高硫铝土矿中硫的赋存状态及除硫
来源期刊:中国有色金属学报(英文版)2011年第7期
论文作者:胡小莲 陈文汨 谢巧玲
文章页码:1641 - 1647
关键词:高硫铝土矿;硫化物相;氧化焙烧;碳酸钠洗涤
Key words:high sulfur-containing bauxite; sulfur phase; oxidizing-roasting; sodium carbonate solution washing technology
摘 要:
采用X射线衍射分析和化学物相定量法研究高硫铝土矿中硫的赋存状态,并对不同形态的硫的脱除方法进行研究。结果表明:高硫铝土矿中硫的存在形态有的以硫化物为主(主要是黄铁矿),有的以硫酸盐为主,不同产地的铝土矿中硫的主要存在形态是不同的,进一步补充了铝土矿中硫的物相存在的规律性。采用X射线衍射结合化学物相定量法能更准确确定高硫铝土矿中硫的存在形态。确定高硫铝土矿中硫的主要存在形态对除硫方法的选择具有理论指导意义。不同形态的硫可以采用不同的除硫方法。采用氧化焙烧能有效脱除高硫铝土矿中硫化物型硫,原矿溶出液中S2?的含量均在1.7 g/L以上,而焙烧矿的溶出液中S2?的含量均在0.18 g/L以下。通过N2CO3溶液洗涤可以有效脱除矿石中硫酸盐型硫,原矿中全硫含量下降到0.2%以下,符合生产中对矿石中硫含量的要求。
Abstract:
The sulfur phase in high sulfur-containing bauxite was studied by an X-ray diffraction analysis and a chemistry quantitative analysis. The methods for the removal of different shaped sulfur were also discussed. The results show that sulfur phases in high sulfur-containing bauxites exist in the main form of sulfide sulfur (pyrite) or sulfate sulfur, and the main sulfur forms of bauxites from different regions are not the same. Through a combination of an X-ray diffraction analysis and a chemistry quantitative analysis, the sulfur phases of high sulfur-containing bauxite could be accurately investigated. Deciding the main sulfur form of high sulfur-containing bauxite could provide theoretical instruction for choosing methods for the removal of sulfur from bauxite, and an oxidizing-roasting process is an effective way to remove sulfide sulfur from high sulfur-containing bauxite, the content of S2? in crude ore in the digestion liquor is above 1.7 g/L, but in the roasted ore digestion liquor, it is below 0.18 g/L. Using the sodium carbonate solution washing technology to wash bauxite can effectively remove sulfate sulfur, the content of the total sulfur in ore is lowered to below 0.2% and can meet the production requirements for the sulfur content.

HU Xiao-lian1, 2, CHEN Wen-mi1, XIE Qiao-ling1
1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Chemistry and Chemical Engineering, Hunan University of Science and Technology,
Xiangtan 411201, China
Received 4 August 2010; accepted 3 November 2010
Abstract: The sulfur phase in high sulfur-containing bauxite was studied by an X-ray diffraction analysis and a chemistry quantitative analysis. The methods for the removal of different shaped sulfur were also discussed. The results show that sulfur phases in high sulfur-containing bauxites exist in the main form of sulfide sulfur (pyrite) or sulfate sulfur, and the main sulfur forms of bauxites from different regions are not the same. Through a combination of an X-ray diffraction analysis and a chemistry quantitative analysis, the sulfur phases of high sulfur-containing bauxite could be accurately investigated. Deciding the main sulfur form of high sulfur-containing bauxite could provide theoretical instruction for choosing methods for the removal of sulfur from bauxite, and an oxidizing-roasting process is an effective way to remove sulfide sulfur from high sulfur-containing bauxite, the content of S2- in crude ore in the digestion liquor is above 1.7 g/L, but in the roasted ore digestion liquor, it is below 0.18 g/L. Using the sodium carbonate solution washing technology to wash bauxite can effectively remove sulfate sulfur, the content of the total sulfur in ore is lowered to below 0.2% and can meet the production requirements for the sulfur content.
