高砷烟尘NaOH-Na2S碱浸过程的金属元素浸出行为
来源期刊:中国有色金属学报(英文版)2016年第2期
论文作者:郭学益 易宇 石靖 田庆华
文章页码:575 - 580
关键词:高砷烟尘;碱性浸出;浸出行为;砷
Key words:high arsenic dust; alkaline leaching; leaching behavior; arsenic
摘 要:采用NaOH-Na2S碱浸方法实现了高砷烟尘中砷的选择性浸出。在砷浸出过程中,探讨了碱料比、硫化钠用量、浸出温度、浸出时间和液固比对各金属元素浸出率的影响。结果表明:在碱料质量比为0.5,硫化钠用量为0.25 g/g,浸出温度为90 °C,浸出时间为2 h,液固比为5:1的优化条件下实现了砷与其他金属的有效分离。在优化条件下,砷、锑、铅、锡和锌的平均浸出率为92.75%, 11.68%, 0.31%, 29.75%和36.85%。NaOH-Na2S碱性浸出过程提供了一种从含砷烟尘中脱除砷的简单有效的方法,同时产生具有利用价值的铅渣。
Abstract: Arsenic is selectively extracted from high-arsenic dust by NaOH-Na2S alkaline leaching process. In the leaching arsenic process, the effects of alkali-to-dust ratio, sodium sulfide addition, leaching temperature, leaching time and liquid-to-solid ratio on metals leaching efficiencies were investigated. The results show that the arsenic can be effectively separated from other metals under the optimum conditions of alkali/dust mass ratio of 0.5, sodium sulfide addition of 0.25 g/g, leaching temperature of 90 °C, leaching time of 2 h, and liquid-to-solid ratio of 5:1 (mL/g). Under these conditions, the average leaching efficiencies of arsenic, antimony, lead, tin and zinc are 92.75%, 11.68%, 0.31%, 29.75% and 36.85%, respectively. The NaOH-Na2S alkaline leaching process provides a simple and highly efficient way to remove arsenic from high-arsenic dust, leaving residue as a suitable lead resource.
Trans. Nonferrous Met. Soc. China 26(2016) 575-580
Xue-yi GUO1,2, Yu YI1,2, Jing SHI1, Qing-hua TIAN1,2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Cleaner Metallurgical Engineering Research Center, Nonferrous Metal Industry of China, Central South University, Changsha 410083, China
Received 9 February 2015; accepted 1 July 2015
Abstract: Arsenic is selectively extracted from high-arsenic dust by NaOH-Na2S alkaline leaching process. In the leaching arsenic process, the effects of alkali-to-dust ratio, sodium sulfide addition, leaching temperature, leaching time and liquid-to-solid ratio on metals leaching efficiencies were investigated. The results show that the arsenic can be effectively separated from other metals under the optimum conditions of alkali/dust mass ratio of 0.5, sodium sulfide addition of 0.25 g/g, leaching temperature of 90 °C, leaching time of 2 h, and liquid-to-solid ratio of 5:1 (mL/g). Under these conditions, the average leaching efficiencies of arsenic, antimony, lead, tin and zinc are 92.75%, 11.68%, 0.31%, 29.75% and 36.85%, respectively. The NaOH-Na2S alkaline leaching process provides a simple and highly efficient way to remove arsenic from high-arsenic dust, leaving residue as a suitable lead resource.
Key words: high arsenic dust; alkaline leaching; leaching behavior; arsenic
1 Introduction
Arsenic is toxic and carcinogenic to the human beings [1,2]. With the improving of environmental standard, the arsenic containing substances have to be treated as safe materials [3]. The high-arsenic dust is generated in pyrometallurgical process, which contains normally large amounts of valuable metals such as lead, antimony, zinc and indium. The high-arsenic dust not only represents potential risk of environmental contamination but also is a secondary resource, which should be properly disposed and comprehensively utilized to obtain the maximum economic and environmental benefits [4,5]. If the dust is directly sent back to the smelter to recycle the valuable metals, it will reduce the efficiency of equipment and enhance the required energy consumption of smelting process [6-8]. Thus, it is necessary to treat the high-arsenic dust individually to remove arsenic and recover the valuable metals.
The conventional treatment of arsenic containing mineral can be classified into two methods: pyrometallurgical and hydrometallurgical processes. The pyrometallurgical process generally involves anoxic roasting or reducing roasting at 300-600 °C, in which arsenic is volatilized as the form of As2O3 and then collected [9,10]. Nevertheless, the collected As2O3 dust requires further processing, and the volatilized arsenic may result in a source of secondary pollution [11,12]. Therefore, most studies focus on hydrometallurgical treatments of the dusts, using acid or alkaline reagents [13]. In most hydrometallurgical processes, the arsenic is extracted into solution by hot water leaching, acid leaching, alkali leaching and pressure leaching [14-16], then the arsenic is separated and precipitated from the leaching liquid by lime, ferric salt and sodium sulfide [17-19].
