Thermodynamic optimization of Bi-Ni binary system
来源期刊:中国有色金属学报(英文版)2011年第1期
论文作者:王江 孟凡桂 刘立斌 金展鹏
文章页码:139 - 145
关键词:锡基合金;相图;热力学;相图计算;Bi-Ni二元系
Key words:Sn-based alloy; phase diagram; thermodynamics; CALPHAD; Bi-Ni binary system
摘 要:基于文献报导的实验数据,采用相图计算(CALPHAD)方法,热力学优化了Bi-Ni二元系相图。该二元系的液相、fcc_A1(Ni) 相和rhombohedral_A7(Bi)相用替换溶液模型来描述,其过剩吉布斯自由能用Redlich-Kister多项式来表达。考虑到晶体结构(NiAs型)以及与多组元体系热力学数据库的兼容性,中间化合物BiNi相采用亚点阵模型:(Bi)(Ni,Va)(Ni,Va);Bi3Ni相处理为化学计量比化合物。最后,通过优化该二元系实测的相图和热力学数据,获得一组能够表达各相吉布斯自由能的自洽的热力学参数。根据这些热力学参数计算的相图和热力学数据与报导的实验数据吻合良好。
Abstract: Based on the available experimental data, the Bi-Ni binary system was optimized thermodynamically by the CALPHAD method. The solution phases, including liquid, fcc_A1(Ni) and rhombohedral_A7(Bi), were described as substitutional solution phases, of which the excess Gibbs energies were expressed with the Redlich-Kister polynomial. The intermetallic compound, BiNi, was modeled using three sublattices (Bi)(Ni,Va)(Ni,Va) considering its crystal structure (NiAs-type) and the compatibility of thermodynamic database in the multi-component systems, while Bi3Ni was treated as a stoichiometric compound. Finally, a set of self-consistent thermodynamic parameters formulating the Gibbs energies of various phases in this binary system were obtained. The calculated results are in reasonable agreement with the reported experimental data.

WANG Jiang1, 2, 3, MENG Fan-gui4, LIU Li-bin2, JIN Zhan-peng2
1. School of Materials Science and Engineering, Guilin University of Electronic Technology, Guilin 541004, China;
2. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
3. Swiss Federal Laboratories for Materials Testing and Research, Laboratory for Joining and Interface Technology, ?berlandstrasse 129, CH-8600, Dübendorf, Switzerland;
4. School of Materials Science and Engineering, Central South University of Forestry and Technology,
Changsha 410004, China
Received 21 April 2010; accepted 28 July 2010
Abstract: Based on the available experimental data, the Bi-Ni binary system was optimized thermodynamically by the CALPHAD method. The solution phases, including liquid, fcc_A1(Ni) and rhombohedral_A7(Bi), were described as substitutional solution phases, of which the excess Gibbs energies were expressed with the Redlich-Kister polynomial. The intermetallic compound, BiNi, was modeled using three sublattices (Bi)(Ni,Va)(Ni,Va) considering its crystal structure (NiAs-type) and the compatibility of thermodynamic database in the multi-component systems, while Bi3Ni was treated as a stoichiometric compound. Finally, a set of self-consistent thermodynamic parameters formulating the Gibbs energies of various phases in this binary system were obtained. The calculated results are in reasonable agreement with the reported experimental data.
Key words: Sn-based alloy; phase diagram; thermodynamics; CALPHAD; Bi-Ni binary system
1 Introduction
Increasing efforts have been conducted recently on the search for suitable Pb-free solders due to the harmful influence of Pb on both environment and human health[1-4]. Sn-based alloys with different additives, such as Ag, Bi, Cu, In, Sb and Zn, are potential candidates as Pb-free solders[5-7]. The Sn-Bi alloys with low melting point are developed for soldering the temperature-sensitive components in the step-by-step packaging process[7-9]. On the other hand, Ni is plated as an under bump metallurgy (UBM) layer on the substrate prior to soldering in modern microelectronic package[7-10]. Interfacial reactions between Sn-Bi solders and Ni play an important role on the reliability of the solder joints. Phase diagrams and thermodynamic properties of the related systems involving Sn-Bi solder and Ni are helpful to control interfacial microstructure during soldering[11-13]. In view of these, the phase relation of the Bi-Ni binary system as a binary sub-
system was considered here.
