
Thermodynamic assessment of Cu-Cd system
CHEN Xiao-ming(陈小明), LIU Li-bin(刘立斌), ZHANG Li-gang(章立钢),
BO Hong(薄 宏), JIN Zhan-peng(金展鹏)
School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 27 February 2009; accepted 3 June 2009
Abstract: A thermodynamic assessment of the Cu-Cd system has been carried out by CALPHAD technique. The liquid phase, FCC_A1(Cu) and HCP_A3(Cd) terminal phases are described by a simple substitutional model, their excess Gibbs energy is formulated with the Redlich-Kister expression. The system contains four intermediate compounds, including β(Cu2Cd), γ(Cu4Cd3), δ(Cu5Cd8) and ε(Cu3Cd10). The δ phase is described by a two-sublattice model and the other three intermediate phases are treated as stoichiometric phases. The optimization is carried out in the Thermo-Calc package. A set of self-consistent thermodynamic parameters are obtained. The calculated phase diagram and thermodynamic properties agree well with the available experimental data.
Key words: Cu-Cd; phase diagram; thermodynamic assessment
1 Introduction
The copper based alloys with high strength and conductivity are the promising functional materials. Recently, there has been a remarkable progress for their research and development. So far, a series of copper based alloys with high strength and high conductivity have been developed, and Cu-Cd alloy is a typical one which shows the stage of the history to develop this kind of Cu-based alloy. According to Ref.[1], with 2% Cd (mass fraction) added, the crystal lattice of copper keeps unchanged, however, nor the conductivity of copper, but the strength has been increased considerably. Moreover, the Cu-Cd system is a subsystem of ternary Al-Cu-Cd system. Addition of a small amount of Cd into the Al-Cu alloy could significantly increase the nucleation rate of θ′ phase, this results in finer structure, and then the aging strengthening is improved. LI et al[2] studied the effect of Cd on the aging process of Al-Cu-Mg-Si system. Their result shows that the strength after aging is increased by 20% with 0.27% Cd (mass fraction) added.
The Cu-Cd system has been reviewed by several authors[3-4], they gave a complete Cu-Cd phase diagram. Knowledge of phase relations in temperature- composition space of the Cu-related systems is particularly helpful to comprehend the interaction between different elements and promote the development of new Cu-based materials with superior properties. However, no thermodynamic assessment is available, and the purpose of this work is to optimize it through CALPHAD approach to obtain a set of self- consistent thermodynamic parameters.
2 Experimental
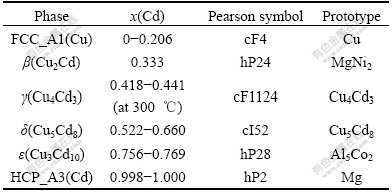
The Cu-Cd phase diagram was constructed by Hansen et al[3] and Subramanian et al[4]. Fig.1 shows the result based on the phase equilibria assessed by Subramanian et al[4]. The following phases are included in the Cu-Cd system: liquid, FCC_A1(Cu), β(Cu2Cd), γ(Cu4Cd3), δ(Cu5Cd8), ε(Cu3Cd10) and HCP_A3(Cd). The crystal structure data of these phases are given in Table 1.
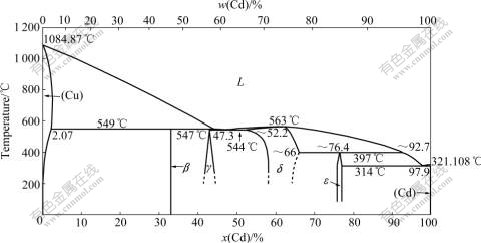
Fig.1 Cu-Cd phase diagram based on assessed phase equilibria from Ref.[4]
Table 1 Condensed phases in Cu-Cd binary system[4]
The liquidus was investigated by several researchers [5-7]. Heycock and NEVILLE[7] first investigated the liquidus of the Cd-rich region ranging from 97.9% to 100% Cd (mole fraction, the same below if not mentioned), while Sahmen[5] and Jenkins and HANSON[6] investigated the entire composition range. The solidus and solvus in the Cu-rich region were determined by X-ray, metallography and microhardness measurements[6-10]. Sulonen[10] determined the boundary by lattice constant measurements, and the assessed maximum solubility of Cd in FCC_A1(Cu) is 2.56% Cd at 650 ℃. The data from RAUB[9], Sulonen[10], and GLAZOV et al[11] were in good agreement with each other, but their results are different from those of Pogodin et al[12]. So the latter was not used in this optimization. The solubility of Cu in HCP_A3 (Cd) was reported by Jenkins and HANSON [6] as 0.12% Cu at 300 ℃ and 0.17% Cu at 270 ℃ by Tammann and HEINIEL[13]. Massalski et al[14] reported the equilibrium solid solubility of Cu in (Cd) is about 0.2% Cu.
