Trans. Nonferrous Met. Soc. China 22(2012) s157-s161
Electrochemical performance of LiVPO4F/C synthesized by different methods
LIU Jie-qun1, ZHONG Sheng-kui1, 2, WU Ling1, WAN Kang1,  Fan1
Fan1
1. Shagang School of Iron and Steel, Soochow University, Suzhou 215021, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China
Received 9 July 2012; accepted 6 August 2012
Abstract: LiVPO4F/C samples were synthesized by one-step solid-state reaction and two-step solid-state reaction methods, respectively. The X-ray diffraction (XRD), scanning electron microscopy (SEM) and electrochemical performance tests were adopted to characterize the properties of LiVPO4F/C. XRD results show that the LiVPO4F/C samples prepared by one-step solid-state reaction method have the same triclinic structure (space group 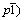 as that synthesized by conventional two-step solid-state reaction. SEM image exhibits that the particle size of LiVPO4F/C prepared by one-step solid-state reaction method is smaller than that of the sample synthesized by two-step solid-state reaction. The improved electrochemical properties of the LiVPO4F/C are attributed to the depressed grain size and enhanced electrical conductivity produced via one-step solid-state reaction method using oxalic acid as both reduction agent and carbon sources. AC impedance measurements also show that the LiVPO4F/C synthesized by one-step solid-state reaction route significantly decreases the charge-transfer resistance.
as that synthesized by conventional two-step solid-state reaction. SEM image exhibits that the particle size of LiVPO4F/C prepared by one-step solid-state reaction method is smaller than that of the sample synthesized by two-step solid-state reaction. The improved electrochemical properties of the LiVPO4F/C are attributed to the depressed grain size and enhanced electrical conductivity produced via one-step solid-state reaction method using oxalic acid as both reduction agent and carbon sources. AC impedance measurements also show that the LiVPO4F/C synthesized by one-step solid-state reaction route significantly decreases the charge-transfer resistance.
Key words: lithium-ion batteries; cathode material; LiVPO4F; one-step solid-state reaction; two-step solid-state reaction
1 Introduction
Rechargeable lithium-ion batteries are considered one of the most advanced energy storage systems. However, the development of lithium-ion batteries cannot keep up with the fast development of the current information-rich mobile society. Therefore, more efforts have been made to explore novel Li+ insertion materials for both cathodes and anodes [1-3]. In commercial lithium-ion batteries, cobalt-based oxide is utilized as the cathode material [4], but its high cost hinders the large-scale use. Recently, lithiated transition metal phosphates, such as Li3V2(PO4)3 [5-9], LiVPO4F [10-13] and LiFePO4 [14-16], have been proposed as a new class of cathode materials for lithium-ion batteries. Among those, LiVPO4F, proposed by BARKER et al [10], is considered a novel 4 V cathode material to substitute LiCoO2 in lithium ion batteries, owing to its good electrochemical performance, high voltage platform, high energy density and excellent thermal stability. It is isostructural with the naturally-occurring mineral tavorite, LiFePO4��OH or ambylgonite, LiAlPO4F, crystallizing with a triclinic structure (space group 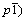 [11]. The reversible Li extraction/insertion reaction for Li1-xVPO4F, based on the V3+/V4+ redox couple, operates at about 4.2 V (vs Li/Li+). However, the conductivity of lithiated transition metal phosphates is not so good. To improve the conductivity of lithiated transition metal phosphates, an effective way is to decrease the particle size. The reduction of particle size may shorten the diffusion path of Li ions, and accordingly enhance the electrochemical performance.
[11]. The reversible Li extraction/insertion reaction for Li1-xVPO4F, based on the V3+/V4+ redox couple, operates at about 4.2 V (vs Li/Li+). However, the conductivity of lithiated transition metal phosphates is not so good. To improve the conductivity of lithiated transition metal phosphates, an effective way is to decrease the particle size. The reduction of particle size may shorten the diffusion path of Li ions, and accordingly enhance the electrochemical performance.