The Bi-Ni binary system was reviewed critically by NASH[14] in 1985. The assessments of this binary system were performed by VASSILEV et al[15-17] and SEO et al[18]. However, there are still some problems in their assessments[15-18]. Firstly, the miscibility gap of liquid phase is stable at above 1630 K from the assessment by SEO et al[18], resulting in the inconsistence with the experimental phase diagram, but it was not noticed in the original publication. Secondly, the different thermodynamic models of intermetallic compound BiNi were used in their assessment[15-18], and they are thus not compatible with the extrapolation of the multi-component system. Finally, it is not reasonable that the heat capacity of BiNi measured by PERRING et al[19] was not employed to optimize its thermodynamic parameters. Therefore, the present work was to obtain a set of self-consistent and reliable thermodynamic description of the Bi-Ni binary system using the CALPHAD method[20-21] and Thermo-calc? software package[22].
2 Experimental
2.1 Phase diagram data
The liquidus of the Bi-Ni binary system was measured by PORTEVIN[23] and VOSS[24] using thermal analysis. The experimental data on the liquidus are in good agreement with each other, while there is a discrepancy of the peritectic temperature of intermetallic compound BiNi. NASH[14] pointed out that the peritectic temperature (911 K) remarked by VOSS[24] is too low due to the supercooling effect of liquid alloys compared with the experimental data (927 K) obtained by PORTEVIN[23]. SHAVINSKI et al[25] reported that the composition and temperature of the eutectic reaction, L?Bi3Ni+rhombohedral(Bi), are 99.47% Bi (molar fraction) and 543 K, respectively. FESCHOTTE and ROSSET[26] investigated the Bi-Ni binary system and determined the eutectic and peritectic temperatures in this binary system. Their experimental results reveal that the intermetallic compound BiNi has a narrow homogeneity range and Bi3Ni is a stoichiometric compound. Recently, VASSILEV et al[15] determined the solidus of fcc_A1(Ni) phase as well as the compositions and temperatures of the invariant reactions by electron microprobe analyses, optical microscopy and differential scanning calorimetry. It was found that the narrow homogeneity region of BiNi is 50%-52% Bi and Bi3Ni is a stoichiometric compound. Using the diffusion couple method, SEO et al[18] measured accurately the homogeneity region of BiNi at different temperatures (473-873 K) through X-ray spectroscope and electron microprobe analyzer. The experimental data mentioned above in Refs.[15, 18, 23-26] were taken into account in the present optimization.
2.2 Thermodynamic data
Up to now, no experimental enthalpy of mixing of liquid Bi-Ni alloys was reported in the published literature. Activities of Bi and Ni in liquid phase were investigated by IWASE and MCLEAN[27] and VASSILEV et al[16]. IWASE and MCLEAN[27] determined the activities of Ni in liquid phase at 1 073 K and 1 773 K by electromotive force (EMF) method. Recently, VASSILEV et al[16] measured the activities of Bi in liquid phase at different temperatures (1 723 K, 1 753 K and 1 773 K) using the modified isothermal isopiestic method. On the other hand, the enthalpy of formation of intermetallic compound BiNi was determined by PREDEL and RUGE[28] using calorimetric technique. PERRING et al[19] measured the heat capacities of BiNi in the temperature range of 310-850 K by differential scanning calorimetry. The experimental data in Refs.[16, 19, 27-28] were used in the present optimization.
3 Thermodynamic model
3.1 Pure elements
The stable forms of the pure elements at 298.15 K and 100 kPa are chosen as the reference states. The Gibbs energy for the element i in ![]() status is given as
status is given as
![]()
![]() (1)
(1)
where ![]() is the enthalpy of the element i in its standard reference state (SER) at 298.15 K and 100 kPa;
is the enthalpy of the element i in its standard reference state (SER) at 298.15 K and 100 kPa; ![]() is the temperature;
is the temperature; ![]() is the Gibbs energy of the element i with structure
is the Gibbs energy of the element i with structure ![]() ;
; ![]() is the molar Gibbs energy of the element
is the molar Gibbs energy of the element ![]() with the structure of
with the structure of ![]() referred to the enthalpy of its stable state at 298.15 K and 100 kPa. In the present work,
referred to the enthalpy of its stable state at 298.15 K and 100 kPa. In the present work, ![]() and
and ![]() are taken from the SGTE (Scientific Group Thermodata Europe) database[29].
are taken from the SGTE (Scientific Group Thermodata Europe) database[29].