As for thermodynamic properties, the mixing enthalpy of liquid Cu-Cd alloys was measured through calorimetry at 723 K by KLEPPA[15]. The activities of Cd in liquid Cu-Cd alloys were measured by Nikolskaya et al[16] with the EMF method at 575, 600, 625 and 650 ℃, respectively. Also, Riccoboni et al[17] (EMF method) as well as Jellinek and ROSNER[18] and Azakami and YAZAWA[19] (vapor pressure measurements) measured the activities of Cd in liquid. Most activity values selected or calculated by Hultgren et al[20] were reliable and used for comparison.
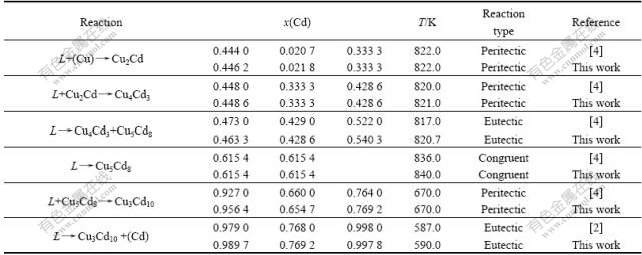
The Cu-Cd binary system reported by Subramanian et al[4] had two eutectic reactions, three peritectic reactions and one congruently melting reaction. The (Cu)+Liquid β peritectic reaction occurred between 545 and 552 ℃ was first reported by Sahmen et al[5] using DTA measurements. The peritectic temperature was confirmed at 547℃ by Jenkins and HANSON[6], SULONEN[10] and Glazov et al [11] through thermal, lattice parameter, and microhardness measurements, respectively. Jenkins and HANSON[6] also reported the peritectic reaction L+β
β peritectic reaction occurred between 545 and 552 ℃ was first reported by Sahmen et al[5] using DTA measurements. The peritectic temperature was confirmed at 547℃ by Jenkins and HANSON[6], SULONEN[10] and Glazov et al [11] through thermal, lattice parameter, and microhardness measurements, respectively. Jenkins and HANSON[6] also reported the peritectic reaction L+β γ, which was described as 44.8% Cd of liquid at 547 ℃. Sahmen et al[5] and Jenkins and HANSON[6] reported the congruent formation of δ at 563 ℃. And they first described δ as Cu2Cd3, while recent studies showed that δ has Cu5Cd8 prototype. The peritectic reaction L+δ
γ, which was described as 44.8% Cd of liquid at 547 ℃. Sahmen et al[5] and Jenkins and HANSON[6] reported the congruent formation of δ at 563 ℃. And they first described δ as Cu2Cd3, while recent studies showed that δ has Cu5Cd8 prototype. The peritectic reaction L+δ ε was first reported by Jenkins and HANSON[6], who showed that ε-phase forms at 397 ℃ and 92.7% Cd of liquid. He also reported the eutectic points near pure Cd and in the central part of the phase diagram, which was 314 ℃, 97.9% Cd and 544 ℃, 47.3% Cd respectively. All data concerning invariant reactions used for the assessment procedure are shown in Table 2.
ε was first reported by Jenkins and HANSON[6], who showed that ε-phase forms at 397 ℃ and 92.7% Cd of liquid. He also reported the eutectic points near pure Cd and in the central part of the phase diagram, which was 314 ℃, 97.9% Cd and 544 ℃, 47.3% Cd respectively. All data concerning invariant reactions used for the assessment procedure are shown in Table 2.
Table 2 Invariant reactions in Cu-Cd system
3 Thermodynamic modeling
The Cu-Cd system includes seven phases: three solution phases such as liquid, FCC_A1(Cu), and HCP_A3(Cd), one solution compound δ(Cu5Cd8), and three stoichiometric compounds such as β(Cu2Cd), γ(Cu4Cd3), and ε(Cu3Cd10).
