
Bioleaching of anilite by Acidithiobacillus ferrooxidans
CHENG Hai-na(�̺���)1, 2, HU Yue-hua(������)1, 2, GAO Jian(�� ��)1, MA Heng(�� ��)1
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Biometallurgy of Ministry of Education, School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 20 September 2008; accepted 5 November 2008
Abstract: In order to characterize the efficiency of copper bioleaching from anilite using pure cultures of Acidithiobacillus ferrooxidans in the absence and presence of ferrous sulphate, the experiments were carried out in shake flasks with or without 4 g/L ferrous sulphate (FeSO4��7H2O) at pH 2.0, 150 r/min and 35 ��. The tests show that Acidithiobacillus ferrooxidans is unable to attack anilite in iron-free 9K medium. Anilite is rapidly oxidized by bacterial leaching when ferrous sulphate is added. Chemical oxidation of anilite is slow compared with Acidithiobacillus ferrooxidans initiated solubilization in the presence of iron. The EDAX analysis of the surfaces of anilite confirms that sulfur coating layer is present as a reaction product on the surface of the bacterially leached mineral.
Key words: bioleaching; anilite; Acidithiobacillus ferrooxidans
1 Introduction
Naturally occurring copper is commonly found as sulfide minerals. The copper sulfur system is complex and there are a large number of phases in Cu2S�CCuS[1]. In the chalcocite group of Cu2-xS (0��x��0.6), eight compounds exist, that is ��-chalcocite Cu2S (low orthorhombic), ��-chalcocite Cu2S (high, hexagonal), djurleite Cu1.97S, digenite Cu1.80S, roxbyite Cu1.78S, anilite Cu1.75S, geerite Cu1.60S and spionkopite Cu1.40S[2]. Anilite, found by MORIMOTO et al in 1969[3], has an extremely narrow range of composition near Cu7S4. It crystallizes in Pnma, and a, b, c equal 0.789, 0.784 and 1.101 nm, respectively. Anilite can be easily changed to the metastable digenite-type phase by grinding. Anilite is associated with djurleite. The twinning of anilite is governed by the pseudocubic subcell of sulfur close packing. Anilite was found in the Zijinshan Copper Mine, Fujian Province of China by XUE et al[4]. Its matrix phase can be expressed as Cu1.756S in chemical formula, which is consistent with anilite discovered in Japan in 1969. According to the XRD of the matrix and lamellae, the cell dimensions are modified as a=0.787 nm, b=0.785 nm, c=1.108 nm[4].
Bioleaching is an economical and widely used
method for extraction of metals compared with other available technologies[5]. This process has been exploited in the extraction of gold, nickel, copper and cobalt from sulfide ores[6]. The classical leaching bacteria now belong to the genus Acidithiobacillus. Acidithiobacillus ferrooxidans (A. ferrooxidans) plays an important role in bacterial leaching. It is an obligate chemolithoautotrophic bacterium that uses elemental sulfur and/or reduced sulfur compounds as well as ferrous iron as a source of energy and carbon dioxide as a source of carbon[7-9].
The secondary copper sulphides have been successfully used in many bioleaching operations, and chalcocite is the main copper secondary sulphide mineral[10]. Little research on bioleaching of a research-grade chalcocite has been reported in the public domain. The aim of this work was to determine the efficiency of copper bioleaching from anilite using pure cultures of A. ferrooxidans in the absence and presence of ferrous sulphate.
2 Materials and methods
2.1 Bacteria
A strain of A. ferrooxidans used in this study was isolated from an acid mine water at Gaofeng Copper Mine, Guangxi Province of China. Bacteria were grown in 9K medium[11] and harvested from the late logarithmic phase of growth. Iron(��) precipitates were removed by filtration through blue ribbon filter paper. Cells were centrifuged (10 000 r/min), washed several times and suspended in iron-free 9K medium at pH 2.0. The bacterial population in the inocula was about 1��107 cells/mL.
2.2 Substrate
An anilite concentrate was obtained from Zijinshan Copper Mine, Fujian Province of China. It was characterized by X-ray diffraction, which proved the major presence of anilite (75.7%) and small amount of quartz (9.1%), chalcocite (12.0%) and yarrowite (3.2%). The chemical compositions of the sample were 68.84% Cu, 19.07% S, 5.11% Si, 6.54% O and 0.02% Ca. The mineral was ground to pass a 325-mesh size and samples were used for bacterial leaching in shake flasks.