3.2 Solution phases
The solid solution phase![]() (
(![]() =liquid, fcc or rhombohedral) is described with the substitutional solution model. The molar Gibbs energy of the solid solution with the Redlich-Kister polynomial[30] is expressed as follows:
=liquid, fcc or rhombohedral) is described with the substitutional solution model. The molar Gibbs energy of the solid solution with the Redlich-Kister polynomial[30] is expressed as follows:
![]()
![]() (2)
(2)
where R is the gas constant; xBi and xNi are the mole fractions of Bi and Ni, respectively; and ![]() is the interaction parameter dependent on temperature:
is the interaction parameter dependent on temperature:
![]() (3)
(3)
where Ai and Bi are model parameters to be optimized in the present work.
3.3 Intermetallic compounds
On the basis of the experimental result in Ref. [31], the crystal structure of intermetallic compound BiNi is NiAs-type, as shown in Fig.1. The deviations from stoichiometric composition on both Bi-rich side and Ni-rich side are achieved by vacancies in the normal Ni atom sites and interstitial Ni atom in the normal vacancy sites, respectively. Therefore, the sublattice model (Bi)0.3334(Ni,Va)0.3333(Ni,Va)0.3333 is used in the present work. The second sublattice (Ni,Va)0.3333 means the normal Ni site with some vacancies, while the third one (Ni,Va)0.3333 means the normal interstitial site with some
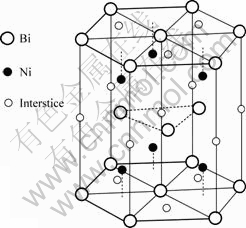
Fig.1 Schematic crystal structure of NiAs-type phase for intermetallic compound BiNi
interstitial Ni atoms. Furthermore, the same model was employed to describe other intermetallic compounds with NiAs-type structure, CoSb and NiSb in the Co-Sb and Ni-Sb binary systems and Co-Ni-Sb ternary system by ZHANG et al[32-34]. The molar Gibbs energy of BiNi can be given as
![]()
![]()
![]()
![]()
![]() (4)
(4)
with
![]()
![]() (5)
(5)
![]() (6)
(6)
![]() (7)
(7)
![]() (8)
(8)
where ![]() and
and ![]() are the Gibbs energies of elements Bi and Ni at their respective standard states, and are taken from Ref.[29];
are the Gibbs energies of elements Bi and Ni at their respective standard states, and are taken from Ref.[29]; ![]() and
and ![]() denote mole fraction of i in the second and third sublattices, respectively;
denote mole fraction of i in the second and third sublattices, respectively;![]() ,
, ![]() ,
, ![]() ,
, ![]() are the interactive parameters, and optimized as dependent temperature. The parameters from C0 to J0 as well as Ci and Di are assessed in the present work.
are the interactive parameters, and optimized as dependent temperature. The parameters from C0 to J0 as well as Ci and Di are assessed in the present work.
On the other hand, intermetallic compound Bi3Ni is treated as a stoichiometric compound because of its limited homogeneity range. The molar Gibbs energy of Bi3Ni is given as follows:
![]() (9)
(9)
where the parameters C4 and D4 are to be optimized.
4 Results and discussion
Using the lattice stabilities of Bi and Ni cited from Ref.[29], the optimization of the model parameters was performed using the PARROT module in the Thermo-calc? software package developed by SUNDMAN et al[22]. This module works by minimizing the square sum of the differences between the experimental data and calculated values. In the optimization procedure, each set of the experimental data is given a certain weight according to the reliability and compatibility of the experimental data. Thermodynamic parameters for all condensed phases in the Bi-Ni binary system are summarized in Table 1. The calculated temperatures and composition of all invariant reactions in this binary system are compared with the experimental data and listed in Table 2.
Fig.2 shows the calculated phase diagram of the Bi-Ni binary system in the present work. The comparison of the calculated phase diagram with the experimental data[15, 18, 23-24] is given in Fig. 3. In combination with Table 2, it can be seen that the calculated liquidus, sodidus, the homogeneity region of intermetallic compound BiNi, as well as temperatures and compositions of the invariant reactions are in good agreement with the experimental data[23-24] and the previous results assessed by Refs.[15-18].
Fig.4 presents the calculated activities of Bi in liquid phase at 1753 K with the limited experimental data[16]. The calculated activities of Ni at 1 073 K and 1 773 K are compared respectively with the experimental data[27] and shown in Fig.5. It is evident that the calculated results are in good agreement with the experimental results[16, 27].