3.1 Solution phases
The solution phases concerning liquid, FCC_A1(Cu) and HCP_A3(Cd) in the system were described by substitutional solution model. The Gibbs energies of the solution phases are expressed as follows:
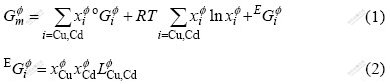
where 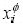 represents the mole fraction of component i in the
represents the mole fraction of component i in the 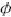 phase;
phase; 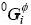 is the mole Gibbs free energy of pure component i;
is the mole Gibbs free energy of pure component i; 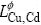 is the binary interaction parameter between Cu and Cd, which can be expressed in the form of a Redlich-Kister polynomial as
is the binary interaction parameter between Cu and Cd, which can be expressed in the form of a Redlich-Kister polynomial as
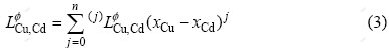
where 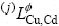 is the interaction parameter and can be expressed as temperature dependent terms as follows:
is the interaction parameter and can be expressed as temperature dependent terms as follows:
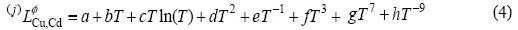
where a, b, c, d, e, f, g and h are the coefficients to be optimized. In most cases, only the first one or two terms of the above equation are used. Lattice stabilities of elements Cu and Cd are referred to Dinsdale[21], but there is no data available for Cd in FCC_A1(Cu), so 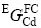 is treated as 5 kJ/mol in the present assessment.
is treated as 5 kJ/mol in the present assessment.
3.2 Compound δ with homogeneity range
The phase δ with Cu5Cd8-type cubic structure exhibits a considerable range of homogeneity. Several researchers have studied its crystal structure[22-24]. Bradley et al[22] and Heidenstam et al[23] showed 20 Cu and 32 Cd atoms distributed in different sites as follows: 16 Cu atoms in the inner and outer tetrahedral sites, the remaining 4 Cu and 32 Cd atoms distributed in the octahedral and distorted cubo- octahedral position, respectively. To simplify the sublattice model, (Cu,Cd)5(Cd,Cu)8 was used to describe δ-phase.
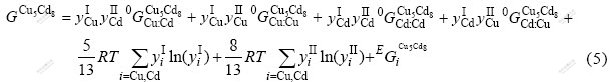
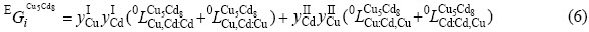
where 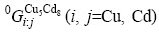 represents the Gibbs free energy of the corresponding compounds in the Cu5Cd8 structure.
represents the Gibbs free energy of the corresponding compounds in the Cu5Cd8 structure. 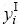 and
and 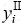 are the site fractions of component i (i=Cu, Cd) in the first and second sublattices.
are the site fractions of component i (i=Cu, Cd) in the first and second sublattices. 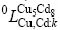 and
and 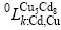 (k represents Cu or Cd) are the interaction parameters between Cu and Cd in the first and the second sublattices. In order to reduce the number of parameters in the Gibbs energy expression, it is supposed that
(k represents Cu or Cd) are the interaction parameters between Cu and Cd in the first and the second sublattices. In order to reduce the number of parameters in the Gibbs energy expression, it is supposed that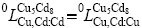 and
and 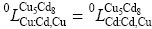 in the present assessment.
in the present assessment.
3.3 Stoichimetric compounds
The β(Cu2Cd) phase was described as a stoichiometric compound because it possesses no composition range. Also, the intermetallic phases, γ(Cu4Cd3) and ε(Cu3Cd10) are treated as stoichiometric compounds due to their small homogeneity range. According to Neumann-Kopp rule, the Gibbs free energy of each compound Cu1-xCdx is formulated as
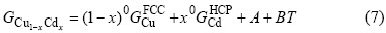
where 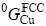 and
and 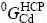 are the Gibbs energies of elements Cu and Cd at their respective standard states, which are taken from Dinsdale[21]. A and B can be evaluated using thermodynamic and phase diagram data.
are the Gibbs energies of elements Cu and Cd at their respective standard states, which are taken from Dinsdale[21]. A and B can be evaluated using thermodynamic and phase diagram data.
