء�Langmuir���������ó����ֹž�֮�䲻���ھ�����������ϵ����qPCR�����ʾ���ڻ�Ͻ����Ĺ����У�S. metallicus��A. brierleyi֮�������ٽ������������о���һ��������ϼ������Ⱦ��ڿ����������������������Ϊ��
�ؼ��ʣ��������ȹž���A. brierleyi��S. metallicus����ͭ������������
(Edited by Wei-ping CHEN)
Foundation item: Project (51774342) supported by the National Natural Science Foundation of China; Project (2016RS2016) supproted by the Hunan Provincial Science and Technology Leader (Innovation Team of Interface Chemistry of Efficient and Clean Utilization of Complex Mineral Resources), China
Corresponding author: Wen-qing QIN; Tel: +86-731-88830884; E-mail: qinwenqing369@126.com
DOI: 10.1016/S1003-6326(18)64900-3

