Effect of ageing temperature on precipitation of Al–Cu–Li–Mn–Zr alloy
来源期刊:中南大学学报(英文版)2018年第6期
论文作者:黄光杰 曹玲飞 邓燕君 吴晓东 黄利
文章页码:1340 - 1349
Key words:ageing temperature; precipitation; Al–Cu–Li–Mn–Zr alloy; T1 phase; χ (Al5Cu6Li2) phase
Abstract: The precipitation behaviors of an Al–Cu–Li–Mn–Zr alloy at different ageing temperatures (120, 160 and 200 oC) were investigated using Vickers hardness measurements and transmission electron microscopy (TEM) characterization. Age hardening curves show an increase in precipitation kinetics with increasing ageing temperature. The results of TEM show that for the samples peak aged at 120 oC, the amount of δ'''' (Al3Li), GP zones/θ'''' (Al2Cu) and χ (Al5Cu6Li2) phases is obviously higher than that of T1 (Al2CuLi) precipitates; while the samples peak aged at 160 and 200 oC are usually dominated by T1 phase with a minor fraction of GP zones/θ'''' and δ'''', and the χ phase almost does not form. In addition, quantitative analysis on the T1 platelets demonstrates that the samples peak aged at 200 oC have larger plate diameter and smaller area fraction of T1, as compared to the samples peak aged at 160 oC. Correspondingly, the possible reasons for such phenomena are discussed.
Cite this article as: DENG Yan-jun, HUANG Guang-jie, CAO Ling-fei, WU Xiao-dong, HUANG Li. Effect of ageing temperature on precipitation in Al–Cu–Li–Mn–Zr alloy [J]. Journal of Central South University, 2018, 25(6): 1340–1349. DOI: https://doi.org/10.1007/s11771-018-3830-8.

J. Cent. South Univ. (2018) 25: 1340-1349
DOI: https://doi.org/10.1007/s11771-018-3830-8
DENG Yan-jun(邓燕君), HUANG Guang-jie(黄光杰), CAO Ling-fei(曹玲飞),WU Xiao-dong(吴晓东), HUANG Li(黄利)
International Joint Laboratory for Light Alloys of Ministry of Education, College of Materials Science and Engineering, Chongqing University, Chongqing 400044, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: The precipitation behaviors of an Al–Cu–Li–Mn–Zr alloy at different ageing temperatures (120, 160 and 200 oC) were investigated using Vickers hardness measurements and transmission electron microscopy (TEM) characterization. Age hardening curves show an increase in precipitation kinetics with increasing ageing temperature. The results of TEM show that for the samples peak aged at 120 oC, the amount of δ' (Al3Li), GP zones/θ' (Al2Cu) and χ (Al5Cu6Li2) phases is obviously higher than that of T1 (Al2CuLi) precipitates; while the samples peak aged at 160 and 200 oC are usually dominated by T1 phase with a minor fraction of GP zones/θ' and δ', and the χ phase almost does not form. In addition, quantitative analysis on the T1 platelets demonstrates that the samples peak aged at 200 oC have larger plate diameter and smaller area fraction of T1, as compared to the samples peak aged at 160 oC. Correspondingly, the possible reasons for such phenomena are discussed.
Key words: ageing temperature; precipitation; Al–Cu–Li–Mn–Zr alloy; T1 phase; χ (Al5Cu6Li2) phase
Cite this article as: DENG Yan-jun, HUANG Guang-jie, CAO Ling-fei, WU Xiao-dong, HUANG Li. Effect of ageing temperature on precipitation in Al–Cu–Li–Mn–Zr alloy [J]. Journal of Central South University, 2018, 25(6): 1340–1349. DOI: https://doi.org/10.1007/s11771-018-3830-8.
1 Introduction
Al–Cu–Li alloys are extensively used for aerospace applications due to their low density, high elastic modulus and high specific strength [1–3]. The mechanical properties of these alloys depend largely on the type, size and distribution of the precipitates formed upon thermal ageing. In Al–Cu–Li alloys, several types of fine precipitates may be formed, such as T1, θ' and δ' phases [4, 5]. In general, the formation of these precipitates can be significantly affected by alloy composition and heat-treatment parameters.
