
Thermodynamic assessment of Co-Cr-W ternary system
YANG Shu-yu, JIANG Min, LI Hong-xiao, WANG Lei
Key Laboratory for Anisotropy and Texture of Materials, Ministry of Education,
Northeastern University, Shenyang 110819, China
Received 30 October 2010; accepted 27 May 2011
Abstract: The Co-Cr-W ternary system was critically assessed using the CALPHAD technique. The solution phases including the liquid, g-Co, e-Co and a-Cr were described by a substitutional solution model. The σ, μ and R phases were described by three-sublattice models of (Co,W)8(Cr,W)4(Co,Cr,W)18,(Co,Cr,W)7W2(Co,Cr,W)4 and (Co,W)27(Cr,W)14(Co,Cr,W)12, respectively, in order to reproduce their homogeneity ranges. A self-consistent set of thermodynamic parameters for each phase was derived. The calculated isothermal sections at 1 000, 1 200 and 1 350 °C are in good agreement with the experimental data. A eutectoid reaction of R m+g-Co+s in this ternary system was predicted to occur at 1 022 °C.
m+g-Co+s in this ternary system was predicted to occur at 1 022 °C.
Key words: Co-Cr-W system; Co-based superalloys; phase diagram; thermodynamics; σ phase; μ phase; R phase
1 Introduction
Both chromium (Cr) and tungsten (W) are main constituent elements of Co-based superalloys [1-4]. Cr can improve the resistance against high temperature oxidation and corrosion of the alloys [5], while W serves to provide additional strength to the matrix owing to its large atomic size. So the Co-Cr-W system can be regarded as an important subsystem for Co-based superalloys [6-7]. It is also well known that phase diagrams and self-consistent thermodynamic descriptions of alloys are of great value for alloy design and processing [8-13]. Thus, a study on the phase diagram and thermodynamic properties of the Co-Cr-W ternary system is fundamental to develope Co-based superalloys. Until now, several isothermal sections in the Co-rich region in the Co-Cr-W ternary system have been established by experiment [14-16]. However, a precise thermodynamic description for this system, which is crucial for the alloy design of Cr and W in Co-based superalloys, has not been reported. Therefore, in the present study, a thermodynamic assessment of the Co-Cr-W ternary system is carried out by a CALPHAD (CALculation of PHAse Diagram) approach, which is very useful for the development of high quality Co-based superalloys.
2 Available experimental information
The phase equilibria of the Co-Cr-W system, especially in the Co-Cr side, were studied in several works [14-16]. DRAPIER et al [14] first measured the solubility of the γ-Co phase at 1 200 °C and studied the aging characteristic of some Co-Cr-W alloys using optical microscopy (OM), X-ray diffraction (XRD), hardness measurement and transmission electron microscopy (TEM). After that, BARROWS and NEWKIRK [15] investigated the Co-Cr-W system in somewhat more detail by means of OM, microhardness measurement, XRD and electron probe microanalysis (EPMA). In their work, a ternary intermetallic compound R phase was detected around the alloy composition of Co23Cr15W15, and the R phase was found to be in equilibrium with the γ-Co, μ and σ phases. The solubility of the γ-Co phase was determined and the μ phase region was detected up to 21% Cr (mass fraction). An isothermal section at 1 350 °C was then established, as the dotted lines shown in Fig. 1. SATO [16] recently investigated the phase equilibria in the Co-Cr-W system from 1 000 °C to 1 350 °C by scanning electron microscopy (SEM) and XRD analysis. A comparison was done for the isothermal section at 1 350 °C determined by BARROWS and NEWKIRK [15] and SATO [16]. The phase relations in the Co-rich region and the solid solubility of the γ-Co phase detected in both works are nearly the same. The μ phase in the Co-W side and the s phase in the Co-Cr side detected by SATO seem to be more stable than in BARROWS’ work, which drives the three-phase region of m+s+a-Cr towards the higher Cr corner in SATO’s work. SATO also studied the isothermal sections at 1 000 °C and 1 200 °C, where nearly the same phase relationships were obtained at 1 200 °C, while the R phase was not stable at 1 000 °C. The solubility of W in the σ phase was determined up to 25% (mole fraction), while the solubility of Cr in the μ phase can be up to about 50% (mole fraction). All these data will be used in the assessments in this work.