It is difficult to separate arsenic from high-arsenic dust by roasting-volatilizing, since arsenic, lead, zinc and antimony will be volatilized as mixture dust. On the other hand, the highly toxic arsenic gas might be produced during the sulfuric acid leaching [14]. Therefore, a combination of hydrometallurgical and pyrometallurgical processes is chosen for removal of arsenic and comprehensive utilization of valuable metals from high-arsenic dust [20]. Firstly, the arsenic is selectively leached and separated from high-arsenic dust, then the residue is sent back to the smelter to recover lead, antimony and zinc. The arsenic in leach liquor is recovered in the form of sodium arsenate by oxidation-crystallization process, and the filtrate is recycled to leaching step. The obtained sodium arsenate is transformed to calcium arsenate, which is solidified with cement and dumped in landfill. In this work, the NaOH-Na2S alkaline leaching is adopted to extract arsenic from high-arsenic dust. The effects of processing parameters including NaOH/dust mass ratio, sodium sulfide added, leaching temperature, leaching time and liquid-to-solid ratio on leaching behaviors of arsenic, lead, antimony, tin and zinc are investigated.
2 Experimental
2.1 Materials
The high-arsenic dust, which was generated in copper dross pyrometallurgical process in blast furnace, was collected from a lead smelter in Guangxi, China. The dust was dried at 105 °C for 24 h, and ground to pass through a 150 μm sieve. The chemical analysis results of main elements in high-arsenic dust are listed in Table 1. The major elements are lead, antimony, arsenic, sulfur, tin, zinc, iron and copper. The chemical phase analysis of arsenic is shown in Table 2 by selective dissolution procedure [21]. As presented in Table 2, over 50% of arsenic exists in the form of lead arsenate. All chemical reagents used in all experiments were of analytical grade, manufactured by Xi-long Chemical Company, China, and deionized water was used in all experiments.
Table 1 Chemical composition of high-arsenic dust (mass fraction, %)

Table 2 Chemical phase analysis of arsenic in high-arsenic dust
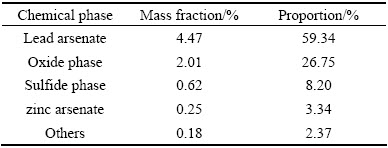
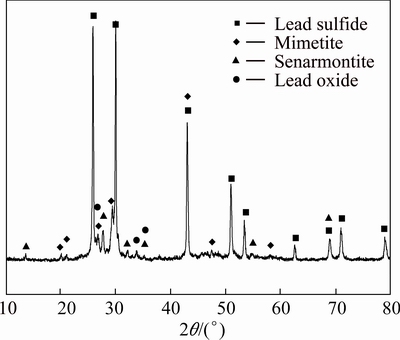
Fig. 1 XRD pattern of high-arsenic dust
As presented in Fig. 1, the XRD pattern of high-arsenic dust shows that the dust consists of lead sulfide (PbS), mimetite (Pb5(AsO4)3 OH), senarmontite (Sb2O3) and lead oxide (Pb3O4).
2.2 Experimental methods
The alkaline leaching experiments were performed in a 500 mL four-necked round-bottomed flask equipped with two-flat-blade mechanical agitators, water-cooled condenser and thermometer. The reflux condenser was used to prevent evaporation of the solution. During experiments, the flask was placed in a thermostatic water bath and the temperature was controlled accurately within ±0.5 °C.
In batch experiments, 60 g high-arsenic dust was mixed with alkaline leaching reagent containing NaOH, Na2S and a certain amount of water, and then the slurry was leached at a stirring speed of 400 r/min at a certain temperature. Finally, the slurry was filtered; the residue was washed by hot water. The leach liquor mixed with the washing water was sent to chemical analysis. The washed residue was dried in an oven at 105 °C overnight, pulverized and sent for analysis. The extraction of arsenic was calculated on the basis of residue composition and the extraction of other metals was calculated on the basis of the mixed solution composition.
2.3 Characterization
The phases of high-arsenic dust and leach residue were analyzed by X-ray diffraction (Siemens D5000, Cu Kα λ=1.542×10-10 m), and the contents of arsenic in mixed solution and residue were determined by atomic fluorescent photometer (AFS-2202E). The contents of other metals were determined by ICP-MS (IRIS Interpid II XSP, Thermo Electron Corporation).