In Al–Cu–Li alloys, the Cu and Li concentrations have significantly influences on the precipitation sequence, since the precipitation phases, such as T1, θ' and δ', all compete for Cu and Li atoms. As reported in Refs. [6–8], a high level of Li usually facilitates the formation of the metastable δ' phase. When the Li concentration is low [9–11], the alloys prefer to form T1 or θ', and which one of these two phases is dominant depending on the Li/Cu atomic ratio. As reported by SILCOCK [12], the θ' phase dominates when the Li/ Cu atomic ratio is less than 1.0, while the T1 phase will be the mainstay on the contrary. Moreover, numerous studies have shown that minor solute additions of Mg, Ag and Zn to Al–Cu–Li alloys can promote the precipitation of the T1 phase and precipitate simultaneously the δ', θ' and S' (Al2CuMg) phases [13–16]. In addition to the precipitates mentioned above, a σ (Al5Cu6Mg2) phase with cubic morphology has also been characterized in an aged Al–Cu–Li–Zn–Mg–Mn–Zr alloy [17–19]. Moreover, PAN et al [20] reported that the Indium microalloyed Al–Cu–Li alloy can cause the precipitate of another similar cubic shaped phase, named χ phase, which is considered as a variant of σ (Al5Cu6Mg2) with Li substituting the Mg positions. However, the formation mechanism of the χ phase is still not clear, which is worth studying further.
Besides, ageing temperature as an important heat treatment parameter also has a great influence on the precipitation in Al–Cu–Li alloys. For example, BLANKENSHIP et al [21] found that lower ageing temperature could promote the precipitation of δ' particles and reduced T1 precipitation on subgrain boundary in the Al–Cu– Li–Mg–Ag–Zr alloy. And as reported in Ref. [22], in the Al–Cu–Li–Mg–Ag–Zr alloy, the major precipitates are δ' and θ' when ageing below 160 °C; while T1 phase becomes the dominant strengthening phase when the ageing temperature is above 160 °C.
Accordingly, since the precipitations are closely associated with mechanical behavior of Al–Cu–Li alloys, it is essential to understand the effects of element added and ageing temperature on the type and amount of precipitations in Al–Cu–Li alloys. However, for the Al–Cu–Li–Mn–Zr alloy, limited information is available on its ageing response at different ageing temperatures. Therefore, the aim of the present work is to thoroughly understand the effect of ageing temperature on the precipitation in the Al–2.8Cu–1.4Li–0.3Mn–0.1Zr (wt%) alloy. To this end, hardness testing has been employed to explore the effect of ageing temperatures on ageing kinetics and ageing responses. Additionally, the TEM technique has also been used to determine the effect of ageing temperature on the types, dimensions and distribution of the precipitation phases at the peak aged state.
2 Experimental
The hot rolled plates of Al–Cu–Li–Mn–Zr alloys were provided by Southwest Aluminum (Group) Co., Ltd., China. The chemical composition of this alloy is Al–2.8Cu–1.4Li– 0.3Mn–0.1Zr (wt%). Tensile samples were cut from the 1/2 depth of the plate and parallel to the rolling direction (RD) of the plate. Each test sample was solution treated at 520 °C for 1 h and then water quenched to room temperature. Within 5 min, 4% plastic deformation was introduced by stretching and subsequently these samples were naturally aged for several days. Artificial ageing was performed at 120, 160 and 200 °C for different times, respectively.
Vickers hardness measurements were performed to determine the ageing responses at the different ageing temperatures. The applied load of hardness measurements was 9.8 N and indentation time was 15 s. The average hardness value was obtained from 20 different measurements.
The specimens for TEM were cut parallel to the RD/ND (normal direction) plane. And the TEM foils were prepared by a twin-jet electro polishing unit with a solution of 30% nitric acid and 70% methanol at –30 °C. High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) was performed on an FEI TECNAI G2 F20 instrument with 200 kV acceleration voltage. Selected area electron diffraction (SAD) patterns and dark field (DF) micrographs were obtained on a JEM-2100 instrument with an operating voltage of 200 kV.
3 Results
3.1 Age hardening curves
The solute treated samples are subjected to a 4% pre-strain and subsequently aged at 120, 160 and 200 °C for different times, respectively. The hardness values of these samples are used to obtain age hardening curves with respect to different ageing temperatures, as shown in Figure 1. It is worth mentioning that a 4% pre-strain before artificial ageing was performed in order to promote the precipitation of T1, which is the major strengthening phase in the aged Al–Cu–Li alloy [23]. As illustrated in Figure 1, the ageing hardening curves show an expected increase in ageing kinetics with increasing ageing temperature. The hardness of the samples aged at 120 and 160 °C behaves in a similar manner. The hardness values increase significantly with ageing time, and reach the peak values of about HV 134 at 144 h and about HV 148 at 36 h for samples aged at 120 and 160 °C, respectively. Subsequently, with a further increase in the ageing time, the hardness values remain steady for both conditions. While, a different scenario occurs when the samples are aged at 200 °C. The hardness values increase sharply and reach its maximum value of HV 142 at 7 h, followed by an over-ageing stage with decreasing hardness, which is attributed to the thickening of T1 precipitates [24]. In addition, it can be observed from Figure 1 that the samples peak aged at 160 °C exhibit the highest Vickers hardness. In order to explore the possible reasons for the different ageing responses at 120, 160 and 200 °C, the corresponding microstructures of the samples at the peak aged stage were characterized, as shown below.
