J. Cent. South Univ. Technol. (2011) 18: 83-88
DOI: 10.1007/s11771-011-0663-0
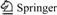
Thermodynamics of chromite ore oxidative roasting process
QI Tian-gui(齐天贵), LIU Nan(刘楠), LI Xiao-bin(李小斌),
PENG Zhi-hong(彭志宏), LIU Gui-hua(刘桂华), ZHOU Qiu-sheng(周秋生)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2011
Abstract: To explicate the thermodynamics of the chromite ore lime-free roasting process, the thermodynamics of reactions involved in this process was calculated and the phrases of sinter with different roasting times were studied. The thermodynamics calculation shows that all the standard Gibbs free energy changes of the reactions to form Na2CrO4, Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 via chromite ore and Na2CO3 are negative, and the standard Gibbs free energy changes of the reactions between MgO, Fe2O3 and SiO2 released from chromite spinel to form MgO・Fe2O3 and MgO・SiO2 are also negative at the oxidative roasting temperatures (1 173- 1 473 K). The phrase analysis of the sinter in lime-free roasting process shows that Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 can be formed in the first 20 min, but they decrease in contents and finally disappear with the increase of roasting time. The final phase compositions of the sinter are Na2CrO4, MgO・Fe2O3, MgO・SiO2 and MgO. The results indicate that Na2CrO4 can be formed easily via the reaction of Na2CO3 with chromite ore. Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 can be formed as intermediate compounds in the roasting process and they can further react with chromite ore to form Na2CrO4. MgO released from chromite ore may react with iron oxides and silicon oxide to form stable compounds of MgO・Fe2O3 and MgO・SiO2, respectively.
Key words: chromite ore; oxidative roasting; thermodynamics; sodium chromate
1 Introduction
Soda-ash roasting of the chromite mineral is almost the unique method used worldwide for the production of water soluble sodium chromate [1]. The soda-ash roasting process is divided into lime-based roasting process and lime-free roasting process according to the composition of filler materials. In the roasting process, chromite ore and soda ash are mixed and subsequently roasted at 1 173-1 473 K in oxidative atmosphere to oxidize insoluble trivalent chromium spinels to water-soluble hexavalent chromate [2-3]. In traditional lime-based roasting process for manufacturing chromate, a mass of calcium-contained materials are used as fillers and large quantities of chromite ore processing residues (COPR) containing a lot of highly toxic, carcinogenic and mutagenic hexavalent chromium are produced, causing serious environmental pollution [4-5]. The alternative lime-free roasting process can overcome the disadvantages of low resource efficiency and the pollution of toxic residues in lime-based roasting process because there are no calcium-containing materials in this process, which makes the lime-free process become the most popular chromate production method [1, 6].
The chromite ore oxidative reactions in lime-free roasting process are complicated gas-liquid-solid heterogeneous reaction processes. When the roasting temperature is as high as 1 373 K, the impurities of silicon, ferrate, aluminum and magnesium present in chromite ore can also react with Na2CO3 as well as chromium. These subsidiary reactions affect the chromium oxidation seriously and make the roasting process more complex, so it is very important to reveal the rule of reactions in lime-free roasting process. In the past, the lime-free roasting process have been studied, the preliminary thermodynamics of the reactions of chromite ore with Na2CO3 has been investigated, and the molten salt phrases during the roasting process have been estimated [2, 6-7]. However, the main thrusts of previous research are to optimize the process parameters for improving kiln performance and to detail the kinetics, the reaction thermodynamics and the mechanism of the reactions between Na2CO3 and chromite ore. In this work, the thermodynamics of the reactions involved in lime-free roasting process is concentrated to make the reaction mechanism more clear.