Key words: high sulfur-containing bauxite; sulfur phase; oxidizing-roasting; sodium carbonate solution washing technology
1 Introduction
China is rich in bauxite, the reserve of which has been proved to reach 2.3×109 t [1]. The amount of high sulfur-containing diasporic bauxite has reached 1.5×108 t [2]. These ores are mainly composed of aluminum of middle-high proportion, silicon of middle-low proportion, sulphur of high proportion and middle-high Al/Si ratio (A/S) ores. Most of the ores are of high-grade alumina, but they can only be used after the removal of sulfur from the high sulfur-containing bauxite. Therefore,developing an economical and practical method for the removal of the sulfur is of great importance for industry production. Moreover, in the process of alumina production, the sulfur in the ore can cause not only the loss of Na2O, but also leads to the corrosion of steel material and the rise in the iron concentration of the solution as a result of increasing the concentrate of S2- of the solution. For example, when the sulfur content of the bauxite exceeds 0.8%, it will result in the decline of alumina quality for the existence of Fe, the damage of the equipment in the evaporation process and decomposition process for the corrosion of the steel equipment. It can even decrease the digestibility of the alumina [3].
The removal of sulfur from bauxite has attracted considerable attention in recent years with the rapid development of the alumina industry [3-7]. There are two basic processes for extracting alumina from bauxite, namely, the sintering process and the Bayer process. The disadvantage of the sintering process is its low efficiency (low to 33% or less). Due to its low cost, the Bayer process is the most commonly-used method for extracting alumina from bauxite [8-10]. In the Bayer process, the research area of the removal of sulfur from bauxite is mainly the removal of sulfur from sodium aluminate solutions or Bayer liquor. The methods are: 1) the removal of sulfur by means of wet air oxidation and 2) adding a desulfurizing agent. But by former this method problems for attention are that it should avoid generating thiosulfate and increasing corrosion of the equipment during air oxidation, and it also has a certain risk by means of wet air oxidation.
It has been found that the removal of sulfur is achieved by adding a de-sulfurizing agent, which is mainly zinc oxide or barium oxide, to the liquor, but the basic principles of both methods are not the same. In terms of the former process, the removal of sulfur is capable of separating divalent sulfur ions from the liquor by forming insoluble zinc sulfide and the latter concerns the removal of sulfur from the Bayer process liquors by precipitation with barium sulfate. However, in order to improve the pertinent choice of de-sulfurizing agent, the phase of the sulphur should be known first. To resolve this issue, it is necessary to be aware of the phase of the sulphur and its content of bauxite so that the appropriate methods can be chosen for the removal of the sulfur.
So far, studies on the phase of sulfur of high-sulfur bauxite have seldom been reported, and although there are many kinds on the research methods for the phases, the method for the study of the bauxite phase is the use of an X-ray diffraction analysis and a scanning electron microscopy [11-12]. ZHANG et al [11] investigated the existence of sulfur mineral in high-sulfur bauxite from Guizhou Province, China, by an X-ray diffraction analysis and a scanning electron microscopy. Due to the limitations of the instrument, the X-ray diffraction phase analysis method is imperfect in many aspects. Firstly, it mainly analyzes crystal, and is not suitable for the analysis of non-crystal, which means that the diffraction peak of sulfate, which does not crystallize well, cannot be shown in the X-ray diffraction pattern, and cannot demonstrate whether or not sulfate is present in the ore. Secondly, XRD cannot quantitatively and accurately measure the content of sulfur at various phases, and a scanning electron microscopy can only perform a topographical observation and cannot be used as a method of the phase.
Therefore, using seven kinds of high sulfur-containing bauxites from three provinces, namely, Henan province, Guizhou province and Guangxi province, China, by means of both XRD and a chemistry quantitative analysis, the sulfur-presence phase of the ore was shown by X-ray diffractometry, the total sulfur content was measured by chemical analysis, and a quantitative measurement of the content of various phase sulfur was carried out. On the basis of these, the further law of the sulfur phase of high sulfur-containing bauxite was explored. Moreover, different methods for the removal of different phase sulfur were also investigated.
2 Experimental
2.1 High-sulfur bauxite
Experiment materials included seven types of high- sulfur bauxites, namely, Guizhou ores A and B, Henan ores A, B and C, Guangxi ore A and B. Those ores were ground using a vibrating grinding mill. The main chemical compositions of these bauxites are shown in Table 1.
Table 1 Chemical compositions of seven bauxites

It can be seen from Table 1 that the sulfur contents of Henan bauxite samples A, B, C and Guangxi bauxite sample A are relatively high, respectively 0.97%, 0.85%, 0.78% and 2.28%. When the sulfur content of the bauxite exceeds 0.8%, it will result in the decline of the quality of the alumina for the pollution of Fe, the damage of the equipment in evaporation process and the decomposition process, as a result of the corrosion of the steel equipment [3]. In order to meet this specification, it is necessary to remove the sulfur from the high-sulfur bauxite. It can be seen from aluminum silicon ratio that Guizhou ores A, B and Henan ore A belong to the middle-grade bauxites (A/S of 4-7), but Henan ores B, C and Guangxi ore are high-grade bauxites (A/S> 11).