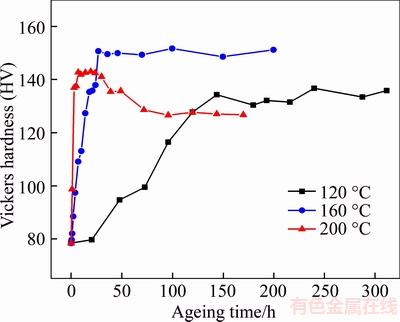
Figure 1 Age hardening curves as a function of ageing temperature.
3.2 TEM analysis
It is well known that the precipitates in Al–Cu–Li alloys can be identified by the diffraction patterns taken along the <110>Al zone axis [7, 25–27]. However, the SAD patterns obtained from Al–Cu–Li alloys are generally complicated due to the existence of several phases. Therefore, for a better understanding of the actual SAD patterns obtained from our samples, the typical SAD patterns of T1, θ'/GP zones and δ'/β' (Al3Zr) phases taken along the <110>Al zone axis are sketched and presented in Figure 2. Moreover, the placement of the aperture marked by the yellow empty circle in Figure 2 will be used for the DF micrographs, which is in order to analyze the size and distribution of δ' precipitates.
Figure 3 shows the microstructures of the Al–Cu–Li–Mn–Zr alloy aged at 120 °C for 144 h corresponding to peak aged condition. According to the diffraction pattern in Figure 3(a), it is easy to identify that the sample peak aged at 120 °C contains T1, θ'/GP zones and δ' phases. Obviously, the diffraction spots at 1/2 {002} positions corresponding to δ' phase are much stronger. The corresponding DF image of δ' precipitates is shown in Figure 3(b), confirming the presence of a large number of δ' precipitates. The δ' precipitates are lenticular or long in shape and distribute along the {100} planes, which can be deduced that δ' are flanking θ' or GP zones [5, 7]. These δ' precipitates are approximately 20–50 nm in length.
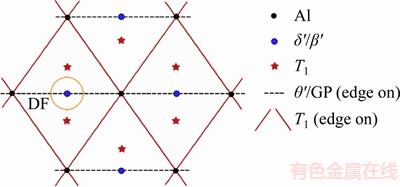
Figure 2 Scheme of spots and streaks expected from main precipitate phases in <110>Al zone axis
Moreover, the intense streaks along {002} reflections corresponding to θ'/GP zones are also observed in Figure 3(a). It is worthy to mention that since the θ' and GP zones are difficult to be distinguished at low magnification STEM image or by SAD patterns, these precipitates are referred to as θ'/GP zones in this work. To further characterize their size and distribution, the STEM imaging was conducted along the <100>Al zone axis, as shown in Figure 3(d). This STEM image in Figure 3(c) indicates the existence of a high density of θ'/GP zones marked by the red arrows. Furthermore, it should be noted that there also exist large quantities of cubic phase in Figure 3(c), as marked by the yellow arrow. The cubic phase is parallel with needle-like θ'/GP phase, so its habit plane is also along the {100}Al matrix plane. The similar cubic phase has been reported previously in the aged Al–Cu–Li–Zn–Mg–Mn–Zr alloy [17–19], in which such cubic phase is considered to be σ (Al5Cu6Mg2) phase. In the aged Al–Cu–Li–In alloy, the similar cubic phase was also found by PAN et al [20] with the combination of high-resolution electron microscopy (HREM) and atom probe tomography (APT). They suggested the cubic phase is Al5Cu6Li2 (named χ phase), which is a variant of Al5Cu6Mg2 with Li substituting the Mg positions. In our alloy, since the chemical composition is Al–2.8Cu–1.4Li– 0.3Mn–0.14Zr (wt%) without any Mg element added, it can be deduced that the cubic phase appeared in Figure 3(c) may be the χ phase. While the corresponding diffraction pattern of the χ phase is not shown in Figure 3(a), which may be owing to its tiny dimension.

Figure 3 TEM images of samples aged at 120 °C for 144 h (T8 condition):(T1, θ'/GP zones, χ and β ' phases are marked by blue, red, yellow and green arrows, respectively)
Finally, the diffraction streaks along {111} reflections and the weak diffraction spots at 1/3 {022} positions suggest that the sample contains a relatively small amount of T1 precipitates as compared to θ'/GP zones and δ' phases, which is also consistent with the observation in Figure 3(d). In addition, some χ phase and β' phase are also characterized in Figure 3(d). It is well known that the distribution of β' phase is usually very heterogeneous in TEM experimental observations [28], showing that high density of β' phase may appear in some places, while few such precipitations can be seen in other places.
