
Indium recovery from zinc oxide flue dust by oxidative pressure leaching
LI Xuan-hai(���纣)1, ZHANG Yan-juan(�����)1, QIN Quan-lun(��ȫ��)1,
YANG Jian(�� ��)1, WEI Yan-song(����)2
1. School of Chemistry and Chemical Engineering, Guangxi University, Nanning 530004, China;
2. Department of Chemistry and Biology, Hechi College, Yizhou 546300, China
Received 6 July 2009; accepted 30 December 2009
____________________________________________________________________________________________
Abstract: Indium was recovered from zinc oxide flue dust (ZOFD) with sulfuric acid by oxidative pressure leaching in an autoclave, and the effects of different technological conditions on indium leaching were studied. Potassium permanganate and hydrogen peroxide were used as oxidants. The atmospheric pressure leaching experiments were also carried out. The experimental results show that the leaching rate of indium can be effectively improved by oxidative pressure leaching. The optimum conditions of pressure leaching are determined as sulfuric 5.10 mol/L acid, leaching time 150 min, temperature 90 ��, and the H2O2 dosage of 0.5 mL/g or 2.5% KMnO4. The leaching rate of indium is more than 90%, which is increased by 13% compared with that of atmospheric pressure leaching process without oxidant under the optimum conditions.
Key words: indium; zinc oxide flue dust; oxidative pressure leaching; potassium permanganate; hydrogen peroxide
____________________________________________________________________________________________
1 Introduction
Indium has a wide range of applications due to its unique physical and chemical properties[1], which has been widely used in optoelectronic devices, transparent electrodes for liquid crystal display and solar cells[2-6]. Indium is most commonly found in zinc-bearing materials such as sphalerite or marmatite[7-8]. At present, many raw materials for recovering indium are the previously abandoned zinc smelter slag and leaching residue. These waste residues are calcined at high temperature, the flue dust was collected and then the zinc oxide flue dust (ZOFD) containing 0.1%-0.3% (mass fraction) indium was got[9-10]. Sphalerite and marmatite are sulphur-bearing minerals. When being calcined at high temperature, indium sulfide (In2S3) will be formed and enter into the crystal lattice of zinc ferrite (franklinite, ZnFe2O4) or other solid solutions. The stability of indium sulfide, zinc ferrite and solid-melt in sulphuric acid creates problems in indium recovery by hydrometallurgical processes.
Pressure leaching can increase the temperature to more than 100 ��, which cannot be achieved under atmospheric pressure. In addition, the oxidative effect of oxidants can be enhanced under the pressure leaching. In recent investigations, oxidative pressure leaching was widely used in the leaching of sulphur-bearing nickel minerals[11-14], sulphur-bearing zinc minerals[15-16], sulphur-bearing copper minerals[17-18] and other sulphide concentrates or slag[19-21]. The oxidative pressure leaching has very significant effect on recovering metals from sulphur-bearing minerals or slag. Using hydrogen peroxide (H2O2) and potassium permanganate (KMnO4) as oxidant in pressure leaching has been reported[19, 22-24]. In the presence of oxidant, the sulfides and other stable phases can be dissolved efficiently, then the valuable metals are recovered. In order to efficiently extract indium from ZOFD, potassium permanganate and hydrogen peroxide are used as oxidants, and the effect of oxidative pressure leaching on indium extraction in the autoclave is studied in this work.
2 Experimental
2.1 Materials
The ZOFD sample used was got from a zinc production plant in Liuzhou, Guangxi, China, which was obtained by roasting the smelting residues in the rotary kiln at high temperature. The sample was characterized by X-ray diffractometry (XRD, Rigaku model D/max-2500) and the result is shown in Fig.1. The composition of the ZOFD is very complex, including wurtzite (ZnS), zincite (ZnO), quartz (SiO2), galenite (PbS), aluminium oxide (Al2O3), franklinite (ZnFe2O4), lead silicate (Pb2SiO4), zinc antimony oxide (Zn2.33Sb0.67O4), and so on. The chemical composition is presented in Table 1. Industrial sulphuric acid (89.5%) was used for leaching. Two oxidants, potassium permanganate (KMnO4) and hydrogen peroxide (H2O2), were of analytical grade.