2.2 Pregnant liquor
The pregnant liquor in the digestion experiment came from an aluminate solution of one alumina factory by the Bayer process in China. The main chemical composition is shown in Table 2. It is composed of total alkali (NT), caustic alkali (NK) and Al2O3, and the concentration ratio of Al2O3 to caustic alkali (RP) was 0.61.
Table 2 Chemical composition of aluminate solution
![]()
2.3 Methods
2.3.1 Analysis of main chemical composition of solid phase and ores
The main chemical compositions, Al2O3 and SiO2, of the solid phase and ores were analyzed by a wavelength dispersive-X-ray fluorescence spectrometer, and the total sulfur of bauxite was analyzed by the national standard “GB3257.18―82”, combustion- iodimetry of bauxite chemical analysis method.
2.3.2 Analysis of main chemical composition of liquid phase
After the digestion process was finished, the liquid phase was separated from the solid phase by filtration. The composition of liquid phase Al2O3 was analyzed by an EDTA compleximetry method and the caustic alkali was analyzed by acid-base titration. The concentration of S2- in the aluminate solution was analyzed according to the literature method [13]. The sample was firstly acid-decomposed by adding the SnCl2 hydrochloric acid solution and releasing H2S, and the sulphur of H2S was determined.
2.3.3 Study on phase of sulfur
The study on the sulfur phase of high-sulfur bauxite used two methods, namely, XRD and chemistry phase analysis of sulfur. The sulfur in bauxite mainly exists in the form of pyrite, with maybe a small quantity of sulfate. Therefore, the determination of the sulfur was mainly composed of the determination of the total sulfur, the contents of sulfide sulfur and sulfate sulfur. The analysis of the total sulfur in the bauxite was based on the national standard “GB3257.18―82”, combustion- iodimetry of bauxite chemical analysis method. The sample with a fusing agent was burned at (1 300±20) °C with oxygen, and SO2 was released, which was absorbed into H2SO3 in water. The sulphur of H2SO3 in water was determined by a titration of standard iodine with starch as an indicator.
To determine the content of the sulfate sulfur and sulfide sulfur [14], the ore was washed with 10% sodium carbonate solution, and 10% BaCl2 solution was added to the filtrate after it was separated from the filter cakes by filtration. Then, the barium sulfate was determined by means of barium sulfate gravimetry and was converted into sulfur, which was the content of the sulfate sulfur. The sulfur in the dried filtrate cake, namely, sulfide sulfur was determined by burning iodimetry.
2.3.4 Roasting experiment and digestion experiment
The roasting experiment was carried out in a muffle furnace SX2-5-12, with a temperature control accuracy of ±5 °C. After spreading the powder-like bauxite in a porcelain dish, it was quickly removed into a muffle when the furnace reached the specified temperature, and then quickly taken out and cooled down after pre-determined roasting time to begin to carry out the digestion experiment.
The digestion experiment was carried out on a molten salt furnace, using an automatic temperature control device, with a temperature accuracy of ±1 °C. The digestion experiment temperature was adjusted according to the experimental requirements, the dissolution time was 60 min and the stirring speed was 60 r/min. The mineral powder and the pregnant liquor were placed in a 100 mL-steel bomb, which was sealed in the experiment. When the furnace reached the specified temperature for 5 min, the steel bomb was placed in it and the dissolution process was completed within the predetermined time. When the experiment was over, the sodium aluminate solution was filtered. Then the contents of Al2O3 and SiO2 in the dried filter cake were sampled and analyzed, and the contents of Al2O3, caustic alkali and S2- in filtrate were also analyzed.