Figures 4 and 5 illustrate the microstructures of the Al–Cu–Li–Mn–Zr alloy aged at 160 °C for 36 h and 200 °C for 7 h, respectively, corresponding to the peak aged stages. The SAD patterns with <110>Al zone axis in Figures 4(a) and 5(a) reveal the presence of the precipitates of T1, δ' and θ'/GP zones in these two conditions. However, in both conditions, the reflections of T1 phase are much more intense than those of the δ' and θ'/GP zones, indicating that the amount of the T1 phase is much larger than that of δ' and θ'/GP zones. Such microstructural features can be further confirmed by the observations in the DF images and the STEM images in Figures 4(b)–(d) and 5(b)–(d). Moreover, it should be noted that compared to the precipitation of χ phase at 120 °C samples, the amount of χ phase in 160 and 200 °C samples seems to be rather smaller. As a consequence, the χ phase in these two conditions is not easy to be characterized. Therefore, the microstructures of the samples peak aged at 160 and 200 °C are dominated by T1 precipitates with a minor fraction of δ' and θ'/GP zones. Additionally, it is also worthy to mention that some distinct difference exists between these 160 and 200 °C samples in terms of the size and fraction of T1 platelets, therefore the quantitative analysis on the T1 platelets is necessary.

Figure 4 TEM images of samples aged at 160 °CC for 36 h (T8 condition):

Figure 5 TEM images of samples aged at 200 °C for 7 h (T8 condition):
Measurements of the T1 plates’ diameter and area fraction with increasing ageing temperature were made manually from STEM-HAADF images, and such measurement way was also used in some previous studies [29, 30]. Figures 6(a)–(c) show the diameter distribution of T1, which exhibits an increase in the width of the precipitate diameter frequency distributions with increasing ageing temperature, suggesting a decrease in the homogeneity of the diameter of the T1 precipitates. Figure 6(d) shows the evolution of T1 plates’ average plate diameter and area fraction in these peak aged samples with increasing ageing temperature. This reveals an apparent increase in the average plate diameter of T1 with increasing ageing temperature. While the area fraction of T1 significantly increases from 120 to 160 °C first, and then slightly drops from 160 to 200 °C. In order to accurately measure the area fraction, we took care to use comparable sample thicknesses for the three peak aged samples, i.e., the vicinity of the thin foil edge at similar magnification. Besides, a higher magnification is also necessary to detect the thickness of T1. The results present that in the three T8 conditions a few of multiple cell thickness precipitates exist in every image, which is consistent with the study of GUMBMANN et al [13]. Nevertheless, to keep the calculation of area fraction simple, we consider T1 has a single cell thickness of 0.94 nm [31].
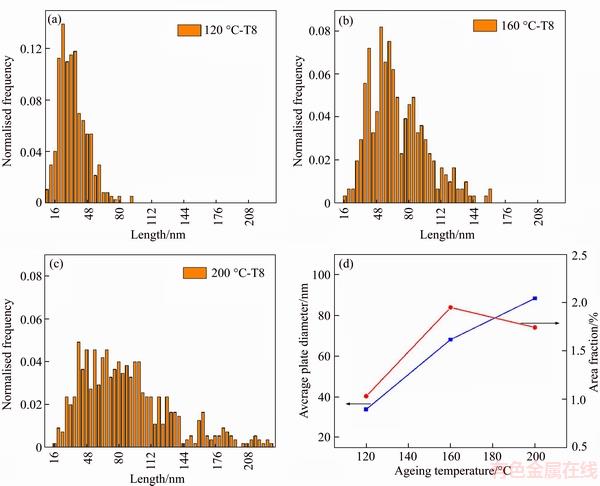
Figure 6 Evolution of diameter distribution of T1 for three ageing temperatures in T8 conditions at 120 °C (a), 160 °C (b), 200 °C (c) and evolution of average plate diameter and area fraction with increasing ageing temperature (d)
4 Discussion
4.1 Precipitates in Al–Cu–Li–Mn–Zr alloy aged at 120 °C
It is generally known that the chemical composition, pre-deformation and ageing treatment can significantly affect the precipitation of Al–Cu– Li alloys. In this work, it is found that the ageing temperature could significantly impact the precipitates of the Al–2.8Cu–1.4Li–0.3Mn–0.1Zr (wt%) alloy. When aged at 120 °C for 144 h, this alloy prefers to form the precipitations of δ', GP zones/θ' and χ phases rather than T1, as illustrated in Figure 3. While when aged at 160 °C for 36 h and 200 °C for 7 h, the precipitation for these samples is dominated by a large amount of T1 phases (Figures 4 and 5).