The enthalpies of mixing of liquid alloys at 1 773 K are calculated in the present work, as shown in Fig.6. It can be seen that, the calculated values are positive in Ni-rich side, but are negative in the Bi-rich side. The positive enthalpy of mixing of liquid phase in the Ni-rich side is corresponding with the smooth liquidus of the Bi-Ni phase diagram in Fig.2, which results in a possible metastable liquid miscibility gap at the low temperature. However, the calculated results need further experimental
Table 1 Thermodynamic parameters of Bi-Ni binary system

Table 2 Invariant reactions in Ni-Bi binary system
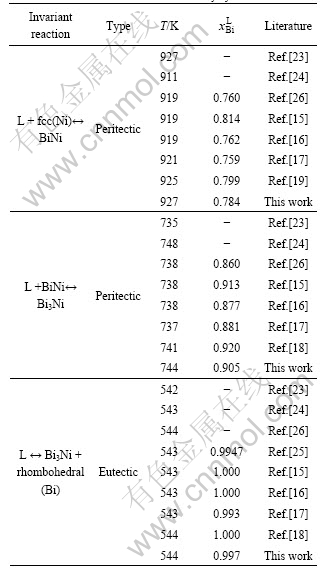
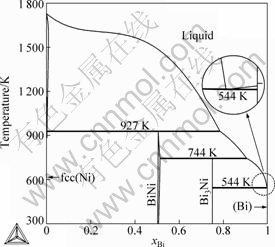
Fig.2 Calculated phase diagram of Bi-Ni binary system in present work
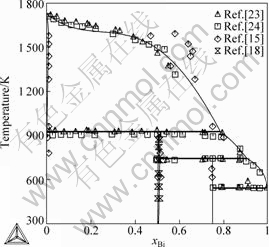
Fig.3 Calculated phase diagram of Bi-Ni binary system in present work compared with experimental data[15, 18, 23-24]
verification because there is no reported experimental data on the mixing enthalpy of liquid phase in published literature.
Fig.7 shows the calculated enthalpies of formation of the intermetallic compounds and the experimental
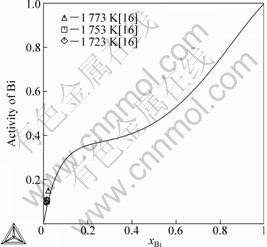
Fig.4 Calculated activity of Bi at 1 753 K with experimental data in Ref.[16] (Referred state: liquid Bi)
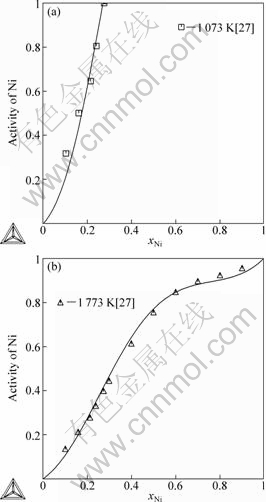
Fig.5 Calculated activities of Ni at 1 073 K (a) and 1 773 (b) with experimental data in Ref.[27] (Referred state: liquid Ni)
data[28]. The calculated heat capacities of BiNi are compared with the experimental data[19] and shown in Fig.8. Obviously, the calculated results are in good
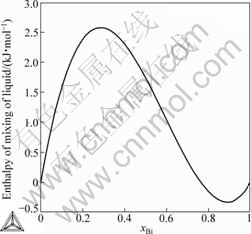
Fig.6 Calculated enthalpies of mixing of liquid alloys at 1 773 K in present work (Referred states: liquid Bi and liquid Ni)
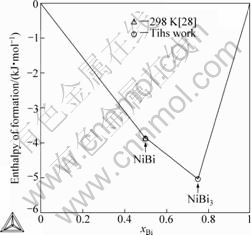
Fig.7 Calculated enthalpies of formation of intermetallic compounds in comparison with experimental data in Ref.[28] at 298 K (Referred states: solid Bi and solid Ni)
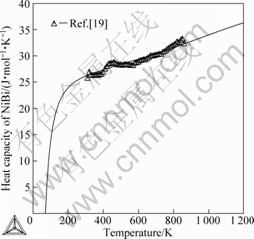
Fig.8 Calculated heat capacity of intermetallic compound BiNi with experimental data in Ref.[19]
agreement with the experimental data.
5 Conclusions
1) The present work reviewed critically the experimental information on phase diagram and thermodynamic properties of the Bi-Ni binary system in published literature.