3 Results and discussion
3.1 XRD analysis
According to Ref. [15], the sulfur in bauxite is mainly FeS2 and most of it exists in the form of pyrite. Since the content of sulfur in bauxite is very low, generally below 3%, and most of it appears in the form of pyrite, other types of sulfur mineral scatter, such as sulfate, and they are difficult to form crystal. It can be predicted that no sulfate exists in the XRD patterns of bauxite. According to the standard XRD pattern of pyrite, a strong diffraction peak appears at 2θ=33.08°. Moreover, such a diffraction peak does not overlap with the other diffraction peak of the bauxite. In addition, there are some weak peaks, but such weak peaks often overlap with the other diffraction peak of the bauxite. Therefore, it can be basically judged whether or not the sulphur stays in the form of pyrite according to whether or not a diffraction peak of pyrite emerges, and the content of pyrite can be judged according to the intensity of the diffraction peak. The XRD pattern of high sulfur bauxite is shown in Fig. 1.
As shown in Fig. 1, Henan ore A and Guangxi ore A have obvious characteristic diffraction peaks of pyrite crystal at 2θ=33.08° and no other phase of S appears in the XRD pattern. This indicates that the primary phases of sulfur in two types of high-sulfur bauxites are pyrite. In addition, Guizhou ore B only has a weak diffraction peak at 2θ=33.08°, while other ores have no peaks of pyrite crystal at all. This indicates that there is very small content of pyrite in these bauxites. Although Henan bauxites B and C have very high content of total sulphur, respectively 0.78% and 0.85%, there is no obvious characteristic diffraction peaks at 2θ=33.08°, and this may be due to the small content of pyrite. In addition, XRD verifies that these seven bauxites are all diaspore (β-AlOOH) and contain a different amount of titanium-containing minerals, such as TiO2.
3.2 Chemical phase analysis of sulfur
The contents of the total sulfur, sulfide sulfur and sulfate sulfur in high-sulfur bauxite were determined by a chemistry phase analysis of sulfur, and the results are listed in Table 3.
As shown in Table 3, the main existing form of sulfur is sulfide sulfur, which accounts for above 80% of the total sulfur in both the Guangxi bauxite A and Henan bauxite A, and XRD pattern in Fig. 1 verifies that both bauxites have an obvious characteristic diffraction peak of pyrite. However, in Henan bauxites B, C and Guizhou bauxite A, the main existing forms of sulfur are sulfate sulfur which occupies above 70%. Correspondingly, there is almost no diffraction peak of pyrite in XRD patterns. The pyrite content in Guizhou B is very small, and occupies about 40% of the total and there is only a weak diffraction peak in the XRD pattern. The chemistry phase analysis of sulfur is identical to XRD analysis, but it can also be used for a simultaneous quantitative analysis of the content of various form sulfur. The results show the main sulfur phases of the high-sulfur bauxites from different areas are not the same, since some ores stayed in sulfide sulfur (pyrite), and some in sulfate sulfur, and some in both. Such research results of the sulfur phases of the high-sulfur bauxites are further supplements for the theory “sulfur of bauxite exists in the main form of iron sulfide FeS2, mainly pyrite” [15]. By combining X-ray diffraction with a quantitative chemical phase, the sulfur phase of high-sulfur bauxite can be accurately investigated.
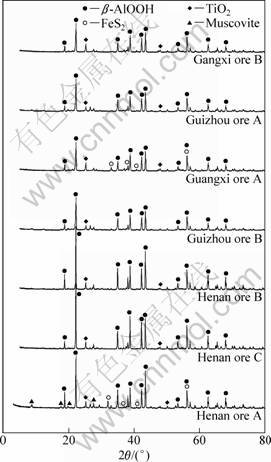
Fig. 1 X-ray diffraction patterns of high-sulfur bauxite
Table 3 Sulfur phase analytic results of high sulfur bauxite

According to Ref. [16], the weathering of a bauxite surface layer which leads to the occurrence of a physical chemistry change of pyrite may initially be the primary pathway for the sulfate formation of the ore, but suitable methods for the removal of sulfur can be chosen by ascertaining the main existing form of sulfur.