3 Results and discussion
3.1 Effect of leaching system
Mimetite, arsenoxide and lead oxide in high-arsenic dust will dissolve in alkaline media, and the metal leaching efficiency is influenced by the conditions. The probable main reactions in the leaching process are as follows:
Pb5(AsO4)3OH+19NaOH=5Na2PbO2+3Na3AsO4+10H2O (1)
Pb3O4+8NaOH=Na2PbO2+2Na3PbO3+4H2O (2)
As2O3+6NaOH=2Na3AsO3+3H2O (3)
As2O5+6NaOH=2Na3AsO4+3H2O (4)
Firstly, the single alkaline leaching of high-arsenic dust was carried out under the following conditions: the alkali-to-dust mass ratio of 0.5, the reaction temperature of 80 °C, the reaction time of 2 h, the liquid-to-solid ratio of 5:1 (mL/g), and the stirring speed of 400 r/min. Under these conditions, the extraction rates of arsenic, lead, antimony, zinc and tin are 66.36%, 11.05%, 9.03%, 21.28% and 25.85%, respectively. The contents of arsenic and lead in the leach liquor are 8.71 g/L and 9.55 g/L, respectively. Due to the ionic equilibrium existed between As(V) and Pb(II), it is difficult to reach the high arsenic extraction and the low leaching of valuable metals simultaneously [19]. Thus, the key to effectively extract arsenic from high-arsenic dust is to decrease the concentrations of Pb(II) in the solution. Metal sulphide precipitation is an important method for recovering valuable metals due to the low solubility of metal sulphide precipitates [22]. The addition of sodium sulfide can effectively decrease the concentrations of lead in the solution.
Na2PbO2+Na2S+2H2O=PbS+4NaOH (5)
2Na3PbO3+3Na2S+6H2O=2PbS+12NaOH+S (6)
So, much higher extraction of arsenic and separation arsenic from valuable metals cannot be easily achieved under the single alkaline leaching conditions. The mixed alkaline leaching experiments are further conducted.
3.2 Effect of alkali/dust mass ratio on metal extraction rate
Alkali/dust mass ratio is the most important factor in metals extraction. Figure 2 shows the effects of alkali/dust ratio on the extraction rate of metals in the leaching process in the alkali/dust ratio range of 0.1-1.
It can be seen from Fig. 2 that the extraction rate of tin increases with the increase of alkali/dust ratio from 0.1 to 0.3 and raise slightly with further increasing alkali/dust ratio. The extraction rates of lead, zinc and antimony increase steadily with the increase of alkali/dust ratio. The arsenic extraction efficiency obviously increases with the increase of alkali/dust ratio from 0.1 to 0.6 and reaches the maximum of 89.26%, and then decreases with the further increasing alkali/dust ratio. On one hand, since the solubility of lead or zinc arsenite and arsenate is very low, for example, 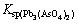 =4.0×10-36,
=4.0×10-36, 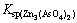 =2.8×10-28, an ionic equilibrium between As(V) and metal ions Pb(II) and Zn(II) exists in the solution [19]. On the other hand, the solubility of As2O5 decreases when content of Na2O increases [14]. Therefore, the extraction of arsenic is inhibited at high alkali/dust ratio. For the purpose of high extraction of arsenic and effective separation of arsenic from other elements, the appropriate alkali/dust mass ratio is determined as 0.5.
=2.8×10-28, an ionic equilibrium between As(V) and metal ions Pb(II) and Zn(II) exists in the solution [19]. On the other hand, the solubility of As2O5 decreases when content of Na2O increases [14]. Therefore, the extraction of arsenic is inhibited at high alkali/dust ratio. For the purpose of high extraction of arsenic and effective separation of arsenic from other elements, the appropriate alkali/dust mass ratio is determined as 0.5.
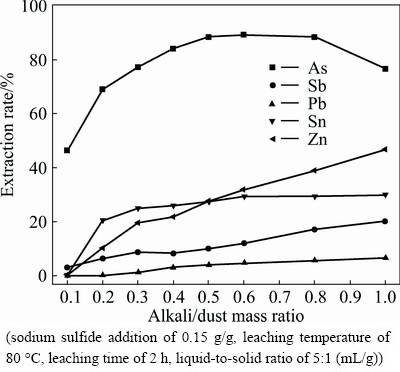
Fig. 2 Effect of alkali/dust mass ratio on metal extraction rate
3.3 Effect of sodium sulfide on metal extraction rate
The addition of sodium sulfide also plays a great significant role in leaching process. Sodium sulfide effects on the extraction rates of metals in the leaching process are shown in Fig. 3.