Usually, the LiVPO4F is synthesized by two-step solid-state reaction [10-12] and sol-gel method [13]. The two-step solid-state reaction method is complicated because of the preparation of VPO4 intermediate, which is used to react with LiF to produce the final LiVPO4F. Moreover, it is really hard to get the LiVPO4F with high purity by two-step solid-state reaction route. It is also found that the particle size of LiVPO4F synthesized by this method is relatively large and ununiform, which results in the poor electrochemical performance of the material. Though the particle size of the LiVPO4F synthesized by sol-gel method is small and the electrochemical performance is good, the synthesis needs high cost and long time, leading to the difficulty for the realization of industrialized production [13]. In this work, LiVPO4F was synthesized by a novel one-step solid-state reaction method using oxalic acid as both reduction agent and carbon sources. Compared with the two-step solid-state reaction method and sol-gel method, this method is superior in the simple synthesis process and industrialized production. In addition, the raw material oxalic acid will decompose during the heat treatment, which can restrict the growth of LiVPO4F particles, and consequently enhance the electrochemical performance.
2 Experimental
LiVPO4F/C samples were synthesized by one-step solid-state reaction method. Firstly, appropriate amounts of V2O5, NH4H2PO4, LiF and oxalic acid were initially ground in a mortar and then thoroughly mixed by ball milling for 4 h. Finally, the precursor was calcined at 600 ��C for 2 h to yield LiVPO4F/C. To avoid the oxidation of vanadium, the process was carried out under flowing Ar atmosphere.
LiVPO4F/C samples were also synthesized by two-step solid-state reaction method [12]. It followed two main processes. Firstly, appropriate amounts of V2O5, NH4H2PO4 and acetylene black were ground in a mortar and then thoroughly mixed by ball milling for 4 h. The mixed precursor was decomposed at 300 ��C for 4 h to remove NH3 and H2O, and then calcined at 750 ��C for 6 h to obtain VPO4. Secondly, appropriate amounts of VPO4 and LiF were thoroughly mixed by ball milling for 2 h, and calcined at 750 ��C for 30 min to yield the LiVPO4F material. To avoid the oxidation of vanadium, each step of the process was carried out under flowing Ar atmosphere. The samples synthesized by two-step solid-state reaction and one-step solid-state reaction are abbreviated to LiVPO4F/C(T) and LiVPO4F/C(O), respectively.
The powder X-ray diffraction (XRD, Rint-2000, Rigaku) measurement using Cu K�� radiation was employed and recorded at room temperature to identify the crystalline phase of the synthesized materials. The particle size and morphology of the LiVPO4F powders were observed by scanning electron microscope (JEOL, JSM-5600LV) with an accelerating voltage of 20 kV. The carbon contents of samples were determined by a carbon-sulfur analyser (Mlti EA2000).
Charge/discharge tests were performed using CR2025 coin-type cell. The cells were charged and discharged over a voltage range from 3.0 to 4.4 V (vs Li/Li+) at room temperature. The cyclic voltammetric (CV) measurements were carried out with a CHI 600A electrochemical analyzer. The CV curves for the above test cells were recorded in the potential range of 3.0-4.9 V at a scanning rate of 0.1 mV/s. The EIS spectra were recorded by applying an AC voltage of 5 mV amplitude in the frequency range of 0.01 Hz-10 kHz. The CV and EIS experiments were performed in a three-electrode system using metallic foils as both counter and reference electrode.
3 Results and discussion
In the one-step solid-state route, oxalic acid is selected as a reductant and a carbon source, which can reduce the V5+ to V3+ during the heat treatment. Figure 1 shows the XRD patterns of the prepared LiVPO4F/C(T) and LiVPO4F/C(O). It is evident that all fundamental peaks can be indexed to the triclinic structure [11] with space group 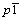 (JCPDS card No.42-1412) for both samples. In addition, the XRD patterns of both samples are similar, indicating that different synthesis routes do not affect the crystalline phase. The mass fraction of carbon in LiVPO4F/C(T) and LiVPO4F/C(O) are 1.83% and 1.95%, respectively. However, no diffraction peaks are observed for the crystalline carbon, indicating that carbon in the samples is amorphous or the carbon amount is too small.
(JCPDS card No.42-1412) for both samples. In addition, the XRD patterns of both samples are similar, indicating that different synthesis routes do not affect the crystalline phase. The mass fraction of carbon in LiVPO4F/C(T) and LiVPO4F/C(O) are 1.83% and 1.95%, respectively. However, no diffraction peaks are observed for the crystalline carbon, indicating that carbon in the samples is amorphous or the carbon amount is too small.