2 Assumptions and method
It is assumed that the whole roasting process and all reactions are carried out at a constant temperature and
under the atmospheric pressure, and all compounds and substances in chromite ore are pure spinels. So, the standard Gibbs free energy change of all reactions can be calculated according to the classical thermodynamics theory related in Ref.[8]. Most of the thermodynamic data required for the calculation are drawn from Refs.[9-11]. For some complicated compounds, the thermodynamic data are calculated and estimated by the simulation models proposed in Refs.[12-13].
3 Thermodynamic calculation and discussion
3.1 Thermal decomposition of chromite ore
Natural chromite ore with spinel structure, has a formula of (Fe2+,Mg)(Cr,Al,Fe3+)2O4, which can be regard as a solid solution of FeO・Cr2O3, MgO・Cr2O3, FeO・Al2O3 and MgO・Al2O3. Meanwhile, some impurities such as 3MgO・2SiO2・2H2O, SiO2 and Fe2O3 are packed in the ore [14]. Generally, the chromite oxidative roasting process is operated at a high temperature, and some thermal decomposition reactions would be carried out under this condition. To determine all the original reactants in roasting process, it is necessary to probe the thermal decomposition reactions of chromite ore. The main decomposition reactions in this process can be described as reactions (1)-(4):
FeO・Cr2O3=FeO+Cr2O3 (1)
MgO・Cr2O3=MgO+Cr2O3 (2)
FeO・Al2O3=FeO+Al2O3 (3)
MgO・Al2O3=MgO+Al2O3 (4)
FeO・Cr2O3+1/4O2=1/2Fe2O3+Cr2O3 (5)
FeO・Al2O3+1/4O2=1/2Fe2O3+Al2O3 (6)
The relationship between standard Gibbs free energy change of reactions (1)-(6) and temperature are plotted in Fig.1. Fig.1 shows that the standard Gibbs free energy changes of decomposition reactions of spinel compounds of FeO・Cr2O3, MgO・Cr2O3, FeO・Al2O3 and MgO・Al2O3 in neutral or reductive atmosphere are all positive. It is evident that chromite ore is hard to decompose without oxygen. Actually, the chromite roasting process is carried out in oxidative atmosphere. Under oxidative roasting condition, decomposition of chromite can be described as reactions (5) and (6). Fig.1 shows that standard Gibbs free energy change of reaction (5) is negative while that of reaction (6) is positive. This suggests that FeO・Cr2O3 may be decomposed to Fe2O3 and Cr2O3 in oxidative roasting process, but FeO・Al2O3 cannot be decomposed under oxidative, neutral and reductive roasting atmosphere.
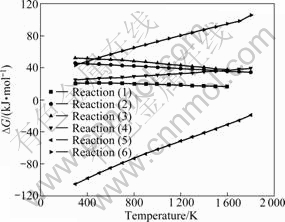
Fig.1 Relationships between standard Gibbs free energy change of reactions (1)-(6) and temperature
3.2 Thermodynamics of chromium oxidation
The main target of chromite ore oxidative roasting process is to make the insoluble chromium transform to water-soluble sodium chromate. The possible oxidation reactions of chromite spinel in roasting process are listed as reactions (7)-(12). The relationships between standard Gibbs free energy changes of these reactions and temperature are plotted in Fig.2.