The influence of ageing temperature on the precipitates of Al–Cu–Li alloys has been reported previously [21, 22, 32]. According to these publications, lower ageing temperatures would result in the major precipitates of δ' and θ' phases, while higher ageing temperatures lead to the precipitate of T1 phase in large numbers. As proposed by CHEN et al [32], such phenomenon may be ascribed to the fact that when aged at low ageing temperatures, the nucleation of δ' and GP zones is very rapid due to their lower nucleation activation energy, while the nucleation of T1 and θ' phases usually needs longer time. Accordingly, at the early stage of ageing treatment, a large amount of δ' and GP zones would precipitate, which reduces the solute concentration in the matrix. As a consequent, the precipitate of T1 is diminished. Additionally, with increasing the ageing time, the amount of θ' phase might increase, as some GP zone could act as the nucleation sites of θ'' phase, which could evolve into θ' phase subsequently [33].
Except for δ' and GP zones/θ', it should be noted that a large amount of χ phase is also formed when peak aged at 120 °C, as shown in Figure 3. According to Ref. [20], the χ phase can be considered as a variant of σ (Al5Cu6Mg2) with Li substituting the Mg positions. Since the σ is an equilibrium phase [34], it can be deduced that the χ phase may also be an equilibrium phase. As a consequence, the nucleation of the χ phase would need longer time, just similar with that of T1 and θ' phases [32]. Therefore, for the alloy studied in this work, when aged at 120 °C, a large amount of δ' and GP zones are formed first, and then T1, θ' and χ are precipitated. Based on the formulations of these phases, the Li: Cu atomic ratio of T1, θ' and χ is 1:1, 0:1 and 1:3, respectively. Therefore, when the Li: Cu atomic ratio is greater than 1.0, the alloy prefers to form the precipitations of T1 phases [12], while the θ' and χ phases will be the mainstay on the contrary. In other words, which ones of these three phases would be dominated depending on the remaining Li: Cu atomic ratio after the precipitates of δ' and GP zones. In this work of the Al–2.8Cu–1.4Li–0.3Mn–0.14Zr (wt%) alloy, by calculation and analysis, the Li: Cu atomic ratio is approximately 3:1. The calculation of atomic ratio is based on the following assumptions that: 1) the Li solubility is 2.0 at% and the Cu solubility is negligible [35]; 2) the Cu content is adjusted due to the presence of Mn, which is used to form Al20Mn3Cu2 dispersoid. When aged at 120 °C, a large amount of δ' and GP zones are formed first. As the formation of δ' and GP zones occurs and matures by continuously absorbing Cu and Li, the solute concentration in matrix would decrease significantly, especially the Li solute concentration due to the lower diffusion coefficient for Li atom in Al matrix [36]. This may result in the Li: Cu atomic ratio less than 1.0 and in turn promotes the precipitation of θ' and χ. As such, the T1 phase has the lowest area fraction and the smallest average plate diameter when peak aged at 120 °C, which is also consistent with the static analysis in Figure 6.
4.2 Precipitates in Al–Cu–Li–Mn–Zr alloy aged at 160 °C and 200 °C
It is generally known that the chemical composition, pre-deformation and ageing treatment can significantly affect the precipitation of Al–Cu–Li alloys. In this work, it is found that the ageing temperature could significantly impact the precipitates of the Al–2.8Cu–1.4Li–0.3Mn–0.1Zr (wt%) alloy. When aged at 120 °C for 144 h, this alloy prefers to form the precipitations of δ', GP zones/θ' and χ phases rather than T1, as illustrated in Figure 3. While when aged at 160 °C for 36 h and 200 °C for 7 h, the precipitation for these samples is dominated by a large amount of T1 phases (Figures 4 and 5).
Unlike the sample peak aged at 120 °C, the samples peak aged at 160 °C and 200 °C are both dominated by T1 phase with a minor fraction of GP zones/θ' and δ'. However, the distinct difference between these two samples is the average plate diameter and area fraction of T1 phase. As shown in Figure 6, the samples peak aged at 200 °C usually exhibit a larger average plate diameter and a smaller area fraction of T1 phase, as compared to the samples peak aged at 160 °C. Such phenomenon may be explained from the perspective of the nucleation rate of T1. In this work, the T1 precipitates would nucleate mostly on the dislocations introduced by a pre-stretching operation. Hence, the nucleation rate of T1 may be estimated based on the equation for heterogeneous nucleation rate [21]
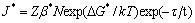 (1)
(1)
where J* is nucleation rate and ΔG* represents the driving force (volume free energy change). In this equation, only ΔG* would be affected by ageing temperature. As the driving force increases, the nucleation rate of T1 will be promoted. Generally, the low ageing temperature (160 °C) would result in the large driving force for the precipitation because of the high undercooling. As a consequence, the nucleation of T1 would be easier at ageing temperature of 160 °C than that at 200 °C. This would result in a smaller average plate diameter of T1 and a smaller average distance between T1 plates at ageing temperature of 160 °C [35]. Moreover, as proposed by DORIN et al [37], there exists a significant supersaturation in Cu and Li between the T1 precipitating phases even in T8 condition. Therefore, it can be further deduced that since the samples peak aged at 200 °C possess a greater average distance between T1 plates, more supersaturation in Cu and Li would be still present between the T1 plates, which is not used to precipitate the T1 phase. This phenomenon in turn leads to the lower area fraction of T1 precipitates in the samples peak aged at 200 °C.