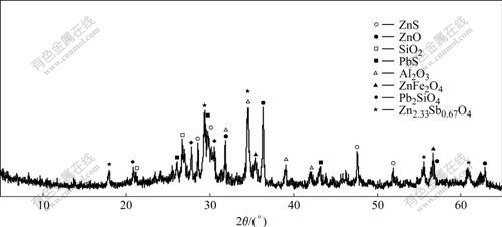
Fig.1 XRD pattern of zinc oxide flue dust
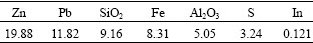
Table 1 Main chemical composition of zinc oxide flue dust (mass fraction, %)
2.2 Leaching procedure
The atmospheric pressure leaching experiments were carried out in a 500 mL three-neck flask immersed in thermostatically controlled water bath, equipped with a reflux condenser, mechanical stirrer and thermometer. The temperature could be controlled within ��1 ��. Deionized water and sulphuric acid were used to prepare the desired leaching solutions (400 mL) in the three-neck flask. When the temperature reached the pre-set value, 50.0 g of ZOFD and variable dosage of oxidant were added. After reaction for 150 min, the slurry was filtered and the residue was washed adequately with deionized water. The indium concentration in the filtrate was analyzed by polarography (Model JP-303, China)[25].
The pressure leaching tests were performed in a 1 L 1Cr18Ni9Ti stainless steel autoclave (GSH-1) with polytetrafluoroethylene lining, equipped with a PID temperature controller, a heating mantle, a variable speed stirrer, and a feeder. In a typical experiment, the slurry of mixing solid sample and distilled water was added into the autoclave, then the autoclave was sealed and heated. When the pre-set temperature was approached, sulphuric acid and oxidant were fed into the autoclave by the air compressor device. The pressure in the autoclave was kept constant at 0.5 MPa (gage pressure). At the end of the experiment, the autoclave was rapidly cooled by cold water through the feeder. The slurry was filtered and the residue was washed adequately with deionized water. The filtrate was analyzed for indium concentration by polarography. In these atmospheric pressure and pressure leaching experiments, the reaction temperature, reaction time, initial concentration of sulfuric acid, solid/liquid ratio, and stirring speed were fixed at 90 ��, 150 min, 5.10 mol/L, 1?8, and 575 r/min, respectively.
3 Results and discussion
The oxidative pressure leaching experiments were first carried out, then the atmospheric pressure leaching experiments were performed for comparison.
3.1 Effect of dosage of H2O2 on oxidative pressure leaching
The experiments were carried out with the H2O2 dosage of 0.25, 0.50, 0.75, and 1.0 mL/g. The results are shown in Fig.2. It can be seen that the leaching rate of indium increases obviously with the increase of the dosage of hydrogen peroxide when the dosage is less than 0.50 mL/g. Above 0.50 mL/g, the dosage of H2O2 has slight influence on the leaching rate. The leaching rate of indium is more than 90% with H2O2dosage of 0.50 mL/g.
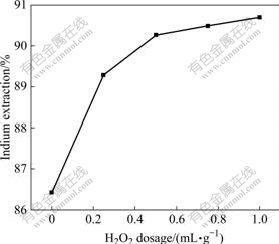
Fig.2 Effect of H2O2 dosage on indium extraction in oxidative pressure leaching
3.2 Effect of sulfuric acid concentration with H2O2 as oxidant
The effect of sulfuric acid concentration on indium leaching rate was studied in the range of 2.04-6.12 mol/L with the H2O2 dosage of 0.50 mL/g. As seen from Fig.3, indium leaching rate is increased from 63.7% to 91.3% with increasing H2SO4 concentration from 2.04 to 6.12 mol/L. It is also observed that the leaching rate is enhanced slowly when the H2SO4 concentration is higher than 5.10 mol/L. Increasing the H2SO4 concentration from 5.10 to 6.12 mol/L, the leaching late of indium is slightly improved from 90.3% to 91.3%. Therefore, an initial sulfuric acid concentration of 5.10 mol/L is sufficient for the recovery of indium.