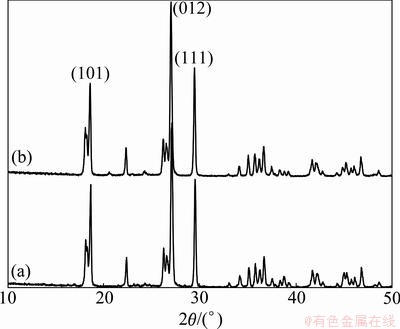
Fig. 1 XRD patterns of LiVPO4F/C(O) (a) and LiVPO4F/C(T) (b)

Fig. 2 SEM images of LiVPO4F/C(O) (a) and LiVPO4F/C(T) (b)
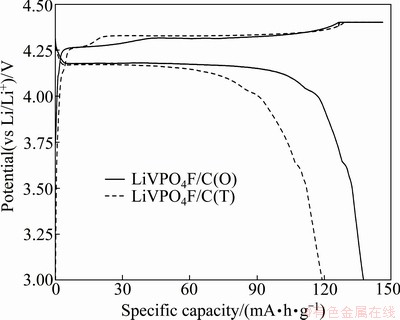
Fig. 3 First charge-discharge curves of LiVPO4F/C(O) and LiVPO4F/C(T) at rate of 0.2C
SEM images of LiVPO4F/C(T) and LiVPO4F/C(O) are shown in Fig. 2. It is obvious that the particle size of the two particles are different. LiVPO4F/C(T) consists of large and ununiform particles. Contrast to LiVPO4F/C(T), LiVPO4F/C(O) shows small particle in the size of about 200 nm and a narrow size distribution. This indicates that the one-step solid-state reaction may inhibit the particle growth during the sintering process. Reducing the particle size can shorten the diffusion path of lithium ions, and consequently improve the electrochemical performance of LiVPO4F/C.
The first charge-discharge curves of LiVPO4F/C(T) and LiVPO4F/C(O) between 3.0 V and 4.4 V at the rate of 0.2C are shown in Fig. 3. For both the samples, the shapes of charge-discharge curves are similar. However, the initial capacities of LiVPO4F/C(T) and LiVPO4F/ C(O) are obviously different. The first charge and discharge capacities of LiVPO4F/C(T) are 140 and 119 mA��h/g, respectively, showing that the columbic efficiency is 85.0%. While, the capacities of LiVPO4F/ C(O) are 146 and 138 mA��h/g, respectively, and the columbic efficiency is 94.5%. Obviously, the initial charge-discharge performance of LiVPO4F/C(T) is worse than that of LiVPO4F/C(O).
The cycling performances of LiVPO4F/C(T) and LiVPO4F/C(O) were evaluated in the Li/LiVPO4F cell configuration in the voltage range of 3.0-4.4 V at room temperature. Figure 4 shows the cyclic discharge profiles for the LiVPO4F cathode materials at different rates. After 30 cycles, the LiVPO4F/C(T) sample exhibits the discharge capacities of 89, 79 and 74 mA��h/g at 0.2C, 0.5C and 1.0C rates, with the capacity losses of 25.20%, 33.61%, and 37.82%, respectively. However, under the same condition, the LiVPO4F/C(O) sample delivers discharge capacities of 132, 129 and 124 mA��h/g, respectively. Namely, the capacity loss is 4.35% at 0.2C, 6.52% at 0.5C, and 10.14% at 1.0C. From the above results, it can be concluded that the LiVPO4F/C(O) sample synthesized by one-step solid-state reaction has better cycling performance than LiVPO4F/C(T) sample synthesized by two-step solid-state reaction at room temperature.