In addition, it is worth mentioning that the fraction evolution of T1 with ageing temperature here is in contradiction with previous studies [24, 37], where an increase in volume fraction of T1 occurs with raising ageing temperature. In their studies, a duplex ageing treatment was performed in 2198 alloy, i.e. the samples heat treated at 155 °C for 18 h and then at 190 °C for different time. When the ageing temperature increases to 190 °C, the alloy is thought to be in over-aged state, and the thickening process of T1 phase is activated which is accomplished by absorbing the remaining Cu and Li solute atoms. This can lead to the increase in volume fraction of T1 precipitate when the ageing temperature is increased to 190 °C. However, in this work, the microstructure of the peak aged state at 200 °C was characterized, in which the thickening process of T1 phase did not occur.
5 Conclusions
The effect of ageing temperatures on precipitation in Al–Cu–Li–Mn–Zr alloy has been investigated by using a combination of TEM and Vickers hardness tester.
1) In the peak aged condition, the samples peak aged at 120 °C contain a large number of δ', θ'/GP zones and χ phases, which are obviously more than T1 phase; whereas the samples peak aged at 160 °C and 200 °C are dominated by T1 with a small fraction of δ' and θ'/GP zones. The different ageing responses at 120 °C are due to the formation of a large amount of δ' precipitates first, which leads to the lower Li: Cu atomic ratio in Al matrix and in turn promotes the precipitation of θ' and χ.
2) The T1 precipitations in the samples peak aged at 120 °C exhibit the lowest area fraction and the smallest average plate diameter, which can be attributed to the fact that a majority of Cu and Li atoms have been used for the precipitation of δ', θ'/GP zones and χ phases.
3) For T1 precipitations in the samples peak aged at 160 °C and 200 °C, with decreasing ageing temperature, the average plate diameter decreases, while the area fraction increases. Such phenomenon is thought to be the consequence of improved nucleation rate of T1 due to the greater driving force for the precipitation at 160 °C.
References
[1] RIOJA R J. Fabrication methods to manufacture isotropic Al-Li alloys and products for space and aerospace applications [J]. Materials Science and Engineering A, 1998, 257(1): 100–107.
[2] LIU Xiao-yan, WANG Zhao-peng, LI Qing-shuai, ZHANG Xi-liang, CUI Hao-xuan, ZHANG Xiao-liang. Effects of pre-deformation on microstructure and properties of Al–Cu–Mg–Ag heat-resistant alloy [J]. Journal of Central South University, 2017, 24(5): 1027–1033.
[3] STARKE E A Jr, STALEY J T. Application of modern aluminum alloys to aircraft [J]. Progress in Aerospace Sciences, 1996, 32(2, 3): 131–172.
[4] YU Cheng, YIN Deng-feng, ZHENG Feng, YU Xin-xiang. Effects of solution treatment on mechanical properties and microstructures of Al–Li–Cu–Mg–Ag alloy [J]. Journal of Central South University, 2013, 20(8): 2083–2089.
[5] YOSHIMURA R, KONNO T J, ABE E, HIRAGA K. Transmission electron microscopy study of the evolution of precipitates in aged Al–Li–Cu alloys: The θ' and T1 phases [J]. Acta Materialia, 2003, 51(14): 4251–4266.
[6] DECREUS B, DESCHAMPS A, de GEUSER F, DONNADIEU P, SIGLI C, WEYLAND M. The influence of Cu/Li ratio on precipitation in Al–Cu–Li–x alloys [J]. Acta Materialia, 2013, 61(6): 2207–2218.
[7] YOSHIMURA R, KONNO T J, ABE E, HIRAGA K. Transmission electron microscopy study of the early stage of precipitates in aged Al–Li–Cu alloys [J]. Acta Materialia, 2003, 51(10): 2891–2903.
[8] KHAN A K, ROBINSON J S. Effect of silver on precipitation response of Al–Li–Cu–Mg alloys [J]. Materials Science and Technology, 2008, 24(11): 1369–1377.