2.3 Leaching experiments
Leaching experiments were carried out in 150 mL cultures in 250 mL shake flasks containing anilite (0.2% w/v) in iron-free 9K medium at an initial pH of 2.0. The flasks were sterilized by autoclaving (30 min, 120 ��). The cultures in shake flasks were incubated with 150 r/min at 35 ��.
Bioleaching of anilite was carried out using a pure culture of A.ferrooxidans in the absence and presence of 4 g/L Fe2+ (FeSO4��7H2O). The bacterial population in the inocula was about 1��107 cells/mL. All experiments were conducted at least in duplicate.
2.4 Analytical techniques
Copper concentrations in solution were measured by atomic absorption spectrophotometry (Varian SpectrAA-300). The ferrous iron was analyzed by a volumetric method by titration with potassium dichromate. The pH of the supernatant at ambient temperature was also measured with a pH meter. Free bacteria in solution were counted in a Petroff-Hausser chamber under an optical microscope. Solid residues were filtered, washed, dried and finally analyzed by X-ray diffractometry. Samples of these solid residues were examined under a scanning electron microscope with an energy dispersive X-ray(EDAX) probe.
3 Results and discussion
3.1 Leaching of anilite without ferrous sulphate
Changes in copper(��) concentration, pH and bacterial population with time during bioleaching of anilite in iron-free 9K medium are shown in Fig.1. The concentrations of copper released in the bioleaching are nearly the same as those in the sterile control. A. ferrooxidans seems to have no ability to initiate anilite solubilization. The same results are obtained using three different strains of A. ferrooxidans to bioleach anilite. The pH values of inoculated and sterile systems increase from 2 to 3.5 during bioleaching. This suggests that anilite dissolution is caused by proton attack (Reaction (1)). The A. ferrooxidans population decreases initially due to the bacterial attachment to anilite. Bacteria population is very sparse throughout bioleaching and does not exhibit a logarithmic phase. The results conclude that A. ferrooxidans can not use anilite as substrate to grow in iron-free 9K medium.
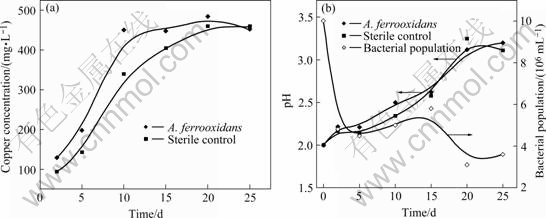
Fig.1 Changes in copper(��) concentration (a), pH and bacterial population (b) during bioleaching of anilite in iron-free 9K medium
In inoculated and sterile systems, X-ray diffraction patterns of the solid residues show the presence of copper sulphide (CuS) in addition to anilite (Cu7S4) and yarrowite (Cu9S8) (Fig.2). It is clearly found that the copper sulphide (CuS) is produced by proton attack on anilite (Reaction (1)). ELSHERIEF et al[12] reported that the anodic leaching of chalcocite (Cu2S) in H2SO4 electrolyte solution led to the formation of copper- deficient compounds described as Cu2-xS, and finally to copper sulphide (CuS).

Fig.2 XRD patterns of untreated anilite (a), solids residues after 15 d of contact time in sterile control with 4 g/L ferrous sulphate (b) and in A. ferrooxidans cultures incubated supplemented with (c) or without (d) 4 g/L ferrous sulphate
2Cu7S4+3O2+6H2SO4��8CuS+6CuSO4+6H2O (1)
3.2 Leaching of anilite with ferrous sulphate
In the presence of iron, the copper dissolution is presented in Fig.3. After 15 d bioleaching, the dissolved copper reaches about 1 388 mg/L in the presence of ferrous sulphate, which is much higher than that in the absence of ferrous sulphate (about 447 mg/L). This suggests that the effect of supplemental iron is important in the A. ferrooxidans bioleaching of anilite. The presence of iron also slightly enhances copper dissolution in the sterile control because the ferrous iron is partially oxidized under the action of air. It was also proved by other researchers using synthetic copper sulphide[13].
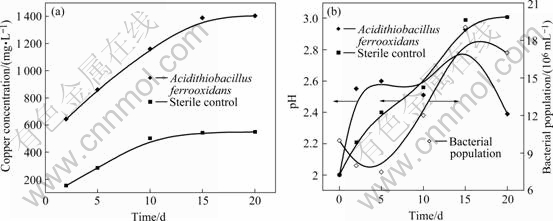
Fig.3 Recovery of copper (a), pH and bacteria population change (b) during bioleaching of anilite in presence of ferrous sulphate
It is determined that the A. ferrooxidans culture can completely oxidize ferrous iron in 20 h and only about 4.4% ferrous iron is oxidized by air in the sterile control.