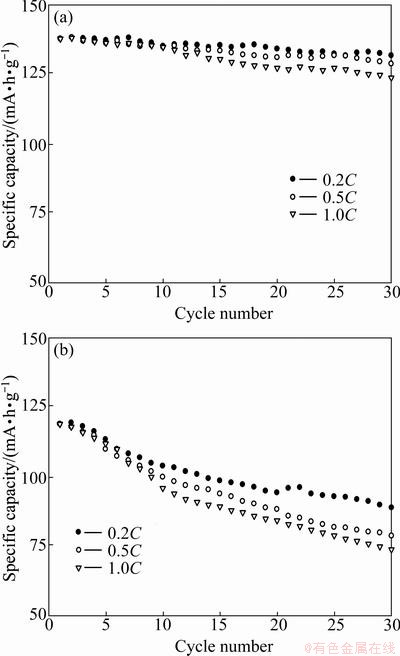
Fig. 4 Cycling performance of LiVPO4F/C(O) (a) and LiVPO4F/C(T) (b) at room temperature
The electrochemical cycling performance of LiVPO4F/C was also tested at 55 ��C in the voltage range of 3.0-4.4 V at different rates. As shown in Fig. 5, the LiVPO4F/C(T) sample has the capacity loss of about 23.52% at 0.2C, 31.09% at 0.5C, and 36.97% at 1.0C after 30 cycles. In contrast, the LiVPO4F/C(O) sample still exhibits better cyclic performance with the capacity loss of about 3.62%, 5.07%, and 7.25%. It also indicates that the two samples have better cycling performance at 55 ��C than at room temperatures. The improved cycle performance is attributed to the increased diffusion of lithium ions at the elevated temperatures.
The electrochemical impedance spectra (EIS) of LiVPO4F/C(O) and LiVPO4F/C(T) electrodes were measured at different charging states, as presented in Fig. 6. Similar EIS patterns are observed for LiVPO4F/ C(O) and LiVPO4F/C(T) samples. A semicircle is observed to center on the real axis in the high frequency range. In the low frequency range, a straight line with an angle of about 45�� to the real axis corresponds to the Warburg impedance. The high frequency semicircle is related to the charge-transfer resistance (Rct) and the double-layer capacitance. The low frequency tail results from the diffusion of lithium ions in the bulk active mass. In the case of LiVPO4F/C(O), the diameter of the semicircle significantly depends on the potential during charging, indicating that the film formation process is dependent on the lithium ion content. On the other hand, the charge transfer resistance, Rct, shows a greater dependence on the lithium insertion and extraction levels. In the highly charged states, the sample is found to have lower Rct values. Comparing the diameter of semicircles of the above two systems, it can be found that LiVPO4F/ C(O) shows lower Rct value than LiVPO4F/C(T), indicating that the one-step solid-state reaction may increase the electronic conductivity and improve the Li+ kinetic behavior.
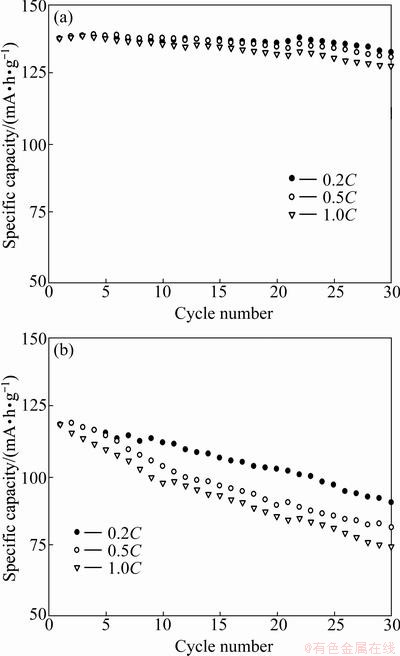
Fig. 5 Cycling performance of LiVPO4F/C(O) (a) and LiVPO4F/C(T) (b) at 55 ��C
The cyclic voltammetry (CV) curves for LiVPO4F/ C(O) and LiVPO4F/C(T) electrodes at a scanning rate of 0.1 mV/s are shown in Fig. 7. The two CV curves are similar, suggesting that the reaction mechanism does not change during the lithium extraction/insertion process. However, the redox peak potentials are different for the two samples. LiVPO4F/C(T) exhibits oxidation and reduction peaks at around 4.48 and 4.09 V, respectively. For LiVPO4F/C(O), the oxidation peak potential decreases to 4.42 V, and the reduction peak potential increases to 4.15 V. Moreover, the potential difference of the redox peaks for LiVPO4F/C(T) is about 0.39 V, whereas that of the LiVPO4F/C(O) decreases to 0.27 V. Therefore, the reversibility of the LiVPO4F/C(O) electrode reaction is better than that of LiVPO4F/C(T).
