文章编号:1004-0609(2014)04-1076-08
巯基化改性麦糟对Zn(Ⅱ)的吸附特性
李青竹1, 2, 3,覃文庆2,柴立元1, 3,王庆伟1, 2, 3
(1. 中南大学 冶金与环境学院,长沙 410083;
2. 中南大学 资源加工与生物工程学院,长沙 410083;
3. 中南大学 国家重金属污染防治工程技术研究中心,长沙 410083)
摘 要:采用巯基化改性麦糟去除废水中的锌离子,研究溶液pH值、反应时间和温度以及Zn(Ⅱ)初始溶度对巯基化改性麦糟吸附效果的影响;借助吸附平衡等温线及吸附动力学模型拟合,结合傅立叶变换红外光谱(FTIR)分析阐明吸附机制。结果表明:在较宽的pH值范围(6~9)内,巯基化改性麦糟表现出对Zn(Ⅱ)良好的吸附性能。由Langmuir吸附等温线方程计算得到该吸附剂对Zn(Ⅱ)的理论饱和吸附量为353.36 mg/g,高于改性木质纤维素类吸附剂的吸附量(17.88~156 mg/g)。巯基化改性麦糟对Zn(Ⅱ)的吸附动力学特性表明吸附反应很快在30 min 内达到平衡,吸附符合拟二级动力学方程,活化能的计算结果表明吸附为活性化学吸附。FTIR分析可知:由巯基化改性麦糟吸附Zn(Ⅱ)主要是羟基和巯基中S―H基团的硫原子与Zn(Ⅱ)配合的结果。
关键词:巯基化改性麦糟; 吸附; Zn(Ⅱ); 动力学; 吸附特性
中图分类号:O647.3
Adsorption characteristics of Zn(Ⅱ) on thiol-functionalized spent grain
LI Qing-zhu1, 2, 3, QIN Wen-qing2, CHAI Li-yuan1, 3, WANG Qing-wei1, 2, 3
(1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
3. Chinese National Engineering Research Center for Control and Treatment of Heavy Metal Pollution,
Central South University, Changsha 410083, China)
Abstract: The thiol-functionalized spent grain (TSG) was used to remove Zn(Ⅱ) from wastewater. The effects of solution pH, reaction time and temperature and initial Zn(Ⅱ) concentration on the adsorption were investigated. The adsorption mechanism was clarified by the adsorption equilibrium isotherm and adsorption kinetics model fitting combined with Fourier transform infrared spectroscopy (FTIR) analysis. The results show that the thiol-functionalized spent grain exhibits good adsorption performance over the wide pH range of 6-9. The maximum theoretical Zn(Ⅱ) adsorption capacity of TSG calculated from Langmuir isotherm is 353.36 mg/g, which is higher than those of other modified lingocellulosic adsorbents reported (17.88-156 mg/g). In addition, the results of Zn(Ⅱ) sorption kinetics by TSG indicate that the sorption process is fast and the equilibrium is established in 30 min. The sorption kinetics is well described by the pseudo-second order kinetic model. The calculated activation energy (Ea) implies that the adsorption of Zn(Ⅱ) on TSG is a chemical adsorption. FTIR analysis demonstrates that the hydroxyl groups and sulphur atoms of thiol groups (S―H) of TSG complexing with the zinc ion leads to the adsorption.