3.3 Instructional role of sulfur phase analysis for choosing methods for removal of sulfur
3.3.1 Removal of sulfide sulfur
In the process of alumina production, the harm caused by the sulfur of different existing forms is not the same. ABLAMOFF [15] believed that the main harm of sulfur is the decline of the alumina quality for the pollution of Fe. Under a high temperature and high concentration of Na2O, the dissolution rate and equilibrium concentration of iron greatly increased the content of sodium sulfide in Bayer solution, and hydrated sodium thiosulfate Na2[FeS2(OH)2・2H2O] was formed. Sodium sulfide is formed under a condition of high temperature and a high alkali solution. Therefore, the removal of sulfide sulfur was mainly studied. The removal of pyrite from bauxite was achieved by oxidation roasting. According to Ref. [17] FeS2 can evoke a series of reactions during roasting in an oxygen atmosphere:
(1-x)FeS2+(1-2x)O2=Fe1-xS+(1-2x)SO2 (1)
2Fe1-xS+(3-x)O2=2(1-x)FeO+2SO2 (2)
3FeO+1/2O2=Fe3O4 (3)
2FeS2=2FeS+S2 (4)
S+2O2=2SO2 (5)
4FeS+7O2=2Fe2O3+4SO2 (6)
Fe3O4 and FeO were oxidized to form Fe2O3, and in addition, some of the SO2 produced by the reaction was dispersed in the air, and some reacted with alkalinity in the ore to form sulfate. In order to explore the mechanism of roasting, the XRD analysis of ore roasted at 600 °C for 60 min was carried out, and the results are shown in Fig. 2.
As shown in Fig. 2, the disappearance of the characteristic diffraction peak of pyrite and the emergence of the Fe2O3 peak are in contrast with Figs. 1 and 2, which indicates that pyrite reacted to form Fe2O3 during the roasting. Therefore, roasting could lower not only the total sulfur of ore obviously, but also the sulfide sulfur.
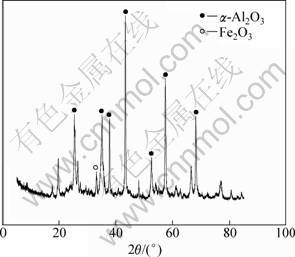
Fig. 2 X-ray diffraction pattern of roasted Henan bauxite
Moreover, by comparing Fig. 2 and Fig. 1, it can be seen that the diffraction peak of diasporite β-AlOOH disappears, and a peak of α-Al2O3 appears. The formula of the phase transition can be expressed as follows:
β-AlOOH→α-Al2O3+H2O (7)
According to Ref. [12], the crystal phase of diasporite does not change when baked at low temperatures below 500 °C. At 500 °C, part of the diasporite changes to α-Al2O3. At 600 °C, the diasporite alumina β-AlOOH completely changes to α-Al2O3. The crystal cell makes a transition from octahedral Al(O,OH)6- of orthogonal crystal system to the octahedral AlO6- of trigonal system, namely, perfect crystalline diasporite β-AlOOH transforms into incomplete crystalline corundum (α-Al2O3). Since such incomplete crystalline corundum (α-Al2O3) activities are better than those of the diasporite, it can be confirmed that the dissolution rate of roasted ore at 600 °C is higher than that of crude ore.
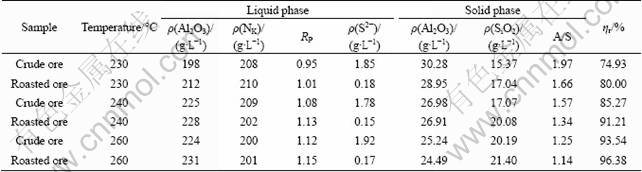
In order to verify the effect of roasting and determine whether or not the digestibility was affected by roasting, a digestion experiment was carried out using roasted ore. Henan high sulfur-containing bauxite A was firstly roasted in the muffle roaster at 600 °C for 60 min, and then the digestion experiment was simultaneously carried out in a molten salt furnace by the Bayer process for both the crude ore and the roasted ore at dissolution temperatures of 230, 240, 260 °C for dissolution time of 60 min. The aluminate solution is shown in Table 2. After the experiment, the divalent sulfur ion (S2-) concentration was analyzed, and according to results, the extraction rate of aluminum was calculated to test whether or not digestibility was affected by roasting. The dissolution results are shown in Table 4.
![]()
According to the experimental results, the content of S2- in the roasted ore digestion liquor is significantly decreased. The content of S2- in crude ore in the digestion liquor is above 1.7 g/L, but in the roasted ore digestion liquor, it is below 0.18 g/L. In addition, the digestion liquor of crude ore with a high content of S2- is black, yet roasted ore digestion liquor is yellow. On the other hand, the relative extraction rate of aluminum (ηr) of roasted ore is higher than that of crude ore, especially since the lower the dissolution temperature, the larger the distinction. The experimental results indicate that the roasting cannot only significantly decrease the content of S2- in digestion liquor, but also greatly improve the digestibility of ore. Thus, using oxidizing roasting is an effective way to remove the sulfide sulfur from high-sulfur bauxite.