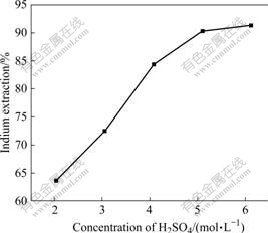
Fig.3 Effect of H2SO4 concentration on indium extraction in hydrogen peroxide oxidative pressure leaching
3.3 Effect of dosage of KMnO4 on oxidative pressure leaching
Another oxidant, potassium permanganate, was used to study the effect of dosage of oxidant on the indium leaching rate. The experiments were performed with the KMnO4 dosage of 0, 1.25%, 2.5%, 3.75%, 5%, and 6.25%. The results are plotted in Fig.4. Not surprisingly, the leaching rate of indium increases with increasing the dosage of potassium permanganate. It can also be observed that the leaching rate almost increases linearly increasing when the KMnO4 dosage is less than 2.5%, while above 2.5%, the leaching rate is not increased obviously. The leaching rate of indium reaches 93.9% with the KMnO4 dosage of 2.5%, only 94.3% of indium is extracted with 6.25% KMnO4. Therefore, the KMnO4 dosage of 2.5% is chosen for the recovery of indium.
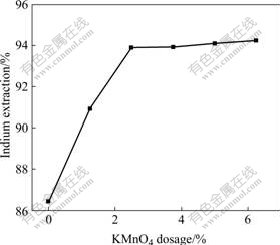
Fig.4 Effect of KMnO4 dosage on indium extraction in oxidative pressure leaching
3.4 Effect of H2SO4 concentration with KMnO4 as oxidative
The effect of H2SO4 acid concentration on indium leaching rate was studied in the range of 2.04-6.12 mol/L with the KMnO4 dosage of 2.5%. It can be observed from Fig.5 that the extraction of indium increases sharply with the H2SO4 concentration increasing from 2.04 to 3.06 mol/L. The increasing trend becomes slight while the H2SO4 concentration is above 3.06 mol/L, and the leaching efficiency of indium is about 90% with acid concentration of 3.06 mol/L. Increasing the H2SO4 concentration from 3.06 to 6.12 mol/L slightly improves the recovery of indium from 89.5% to 94.4%. This shows that the extraction of indium is not strongly dependent on the acid concentration while the H2SO4 concentration is above 3.06 mol/L with potassium permanganate as oxidant.
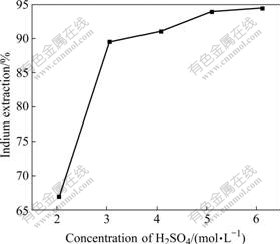
Fig.5 Effect of H2SO4 concentration on indium extraction in KMnO4 oxidative pressure leaching
3.5 Comparison of two leaching technologies
In order to study the different effects of atmospheric oxidative leaching and oxidative pressure leaching, the experiments were carried out in a 500 mL three-neck flask with potassium permanganate as oxidant. The conditions of atmospheric oxidative leaching were the same as those of the experiments of changing the dosage of potassium permanganate in oxidative pressure leaching. The results are shown in Fig.6. The results demonstrate that the pressure leaching has obvious influence on the extraction of indium. Without oxidant, the leaching rate of indium is 77.6% in atmospheric oxidative leaching, while 86.4% in pressure leaching, so the recovery of indium is increased by 8.8% just through enhancing the pressure. In these experiments, the leaching rate of indium is significantly increased with the increase of the dosage of potassium permanganate, and the recovery of indium in pressure leaching is about 8% higher than that in atmospheric pressure leaching. These results indicate that both the oxidant and pressurizing can strengthen the indium extraction from zinc oxide flue dust.