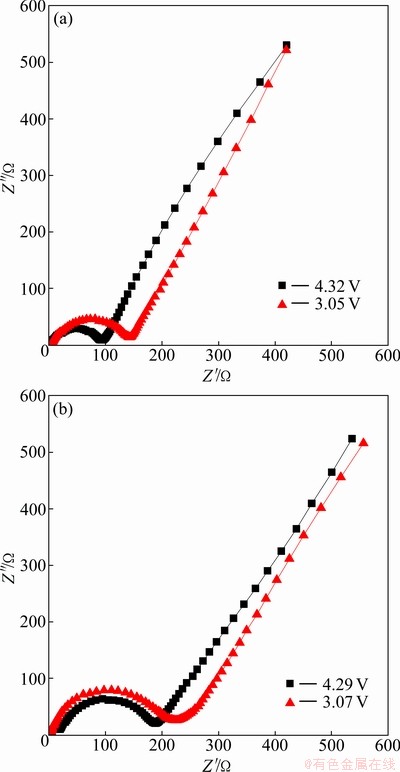
Fig. 6 Nyquist plots of LiVPO4F/C(O) (a) and LiVPO4F/C(T) (b) at different charging and discharging states
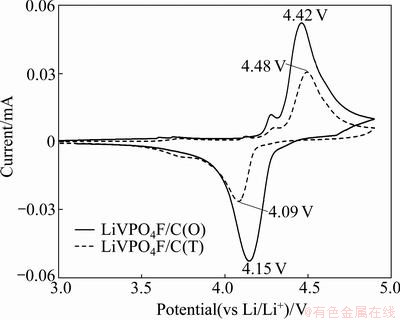
Fig. 7 Cyclic voltammetry curves of LiVPO4F/C(T) and LiVPO4F/C(O) electrodes in the first cycle
4 Conclusions
1) The grains of LiVPO4F/C synthesized through one-step solid-state route are significantly small. The LiVPO4F/C(O) sample exhibits a much higher discharge capacity and lower capacity loss than LiVPO4F/C(T) after 30 cycles. It is confirmed that the sample prepared by one-step solid-state route show smaller resistances of the lithium-ion migration and charge transfer. The improved electrochemical properties of the LiVPO4F/C can be attributed to the depressed grain size and enhanced electronic conductivity produced via one-step solid-state reaction method using oxalic acid as both reduction agent and carbon sources.
2) One-step solid-state route proposed in this investigation must increase the possibility of the application LiVPO4F/C to commercial lithium-ion batteries.
References
[1] TARASCON J M, ARMAND M. Issues and challenges facing rechargeable lithium batteries [J]. Nature, 2001, 414: 359-367.
[2] ZHOU Z, ZHAO J J, GAO X P, CHEN Z F, YAN J, SCHLEYER P V, MORINAGA M. Do composite single-walled nanotubes have enhanced capability for lithium storage [J]. Chem Mater, 2005, 17(5): 992-1000.
[3] PU W H, HE X M, REN J G, WAN C R, JIANG C Y. Electrodeposition of Sn-Cu alloy anodes for lithium batteries [J]. Electrochimica Acta, 2005, 50(25): 4140-4145.
[4] KERR T, GAUBICHER J, NAZAR L F. Highly reversible Li insertion at 4 V in epsilon-VOPO4/alpha-LiVOPO4 cathodes [J]. Electrochem Solid-State Lett, 2000, 3(10): 460-462.
[5] BARKER J, SAIDI M Y. Lithium-containing phosphates, method of preparation, and use thereof. US: 005871866A [P]. 1999.
[6] LI Yu-zhan, LIU Xin, YAN Jie. Study on synthesis routes and their influences on chemical and electrochemical performances of Li3V2(PO4)3/carbon [J]. Electrochim Acta, 2007, 53(2): 474-479.
[7] WANG Li-juan, ZHOU Xue-chou, GUO Yong-lang. Synthesis and performance of carbon-coated Li3V2(PO4)3 cathode materials by a low temperature solid-state reaction [J]. J Power Sources, 2010, 195(9): 2844-2850.