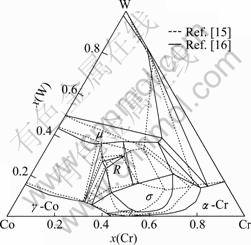
Fig. 1 Isothermal section of Co-Cr-W system at 1 350 °C determined by BARROWS and NEWKIRK [15] and SATO [16]
3 Thermodynamic modeling
3.1 Solution phases
The substitutional solution model is used to describe the solution phases of the liquid and the solid phases of g-Co, e-Co and a-Cr, which have the FCC, HCP and BCC structures, respectively. The Gibbs free energy of the phase f (f =liq, FCC, BCC and HCP?) is described by the following expression:
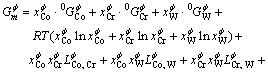
 (1)
(1)
where 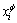 (i = Co, Cr, W) is the mole fraction of component i in the f phase; R is the gas constant; T is the thermodynamic temperature;
(i = Co, Cr, W) is the mole fraction of component i in the f phase; R is the gas constant; T is the thermodynamic temperature; 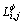 (i, j = Co, Cr, W, and i ≠ j) is the binary interaction parameter between the components i and j;
(i, j = Co, Cr, W, and i ≠ j) is the binary interaction parameter between the components i and j;  is the molar Gibbs free energy of the pure component i;
is the molar Gibbs free energy of the pure component i; 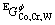 is the ternary excess Gibbs free energy; and
is the ternary excess Gibbs free energy; and  is the magnetic contribution to the Gibbs free energy. The binary interaction parameter
is the magnetic contribution to the Gibbs free energy. The binary interaction parameter 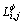 is written as follows:
is written as follows:
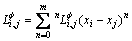 (2)
(2)
The ternary excess Gibbs free energy can be expressed by:
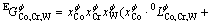
 (3)
(3)
The binary interaction parameters  are taken from the previous thermodynamic calculations [17-19] and the ternary interaction parameters
are taken from the previous thermodynamic calculations [17-19] and the ternary interaction parameters  will be optimized in this work.
will be optimized in this work.
The  in Eq. (1) is a magnetic contribution to the Gibbs free energy [20]. In this work, no ternary magnetic parameters are introduced.
in Eq. (1) is a magnetic contribution to the Gibbs free energy [20]. In this work, no ternary magnetic parameters are introduced.
3.2 σ, μ and R phases
The σ phase is D83 structure with the CrFe type [21]. In the Co-Cr binary system, the σ phase was modeled by (Co)8(Cr)4(Co,Cr)18 [17]. As shown in Fig. 1, the s phase can dissolve a large amount of W, where W substitutes partial Co and Cr. Therefore, in this work, we assume that W can enter all the three sublattices, and model the ternary σ phase as (Co,W)8(Cr,W)4(Co,Cr,W)18.
The μ phase is D85 structure with the Fe7W6 type [22]. In the Co-W binary system, the μ phase was modeled by a three-sublattice model of (Co,W)7W2- (Co,W)4 [18]. More than 50% Cr (mole fraction) can dissolve in the m phase in the ternary system, substituting both Co and W according to its homogeneity range. Therefore, in this work, we assume that Cr can occupy both the first and the third sublattices, and model the ternary m phase as (Co,Cr,W)7W2(Co,Cr,W)4.
A ternary intermetallic compound, R phase, has been reported in the Co-Cr-W system around the composition of Co23Cr15W15 [15]. It has been known that the R phase has the same structure with that in the Fe-Mo binary system [23], which has been modeled as (Fe)27(Mo)14(Fe,Mo)12. As we know, Co and Fe have very similar physical and chemical properties, thus, in the modeling work we assume that Co occupies the sublattices of Fe. Meanwhile, the elements Cr and Mo are in the same group in the periodic table and we assume that Cr occupies the sublattices of Mo. Finally, W atom may occupy all three sublattices. Therefore, the ternary R phase is described by a three-sublattice model of (Co,W)27(Cr,W)14(Co,Cr,W)12.