4 Results and discussion
Using the lattice stability parameters of the element Cu and Cd cited from Dinsdale[21], the phase diagram of the Cu-Cd binary system was optimized by Thermo-calc software developed by Sundman et al [25]. The phase diagram and thermochemical data were used as input to the program for the optimization. Each piece of selected information was given a certain mass by personal judgment, and verified by trial and error method during the assessment, until most of the selected experimental information was reproduced within the selected uncertainty limits.
Firstly, the thermodynamic parameters for the liquid phase were optimized by using the mixing enthalpy of liquid and the activities of Cd in liquid, then the parameters of the intermetallic compounds and the terminal solution phases were optimized by using phase diagram data. All the parameters were finally optimized simultaneously to give a reasonable description of this system. The same set of parameters was verified with Pandat software[26], and it is found that the calculated results by Pandat software are in good agreement with those by Thermo-Calc. All the evaluated parameters are listed in Table 3.
The calculated phase diagram of the Cu-Cd system
compared with experimental data is illustrated in Fig.2. It is clear that the calculated results are in good agreement with experimental data reported by Subramanian et al[4]. Table 2 lists the invariant reactions in the Cu-Cd system. An agreement within 5 K between the calculated and experimental temperatures has been obtained. As shown in Fig.3, the calculated mixing enthalpies of liquid at 723 K agree well with the experimental data.
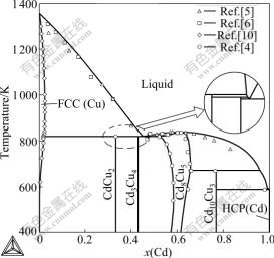
Fig.2 Calculated Cu-Cd phase diagram together with experimental data[4-6, 10]
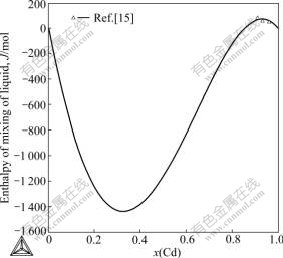
Fig.3 Enthalpies of mixing of liquid Cu-Cd alloy at 723 K (Reference states are liquid components[15])
Table 3 Thermodynamic parameters of Cu-Cd system
Fig.4 shows the calculated activities of Cd in liquid at 873 K together with the experimental data, and they are in good agreement with each other. Enthalpies of formation of intermetallic alloys at 298 K are shown in Fig.5.
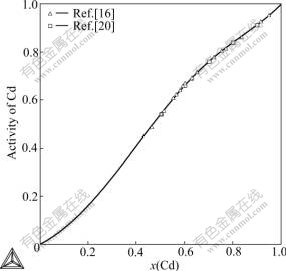
Fig.4 Calculated activity of Cd in liquid Cu-Cd alloys at 873 K compared with experimental data referred to liquid Cd[16, 20]
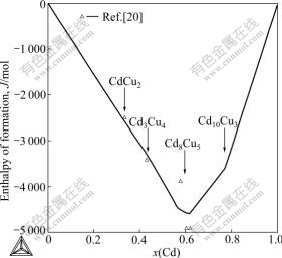
Fig.5 Enthalpies of formation of intermetallic alloys at 298 K compared with experimental data referred to FCC(Cu) and HCP(Cd)[20]
5 Conclusions
1) On the basis of the reported experimental data, using the CALPHAD method, a self-consistent thermodynamic description describing all the phases of Cu-Cd system is obtained.
2) Extensive comparisons between the calculated and experimental data are presented. The present description gives a good agreement with most of the experimental data.
References
[1] WANG S Q, CHEN Z Q, PENG D L, AN G Y. A comprehensive survey of high conductivity high strength copper based alloys [J]. China Academic Journal, 1995, 7: 3-6. (in Chinese)
[2] LI R X, LI R D, GUAN H R, HU Z Q. Effect of Cd addition on the aging process of Al-Si-Cu-Mg cast alloy [J]. Acta Metallurgica Sinica, 2004, 40(8): 867-872. (in Chinese)
[3] HANSEN M, ANDERKO K. Constitution of binary alloys [M]. New York: McGraw-Hill Book Comp, 1958.
[4] SUBRAMANIAN P R, LAUGHLIN D E. The Cd-Cu (cadmium- copper) system [J]. Bull Alloy Phase Diagrams, 1990, 11: 160-169.