Bacterial population in solution increases as the copper is solubilized. A. ferrooxidans population initially grows as a consequence of ferrous iron oxidation. When ferrous iron is consumed, growth of A. ferrooxidans is further produced by copper sulphide (CuS). After 15 d, the free bacterial population starts to decrease due to the attachment to precipitate[13].
The pH of leaching liquor is higher in A. ferrooxidans than that in the sterile control as a result of ferrous iron oxidation at first 5 d. After 15 d, pH decreases progressively as a result of iron hydrolysis and the consequent precipitation of ferric compounds. The gradual increase of pH value for the sterile control is caused by the progressive ferrous iron oxidation and the acid dissolution of anilite. This is in agreement with previous results[13-14].
Solid residues of the sterile control and A. ferrooxidans leaching systems after 15 d were examined by the XRD and the results are given in Fig.2. Jarosite is confirmed as the main phase of solid residues in A. ferrooxidans bioleaching, in which iron is oxidized and precipitated as jarosite because of the enhancing effects of the increased pH and iron concentration[15]. Sulfur is also detected beside copper sulphide (CuS) in the solid residues when ferrous iron is added. Sample of the sterile control shows that the main phase is copper sulphide (CuS). Oxidation of anilite by ferric iron or proton attack produces copper sulphide. Jarosite is found as new phases towards the end of the experiment.
3.3 SEM and EDAX results of leached residues
Figs.4(a) and (b) show the SEM photographs of bacterially and chemically leached anilite in the presence of 4 g/L ferrous sulphate after 15 d, respectively. Figs.4(a) and (b) indicate that the mineral surface of the chemically leached sample is clear, while that of the bacterially leached sample is covered with a porous layer of sulfur. The EDAX analyses of the surfaces are shown in Figs.4(c) and (d). The EDAX analyses confirm that the surface of chemically leached sample is fresh copper sulfide. The quantity of sulfur is approximately equal to the amount of copper in the chemically leached residue. The EDAX analyses of the surfaces in Fig.4(b) confirm that the sulfur coating layer is present as a reaction product on the surface of the bacterially leached anilite. This suggests that anilite is oxidized and forms a sulfur layer on the mineral (Reactions (2) and (3)). Bacteria can oxidize elemental sulfur to sulphate. However, the oxidation rate appears to be slower than the rate of elemental sulfur formation because the elemental sulfur is often found in solid residues from bacterial leaching systems [15]. The rate of chemical leaching of anilite is partially limited by the diffusion of ferric iron through the porous sulfur layer[13]. Sulfur layer can be removed from the mineral surface by sulfur-oxidizing bacteria according to the previous observation using synthetic copper sulphide[13] and sphalerite[6].

Fig.4 SEM photographs of bacterially and chemically leached anilite with 4 g/L ferrous sulphate cultured for 15 d: (a) Surface of chemically leached anilite; (b) Surface of bacterially leached anilite; (c) EDAX spectrum of surface in Fig.4(a); (d) EDAX spectrum of surface in Fig.4(b)
The low magnification SEM images of bacterially and chemically leached mineral show that there is much precipitate. The EDAX analysis of the precipitate surfaces shows that the quantity of the element iron is 47% of the total mass fraction. According to the X-ray diffraction patterns the precipitate is jarosite. More jarosite precipitations occur in the presence of A. ferrooxidans perhaps because of the rapid oxidation of iron. The more jarosite precipitates may limit the rate of the copper solubilization by hindering the diffusion of the soluble reactants or products through this layer[14].
The results of anilite oxidation suggest that the leaching of anilite is performed in two stages: formation of copper sulphide (CuS) (Reactions (1) and (2)); and the oxidation of copper sulphide (CuS) (Reaction (4)). The stage of copper sulphide (CuS) formation is easy to produce. Proton and a low concentration of ferric iron can oxidize anilite to copper sulphide. According to these results, the ferrous iron is oxidized by A. ferrooxidans to generate ferric iron (Reaction (2)). Anilite is oxidized by ferric iron or proton leading to copper sulphide. Copper sulphide is oxidized by ferric iron forming a porous sulphur layer on the mineral. Jarosite precipitation is produced during leaching, which can hinder the further dissolution of copper.
Cu7S4+3Fe2(SO4)3��4CuS+3CuSO4+6FeSO4 (2)
CuS+Fe2(SO4)3��CuSO4+2FeSO4+S0 (3)
4FeSO4+O2+2H2SO4��2Fe2(SO4)3+2H2O (4)
4 Conclusions
1) A. ferrooxidans in pure culture without iron is unable to leach anilite significantly, but the presence of ferrous iron considerably enhances the copper dissolution. About 93% copper is obtained in A. ferrooxidans culture after15 d.