We then describe all the three three-sublattice models for the phases of σ, μ and R as a general formula (A,B,C)m(A,B)p(A,B,C)q. The Gibbs energy of the phase j (j =σ, μ and R) can be described by the following expression:
 (4)
(4)
where  is the Gibbs free energy of the j phase when the first sublattice is occupied by the element i (i = Co, Cr or W), the second by the element j (j = Cr, W), and the third by the element k (k = Co, Cr or W);
is the Gibbs free energy of the j phase when the first sublattice is occupied by the element i (i = Co, Cr or W), the second by the element j (j = Cr, W), and the third by the element k (k = Co, Cr or W);  is the interaction parameter between element i1 and i2 (i1, i2 = Co, Cr or W and i1 ≠ i2) when the second sublattice is occupied by the element j (j = Cr, W), and the third by the element k (k = Co, Cr or W); the parameters
is the interaction parameter between element i1 and i2 (i1, i2 = Co, Cr or W and i1 ≠ i2) when the second sublattice is occupied by the element j (j = Cr, W), and the third by the element k (k = Co, Cr or W); the parameters 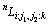 and
and  are described in the same way.
are described in the same way.
4 Optimization
The thermodynamic parameters of the binary
Co-Cr, Co-W and Cr-W systems were taken from the work of OIKAWA et al [17], SATO et al [18] and GUSTAFSON [19], respectively. The ternary interaction parameters were thermodynamically optimized in this work by using the CALPHAD technique with the aid of the PARROT module of the Thermo-Calc package [24]. The optimization began with the γ-Co phase. The determined solid solubility data of the γ-Co phase by BARROWS and NEWKIRK [15] and SATO [16] were used to optimize the parameters of 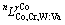 . The model parameters of the σ and μ phases were then considered based on the phase relationships claimed by both BARROWS and NEWKIRK [15] and SATO [16], with a larger weight factor on the data of SATO. The parameter
. The model parameters of the σ and μ phases were then considered based on the phase relationships claimed by both BARROWS and NEWKIRK [15] and SATO [16], with a larger weight factor on the data of SATO. The parameter  was specially evaluated in order to reproduce the σ phase boundary in the Cr-rich region. For the m phase, the parameters
was specially evaluated in order to reproduce the σ phase boundary in the Cr-rich region. For the m phase, the parameters  and
and  were adjusted in order to reproduce the homogeneity range extending to the high Cr region. Finally, the model parameters of the R phase were considered according to the determined phase compositions and the phase stability in Refs. [15] and [16]. The ternary model parameters obtained in this work together with those in the constituent binary systems are listed in Table 1.
were adjusted in order to reproduce the homogeneity range extending to the high Cr region. Finally, the model parameters of the R phase were considered according to the determined phase compositions and the phase stability in Refs. [15] and [16]. The ternary model parameters obtained in this work together with those in the constituent binary systems are listed in Table 1.
Table 1 Thermodynamic parameters of ternary Co-Cr-W system

5 Results and discussion
With the thermodynamic parameters in Table 1, the phase diagrams of the Co-Cr-W system can be calculated. Figure 2 shows the calculated isothermal sections at 1 000, 1 200 and 1 350 °C compared with the experimental data. It can be seen that the calculation can reproduce the ternary solid solubility of the γ-Co and α-Cr determined by experiments. The phase relationships derived by the calculation agree well with the work in Refs. [15] and [16]. The very large solubilities of the m and s phases determined by experiments have been satisfied in the calculation. The R phase is stable at high temperatures of 1 200 and 1 350 °C, and is not stable below 1 000 °C. A eutectoid reaction of R m+g-Co+s is predicted to occur at 1 022 °C, and at a higher temperature of 1 444 °C, the R phase disappears due to a reaction of L+R
m+g-Co+s is predicted to occur at 1 022 °C, and at a higher temperature of 1 444 °C, the R phase disappears due to a reaction of L+R g-Co+σ by our calculation.
g-Co+σ by our calculation.