3.3.2 Removal of sulfate sulfur
The Bayer solution sulfate produces a harmful effect on equipment for scaling. In accordance with some low sulfur minerals, it employs a natural removing sulfur process, which is the evaporation and condensation of the solution when sulfate has accumulated to some extent in alumina production. However, when the content of sulfur in bauxite is higher, and the capability to naturally remove sulfur is not enough, it is necessary to organize a
special process of removing sulfur. As shown in Table 3, sulfur emerges in the form of sulphate sulfur in Henan bauxites B, C and Guizhou bauxites. Since the contents of total sulfur in Henan bauxites B and C are respectively 0.85 % and 0.76 %, and those of sulfur sulfate are respectively 0.66% and 0.61%, the method of removing the sulfur from the two bauxites is mainly focused on removing sulfate. However, the content of sulfur (>0.7%) is higher than one requested (<0.2%) in production, and even though two such bauxites belong to high-grade alumina bauxite for Al/Si ratio above 11. They cannot be used until the sulfur has been removed from the bauxite. Due to the sulfate sulfur, the sulfate in the ore is entirely dissolved in the solution and separated from the ore, the main phase of ore β-AlOOH will not be affected when the sodium carbonate solution is added to wash the ore. There are some sulfates in bauxite such as Na2SO4, K2SO4 and FeSO4, which are soluble in water and can be separated directly from ore. There are also some sulfate basically CaSO4 which are insoluble in water and can react with sodium carbonate, the main chemical reaction occurs as follows:
CaSO4+Na2CO3=CaCO3+SO42-+2Na+ (8)
Table 4 Results of digestion of Henan high-sulfur bauxite A by Bayer process
So such sulfate can be separated from ore, the results are shown in Table 5.
Table 5 Results of scrubbing of Na2CO3
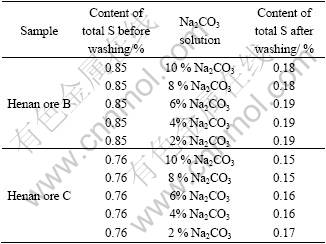
As shown in Table 5, 2% sodium carbonate solution can meet the requirements of washing. The content of the total sulfur in ore is lowered from 0.75 % to below 0.2% and can meet the production requirements for the sulfur content. Therefore, this type of ore can be used again.
4 Conclusions
1) Sulfur in the high sulfur-containing bauxite mainly exists in the form of sulfide sulfur (pyrite) or sulfate sulfur, and the main existing forms of sulfur bauxites from different areas are not the same. These are the further supplements from the previous theory.
2) The sulfur phase of high-sulfur bauxite can be accurately determined by means of a combination of X-ray diffraction and a quantitative chemical analysis. Ascertaining the main form of sulfur in high-sulfur bauxite can provide a theoretical instruction for choosing methods for the removal of sulfur.
3) This is an effective way of using oxidizing roasting to remove sulfide sulfur from high-sulfur bauxite, the roasting ore cannot only significantly decrease the content of S2- in digestion liquor, but also greatly improve the digestibility of ore.
4) This is an effective way of using the sodium carbonate solution washing to remove sulfate sulfur, the content of the total sulfur in ore is lowered from 0.75 % to below 0.2% and can meet the production requirements for the sulfur content.
References
[1] LI Tian-geng, LI Zi-ru, ZHANG Qie-fan. Development of the world aluminum industry and the countermeasure of China [J]. World Nonferrous Metal, 1999(10): 64-68. (in Chinese)
[2] HE Bo-quan. Discussion on the new de-sulfuration method of Chinese high sulfur bauxite [J]. Light Metals, 1996(12): 3-5. (in Chinese)
[3] BI Shi-wen. Technology of alumia production [M]. Beijing: Chemical industry Press, 2006: 96-103. (in Chinese)
[4] HE Run-de, HU Si-chun, LI Zhi-ying. Discussion on the method of hydrometallurgical de-sulfurizing during producing alumina with high grade bauxite containing sulfur [J]. Hydrometallurgy of China, 2004(2): 66-68. (in Chinese)
[5] PADILLA R, VEGA D, RUIZ M C. Pressure leaching of sulfidized chalcopyrite in sulfuric acid-oxygen media [J]. Hydrometallurgy, 2007, 86(1-2): 80-88.
