
Effect of Cr on high temperature oxidation of TiAl
DONG Zi-qiang(����ǿ), JIANG Hui-ren(������), FENG Xiao-ran(����Ȼ), WANG Zhong-lei(������)
School of Materials Science and Engineering,
Beijing University of Aeronautics and Astronautics, Beijing 100083, China
Received 28 July 2006; accepted 15 September 2006
Abstract: The oxidation behavior of TiAl-Cr(mole fraction of Cr?0-20%) was investigated at 1 173 K in air. The microstructure and composition of the oxide scale were studied by X-ray diffractometry(XRD), scanning electron microscopy(SEM) and electro-probe micro-analyses(EPMA). The results show that with the addition of Cr content from 0 to 8%, the oxidation resistance decreases, especially at 3%, which is mainly attributed to the doping effect of Cr3+. TiAl-Cr(mole fraction of Cr?15%-20%) has good oxidation resistance, and the protective alumina layer is preferentially formed on the surface of TiAl alloy, which is due to an increase of mole ratio of Al to Ti in TiAl-Cr alloys.
Key words: TiAl; chromium; oxidation
1 Introduction
TiAl-based intermetallics are considered construc- tion materials for high temperature components, e.g. in aircraft and car engines, due to their low density and substantial mechanical strength at high temperatures[1-2]. Unfortunately, its oxidation resistance at high temperatures is considerably inferior to that of conventional superalloys. Improvement of oxidation resistance and understanding of its mechanisms are important in practical applications of TiAl. Various attempts have been made to find a method to improve the oxidation resistance of TiAl[3-6]. It was shown that alloying is a simpler and less expensive method which can effectively improve the oxidation resistance of TiAl, e.g. by addition of Nb[7-10], Cr[11], W[12], Mo[13], and Si[14]. Among these elements, Cr shows different effects in different additions. Small additions of Cr significantly improve the room-temperature mechanical properties of the ��+��2 titanium aluminides, but degrade their oxidation resistance. Larger additions of Cr significantly improve oxidation resistance, and have shown potential as oxidation-resistant coatings for the ��+��2 titanium aluminides. The mechanism responsible for their excellent oxidation resistance is only partly understood. In this study, the effect of Cr on the oxidation behavior of Ti-Al alloys at elevated temperatures was investigated, and the mechanism of oxidation was discussed. Two series of alloys were selected, i.e. Ti-xCr and TiAl-xCr (mole ratio of Ti to Al is 1?1), each of them with additions of Cr from 0 to 20%.
2 Experimental
The experimental alloys were prepared by high purity Ti, Al and Cr powders. The TiAl-xCr alloys had a fixed mole ratio of Ti to Al of 1?1 and varied chromium content up to 20%(mole fraction). The ingots were prepared by non-consumable electrode arc melting in an argon gas atmosphere. Each alloy button specimen weighed approximately 20 g and was remelted and turned over for three times to ensure homogeneity. The size of specimen for oxidation test was about 10 mm��10mm��1 mm, and the surface was polished with emery papers up to 1000#, and cleaned with acetone to remove grease.
The isothermal oxidation tests were performed at 900 �� in a box furnace in static air at atmospheric pressure for 24 h. At the end of the test, the specimens were furnace cooled. The phases formed in the scales were identified by Rigaku D/max-2200 X-ray diffractometry( Cu K��, 40 kV, 40 mA). The morphology of the scale and the distribution of the elements were examined by scanning electron microscopy(SEM) (JSM-5800, JEOL) and electro-probe micro-analyses
(EPMA)( JXA-8100, JEOL).
3 Results and discussion
3.1 Thickness and phase analysis of oxide scale
Fig.1 shows the variation of oxide scale thickness with mole fraction of Cr(x(Cr)) in the series of TiAl-xCr alloys. The scale thickness of the alloys first increases with the increase of x(Cr) up to a maximum value. Beyond the maximum value, thickness values decrease with the increase of x(Cr), which results in a concave downward characteristic curve. The maximum thickness for TiAl-xCr alloys occurs at 3%. When x(Cr) increases to 15%, the oxide scale is about 7-8 ��m, which is thinner than TiAl-0Cr alloy. When x(Cr) increases to 20%, the scale is about 2 ��m, which shows better oxidation resistance.
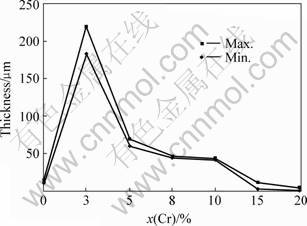
Fig. 1 Variation of thickness of scale with x(Cr)
The X-ray diffraction(XRD) patterns obtained from the high temperature oxidized specimen surfaces are shown in Fig.2. When Cr content increases to 8%, Al2O3 is detected. Al2O3 peaks are relatively weak compared with TiO2 peak. It is believed that the Al2O3 layer formed on the TiAl-20Cr alloy is thin, but protective. In this TiAl-xCr alloy, no chromium oxide is detected by XRD.