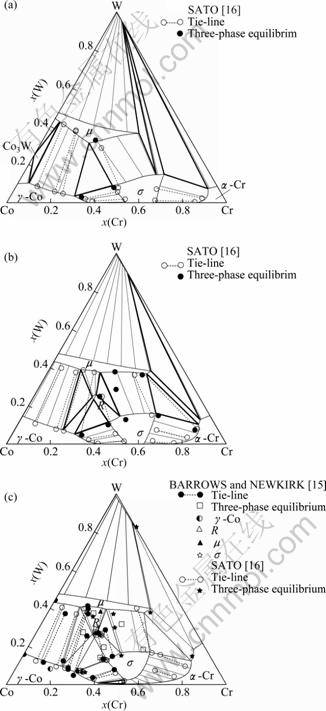
Fig. 2 Calculated isothermal sections at 1 000 °C (a), 1 200 °C (b) and 1 350 °C (c) compared with experimental data by BARROWS and NEWKIRK [15] and SATO [16]
6 Conclusions
1) A thermodynamic analysis of the Co-Cr-W system was carried out and a self-consistent set of thermodynamic parameters was obtained. The calculated isothermal sections are in good agreement with the available experimental data.
2) The phase relationships and the solid solubilities of the g-Co, a-Cr, μ and σ phases determined by experiments have all been reproduced reasonably.
3) The ternary R phase is stable from 1 022 °C to 1 444 °C in the Co-Cr-W system by the present calculation. All these provide useful information for the alloying of Cr and W in Co-based superalloys design.
References
[1] LIU R, XI S Q, KAPOOR S, WU X J. Investigation of solidification behavior and associate microstructures of Co-Cr-W and Co-Cr-Mo alloy systems using DSC technique [J]. Materials Science, 2010, 45: 6225-6234.
[2] GUPTA R K, KARTHIKEYAN M K, BHATIA D N, GHOSH B R, SINHA P P. Effect of microstructure on mechanical properties of refractory Co-Cr-W-Ni alloy [J]. Metal Science and Heat Treatment, 2008, 50(3): 175-179.
[3] GUPTA R K, KARTHIKEYAN M K, BHATIA D N, GHOSH B R, SINHA P P. Effect of microstructure on mechanical properties of Co-Cr-W-Ni superalloy [J]. High Temperature Materials and Processes, 2008, 27(3): 185-191.
[4] BIROL Y. Ni- and Co-based superalloys as potential tool materials for thixoforming of steels [J]. International Journal of Material Forming, 2010, 3(1): 739-742.
[5] FRISK K, MARKSTROM A. Effect of Cr and V on phase equilibria in Co-WC based hardmetals [J]. International Journal of Materials Research, 2008, 99(3): 287-293.
[6] ESPEVIK S, RAPP R A, DANIEL P L, HIRTH J P. Oxidation of ternary Co-Cr-W alloys [J]. Oxidation of Metals, 1983, 20: 37-65.
[7] GUPTA K P. The Co-Cr-W (cobalt-chromium-tungsten) system [J]. Journal of Phase Equilibria and Diffusion, 2006, 27(2): 178-183.
[8] RETTENMAYR M. Alloy development using modern tools [J]. International Journal of Materials Research, 2009, 100(2): 153-159.
[9] OIKAWA K, OHNUMA I, KAINUMA R, ISHIDA K. Alloy phase diagram study and its application for new alloy development [J]. Journal of the Japan Institute of Metals, 2008, 72(8): 545-556.
[10] QIAO Zhi-yu, HAO Shi-ming. New progress of CALPHAD approach [J]. Journal of Materials and Metallurgy, 2005, 4(2): 83-90.(in Chinese)
[11] AVRAHAM S, MAOZ Y, BAMBERGER M. Application of the CALPHAD approach to Mg-alloys design [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 2007, 31(4): 515-521.