Na2CO3+1/2FeO・Cr2O3+7/8O2=1/4Fe2O3+Na2CrO4+CO2 (7)
Na2CO3+1/2MgO・Cr2O3+3/4O2=1/2MgO+Na2CrO4+CO2 (8)
Na2CO3+1/2Cr2O3+3/4O2=Na2CrO4+CO2 (9)
Na2CO3+FeO・Cr2O3=FeO+Na2O・Cr2O3+CO2 (10)
Na2CO3+MgO・Cr2O3=MgO+Na2O・Cr2O3+CO2 (11)
Na2CO3+Na2O・Cr2O3+3/2O2=2Na2CrO4+CO2 (12)
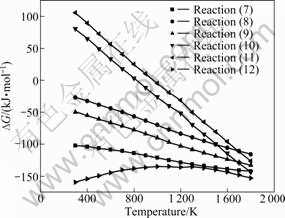
Fig.2 Relationships between standard Gibbs free energy change of reactions (7)-(12) and temperature
Fig.2 shows that all the standard Gibbs free energy changes of reactions (7)-(9) are negative at normal roasting temperatures (1 173-1 473 K), which indicates that chromite spinel and chrominum oxide can be decomposed and oxidized to form Na2CrO4 under oxidizing condition with sodium carbonate. As we all know, the more negative the Gibbs free energy change is, the higher the reaction tendency will be. With this thermodynamic criterion, the thermal oxidative reaction tendency sequence of chromite spinel is FeO・Cr2O3> Cr2O3>MgO・Cr2O3. The standard Gibbs free energy changes of reactions (10) and (11) shown in Fig.2 are reduced rapidly with the temperature rising. All the values are negative at the roasting temperature, which proves that the chromite spinel may react with Na2CO3 to form Na2O・Cr2O3. The standard Gibbs free energy change of reaction (12) shown in Fig.2 suggests that Na2O・Cr2O3 can be oxidized easily when oxygen and sodium carbonate are sufficient.
3.3 Thermodynamics of sodium carbonate
Sodium carbonate is the most important reactant in the roasting process and its reaction rules will affect the roasting process directly. Under the conventional lime-free roasting conditions, sodium carbonate not only reacts with chromite spinel but also reacts with the impurities present in chromite ore. Among them, the reactions of sodium carbonate with the impurities of FeO・Al2O3, MgO・Al2O3, Fe2O3 and SiO2 to form Na2O・Al2O3, Na2O・Fe2O3 and Na2O・SiO2 have received extensive attention. The subsidiary reactions between Na2CO3 and impurities are listed as reactions (13)-(16), and the relationships between changes of standard Gibbs energy and temperature for those reactions are plotted in Fig.3.
Na2CO3+FeO・Al2O3+1/4O2=1/2Fe2O3+Na2O・Al2O3+CO2 (13)
Na2CO3+MgO・Al2O3=MgO+Na2O・Al2O3+CO2 (14)
Na2CO3+Fe2O3=Na2O・Fe2O3+CO2 (15)
Fig.3 Relationships between standard Gibbs free energy change of reactions (13)-(16) and temperature
Na2CO3+SiO2=Na2O・SiO2+CO2 (16)
Fig.3 shows that all the standard Gibbs free energy changes of reactions (13)-(16) become lower with the increase of temperature and all of them are negative above 1 200 K, which indicates that the formation reactions of Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 via Na2CO3 and impurities are possible under the normal chromite roasting temperature.
By comparing the values of standard Gibbs free energy change of reactions (13)-(16) in Fig.3 and those of reactions (7)-(9) in Fig.2, it will be found that the standard Gibbs free energy changes of reactions (7)-(9) are more negative than those of reactions (13)-(16), which indicates that the reaction tendency for Na2CO3 to form Na2CrO4 is higher than the tendency to form Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2. This means that Na2CrO4 is more stable than Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 in roasting process. But, some dynamics research shows that the formation reactions of Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 have large reaction rates [7,15]. So, the thermodynamic and kinetic sequence may make some Na2CO3 form the intermediate compounds of sodium salts of ferrite, aluminate and silicate and then those intermediate compounds replacing part of Na2CO3 react with chromite to form Na2CrO4 in oxidative atmosphere. Those reactions can be described as reactions (17)-(22). The relationships between standard Gibbs energy change and temperature for those reactions are plotted in Fig.4.