Key words: thiol-functionalized spent grain; adsorption; Zn(Ⅱ); kinetics; adsorption characteristic
锌是人体必需的微量元素,但其含量甚微。摄入过量的锌,会对人体造成伤害,引起中毒。锌中毒的主要症状有脱水、电解质失衡、腹痛、恶心、头晕等[1]。锌资源广泛应用于冶炼、采矿、化工、制药及食品行业中,与此同时,这些行业也会产生大量的含锌废水,造成严重的环境污染和资源浪费,威胁人体健康。含锌废水的处理方法主要包括化学沉淀法、离子交换法、膜分离法、电解法和吸附法等[2],其中吸附法处理含锌废水因其操作简单、成本低、不产生二次污染等优点成为研究的热点[3]。近年来,以廉价易得的木质纤维素类物质作为主要原料,通过化学改性制备新型、高效的锌离子吸附剂备受瞩目[4-5]。
我国是啤酒生产大国,啤酒生产中80%以上的副产物为啤酒糟,又称麦糟。麦糟是典型的木质纤维素类物质,其表面富含羟基,可以发生酸化、酯化、醚化等一系列的化学反应,从而引入特殊功能基团,实现功能化。木质纤维素类物质的巯基化改性可以提高其对重金属离子的吸附能力[6-7]。但有关巯基化改性麦糟吸附锌离子的研究鲜见报道。
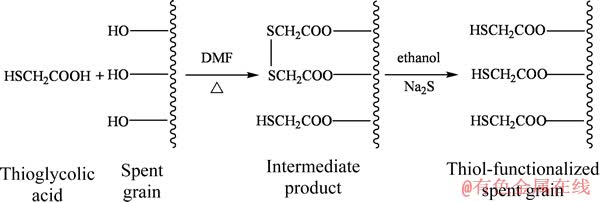
西末雄法[8]是木质纤维素类物质巯基化改性的传统方法,该方法创始于1971年,并一直沿用至今,没有显著改善。西末雄改性法存在反应时间长、巯基易被氧化的缺点。为此,本文作者开发了快速、保护性巯基化改性麦糟新方法[9],优选硫酸氢钠(NaHSO4・H2O)为催化剂、N, N-二甲基甲酰胺(DMF)为反应介质,巯基乙酸与麦糟反应3 h后,加入Na2S・9H2O用于还原被氧化的巯基,即制备了高效的重金属吸附剂,可用于吸附Zn(Ⅱ)。本文作者对巯基化改性麦糟吸附Zn(Ⅱ)的吸附特性、吸附动力学以及吸附机制进行系统研究,对处理含锌废水具有重要的指导意义。
1 实验
1.1 原料与试剂
原料――麦糟取自长沙英博白沙啤酒厂,用超纯水洗去表面附着的泥沙等杂质,于60 ℃烘干后研碎,过300 μm筛,备用。本研究中改性前的麦糟用RSG(Raw spent grain)表示。试剂:硝酸锌(Zn(NO3)2・6H2O)、巯基乙酸、硫酸氢钠(NaHSO4・H2O)、硫化钠(Na2S・9H2O)、N, N-二甲基甲酰胺(DMF)、无水乙醇均为分析纯。
1.2 巯基化改性麦糟的制备与表征
分别取5 g麦糟与0.125 g 催化剂(NaHSO4・H2O)混合,依次加入12.5 mL 反应介质(DMF)和25 mL巯基乙酸,于120 ℃左右反应3 h。反应结束后,冷却至室温。加入30 g Na2S・9H2O在125 mL无水乙醇介质中磁力搅拌反应1 h后减压过滤,用去离子水充分洗涤滤渣,于真空干燥箱中60 ℃烘干,研碎过300 μm筛备用。本研究中巯基化改性后麦糟用TSG (Thiol-functionalized spent grain)表示。
巯基化改性前后吸附剂表面采用英国KRATOS公司的XSAM800光电子能谱仪(XPS)进行元素分析,以Cls(284.6 eV)为电子结合能(B.E.)内标进行修正。改性前后以及吸附前后的样品采用KBr压片法使用美国NICOLET公司IS10型傅立叶变换红外光谱仪测定其红外光谱,波长范围在4000~400 cm-1之间。
1.3 吸附实验方法
1.3.1 pH值的影响
分别取50 mL初始浓度为0.2 mmol/L的Zn(Ⅱ)溶液,加入0.1 g 吸附剂,用0.1 mol/L HNO3或0.1 mol/L NaOH调节溶液的pH值在2~10之间,置于HH-6型恒温水浴磁力搅拌器(江苏常州澳华仪器有限公司)中在25 ℃、转速200 r/min的条件下搅拌吸附30 min。过滤取上清液,滤液用WFX-120型原子吸收分光光度计(北京瑞利分析仪器总厂)检测分析Zn(Ⅱ)浓度,并计算相应的去除率和吸附量。RSG按照上述同样实验条件进行实验与TSG进行对照。