2) Using the CALPHAD method, thermodynamic modelling of the Bi-Ni binary system was performed through the Thermo-calc? software package.
3) A set of self-consistent thermodynamic parameters formulating the Gibbs energies of various phases in the Bi-Ni binary system were obtained, which can reproduce well most of the experimental data on thermodynamic properties and phase diagram.
References
[1] ABTEWA M, SELVADURABY G. Lead-free solders in microelectronics [J]. Mater Sci Eng R, 2000, 27: 95-141.
[2] TU K N, ZENG K. Tin-lead (SnPb) solder reaction in flip chip technology [J]. Mater Sci Eng R, 2001, 34: 1-58.
[3] ZENG K, TU K N. Six cases of reliability study of Pb-free solder joints in electronic packaging technology [J]. Mater Sci Eng R, 2002, 38: 55-105.
[4] LI Fu-quan, WANG Chun-qing. Influence of interfacial reaction between molten SnAgCu solder droplet and Au/Ni/Cu pad on IMC evolution [J]. Transactions Nonferrous Metals Society of China, 2006, 16(1): 18-22.
[5] LI Guo-yuan, SHI Xun-qing. Effects of bismuth on growth of intermetallic compounds in Sn-Ag-Cu Pb-free solder joints [J]. Transactions Nonferrous Metals Society of China, 2006, 16(Suppl 2): s739-s743.
[6] LAURILA T, VUORINEN V, KIVILAHTI J K. Interfacial reactions between lead-free solders and common base materials [J]. Mater Sci Eng R, 2005, 49: 1-60.
[7] LAURILA T, VUORINEN V, PAULASTO-KR?CKEL M. Impurity and alloying effects on interfacial reaction layers in Pb-free soldering [J]. Mater Sci Eng R, 2010, 68: 1-38.
[8] WANG Jiang, LIU Hua-shan, LIU Li-bin, JIN Zhan-peng. Thermodynamic optimization of Sn-Ag-Bi ternary system [J]. Journal of Central South University of Technology, 2007, 14(Suppl 2): 83-89.
[9] WANG Jiang, LIU Hua-shan, LIU Li-bin, JIN Zhan-peng. Interfacial reaction between Sn-Bi alloy and Ni substrate [J]. J Electron Mater, 2006, 35: 1842-1847.
[10] WANG Jiang, LIU Hua-shan, LIU Li-bin, JIN Zhan-peng. Interfacial reaction between Sn-Ag alloy and Ni substrate [J]. J Alloys Compd, 2008, 455: 159-163.
[11] LIU Chun-lei, JIN Zhan-peng, LIU Hua-shan. Application of CALPHAD in soldering of microelectronic materials [J]. Transactions Nonferrous Metals Society of China, 2003, 13(6): 1343-1349.
[12] ISPSER H, FLANDORFER H, LUEF C, SCHMETTERER C, SAEED U. Thermodynamics and phase diagrams of lead-free solder materials [J]. J Mater Sci: Mater Electron, 2007, 18: 3-17.
[13] CHEN S W, WANG C H, LIN S K, CHIU C N. Phase diagrams of Pb-free solders and their related materials systems [J]. J Mater Sci: Mater Electron, 2007, 18: 19-37.
[14] NASH P. The Bi-Ni (bismuth-nickel) system [J]. Bull Alloy Phase Diagrams, 1985, 6: 345-347.
[15] VASSILEV C P, LIU X J, ISHIDA K. Experimental studies and thermodynamic optimization of the Ni-Bi system [J]. J Phase Equil Diffusion, 2005, 26: 161-168.
[16] VASSILEV C P, ROMANOWSKA J, WBUK G. Bismuth activity measurements and thermodynamic re-optimization of the Ni-Bi system [J]. Int J Mat Res, 2007, 98: 468-475.
[17] VASSILEV C P, GANDOVA V, DOCHEVA P. Comments and reconciliation of the Ni-Bi system thermodynamic reassessments [J]. Crst Res Technol, 2009, 44: 25-30.
[18] SEO S K, CHO M G, LEE H M. Thermodynamic assessment of the Ni-Bi binary system and phase equilibria of the Sn-Bi-Ni ternary system [J]. J Electron Mater, 2007, 36: 1536-1544.
[19] PERRING L, KUNTZ J J, BUSSY F, GACHON J C. Heat capacity measurements on the equiatomic compounds in Ni-X(X=Al, In, Si, Ge and Bi) and M-Sb(with M=Ni, Co and Fe) systems [J]. Intermetallics, 1999, 7: 1235-1239.