[12] DUPEN B. A materials engineering technology approach to alloy design [J]. Journal of Engineering Technology, 2007, 24(2): 20-27.
[13] CUI Y F, ZHANG X, XU G L, ZHU W J, LIU H S, JIN Z P. Thermodynamic assessment of Co-Al-W system and solidification of Co-enriched ternary alloys [J]. Journal of Materials Science, 2011, 46(8): 2611-2621.
[14] DRAPIER J M, BROUWER J L, COUTSOURADIS D. Refractory metals and intermetallic precipitates in cobalt-chromium [J]. Cobalt, 1965, 27: 59-72.
[15] BARROWS R G, NEWKIRK J B. Phase equilibria in the Co-Cr-W system with special emphasis on the R-phase [J]. Metallography, 1972, 5: 515-541.
[16] SATO J. Thermodynamic database of Co-based systems [D]. Sendai: Department of Materials, Graduate School of Engineering, Tohoku University, Japan, 2007: 108-110. (in Japanese)
[17] OIKAWA K, QIN G W, IKESHOJI T, KAINUMA R, ISHIDA K. Direct evidence of magnetically induced phase separation in the fcc phase and thermodynamic calculations of phase equilibria of the Co-Cr system [J]. Acta Materialia, 2002, 50: 2223-2232.
[18] SATO J, OIKAWA K, KAINUMA R, ISHIDA K. Experimental verification of magnetically induced phase separation in α-Co phase and thermodynamic calculations of phase equilibria in the Co-W system [J]. Materials Transactions, 2005, 46(6): 1199-1207.
[19] GUSTAFSON P. A thermodynamic evaluation of the Cr-Ni-W system [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 1988, 12(3): 277-292.
[20] HILLERT M, JARL M. A model for alloying effects in fer romagnetic metals [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 1978, 2(3): 227-238.
[21] JOUBRT J M. Crystal chemistry and Calphad modeling of the σ phase [J]. Progress in Materials Science, 2008, 53: 528-583.
[22] HARI KUMAL K C, ANSARA I, WOLLANTS P. Sublattice modeling of the μ-phase [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 1998, 22(3): 323-334.
[23] ANDERSSON J O. A thermodynamic evaluation of the Fe-Mo-C system [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 1988, 12(1): 9-23.
[24] SUNDMAN B, JANSSON B, ANDERSSON J O. The thermo-calc databank system [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 1985, 9(2): 153-190.
[25] DINSDALE A T. SGTE data for pure elements [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 1991, 15(4): 317-425.
[26] DUPIN N, ANSA RA I. On the sublattice formalism applied to the B2 phase [J]. Zeitschrift fur Metallkunde, 1999, 90(1): 76-85
Co-Cr-W三元系相平衡的热力学计算
杨舒宇, 蒋 敏, 李洪晓, 王 磊
东北大学 材料各向异性与织构教育部重点实验室,沈阳 110819
摘 要:用CALPHAD方法评估了Co-Cr-W三元系,计算了1 000, 1 200和1 350 °C的相平衡。采用亚正规溶体模型描述了液相,fcc相,bcc相和hcp相。σ相,μ相,R相分别用模型(Co,W)8(Cr,W)4(Co,Cr,W)18, (Co,Cr,W)7W2(Co,Cr,W)4 和(Co,W)27(Cr,W)14(Co,Cr,W)12来表示;得到了自洽的热力学相互作用参数。计算的1 000, 1 200和1 350 °C 的相图与实验数据吻合。
关键词:Co-Cr-W三元系;Co基高温合金;相图;热力学;σ相;μ相;R相
(Edited by YANG Hua)
Foundation item: Project (50771027) supported by the National Basic Research Program of China; Project (50771027) supported by the National Natural Science Foundation of China
Corresponding author: JIANG Min; Tel: +86-24-83681676; E-mail: jiangm@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(11)61007-8