[6] RAO K A. Studies on recovery of cobalt values by sulfatizing roasting of pyrite from Kalyadi copper ores [J]. Transactions of the Indian Institute of Metals, 1997, 50(2-3): 153-162. (India)
[7] LU Guo-zhi, ZHANG Ting-an, NI pei-yuan, BAO Li, ZHAO Ai-chun, QU Hai-cui. Roasting pretreatment and digestion performance of high-sulfur bauxite [J]. The Chinese Journal of Process Engineering, 2009, 9(1): 71-75. (in Chinese)
[8] LIN Y, LIN C X, WU Y G. Characterization of red mud derived from a combined Bayer process and bauxite calcination method [J]. Journal of Hazardous Materials, 2007, 146(1-2) : 255-261
[9] EYER S I, BHARGAVA S, SUMICH M. Removal of organic from Bayer liquor and wet oxidation [J]. Light Metals, 2000: 45-51.
[10] PIGA L,POCHETTI F, STOPPA L. Application of thermal analysis techniques to a sample of red mud―A by-product of the Bayer process for magnetic separation [J]. Thermochimica Acta, 1995(254): 337-345.
[11] ZHANG Nian-bing, BAI Chen-guang, LI Zhi-ying, MA Mang-yuan. Research on the existence of sulfur mineral in high sulfur bauxite and the desulfurization efficiency [J]. Journal of Chinese Electron Microscopy Society, 2009, 28(3): 229-234. (in Chinese)
[12] LU Guo-zhi, ZHANG Ting-an, NI pei-yuan, BAO Li, ZHAO Ai-chun, QU Hai-cui. Roasting pretreatment of high-sulfur bauxite [J]. The Chinese Journal of Process Engineering, 2008, 8(5): 892-896. (in Chinese)
[13] The drafting group of production alumina by combination process. Control analysis of production alumina by combination process [M]. Beijing: Metallurgical industry Press, 1977: 140-142. (in Chinese)
[14] ZHANG Qi. Application of sulfur phase analytic determination methods [J]. Gold, 2006(11): 52-54. (in Chinese)
[15] ABLAMOFF B R. Physical and chemical principles of comprehensive treatment of aluminum-containing raw materials by basic process [M]. CHEN Qian-de, trans. Changsha: Central South University of Technology Press, 1988: 178-182. (in Chinese)
[16] LU Long, WANG Ru-cheng, XUE Ji-yue, CHEN Jun. Activity of elements during the weathering of pyrite and its environmental effects [J]. Geological Review, 2001, 47(1): 95-101. (in Chinese)
[17] QI Y Q, LI W, CHEN H K. Desulfurization of coal through pyrolysis in a fluidized-bed reactor under nitrogen and 0.6% O2-N2 atmosphere [J]. Fuel and Energy Abstracts, 2004, 45(6) : 705-712. (in Chinese)
胡小莲1, 2,陈文汨1, 谢巧玲1
1. 中南大学 冶金科学与工程学院,长沙410083;
2. 湖南科技大学 化学化工学院,湘潭 411201
摘 要:采用X射线衍射分析和化学物相定量法研究高硫铝土矿中硫的赋存状态,并对不同形态的硫的脱除方法进行研究。结果表明:高硫铝土矿中硫的存在形态有的以硫化物为主(主要是黄铁矿),有的以硫酸盐为主,不同产地的铝土矿中硫的主要存在形态是不同的,进一步补充了铝土矿中硫的物相存在的规律性。采用X射线衍射结合化学物相定量法能更准确确定高硫铝土矿中硫的存在形态。确定高硫铝土矿中硫的主要存在形态对除硫方法的选择具有理论指导意义。不同形态的硫可以采用不同的除硫方法。采用氧化焙烧能有效脱除高硫铝土矿中硫化物型硫,原矿溶出液中S2-的含量均在1.7 g/L以上,而焙烧矿的溶出液中S2-的含量均在0.18 g/L以下。通过N2CO3溶液洗涤可以有效脱除矿石中硫酸盐型硫,原矿中全硫含量下降到0.2%以下,符合生产中对矿石中硫含量的要求。
关键词:高硫铝土矿;硫化物相;氧化焙烧;碳酸钠洗涤
(Edited by YANG Hua)
Foundation item: Project (20971041) supported by the National Natural Science Foundation of China; Project (09B032) supported by Scientific Research Fund of Hunan Provincial Education Department, China
Corresponding author: HU Xiao- lian; Tel: +86-13107120683; E-mail: huxiaolian71@yahoo.com.cn
DOI: 10.1016/S1003-6326(11)60908-4