2) Copper sulphide is found as a new phase of the solid residues by XRD. Additionally, anilite is oxidized by ferric iron, forming a porous sulfur layer on the surface of mineral detected by SEM and EDAX analyses.
References
[1] MOSSELMANS J F W, CHARNOCK J M, CHARNOCK J M, GARNER C D, PATTRICK R A D, VAUGHAN D J. A XAS study of the structural changes undergone by amorphous copper sulfides when precipitated from solution [J]. Physica B, 1995, 208/209: 609-610.
[2] BAL?? P, TAKACSB L, BOLDI??ROV? E, GODO??KOV? E. Mechanochemical transformations and reactivity in copper sulphides [J]. Journal of Physics and Chemistry of Solids, 2003, 64: 1413-1417.
[3] MORIMOTO N, KOTO K, SHIMAZAKI Y. Anilite, Cu7S4, a new mineral [J]. Am Miner, 1969, 54: 1256-1268.
[4] XUE J, SUN T, CAI Y, CHEN W. Discovery of anilite in China and its microstructure [J]. Acta Mineralogica Sinice, 2000, 20(1): 9-12. (in Chinese)
[5] JOHNSON D B, OKIBE N, WAKEMAN K, LIU Y J. Effect of temperature on the bioleaching of chalcopyrite concentrates containing different concentrations of silver [J]. Hydrometallurgy, 2008,94(1/4):42-47.
[6] FOWLER T A, CRUNDWELL F K. Leaching of zinc sulfide by Thiobacillus ferrooxidans: Bacterial oxidation of the sulfur product layer increases the rate of zinc sulfide dissolution at high concentrations of ferrous ions [J]. Applied and Environmental Microbiology, 1999, 65(2): 5285-5292.
[7] CABRERA G, G?MEZ J M, CANTERO D. Kinetic study of ferrous sulphate oxidation of Acidithiobacillus ferrooxidans in the presence of heavy metal ions [J]. Enzyme and Microbial Technology, 2005, 36: 301-306.
[8] PRADHAN N, PRADHAN S K, NAYAK B B, MUKHERJEE P S, SUKLA L B, MISHRA B K. Micro-Raman analysis and AFM imaging of Acidithiobacillus ferrooxidans biofilm grown on uranium ore [J]. Research in Microbiology, 2008, 159: 557-561.
[9] MAHMOUD K K, LEDUC L G, FERRONI G D. Detection of Acidithiobacillus ferrooxidans in acid mine drainage environments using fluorescent in situ hybridization (FISH) [J]. Journal of Microbiological Methods, 2005, 61: 33-45.
[10] WATLING H R. The bioleaching of sulphide minerals with emphasis on copper sulphides �� A review [J]. Hydrometallurgy, 2006, 84: 81-108.
[11] SILVERMAN M P, LUNDGREN D G. Studies on the chemoautotrophic iron bacterium Thiobacillus ferrooxidans (I): An improved medium and a harvesting procedure for securing high cellular yields [J]. Journal of Bacteriology, 1959, 77: 642-647.
[12] ELSHERIEF A E, SABA A E, AFIFI S E. Anodic leaching of chalcocite with periodic cathodic reduction [J]. Minerals Engineering, 1995, 8(9): 967-978.
[13] FALCO L, POGLIANI C, CURUTCHET G, DONATI E. A comparison of bioleaching of covellite using pure cultures of Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans or a mixed culture of Leptospirillum ferrooxidans and Acidithiobacillus thiooxidans [J]. Hydrometallurgy, 2003, 71: 31-36.
[14] QIU M Q, XIONG S Y, ZHANG W M, WANG G X. A comparison of bioleaching of chalcopyrite using pure culture or a mixed culture [J]. Minerals Engineering, 2005, 18: 987-990.
[15] BEVILAQUA D, LEITE A L L C, Jr GARCIA O, TUOVINEN O H. Oxidation of chalcopyrite by Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans in shake flasks [J]. Process Biochemistry, 2002, 38: 587-592.
Foundation item: Project(2004CB619204) supported by the National Basic Research Program of China; Project(50621063) supported by Chinese Science Foundation for Distinguished Group
Corresponding authors: HU Yue-hua; Tel: +86-731-8879815, E-mail: hyh@mail.csu.edu.cn
(Edited by LI Xiang-qun)