锌离子的去除率R(%)和吸附剂的吸附量q(mg・g-1)分别按式(1)和(2)计算:
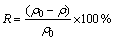 (1)
(1)
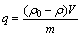 (2)
(2)
式中: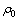 为吸附前溶液中锌离子的质量浓度,mg/L;
为吸附前溶液中锌离子的质量浓度,mg/L;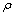 为吸附后溶液中锌离子的质量浓度,mg/L;V为反应溶液体积,L;m为吸附剂干的质量,g。
为吸附后溶液中锌离子的质量浓度,mg/L;V为反应溶液体积,L;m为吸附剂干的质量,g。
1.3.2 吸附动力学实验
向一系列50 mL浓度为20 mg/L的Zn(Ⅱ)溶液中投加0.1 g TSG,在转速200 r/min,温度分别为5、25和45 ℃的恒温水浴磁力搅拌器中吸附一定时间,在不同时间点取样,过滤取上清液,用原子吸收分光光度计测定其中Zn(Ⅱ)的浓度,并计算相应的吸附量用于动力学方程的拟合。
1.3.3 吸附平衡等温线的测定
向一系列50 mL浓度范围在0.2~20 mmol/L的Zn(Ⅱ)溶液中投加0.1 g TSG,在转速200 r/min,温度为25 ℃的恒温水浴磁力搅拌器中反应30 min,过滤取上清液,用原子吸收分光光度计测定其中Zn(Ⅱ)浓度,并计算相应的吸附量,用于吸附平衡等温线的拟合。
2 结果与讨论
2.1 巯基化改性麦糟的制备原理及表征
传统的西末雄法巯基化改性纤维素类物质是将一定量巯基乙酸、乙酸、乙酸酐和少量催化剂浓硫酸混合加入至待改性的纤维素类材料中,置于38 ℃的水浴锅中反应,每隔24 h翻动一次,100 h后取出,用蒸馏水洗至中性,抽滤、烘干[10]。该方法存在反应时间长、反应试剂多、浓硫酸作为催化剂腐蚀性强等缺点。因此,拟研发快速、高效、经济的巯基化改性新方法。
本研究选用DMF作为反应介质,使麦糟的活化前处理过程和有机反应过程同时发生,节省了改性时间。利用巯基乙酸含有的羧基与麦糟表面的羟基发生酯化反应,从而引入巯基。与用浓硫酸作酯化反应的催化剂不同,本研究选用温和、高效的硫酸氢钠作为酯化反应的催化剂,大大降低了反应的活化能,提高了反应速率。在麦糟巯基化制备的过程中,关键是解决巯基不稳定容易被氧化为二硫化物的问题,本研究采用Na2S最终还原的方法确保制备的重金属离子吸附剂含有足够巯基,并且易于保存。麦糟的巯基化改性反应如式(3)所示。可以看出,通过选择合适的反应介质,控制一定的反应温度,在催化剂作用下,巯基乙酸含有的羧基能够和麦糟中羟基上的氢脱水生成酯,并采用Na2S进行还原保护,成功制备了巯基化改性麦糟。
 (3)
(3)
X射线光电子能谱(XPS)可以为成功巯基化改性提供最直观的证据。图1所示为麦糟巯基化改性前后的XPS谱。由图1可以明显地看出,麦糟经巯基化改性后在163.6 eV处出现了S 2p的新峰,这和文献[11]报道的巯基中的S 2p所处位置一致。
2.2 pH值对吸附效果的影响
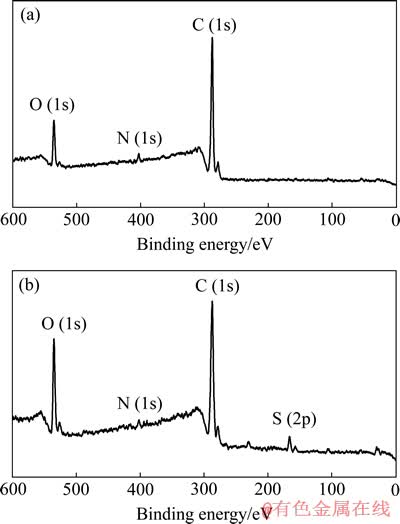
图1 麦糟巯基化改性前后的X射线光电子能谱
Fig. 1 Typical wide-scan XPS spectra of RSG(a) and TSG(b)

图2 pH值对巯基化改性前后麦糟吸附Zn(Ⅱ)的影响
Fig. 2 Effect of pH on adsorption of Zn(Ⅱ) by RSG and TSG