[5] SAHMEN R. On copper-cadmium alloys [J]. Z Anorg Chem, 1906, 49: 301-310. (in German)
[6] JENKINS C H M, HANSON D. The constitution of the alloys of copper and cadmium [J]. J Inst Met, 1924, 31: 257-273.
[7] HEYCOCK C T, NEVILLE F H. On the lowering of the freezing points of cadmium, bismuth, and lead when alloyed with other metals [J]. J Chem Soc, 1892, 61: 888-914.
[8] OEEN E A, PICKUP L. X-ray study of copper-cadmium alloys [J]. Proc R Soc, 1933, A139: 526-541.
[9] RAUB E. Primary binary solid solution [J]. Z Metallkd, 1947, 2: 119-120. (in German)
[10] SULONEN M S. Reinvestigations of the copper-cadmium alloy system: The alpha phase boundary and the crystal structure of Cu2Cd phase [J]. Acta Polytech Scand, 1962, 331(18): 1-22.
[11] GLAZOV V M, PAVLOVA I M, KACHAKHIDZE V V. Investigation and analysis of the retrograde solidus curve in the copper-cadmium system [J]. Sov Non-Ferrous Met Res, 1980, 9(2): 170-174. (in Russian)
[12] POGODIN S A, MIKHEEVA V I, KAGAN G A. α-phase limits in the system copper-cadmium [J]. Izv Inst Fiz-Khim Anal, 1935, 7: 39-47. (in Russian)
[13] Tammann G, Heinzel A. Determination of the solubility curve of mixed crystals at small concentrations and the redistribution of the boundary substance by heating [J]. Z Anorg Chem, 1928, 176: 147-151. (in German)
[14] Samson S. The crystal structure of the intermetallic compound Cu4Cd3 [J]. Acta Crystallogr, 1967, 23: 586-600.
[15] KLEPPA O J. Heat of formation of solid and liquid binary alloys of copper with cadmium, indium, tin, and antimony at 450 ℃ [J]. J Phys Chem, 1956, 60: 852-863.
[16] Nikolskaya A V, Otopkov P P, Gerasimov y I. The investigation of thermodynamic properties of binary metallic systems by electromotive force method (Ⅱ): The copper-cadmium system [J]. Zh Fiz Khim, 1957, 31: 1007-1012. (in Russian)
[17] Riccoboni L, Genta V, Fiorani M, Valenti V. Thermodynamic studies of metallic systems (Ⅰ): The liquid system copper-cadmium [J]. Gazz Chim Ital, 1954, 84: 982-1012.
[18] Jellinek K, Rosner G A. The vapor pressure and the activity of a volatile component in binary alloys at high temperatures [J]. Z Phys Chem, 1931, 152: 67-94. (in German)
[19] Azakami T, Yazawa A. Thermodynamic studies of liquid copper alloys (Ⅲ): Activities of zinc and cadmium in liquid copper-base alloys [J]. J Min Metall Inst Jpn, 1968, 84: 1663-1668. (in Japanese)
[20] Hultgren R, Desai P D, Hawkins D T, Gleiser M, Kelley K K. Selected values of the thermodynamic properties of binary alloys [M]. Ohio: Metals Park, 1973.
[21] Dinsdale A T. SGTE data for pure elements [J]. Calphad, 1991, 15: 317-425.
[22] Bradley A J, Gregory C H. A comparison of the crystal structures of Cu5Zn8 and Cu5Cd8 [J]. Philos Mag, 1931, 12: 143-162.
[23] von Heidenstam O, Johansson A, Westman S. A redetermination of the distribution of atoms in Cu5Zn8, Cu5Cd8, and Cu9Al4 [J]. Acta Chem Scand, 1968, 22(2): 653-661.
[24] Brandon J K, Brizard R Y, Chieh P C, McMillan R K, Pearson W B. New refinements of the γ brass type structures Cu5Zn8, Cu5Cd8, and Fe3Zn10 [J]. Acta Crystallogr B, 1974, 30: 1412-1417.
[25] Sundman B, Jansson B, Andersson J O. The program for optimization [J]. Calphad, 1985, 9: 153-190.
[26] PANDAT software for muticomponent phase diagram calculations by CompuTherm, LLC, Madison, WI, 2000.
Corresponding author: LIU Li-bin; Tel: +86-731-88877732; Fax: +86-731-88876692; E-mail: PDC@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(09)60193-X
(Edited by LI Xiang-qun)