Na2O・Fe2O3+1/2FeO・Cr2O3+7/8O2=5/4Fe2O3+Na2CrO4 (17)
Na2O・Al2O3+1/2FeO・Cr2O3+7/8O2=1/4Fe2O3+Na2CrO4+Al2O3 (18)
Na2O・SiO2+1/2FeO・Cr2O3+7/8O2=1/4Fe2O3+Na2CrO4+SiO2 (19)
Na2O・Fe2O3+1/2MgO・Cr2O3+3/4O2= 1/2MgO+Na2CrO4+Fe2O3 (20)
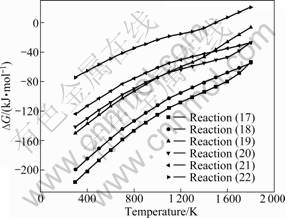
Fig.4 Relationships between standard Gibbs free energy change of reactions (17)-(22) and temperature
Na2O・Al2O3+1/2MgO・Cr2O3+3/4O2= 1/2MgO+Na2CrO4+Al2O3 (21)
Na2O・SiO2+1/2MgO・Cr2O3+3/4O2= 1/2MgO+Na2CrO4+SiO2 (22)
The standard Gibbs free energy changes of reactions (17)-(22) shown in Fig.4 are negative in an extensive range of temperature, suggesting that chromite spinel can seize the alkali in Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 to form Na2CrO4 under oxidative atmosphere.
All the analyses above show that Na2CrO4 is more stable than Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2, while small amounts of Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 can be formed as intermediate compounds in practical roasting process because of the kinetic factors. To explicate the rule of Na2CrO4 formation process, the chromium oxidation rate with different roasting temperatures and roasting times is investigated, then the oxidative reaction kinetic parameters of the reaction of chromite ore with Na2CO3 and Na2O・Fe2O3 in oxidative atmosphere are calculated. The results show that the apparent reaction rate constant of Na2CrO4 formation reaction via Na2CO3 and chromite is about an order larger than that of the reaction via Na2O・Fe2O3 and chromite (5×10-2 min-1 and 3.6×10-3 min-1 at roasting temperature of 1 323 K, respectively). This implies that the reaction rate of Na2CrO4 formation reaction through Na2O・Fe2O3 is significantly lower than that of the reaction through Na2CO3. So, the formation of the intermediate compounds containing alkali (sodium ferrite, etc) may be one of the important causes of low chromium oxidation rate, especially in the later roasting process when the Na2CO3 is exhausted.
3.4 Thermodynamics of magnesium oxide
In the chromite ore oxidative roasting process, the chromite spinels are oxidized and the MgO and iron oxides are released. MgO is the unique alkali compound except Na2CO3 in lime-free roasting process and it can react with most acid compounds at high temperature. The previous study results show that all the FeO, Fe3O4 and Fe2O3 can react with MgO in oxidative atmosphere to form MgO・Fe2O3 with a ferrite structure [16]. These combination reactions are listed as reactions (23)-(25). The relationships between standard Gibbs energy change and temperature for those reactions are plotted in Fig.5.
MgO+Fe2O3=MgO・Fe2O3 (23)
MgO+2/3Fe3O4+1/6O2=MgO・Fe2O3 (24)
MgO+2FeO+1/2O2=MgO・Fe2O3 (25)
Fig.5 shows that standard Gibbs free energy changes of reactions (23)-(25) increase with the tem- perature rising, and all of them are negative and become
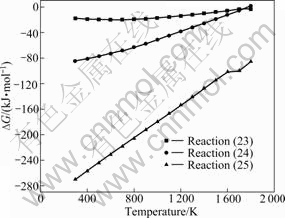
Fig.5 Relationships between standard Gibbs free energy change of reactions (23)-(25) and temperature
more negative with the valence states of iron decreasing, in the chromite roasting temperature range. This suggests that all the reactions between iron oxides and MgO listed above may be carried out and the lower the valence states of iron, the easier the formation of magnesium ferrite compound.
Magnesium oxide can also react with silicon oxide besides the iron oxide, listed as reactions (26)-(27). The standard Gibbs free energy changes of those reactions are shown in Fig.6.