pH值对麦糟巯基化改性前后吸附Zn(Ⅱ)的影响如图2所示。结果表明:改性前的麦糟吸附Zn(Ⅱ)在pH值为8时,去除率达到最大值,pH值低于或高于此值时,去除效果均不佳;巯基化改性麦糟吸附Zn(Ⅱ)的最佳pH值范围较宽,pH值在6~9之间,Zn(Ⅱ)去除率均较高。当pH值小于上述最佳pH值范围时,Zn(Ⅱ)去除率随pH值增加而增大;当pH值大于9时,Zn(Ⅱ)去除率略有降低。这是因为pH值较低时,H3O+与锌离子竞争吸附位点[12],另外,麦糟表面被质子化带正电荷,锌离子表面也带正电荷,两者之间的静电斥力阻碍吸附的顺利进行。锌离子在水溶液中的存在形态对其吸附效果也有一定影响,利用Visual MINTEQ, ver. 2.53 软件[13-14]计算pH值对0.2 mmol/L锌离子在水溶液中形态的影响,如图3所示。由图3可以看出,当pH<6时,Zn主要以Zn(Ⅱ)形式存在;当6<pH<9时,Zn由游离态向ZnOH+和Zn(OH)2(aq)的羟合配离子形态转化;当pH>9时,Zn(OH)2(aq)开始返溶,出现Zn(OH)3-的形态,造成吸附效果略有下降。
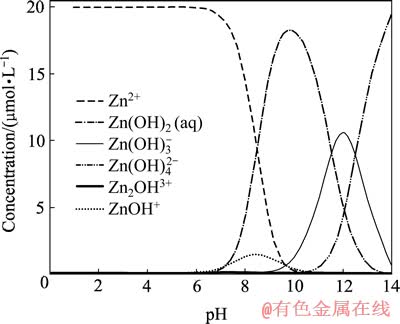
图3 pH值对锌离子在水溶液中形态的影响
Fig. 3 Aqueous speciation of Zn as a function of pH
整个吸附过程中,巯基化改性麦糟对Zn(Ⅱ)的吸附效果均优于未改性麦糟,更重要的是,巯基化改性后吸附Zn(Ⅱ)的最佳pH范围更宽。最佳pH值范围较宽,保证了巯基化改性麦糟处理不同种类不同性质含锌废水的能力。
2.3 吸附动力学
吸附时间对巯基化改性麦糟吸附Zn(Ⅱ)的影响如图4所示。由图4可看出,随着反应时间的延长,Zn(Ⅱ) 的去除率逐渐增大;巯基化改性麦糟吸附Zn(Ⅱ)的速率很快,前5 min内去除率达94%以上,随后缓慢增加至30 min吸附达到平衡。此现象说明改性麦糟表面富含的功能基团能够与Zn(Ⅱ)快速配合,这点通过后面的红外光谱分析也可以得到证实。而Zn(Ⅱ)的去除率随反应温度的升高而增大,表明该吸附反应为吸热反应。

图4 不同温度下吸附时间对巯基化改性麦糟吸附Zn(Ⅱ)的影响
Fig. 4 Effect of absorption time on Zn(Ⅱ) adsorbed by TSG at various temperatures
为全面研究巯基化改性麦糟吸附Zn(Ⅱ)的动力学特性,分别采用拟一级动力学模型(式(4))、拟二级动力学模型(式(5))和内扩散模型(式(6))进行拟合[15](见图5):
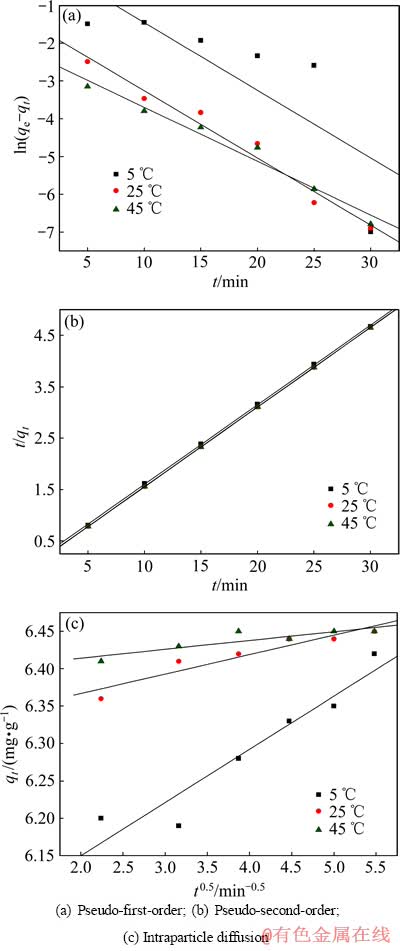
图5 不同温度下巯基化改性麦糟吸附Zn(Ⅱ)的动力学
Fig. 5 Kinetic plots of Zn(Ⅱ) adsorption on TSG at various temperatures
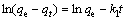 (4)
(4)
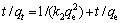 (5)
(5)
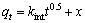 (6)
(6)
式中:qt为t时刻吸附量(mg/g);qe为平衡时吸附量(mg/g);k1(min-1),k2(g・mg-1・min-1)和kint(mg・g-1・min-0.5)为动力学参数。
拟合得到的动力学参数列入表1~3。由拟一级动力学模型拟合得到的理论饱和吸附量与实验值偏差很大。因此,巯基化改性麦糟吸附Zn(Ⅱ)不符合拟一级动力学方程。而采用拟二级动力学方程拟合得到的相关系数均很高,接近1。通过拟二级动力学方程计算得到的理论饱和吸附量与实验值能够很好的吻合。因此,巯基化改性麦糟吸附Zn(Ⅱ)符合拟二级动力学方程,k2为吸附速率常数。吸附速率常数随温度升高而逐渐增大,表明升高温度有利于吸附反应的进行,吸附反应为吸热反应。此外,内扩散模型拟合呈线性关系但不过原点,说明整个吸附过程中存在内扩散作用但内扩散并非唯一的速率控制步骤[16]。总体来看,拟二级动力学模型拟合的线性相关系数最高,因拟二级动力学模型以化学吸附为主,同时包含了吸附的所有过程:外部液膜扩散、表面吸附和颗粒内扩散等[15],所以拟二级动力学模型更真实全面地反映巯基化改性麦糟吸附Zn(Ⅱ)的机制。