[9] van SMAALEN S, MEETSMA A, de BOER J L, BRONSVELD P M. Refinement of the crystal structure of hexagonal Al2CuLi [J]. Journal of Solid State Chemistry, 1990, 85(2): 293–298.
[10] DONNADIEU P, SHAO Y, de GEUSER F, BOTTON G A, LAZAR S, CHEYNET M, de BOISSIEU M, DESCHAMPS A. Atomic structure of T1 precipitates in Al–Li–Cu alloys revisited with HAADF-STEM imaging and small-angle X-ray scattering [J]. Acta Materialia, 2011, 59(2): 462–472.
[11] DWYER C, WEYLAND M, CHANG L Y, MUDDLE B C. Combined electron beam imaging and ab initio modeling of T1 precipitates in Al–Li–Cu alloys [J]. Applied Physics Letters, 2011, 98(20): 201909.
[12] SILCOCK J M. The structural ageing characteristics of aluminium-copper-lithium alloys [J]. J Inst Met, 1960, 88: 357–364.
[13] GUMBMANN E, LEFEBVRE W, de GEUSER F, SIGLI C, DESCHAMPS A. The effect of minor solute additions on the precipitation path of an Al–Cu–Li alloy [J]. Acta Materialia, 2016, 115(15): 104–114.
[14] HUANG B P, ZHENG Z Q. Independent and combined roles of trace Mg and Ag additions in properties precipitation process and precipitation kinetics of Al–Cu–Li–(Mg)–(Ag)– Zr–Ti alloys [J]. Acta Materialia, 1998, 46(12): 4381–4393.
[15] BOUKOS N, FLOUDA E, PAPASTAIKOUDIS C. The effect of Ag additions on the microstructure of aluminium-lithium alloys [J]. Journal of Materials Science, 1998, 33(12): 3213–3218.
[16] ZHENG Z Q, LIANG S Q, XU H, TAN C Y, YUN D F. Effect of 0.5% silver on the age hardening behaviour of an Al–Li–Cu–Mg–Zr alloy [J]. Journal of Materials Science Letters, 1993, 12(14): 1111–1113.
[17] LI Hong-ying, TANG Yi, ZENG Zai-de, ZHENG Zi-qiao, ZHENG Feng. Effect of ageing time on strength and microstructures of an Al–Cu–Li–Zn–Mg–Mn–Zr alloy [J]. Materials Science and Engineering A, 2008, 498(1, 2): 314–320.
[18] CHEN Zhong-wei, ZHAO Kai, FAN Li. Combinative hardening effects of precipitation in a commercial aged Al–Cu–Li–X alloy [J]. Materials Science and Engineering A, 2013, 588: 59–64.
[19] LI Hong-ying, HUANG Da-sheng, KANG Wei, LIU Jiao-jiao, OU Yang-xun, LI De-wang. Effect of different aging processes on the microstructure and mechanical properties of a Novel Al–Cu–Li Alloy [J]. Journal of Materials Science & Technology, 2016, 32(10): 1049–1053.
[20] PAN Zheng-rong, ZHENG Zi-qiao, LIAO Zhong-quan, LI Shi-chen. New cubic precipitate in Al–3.5Cu–1.0Li–0.5In (wt.%) alloy [J]. Materials Letters, 2010, 64(8): 942–944.
[21] BLANKENSHIP C, STARKE E. Structure-property relationships in Al–Li–Cu–Mg–Ag–Zr alloy X2095 [J]. Acta Metallurgica et Materialia, 1994, 42(3): 845–855.
[22] ZHANG Sai-fei, ZENG Wei-dong, YANG Wen-hua, SHI Chun-ling, WANG Hao-jun. Ageing response of a Al–Cu–Li 2198 alloy [J]. Materials & Design, 2014, 63: 368–374.
[23] NOBLE B, THOMPSON G E. T1 (Al2CuLi) precipitation in aluminium–copper–lithium alloys [J]. Metal Science Journal, 1972, 6(1): 167–174.
[24] DORIN T, DESCHAMPS A, de GEUSER F, SIGLI C. Quantification and modelling of the microstructure/strength relationship by tailoring the morphological parameters of the T1 phase in an Al–Cu–Li alloy [J]. Acta Materialia, 2014, 75(15): 134–146.
[25] DENG Yan-jun, BAI Jian-hui, WU Xiao-dong, HUANG Guang-jie, CAO Ling-fei, HUANG Li. Investigation on formation mechanism of T1 precipitate in an Al–Cu–Li alloy [J]. Journal of Alloys and Compounds, 2017, 723(5): 661–666.
[26] LI Qiong, WAWNER F E. Characterization of a cubic phase in an Al–Cu–Mg–Ag alloy [J]. Journal of Materials Science, 1997, 32(20): 5363–5370.