3.2 Microstructure observation and analysis
The surface morphologies of oxide scale formed on TiAl-xCr alloys are shown in Fig.3. Large randomly oriented rod-like grains of TiO2 are formed when Cr content is low. As Cr content increases, oxides formed on alloys become finer and smoother. Since TiO2 crystals grow much faster than Al2O3, the outer surface is subsequently covered with a layer, which is mainly TiO2 at high temperature.
Fig.4 shows the scanning electron microscope(SEM) images and the electro-probe microanalyses(EPMA) line analysis results, which show the cross section of the oxide scale of the TiAl-xCr alloys. The distribution of elements in the oxidation scales is shown in the line analysis. When Cr content is low, the oxide scale is thick, especially for the internal oxidation layer. And continuous and compact alumina scale is not formed (Fig.4(a)). When Cr content is high, the oxide scale becomes much thinner. The protective alumina layer is formed which can impede internal oxidation (Fig.4(b)).
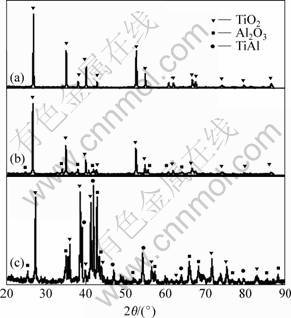
Fig.2 X-ray diffraction patterns of oxidized surfaces of TiAl-xCr: (a) TiAl-3Cr; (b) TiAl-8Cr; (c) TiAl-20Cr
When the foreign metal cations with lower valence than Ti occupy normal Ti-sites, the substitution of two foreign metal cations with valence of 3 accordingly increases one oxygen vacancy and one cation defect, which enhances internal oxidation and the rutile growth. Fig.5 shows the state of the element Cr that is distributed in internal oxide. Cr3+ substitution for Ti4+ in rutile enhances the rutile growth and increases internal oxidation. So the decrease of the oxidation of TiAl alloys with small amount of Cr addition can be interpreted qualitatively in terms of the increased oxygen vacancy concentration and Ti2+ defect.
It has been pointed out that the oxidation resistance of TiAl is expected to be improved either by suppression of rutile growth[15-18] or by enhancement of external alumina layer formation[15-17]. Now the oxidation behavior of TiAl-xCr alloys is discussed in the viewpoint of rutile growth suppression confirmed in Ti-Cr alloys. If the rutile growth suppression is the primary factor for the improvement of oxidation resistance of TiAl-xCr, the oxidation resistance of Ti-xCr alloys should be improved for Cr content in the range from 3% to 20%. Fig.6 shows that there are no changes of the scale of Ti-xCr. So the enhanced formation of external alumina scale may be the main reason for the improvement of oxidation resistance. Enhancement for an external alumina scale formation due to Cr addition can be explained either by WAGNER model or by the variation of activities of Ti and Al in the Ti-Al system[15-16]. According to WAGNER model, large amount of chromium has been achieved by increasing diffusion coefficient for Al(DAl) and simultaneously decreasing solubility of O at 101.325 kPa(No) and diffusion coefficient for O(Do). But there are no data available for the factors such as Do, DAl or No in r-TiAl. NATHAN et al[18] discussed the activity variation of Al and Ti in the Ti-Al-Cr system. From their work, we know that the mole ratio of Al to Ti increases with the increase of Cr. Therefore, the addition of Cr modifies component activities in the direction favoring Al2O3 formation. But the changes in component activities are not sufficient to explain the Cr effect[18]. Because of the lack of necessary data, further discussion is difficult concerning the mechanism of enhancement of the external alumina scale formation on TiAl by Cr addition.

Fig.3 Surface morphologies of oxide scale formed on TiAl-xCr alloys: (a)TiAl-3Cr; (b)TiAl-8Cr; (c) TiAl-15Cr; (d) TiAl-20Cr
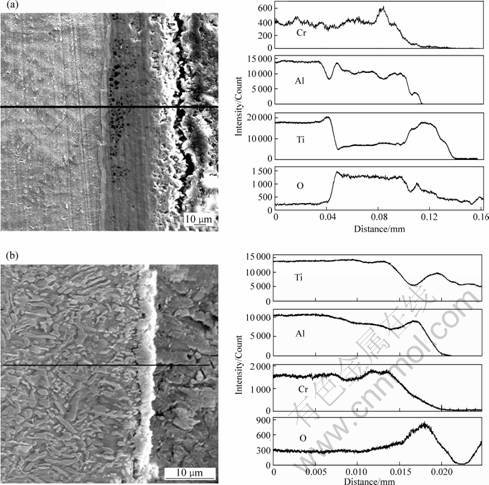
Fig.4 SEM images and EPMA line analysis results: (a) TiAl-5Cr; (b) TiAl-20Cr
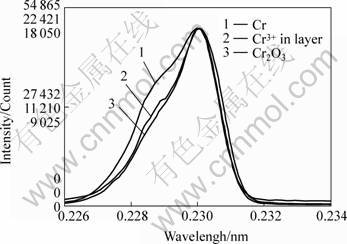
Fig.5 EPMA element state analysis for Cr distribute in TiAl- xCr oxide layer

Fig.6 Cross-sectional micrographs of Ti-xCr treated at 900 ��for 24 h: (a) Ti-5Cr; (b) Ti-10Cr; (c) Ti-20Cr
4 Conclusions
1) A small amount of Cr addition decreases the oxidation of TiAl alloys, which attributes to the increase of concentration of defect led by Cr3+.