表1 不同温度下改性麦糟吸附Zn(Ⅱ)的拟一级动力学参数
Table 1 Pseudo-first-order kinetic parameters for adsorption of Zn(Ⅱ) on TSG at different temperatures

表2 不同温度下改性麦糟吸附Zn(Ⅱ)的拟二级动力学参数
Table 2 Pseudo-second-order kinetic parameters for adsorption of Zn(Ⅱ) on TSG at different temperatures
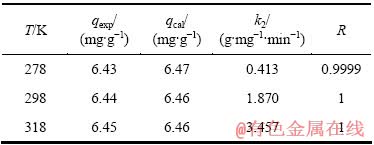
表3 不同温度下改性麦糟吸附Zn(Ⅱ)的内扩散模型参数
Table 3 Intraparticle diffusion parameters for adsorption of Zn(Ⅱ) on TSG at different temperatures

由以上动力学研究可知,k2为吸附速率常数即kads,由Arrhenius方程
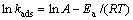 (7)
(7)
计算得到吸附反应的活化能Ea。通常化学吸附力比物理吸附力要强,常见的两种化学吸附类型为活性和非活性化学吸附。活性化学吸附限定活化能在8.4到83.7 kJ/mol之间,非活性化学吸附的活化能接近0[17]。本研究中,巯基化改性麦糟吸附Zn(Ⅱ)的活化能为39.35 kJ/mol,说明巯基化改性麦糟吸附Zn(Ⅱ)是活性化学吸附,这与上述研究结果一致。
2.4 吸附等温线
25 ℃时,巯基化改性麦糟对Zn(Ⅱ)的吸附能力随平衡浓度的增加而增大,当初始浓度为20 mmol/L时,吸附基本达到饱和。平衡吸附量(qe)对锌离子的平衡浓度(ce)的关系采用Langmuir(式(8))和Freundlich(式(9))等温线方程进行拟合,其结果如图6所示。
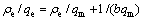 (8)
(8)
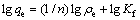 (9)
(9)
式中:b为Langmuir方程表示吸附剂表面覆盖度的经验常数(L/mg),1/b越大,吸附剂对吸附质的吸附量越大;Kf(mg/g)和n分别为Freundlich方程与吸附量和吸附强度有关的经验常数。
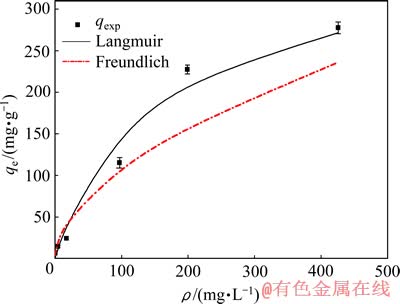
图6 巯基化改性麦糟对Zn(Ⅱ)的吸附等温线
Fig. 6 Adsorption isotherms of Zn(Ⅱ) using TSG as adsorbent
表4所列为25 ℃时Langmuir和Freundlich方程拟合的特征参数值和线性相关系数。Langmuir模型倾向于描述均相表面的单分子层吸附[18],而Freundlich模型适用于非均相表面的复杂吸附。由表2可知,Freundlich吸附等温线的线性相关系数较高,因此,Freundlich方程较Langmuir方程更好地描述巯基化改性麦糟吸附Zn(Ⅱ)的过程,说明巯基化改性麦糟吸附Zn(Ⅱ)的机理并不倾向于单分子层吸附,而是涉及更为复杂的机理。
由Langmuir吸附等温线方程计算得到巯基化改性麦糟对Zn(Ⅱ)的饱和吸附量为353.36 mg/g,远高于文献报道的其它改性木质纤维素类吸附剂对Zn(Ⅱ)的吸附能力(如表5所列),表明巯基化改性麦糟是一种优越的Zn(Ⅱ)吸附剂。
表4 巯基化改性麦糟吸附锌离子的吸附等温线参数