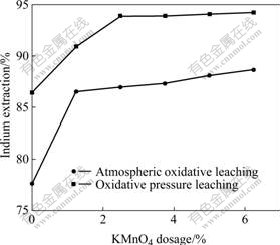
Fig.6 Effect of different leaching processes on indium extraction
These phenomena are related with the composition of the indium-containing minerals. In the pressure leaching, the concentration of oxygen in the solution is higher than that in the atmospheric leaching, so some of the stable indium-bearing phases can be destroyed and then dissolved easily into the solution. The addition of oxidant not only destroys the stable indium-bearing phases, but also oxidizes the indium sulfide which is difficult to dissolve in the normal leaching process, thus increasing the recovery of indium.
4 Conclusions
1) The oxidative pressure leaching can effectively recover indium from zinc oxide flue dust, which is a new process of extracting indium from low indium-bearing minerals.
2) The leaching rate of indium increases with increasing the dosage of oxidant and the concentration of H2SO4. The pressure leaching has obvious influence on the extraction of indium. Both adding the oxidant and enhancing the leaching pressure increase the indium extraction from ZOFD. The effect is more significant using potassium permanganate as oxidant than using hydrogen peroxide. The use of potassium permanganate in pressure leaching can obviously decrease the dependence on the H2SO4 concentration.
3) The optimum conditions of pressure leaching are 5.10 mol/L H2SO4, the H2O2 dosage of 0.5 mL/g or 2.5% KMnO4, at 90 �� for 150 min. The leaching rate of indium is more than 90%.
References
[1] ALFANTAZI A M, MOSKALYK R R. Processing of indium: A review[J]. Minerals Engineering, 2003, 16(8): 687-694.
[2] CHEBOTAREVA A B, UNTILA G G, KOST T N, JORGENSEN S, ULYASHINC A G. ITO deposited by pyrosol for photovoltaic applications[J]. Thin Solid Films, 2007, 515(24): 8505-8510.
[3] CALNAN S, UPADHYAYA H M, THWAITES M J, TIWARI A N. Properties of indium tin oxide films deposited using high target utilisation sputtering[J]. Thin Solid Films, 2007, 515(15): 6045-6050.
[4] JUNG W S, YOON S G, KANG S M, KIM S, YOON D H. Electrical and optical properties of ITO:Ca composite thin films for TEOLED cathode[J]. Thin Solid Films, 2008, 516(16): 5445-5448.
[5] LEE B H, KIM I G, CHO S W, LEE S. Effect of process parameters on the characteristics of indium tin oxide thin film for flat panel display application[J]. Thin Solid Films, 1997, 302(1/2): 25-30.
[6] STANCULESCU A, STANCULESCU F. Investigation of the properties of indium tin oxide-organic contacts for optoelectronic applications[J]. Thin Solid Films, 2007, 515(24): 8733-8737.
[7] SINCLAIR W D, KOOIMAN G J A, MARTIN D A, KJARSGAARD I M. Geology, geochemistry and mineralogy of indium resources at Mount Pleasant, New Brunswick, Canada[J]. Ore Geology Reviews, 2006, 28(1): 123-145.
[8] TONG X, SONG S X, HE J, LOPEZ-VALDIVIESO A. Flotation of indium-beard marmatite from multi-metallic ore[J]. Rare Metals, 2008, 27(2): 107-111.
[9] WANG Hong-jiang, SHEN Li-jun, LI Bo, LEI De-jun. Experimental investigation on continuous blowing of cadmia in fuming furnace[J]. China Nonferrous Metallurgy, 2005, (6): 37-39, 60. (in Chinese)
[10] CHE Yong-xia, LI Jian-cheng. Indium enrichment from low grade indium containing material[J]. Nonferrous Metals (Extractive Metallurgy), 2006, (6): 40-41. (in Chinese)
[11] DAS G K, ANAND S, ACHARYA S, DAS R P. Characterisation and acid pressure leaching of various nickel-bearing chromite overburden samples[J]. Hydrometallurgy, 1997, 44(1/2): 97-111.