[8] KUANG Quan, ZHAO Yan-ming, AN Xiao-ning, LIU Jian-min, DONG You-zhong, CHEN Ling. Synthesis and electrochemical properties of Co-doped Li3V2(PO4)3 cathode materials for lithium-ion batteries [J]. Electrochimica Acta, 2010, 55(5): 1575-1581.
[9] RUI X H, LI C, CHEN C H. Synthesis and characterization of carbon-coated Li3V2(PO4)3 cathode materials with different carbon sources [J]. Electrochimica Acta, 2009, 54(12): 3374-3380.
[10] BARKER J, SAIDI M Y, GOVER R K B, BURNS P, BRYAN A. The effect of Al substitution on the lithium insertion properties of lithium vanadium fluorophosphate, LiVPO4F [J]. J Power Sources, 2007, 174(2): 927-931.
[11] BARKER J, SAIDI M Y, SWOYER J L. Electrochemical insertion properties of the novel lithium vanadium fluorophosphates [J]. J Electrochem Soc, 2007, 150(10): 1394-1398.
[12] ZHONG Sheng-kui, YIN Zhou-lan, WANG Zhi-xing, CHEN Qi-yuan. Synthesis and characterization of the triclinic structural LiVPO4F as possible 4.2 V cathode materials for lithium batteries [J]. J Cent South Univ Techno, 2007, 14(3): 340-343.
[13] LI Yu-zhan, ZHOU Zhen, GAO Xiao-ping, YAN Jie. A novel sol�Cgel method to synthesize nanocrystalline LiVPO4F and its electrochemical Li intercalation performances [J]. J Power Sources, 2006, 160(1): 633-637.
[14] JIN B, JIN E M, HEE K, GU H B. Electrochemical properties of LiFePO4-multiwalled carbon nanotubes composite cathode materials for lithium polymer battery [J]. Electrochem Commun, 2008, 10(10): 1537-1540.
[15] MI C H, ZHANG X G, ZHAO X B. Effect of sintering time on the physical and electrochemical properties of LiFePO4/C composite cathodes [J]. J Alloys Compd, 2006, 424(1/2): 327-333.
[16] YIN Yan-hong, LI Shao-yu, YAN Lin-lin, ZHANG Hui-shuang, YANG Shu-ting. Modified carbothermal reduction method for synthesis of LiFePO4/C composite [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(3): 621-626.
��ͬ�����ϳɵ�LiVPO4F/C�ĵ绯ѧ����
����Ⱥ1����ʤ��1, 2���� ��1���� ��1���� ��1
1. ���ݴ�ѧ ɳ�ָ���ѧԺ������ 215021��
2. ���ϴ�ѧ ��ѧ����ѧԺ����ɳ 410083
ժ Ҫ��ͨ��һ�����෴Ӧ���������෴Ӧ�ֱ�ϳ�LiVPO4F/C������XRD��SEM�͵绯ѧ���ܲ��Զ�LiVPO4F/C�������ܱ�����XRD�о�����һ�����෴Ӧ�ϳɵ�LiVPO4F/C���������෴Ӧ�ϳɵ���Ʒһ������������б��ϵ�ṹ��SEM�о�������һ�����෴Ӧ�ϳɵ�LiVPO4F/C�������������෴Ӧ�ϳɵ���Ʒ����С��һ�����෴Ӧ�ϳɵ�LiVPO4F/C��Ʒ�绯ѧ���ܵõ���������ڲ�����Ϊ��ԭ����̼Դ�ϳɵ���Ʒ������С�������迹�о����������෴Ӧ�ϳɵ�LiVPO4F/C��Ʒ�绯ѧ�迹��С��
�ؼ��ʣ�����ӵ�أ��������ϣ�LiVPO4F��һ�����෨���������෨
(Edited by LONG Huai-zhong)
Foundation item: Project (51164007) supported by the National Natural Science Foundation of China; Project (2011M501288) supported by China Postdoctoral Science Foundation
Corresponding author: ZHONG Sheng-kui; Tel: +86-512-67160032; E-mail: zhongshengkui@suda.edu.cn