Table 4 Isotherm parameters for adsorption of Zn(Ⅱ) on TSG at 25 ℃

表5 改性木质纤维素类吸附剂对锌离子的吸附能力
Table 5 Zn(Ⅱ) adsorption capacity of modified ligno- cellulosic adsorbents

2.5 吸附机理
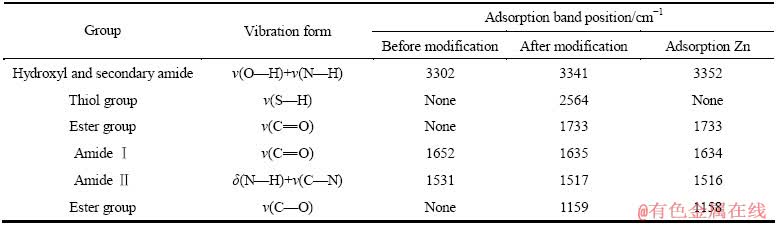
麦糟巯基化改性前后及其吸附Zn(Ⅱ)的红外光谱如图7所示。对比谱图变化较明显的谱带列于表6。对于特征谱带归属如下:3302~3352 cm-1处较宽的吸收带为碳水化合物中结合水的羟基O―H的伸展振动与仲酰胺(RNHCOCH3)中N―H的伸缩振动[27]。1634~1652cm-1处的谱峰来自仲酰胺Ⅰ带,是C―O的伸缩振动引起的;1516~1531 cm-1处的谱峰来自仲酰胺Ⅱ带,是N―H的弯曲振动和C―N的伸缩振动的叠加引起的,这两个峰是蛋白质的特征谱带[28]。
巯基化改性后麦糟在1733和1159 cm-1处出现了两个新的吸收峰,前者是酯基C=O键的特征峰,后者是酯基C―O键的特征峰[29],这说明改性过程中发生了酯化反应。通过巯基乙酸和麦糟上羟基的酯化反应,形成了新的酯键并引入了巯基功能基团,这表现在红外谱图中2564 cm-1处出现的新吸收峰――巯基的伸缩振动峰[30],同时印证了XPS的结果。而且,麦糟巯基化改性后位于3302 cm-1处的宽峰紫移至3341 cm-1处,这说明麦糟上含有的羟基参与了巯基化反应。
从表6巯基化改性麦糟吸附Zn(Ⅱ)前后红外光谱主要特征峰的变化可以看出,羟基O―H吸收峰的位置偏移最大,吸附前羟基吸收峰在3341 cm-1处,吸附Zn(Ⅱ)后羟基吸收峰出现在3352 cm-1处;另外一处非常明显的变化是巯基2564 cm-1处的吸收峰在吸附Zn(Ⅱ)后消失。羟基吸收峰的位置发生偏移是因为羟基本身具有配合重金属离子的能力,由于羟基配合锌离子导致羟基的吸收谱带发生了较大的位移。巯基化改性麦糟在2564 cm-1处的S―H谱带在吸附Zn(Ⅱ)后消失证明了麦糟经巯基化改性处理后生成的巯基在吸附锌离子的过程中发挥了重要作用,主要是巯基S―H键中的硫原子与锌离子配合达到吸附锌离子的目的。其他功能基团如N―H、C―N、C―O基团在吸附锌离子前后红外光谱图中峰位移变化并不明显,说明这些基团对吸附锌离子发挥的作用不大。

图7 麦糟巯基化改性前后及其吸附Zn(Ⅱ)后的红外光谱
Fig. 7 Infrared spectra of spent grain before and after thiol functionalized and TSG adsorption of Zn(Ⅱ)
表6 麦糟巯基化改性前后及其吸附Zn(Ⅱ)后的红外光谱分析
Table 6 Infrared spectra analysis of spent grain before and after thiol functionalized and TSG adsorption of Zn(Ⅱ)
3 结论
1) 巯基化改性麦糟对Zn(Ⅱ)有较好的吸附能力,其理论饱和吸附量达353.36 mg/g,较其他改性木质纤维素类吸附剂对Zn(Ⅱ)的吸附能力要高。
2) 巯基化改性麦糟吸附Zn(Ⅱ)的最佳pH范围较宽为6~9,该吸附反应很快,30 min即达到平衡,为活性化学吸附,有利于其工业应用。
REFERENCES
[1] JAIN C, SINGHAL D, SHARMA M. Adsorption of zinc on bed sediment of River Hindon: Adsorption models and kinetics[J]. Journal of Hazardous Materials, 2004, 114(1): 231-239.