[20] KAUFMAN L, BERNSTEIN H. Computer calculation of phase diagrams [M]. New York: Academic Press, 1970.
[21] SAUNDERS N, MODWNIK A P. CALPHAD-A comprehensive guide [M]. Lausanne: Pergamon, 1998.
[22] SUNDMAN B, JANSSON B, ANDERSON J O. Thermo-calc databank system [J]. CALPHAD, 1985, 9: 153-190.
[23] PORTEVIN M A. The alloys of nickel and bismuth [J]. Rev Met, 1908, 5: 110-120.
[24] VOSS G. The nickel-bismuth system [J]. Z Anorg Chem, 1908, 57: 52-58.
[25] SHAVINSKI B M, ARTYKHIN P I, MITAYAKHIN Y L. Directional crystallization of bismuth. III. determination of the eutectic concentration of some metals in the pure bismuth region [J]. Izv Sib Otd Akad Nauk, 1979, 5: 70-74.
[26] FESCHOTTE P, ROSSET J M. Equilibres de phases dans le systeme binaire nickel-bismuth [J]. J Less-Common Met, 1988, 143: 31-37.
[27] IWASE M, MCLEAN A. Activities in nickel-bismuth alloys [J]. Metall Mater B, 1983, 14: 765-767.
[28] PREDEL B, RUGE H. Bildungsenthalpien und bindungsverh?ltnisse in einigen intermetallischen verbindungen vom NiAs-type [J]. Thermochim Acta, 1972, 3:411-418.
[29] DINSDALE A T. SGTE data for pure elements [J]. CALPHAD, 1991, 15: 317-425.
[30] REDLICH O, KISTER A T. Algebaric representation of thermodynamic properties and the classification of solution [J]. Indust Eng Chem, 1948, 40: 345-348.
[31] KUMAR P, SADAMA Y N. Electrodeposition of alloys IV: Electrodeposition and X-ray structure of bismuth-nickel alloys from aqueous solution [J]. J Less-Common Met, 1975, 43: 259-265.
[32] ZHANG Y B, LI C R, DU Z M, GENG T. The thermodynamic assessment of the Co-Sb system [J]. CALPHAD, 2008, 32: 56-63.
[33] ZHANG Y B, LI C R, DU Z M, GUO C P. A thermodynamic assessment of the Ni-Sb system [J]. CALPHAD, 2008, 32: 378-388.
[34] ZHANG Y B, LI C R, DU Z M, GUO C P, TEDENAC J C. The thermodynamic assessment of the ternary Co-Ni-Sb system [J]. CALPHAD, 2009, 33: 405-414.
王 江1, 2, 3,孟凡桂4,刘立斌2,金展鹏2
1. 桂林电子科技大学 材料科学与工程学院,桂林 541004;
2. 中南大学 材料科学与工程学院,长沙 410083;
3. 瑞士联邦材料测试与研究实验室 焊接与界面技术实验室,苏黎世CH-8600;
4. 中南林业科技大学 材料科学与工程学院,长沙 410004
摘 要:基于文献报导的实验数据,采用相图计算(CALPHAD)方法,热力学优化了Bi-Ni二元系相图。该二元系的液相、fcc_A1(Ni) 相和rhombohedral_A7(Bi)相用替换溶液模型来描述,其过剩吉布斯自由能用Redlich-Kister多项式来表达。考虑到晶体结构(NiAs型)以及与多组元体系热力学数据库的兼容性,中间化合物BiNi相采用亚点阵模型:(Bi)(Ni,Va)(Ni,Va);Bi3Ni相处理为化学计量比化合物。最后,通过优化该二元系实测的相图和热力学数据,获得一组能够表达各相吉布斯自由能的自洽的热力学参数。根据这些热力学参数计算的相图和热力学数据与报导的实验数据吻合良好。
关键词:锡基合金;相图;热力学;相图计算;Bi-Ni二元系
(Edited by YANG Hua)
Foundation item: Projects(50371104, 50771106 and 50731002) supported by the National Natural Science Foundation of China; Project(2008K 22) supported by the Scientific Research Foundation of Hunan Provincial Department of Land & Resources, China; Project supported by Geology Exploration Foundation of Hunan Provincial Department of Land & Resources, China
Corresponding author: WANG Jiang; E-mail: wangjiang158@gmail.com
DOI: 10.1016/S1003-6326(11)60690-0