MgO+SiO2=MgO・SiO2 (26)
2MgO+SiO2=2MgO・SiO2 (27)
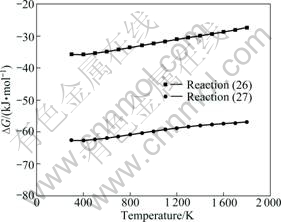
Fig.6 Relationships between standard Gibbs free energy change of reactions (26)-(27) and temperature
From Fig.6, the standard Gibbs free energy changes of reactions (26)-(27) are negative and the values have no obvious change with the increase of temperature. So, the MgO and silicon oxide may react to form stable magnesium silicates, 2MgO・SiO2 and MgO・SiO2. The standard Gibbs free energy change for forming 2MgO・
SiO2 is more negative than that of MgO・SiO2, which indicates that 2MgO・SiO2 is more stable than MgO・SiO2.
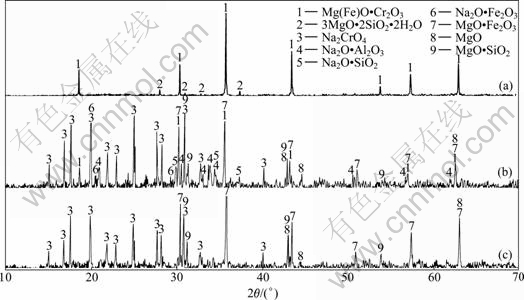
Fig.7 XRD patterns of chromite ore and chromate sinter prepared for different roasting times: (a) Chromite ore; (b) Sinter roasting for 20 min; (c) Sinter roasting for 120 min
3.5 Experimental results
In order to verify the results of the above mentioned thermodynamic analysis, the chromite oxidative roasting experiment was carried out with different roasting time. The main chemical composition of the chromite ore can be characterized as follows (mass fraction): Cr2O3 41.45%, Al2O3 22.95%, Fe2O3 22.29%, SiO2 4.24% and MgO 8.63%. The mixture of this chromite ore and Na2CO3 was roasted at 1 323 K under oxidation atmosphere. The X-ray diffraction (XRD) patterns of chromite ore and chromate sinters with different roasting times are displayed in Fig.7.
XRD analysis reveals that the major phases in chromite ore are chromium spinel with a few serpentine (Fig.7(a)). Na2O・Fe2O3, Na2O・Al2O3, Na2O・SiO2 and unreacted chromium spinel are observed in the sinter with 20 min roasting time (Fig.7(b)), but the contents of those phases decrease and finally disappear with the increase of roasting time. The final phase compositions of oxidization reaction in the roasting process are Na2CrO4, MgO・Fe2O3, MgO・SiO2 and MgO (Fig.7(c)). Combined with thermodynamic analysis, the phase transformation indicates that chromite oxidization reaction may experience two stages. In the first stage, most of Na2CO3 reacts with chromite spinel to form Na2CrO4 and a part of Na2CO3 transforms into Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2. In the second stage, Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 may further react with chromite ore to form Na2CrO4. In the whole roasting process, magnesium compounds transform into MgO・Fe2O3, MgO・SiO2 and MgO.
The results of experiment are consistent with thermodynamic calculation perfectly. Both of them show that the oxidization of the chromite in lime-free roasting process not only take place between chromite ore and Na2CO3, but also take place between chromite and intermediate compounds which are produced by subsidiary reactions. So, we should pay more attention to the intermediate compounds in the roasting process when we want to intensify the chromite oxidizing process.
4 Conclusions
1) The spinel structure of chromite such as FeO・Cr2O3, MgO・Cr2O3, FeO・Al2O3 and MgO・Al2O3 cannot be destroyed by thermal decomposition reactions at normal roasting temperature.
2) Part of Na2CO3 may transform into intermediate compounds such as Na2O・Fe2O3, Na2O・Al2O3 and Na2O・SiO2 by subsidiary reactions in the lime-free roasting process. Those intermediate compounds are less stable than Na2CrO4 and they can further react with chromite to form Na2CrO4.