[2] KURNIAWAN T A, CHAN G, LO W H, BABEL S. Physico-chemical treatment techniques for wastewater laden with heavy metals[J]. Chemical Engineering Journal, 2006, 118(1): 83-98.
[3] MALAMIS S, KATSOU E. A review on zinc and nickel adsorption on natural and modified zeolite, bentonite and vermiculite: Examination of process parameters, kinetics and isotherms[J]. Journal of Hazardous Materials, 2013, 252/253: 428-461.
[4] WAN NGAH WS, HANAFIAH MAKM. Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: A review[J]. Bioresource Technology, 2008, 99(10): 3935-3948.
[5] O'CONNELL D W, BIRKINSHAW C, O'DWYER T F. Heavy metal adsorbents prepared from the modification of cellulose: A review[J]. Bioresource Technology, 2008, 99(15): 6709-6724.
[6] WU Z, CHENG Z, MA W. Adsorption of Pb(Ⅱ) from glucose solution on thiol-functionalized cellulosic biomass[J]. Bioresource Technology, 2012, 104: 807-809.
[7] ZHANG C, SUI J, LI J, TANG Y, CAI W. Efficient removal of heavy metal ions by thiol-functionalized superparamagnetic carbon nanotubes[J]. Chemical Engineering Journal, 2012, 210(1): 45-52.
[8] CELO V, ANANTH R V, SCOTT S L, LEAN D R S. Methylmercury artifact formation during solid-phase extraction of water samples using sulfhydryl cotton fiber adsorbent[J]. Analytica Chimica Acta, 2004, 516(1/2): 171-177.
[9] CHAI L, LI Q, ZHU Y, ZHANG Z, WANG Q, WANG Y, YANG Z. Synthesis of thiol-functionalized spent grain as a novel adsorbent for divalent metal ions[J]. Bioresource Technology, 2010, 101(15): 6269-6272.
[10] LEE Y H, MOWRER J. Determination of methylmercury in natural waters at the sub-nanograms per litre level by capillary gas chromatography after adsorbent preconcentration[J]. Analytica Chimica Acta, 1989, 221(2): 259-268.
[11] LIU J F, ZHANG L G, GU N, REN J Y, WU Y P, LU Z H, MAO P S, CHEN D Y. Fabrication of colloidal gold micro-patterns using photolithographed self-assembled monolayers as templates[J]. Thin Solid Films, 1998, 327: 176-179.
[12] 彭国文, 丁德馨, 胡 南, 杨雨山, 王晓亮. 纳米 Fe3O4负载啤酒酵母菌对铀的吸附性能与机理[J]. 中国有色金属学报, 2012, 22(2): 604-610.
PENG Guo-wen, DING De-xing, HU Nan, YANG Yu-shan, WANG Xiao-liang. Adsportion properties and mechanism of Soccharomyces cererisiac loaded by nano-Fe3SO4 on uranium[J]. The Chinese Journal of Nonferrous Metals, 2012, 22(2): 604-610.
[13] CAO X, MA L, GAO B, HARRIS W. Dairy-manure derived biochar effectively sorbs lead and atrazine[J]. Environmental Science & Technology, 2009, 43(9): 3285-3291.