[12] BROWN J A, PAPANGELAKIS V G. Interfacial studies of liquid sulphur during aqueous pressure oxidation of nickel sulphide[J]. Minerals Engineering, 2005, 18(15): 1378-1385.
[13] RUBISOV D H, KROWINKEL J M, PAPANGELAKIS V G. Sulphuric acid pressure leaching of laterites��Universal kinetics of nickel dissolution for limonites and limonitic/saprolitic blends[J]. Hydrometallurgy, 2000, 58(1): 1-11.
[14] RUBISOV D H, PAPANGELAKIS V G. Sulphuric acid pressure leaching of laterites��A comprehensive model of a continuous autoclave[J]. Hydrometallurgy, 2000, 58(2): 89-101.
[15] LIANG D, WANG J, WANG Y. Difference in dissolution between germanium and zinc during the oxidative pressure leaching of sphalerite[J]. Hydrometallurgy, 2009, 95(1/2): 5-7.
[16] OWUSU G, DREISINGER D B, PETERS E. Effect of surfactants on zinc and iron dissolution rates during oxidative leaching of sphalerite [J]. Hydrometallurgy, 1995, 38(3): 315-324.
[17] PADILLA R, VEGA D, RUIZ M C. Pressure leaching of sulfidized chalcopyrite in sulfuric acid-oxygen media[J]. Hydrometallurgy, 2007, 86(1/2): 80-88.
[18] HUANG K, LI Q, CHEN J. Recovery of copper, nickel and cobalt from acidic pressure leaching solutions of low-grade sulfide flotation concentrates[J]. Minerals Engineering, 2007, 20(7): 722-728.
[19] BANZA A N, GOCK E, KONGOLO K. Base metals recovery from copper smelter slag by oxidising leaching and solvent extraction[J]. Hydrometallurgy, 2002, 67(1/3): 63-69.
[20] AKCIL A, CIFTCI H. Metals recovery from multimetal sulphide concentrates (CuFeS2-PbS-ZnS): Combination of thermal process and pressure leaching[J]. International Journal of Mineral Processing, 2003, 71(1/4): 233-246.
[21] RUIZ M C, ABARZ?A E, PADILLA R. Oxygen pressure leaching of white metal[J]. Hydrometallurgy, 2007, 86(3/4): 131-139.
[22] LIU Jjian-hua, LI A-lin, ZHANG Xin-xian. Experimental study on oxidation leaching of tellurium from tellurium-bearing waste residue [J]. Hydrometallurgy of China, 2007, 26(3): 163-165. (in Chinese)
[23] BOUFFARD S C, RIVERA-VASQUEZ B F, DIXON D G. Leaching kinetics and stoichiometry of pyrite oxidation from a pyrite�Cmarcasite concentrate in acid ferric sulfate media[J]. Hydrometallurgy, 2006, 84(3/4): 225-238.
[24] AYDO?AN S, ARAS A, U?AR G, ERDEMO?LU M. Dissolution kinetics of galena in acetic acid solutions with hydrogen peroxide[J]. Hydrometallurgy, 2007, 89(3/4): 189-195.
[25] YUAN Yu-xia. Determination of indium in CNI-alloy by polarography[J]. Nonferrous Metals, 2003, 55(3): 140-142. (in Chinese)
__________________________
Foundation item: Project(2008105930817D017) supported by Graduate Student Educational Innovation Foundation of Guangxi, China; Project(0728238) supported by the Natural Science Foundation of Guangxi, China
Corresponding author: LI Xuan-hai; Tel: +86-771-3272702; E-mail: xuanhli@gxu.edu.cn
(Edited by YUAN Sai-qian)