[27] DESCHAMPS A, GARCIA M, CHEVY J, DAVO B, de GEUSER F. Influence of Mg and Li content on the microstructure evolution of Al–Cu–Li alloys during long- term ageing [J]. Acta Materialia, 2017, 122(1): 32–46.
[28] TSIVOULAS D, ROBSON J D, SIGLI C, PRANGNELL P B. Interactions between zirconium and manganese dispersoid-forming elements on their combined addition in Al–Cu–Li alloys [J]. Acta Materialia, 2012, 60(13, 14): 5245–5259.
[29] DORIN T, DONNADIEU P, CHAIX J-M, LEFEBVRE W, de GEUSER F, DESCHAMPS A. Size distribution and volume fraction of T1 phase precipitates from TEM images: Direct measurements and related correction [J]. Micron, 2015, 78: 19–27.
[30] RODGERS B I, PRANGNELL P B. Quantification of the influence of increased pre-stretching on microstructure- strength relationships in the Al–Cu–Li alloy AA2195 [J]. Acta Materialia, 2016, 108(15): 55–67.
[31] KANG S J, KIM T H, YANG C W, LEE J I, PARK E S, NOH T W, KIM M. Atomic structure and growth mechanism of T1 precipitate in Al–Cu–Li–Mg–Ag alloy [J]. Scripta Materialia, 2015, 109: 68–71.
[32] CHEN P, BHAT B. Time-temperature-precipitation behavior in Al-Li alloy 2195 [R]. Huntsville: NASA Marshall Space Flight Center, 2002.
[33] LORIMER G, NICHOLSON R. Further results on the nucleation of precipitates in the Al-Zn-Mg system [J]. Acta Metallurgica, 1966, 14(8): 1009–1013.
[34] RINGER S P, SOFYAN B T, PRASAD K S, QUAN G C. Precipitation reactions in Al–4.0Cu–0.3Mg (wt.%) alloy [J]. Acta Materialia, 2008, 56(9): 2147–2160.
[35] GABLE B M, ZHU A W, CSONTOS A A, STARKE E A Jr. The role of plastic deformation on the competitive microstructural evolution and mechanical properties of a novel Al-Li-Cu-X alloy [J]. Journal of Light Metals, 2001, 1(1): 1–14.
[36] del CASTILLO P E J R D, REISCHIG P, van der ZWAAG S. Tailoring of Ostwald ripening behaviour in multicomponent Al alloys [J]. Scripta Materialia, 2005, 52(8): 705–708.
[37] DORIN T, DESCHAMPS A, de GEUSER F, LEFEBVRE W, SIGLI C. Quantitative description of the T1 formation kinetics in an Al–Cu–Li alloy using differential scanning calorimetry, small-angle X-ray scattering and transmission electron microscopy [J]. Philosophical Magazine, 2014, 94(10): 1012–1030.
(Edited by FANG Jing-hua)
中文导读
时效温度对Al–Cu–Li–Mn–Zr合金析出相的影响
摘要:本文采用显微硬度计和透射电子显微镜(TEM)表征手段,研究不同时效温度(120、160、200 °C)对Al–Cu–Li–Mn–Zr合金析出行为的影响。通过分析不同时效温度下的时效硬度曲线可以得出,随着时效温度的升高,析出相的析出速度加快。此外,由TEM表征结果可知,当时效温度为120 °C时,峰值态样品的析出相主要为δ'相(Al3Li)、GP区/θ'相(Al2Cu)和χ相(Al5Cu6Li2),其析出量明显高于T1相;当时效温度为160 °C和200 °C时,样品峰值态的析出相主要为T1相,并有较少的δ'相和GP区/θ'相,而χ相几乎不存在。对T1相进行定量统计可知,与时效温度为160 °C的峰值态样品相比,时效温度为200 °C时峰值态样品的主要析出相T1相的直径更大,且面积分数更小,文本对这些现象的产生原因进行了阐述和讨论。
关键词:时效温度;析出相;Al–Cu–Li–Mn–Zr合金;T1相;χ相(Al5Cu6Li2)
Foundation item: Project(2016YFB0300901) supported by the National Key R&D Program of China; Project(51421001) supported by the National Natural Science Foundation of China; Project(2018CDJDCL0019) supported by the Fundamental Research Funds for the Central Universities, China
Received date: 2017-01-13; Accepted date: 2017-04-05
Corresponding author: HUANG Guang-jie, PhD, Professor; Tel: +86–23–65120787; E-mail: gjhuang@cqu.edu.cn; ORCID: 0000-0003- 4237-1523; CAO Ling-fei, PhD, Professor; Tel: +86–23–65120781; E-mail: caolingfei@cqu.edu.cn; ORCID: 0000-0002-9255-3491