[14] NYBROE O, BRANDT KK, IBRAHIM YM, TOM-PETERSEN A, HOLM PE. Differential bioavailability of copper complexes to bioluminescent pseudomonas fluorescens reporter strains[J]. Environmental Toxicology and Chemistry, 2008, 27(11): 2246-2252.
[15] HO Y, NG J, MCKAY G. Kinetics of pollutant sorption by biosorbents: Review[J]. Separation & Purification Reviews, 2000, 29(2): 189-232.
[16] WEBER W, MORRIS J. Kinetics of adsorption on carbon from solution[J]. Journal of the Sanitary Engineering Division, American Society of Civil Engineers, 1963, 89(17): 31-60.
[17] HAN R, ZHANG L, SONG C, ZHANG M, ZHU H, ZHANG L. Characterization of modified wheat straw, kinetic and equilibrium study about copper ion and methylene blue adsorption in batch mode[J]. Carbohydrate Polymers, 2010, 79(4): 1140-1149.
[18] LANGMUIR I. The adsorption of gases on plane surfaces of glass, mica and platinum[J]. Journal of the American Chemical Society, 1918, 40(9): 1361-1403.
[19] GANJI M, KHOSRAVI M, RAKHSHAEI R. Biosorption of Pb, Cd, Cu and Zn from the wastewater by treated Azolla filiculoides with H2O2/MgCl2[J]. International Journal of Environmental Science & Technology, 2005, 1(4): 265-271.
[20]  . Modified softwood sawdust as adsorbent of heavy metal ions from water[J]. Journal of Hazardous Materials, 2006, 136(2): 266-271.
. Modified softwood sawdust as adsorbent of heavy metal ions from water[J]. Journal of Hazardous Materials, 2006, 136(2): 266-271.
[21] HORSFALL M. Kinetic studies on the adsorption of Cd2+, Cu2+ and Zn2+ ions from aqueous solutions by cassava (Manihot sculenta Cranz) tuber bark waste[J]. Bioresource Technology, 2006, 97(2): 283-291.
[22] SHUKLA S R, PAI R S. Adsorption of Cu(Ⅱ), Ni(Ⅱ) and Zn(Ⅱ) on modified jute fibres[J]. Bioresource Technology, 2005, 96(13): 1430-1438.
[23] NASERNEJAD B, ZADEH T E, POUR B B, BYGI M E, ZAMANI A. Camparison for biosorption modeling of heavy metals (Cr(Ⅲ), Cu(Ⅱ), Zn(Ⅱ)) adsorption from wastewater by carrot residues[J]. Process Biochemistry, 2005, 40(3/4): 1319-1322.
[24] SHUKLA S, PAI R S, SHENDARKAR A D. Adsorption of Ni(Ⅱ), Zn(Ⅱ) and Fe(Ⅱ) on modified coir fibres[J]. Separation and Purification Technology, 2006, 47(3): 141-147.
[25] AMUDA O, GIWA A, BELLO I. Removal of heavy metal from industrial wastewater using modified activated coconut shell carbon[J]. Biochemical Engineering Journal, 2007, 36(2): 174-181.
[26] HOMAGAI P L, GHIMIRE K N, INOUE K. Adsorption behavior of heavy metals onto chemically modified sugarcane bagasse[J]. Bioresource Technology, 2010, 101(6): 2067-2069.
[27] DENG S, TING Y P. Characterization of PEI-modified biomass and biosorption of Cu(Ⅱ), Pb(Ⅱ) and Ni(Ⅱ)[J]. Water Research, 2005, 39(10): 2167-2177.
[28] NOGHABI K A, ZAHIRI H S, YOON S C. The production of a cold-induced extracellular biopolymer by Pseudomonas fluorescens BM07 under various growth conditions and its role in heavy metals absorption[J]. Process Biochemistry, 2007, 42(5): 847-855.
[29] ARSLANOGLU H, ALTUNDOGAN H S, TUMEN F. Heavy metals binding properties of esterified lemon[J]. Journal of Hazardous Materials, 2009, 164(2): 1406-1413.
[30] SOCRATES G. Infrared characteristic group frequencies[M]. New York: Wiley, 1980.
(编辑 李艳红)
基金项目:国家高新技术研究计划资助项目(2011AA061001);国家自然科学基金资助项目(51304251);中国博士后科学基金资助项目(2013M542141);中南大学博士后基金资助项目
收稿日期:2013-08-09;修订日期:2013-12-20
通信作者:王庆伟,讲师,博士;电话:0731-88830875;E-mail: qw_wang@csu.edu.cn