3) The magnesium compounds in chromite ore may transform into stable phases of MgO・Fe2O3, MgO・SiO2 and MgO in the lime-free roasting process.
References
[1] DING Yi. Present status and prospect of chromate production in China [J]. Chemical Industry and Engineering Progress, 2004, 23(4): 345-348. (in Chinese)
[2] ANTONY M P, JHA A, TATHAVADKAR V D. Alkali roasting of Indian chromite ores: Thermodynamic and kinetic considerations [J]. Mineral Processing and Extractive Metallurgy, 2006, 115(2): 71-80.
[3] HAN Deng-lun, ZHANG Zhong-yan, LI Yuan-kui. Non-calcium roasting method of production chromium salts: China Patent, CN1579947 [P]. 2005-02-16. (in Chinese)
[4] JAGUPILLA S C, MOON D H, WAZNE M. Effects of particle size and acid addition on the remediation of chromite ore processing residue using ferrous sulfate [J]. Journal of Hazardous Materials, 2009, 168: 121-128.
[5] MOON D H, WAZNE M, DERMATAS D. Long-term treatment issues with chromite ore processing residue (COPR): Cr6+ reduction and heave [J]. Journal of Hazardous Materials, 2007, 143: 629-635.
[6] TATHAVADKAR V D, ANTONY M P, JHA A. The effect of salt-phase composition on the rate soda-ash roasting of chromite ore [J]. Metallurgical and Materials Transactions B, 2003, 34B(4): 555-563.
[7] TATHAVADKAR V D, ANTONY M P, JHA A. The soda-ash roasting of chromite minerals: Kinetics considerations [J]. Metallurgical and Materials Transactions B, 2001, 32B(4): 593-602.
[8] YE Da-lun, HU Jian-hua. Applicable thermodynamic handbook of inorganic substances [M]. Beijing: Metallurgical Industry Press, 2002: 6-20. (in Chinese)
[9] BARIN I. Thermodynamic data of pure substances [M]. Weinheim: VCH Verlagsgesellschaft, 1995: 563-580.
[10] KNACKE O, KUBASCHEWSKI O, HESSELMANN K. Thermochemical properties of inorganic substance Ⅱ [M]. Berlin: Springer Verlag, 1991: 1160-1172.
[11] LIANG Ying-jiao, CHE Yin-chang. Thermodynamic handbook of inorganic substances [M]. Shenyang: Northeastern University Press, 1993: 150-155. (in Chinese)
[12] WEN Yuan-kai, SHAO Jun, CHEN De-wei. Calculation of Gibbs free energy of oxysalt minerals [J]. Geological Science, 1978(4): 348-357. (in Chinese)
[13] LI Xiao-bin, LI Yong-fang, LIU Xiang-min, LIU Gui-hua. A simple method of estimation of Gibbs free energy and enthalpy of complicate silicates [J]. Journal of the Chinese Ceramic Society, 2003, 29(3): 232-237. (in Chinese)
[14] RAMOS S S, CARBO A D, ADELANTADO J V, VICENTE J P. Analytical and mineralogical studies of ore and impurities from a chromite mineral using X-ray analysis, electrochemical and microscopy techniques [J]. Talanta, 2008, 74: 1592-1597.
[15] NEROSLAVSKAYA L L, SHABALINA O A, IZOTOV V I. Kinetic features of potassium and sodium ferrite formations [J]. Russian Metallurgy, 1986, 2: 220-224.
[16] PAIK J G, LEE M J, HYUN S H. Reaction kinetics and formation mechanism of magnesium ferrites [J]. Thermochimica Acta, 2005, 425: 131-136.
(Edited by YANG Bing)
Foundation item: Project(2009FJ1009) supported by the Major Science and Technology Program of Hunan Province, China
Received date: 2009-10-28; Accepted date: 2010-03-01
Corresponding author: LI Xiao-bin, Professor, PhD; Tel: +86-731-88830453; E-mail: qtg_csu@163.com