2) Large amount of Cr addition can enhance the formation of external alumina scale, which improves the oxidation of TiAl alloy. The protective alumina layer is preferentially formed on the surface of TiAl-20%Cr alloy. The change of the mole ratio of Al to Ti may partially contribute to the formation of external alumina scale.
References
[1] KIM Y W, DIMIDUK D M. Designing gamma TiAl alloys: fundamental, strategy and production[C]// NATHAL M V. Proceed- ings of Structural Intermetallics. Warrendale, PA: TMS, 1997: 531-
543.
[2] AUSTIN C M. Progress in implementation of cast gamma titanium aluminide[C]// Gamma Titanium Aluminides. Warrendale, PA: TMS, 1995: 21-32.
[3] WU Jun, JING He-min, ZHANG Li-wu. Research progress on surface technology of TiAl intermetallics[J]. Corrosion Science and Protection Technology, 2003, 15(1): 24-28.(in Chinese)
[4] TANG Zhao-lin, WANG Fu-hui, WU Wei-tao. Effect of microarc oxidation treatment on oxidation resistance of TiAl alloy[J]. The Chinese Journal of Nonferrous Metals, 1999, 9(S1): 63-68. (in Chinese)
[5] TANG Zhao-lin, WANG Fu-hui. Effect of reactively sputtered Al2O3 films on oxidation resistance of TiAl[J]. The Chinese Journal of Materials Research, 1997, 11(5): 507-510. (in Chinese)
[6] KASAHARA K. Effects of aluminizing on tensile property and high temperature oxidation of TiAl-base alloys [J]. Zairyo to Kankyo Corrosion Engineering, 1998, 47(1): 29-35.
[7] PERKINS R A, MEIER G H. Oxidation behavior and protection of nibium[J]. JOM, 1990, 42(8): 11-16.
[8] VARMA S K, CHAN A, MAHAPATRA B N. Static and cyclic oxidation of Ti-44Al and Ti-44Al-xNb alloys[J]. Oxid Met, 2001, 55: 423-435.
[9] PEREZ P, HAANAPPEL V A C, STROOSNIJDER M F. The effect of niobium on the oxidation behaviour of titanium in N2/20%O2 atmosphere[J]. Mater Sci Eng A, 2000, A284: 126 - 137.
[10] ECKERT M, KATH D, HILPERT K. Thermodynamic activities in the alloys of the Ti-Al-Nb system[J]. Metall Mater Trans A, 1999, 30A: 1315-1326.
[11] BRADY M P, SMIALEK J L, SMITH J, HUMPHREY D L. The role of Cr in promoting protective alumina scale formation by ��-based Ti-Al-Cr alloys(��)��oxidation behavior in air[J]. Acta Mater, 1997, 45: 2371-2382.
[12] SHIDA Y, ANADA H. Role of W, Mo, Nb and Si on oxidation of TiAl in air at high temperature[J]. Mater Trans JIM, 1994, 35(9): 623-631.
[13] SHIDA Y, ANADA H. The effect of various ternary additives on the oxidation behavior of TiAl in high-temperature air[J]. Oxid Met, 1996, 45: 197-219.
[14] MAKI K, SHIODA M, SAYASHI M. Effect of silicon and niobium on oxidation resistance of TiAl intermetallics[J]. Mater Sci Eng A, 1992, A153: 591-596.
[15] CHOUDHURY N S, GRAHAM H C, HINZE J W. Oxidation behavior of titanium aluminides[C]// Properties of High Temperature Alloys with Emphasis on Environmental Effects. Princeton: Electrochemical Society Inc, 1976: 668-680.
[16] BECKER S, RAHMEL A, SCHORR M, SCHUTZE M. Mechanism of isothermal oxidation of the intermetallic TiAl and of TiAl alloys[J]. Oxid Met , 1992, 38: 425 �C 464.
[17] CHEN G L, SUN Z Q, ZHOU X. Oxidation and mechanical behaviour of intermetallic alloy in Ti-Al-Nb ternary system[J]. Mater Sci Eng A, 1992, A153: 597-601.
[18] NATHAN S, MICHAEL P, GOPAL M. Thermodynamics of selected Ti-Al and Ti-Al-Cr alloys[J]. Oxid Met, 1997, 52: 537-556.
(Edited by YANG You-ping)
Corresponding author: DONG Zi-qiang; Tel: +86-10-82311614; E-mail: dzq@mse.buaa.edu.cn