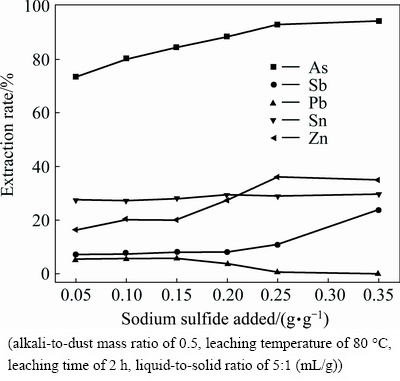
Fig. 3 Effect of sodium sulfide on metal extraction rate
From Fig. 3, the extraction rate of tin does not change obviously with the sodium sulfide. The extraction rate of lead decreases from 5.57% to 0.12% with increasing sodium sulfide. The leaching efficiencies of arsenic and zinc obviously increase with the increase of sodium sulfide and reach 92.81% and 36.03%, respectively, under sodium sulfide amount of 0.25 g/g. Further increasing sodium sulfide addition, the leaching efficiencies remain relatively steady. Along with the increase of sodium sulfide, the precipitation of Pb(II) with S2- releases free OH-(Eqs. (5) and (6)), resulting in the massive dissolution of zinc. On the other hand, the decreased Pb(II) concentration in the solution results in accelerating decomposing of mimetite (Eqs. (1)). However, once the sodium sulfide added surpasses a certain amount, excessive S2- will react with antimony, which leads to substantial increase in antimony concentration. So, the extraction of antimony increases negligibly before sodium sulfide reaching 0.2 g/g, but the leaching efficiency of antimony increases rapidly from 8.19% to 24% when the sodium sulfide increases from 0.2 g/g to 0.35 g/g. Based on the above results, the appropriate addition of sodium sulfide is chosen to be 0.25 g/g.
3.4 Effect of leaching temperature on metal extraction rate
The effect of leaching temperature on the extraction rates of metals is presented in Fig. 4. It can be seen that, the extraction rate of zinc does not change obviously as the leaching temperature increases. The leaching temperature has a significant effect on the lead dissolution, which decreases from 2.53% to 0.19% with leaching temperature increasing. The leaching efficiencies of arsenic, antimony and tin increase with leaching temperature, which can be due to the solubility change of arsenic, antimony and tin at different temperatures. With the increase of leaching temperature from 30 °C to 90 °C, the extraction rates of arsenic, antimony and tin increase from 58.16%, 8.76% and 23.73% to 92.36%, 12.26% and 28.87%, respectively. Given consideration on arsenic extraction and energy consumption, 90 °C is determined to be the optimum leaching temperature.
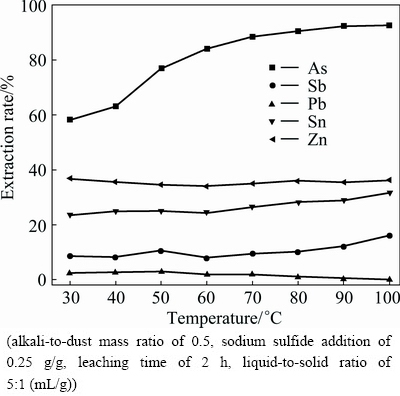
Fig. 4 Effect of leaching temperature on metal extraction rate
3.5 Effect of leaching time on metal extraction rate
Figure 5 presents the effect of leaching time on the leaching efficiencies of arsenic, antimony, lead, zinc and tin. It can be seen that the extractions of antimony and tin do not change obviously with the leaching time. The extraction rates of arsenic and zinc increase apparently with the increase of leaching time and reach 92.88% and 37.7%, respectively, when the time is up to 2 h, and remain relatively stable with further increasing. The extraction rate of lead decreases from 1.56% to 0.22% with the leaching time increasing from 0.5 to 2 h. Further prolonging leaching time, the extraction has only negligible change. Given higher arsenic extraction and lower energy consumption, the leaching time is fixed at 2 h.
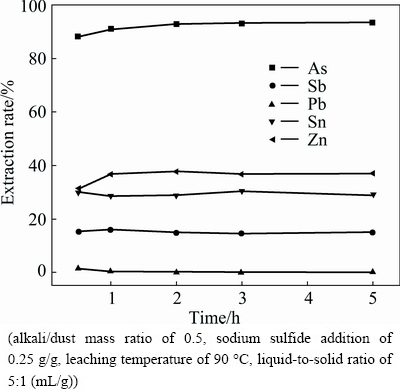
Fig. 5 Effect of leaching time on metal extraction rate
3.6 Effect of liquid-to-solid ratio on metal extraction rate
The effect of liquid-to-solid ratio on the leaching efficiencies of arsenic, antimony, lead, zinc and tin in leaching process is shown in Fig. 6.
It can be seen that the extraction rate of lead does not change obviously with the liquid-to-solid ratio. The extraction rate of arsenic apparently increases with the increase of liquid-to-solid ratio from 3 to 5 and reaches the maximum of 91.24%. Further increasing has a marginal effect on arsenic extraction rate. Meanwhile, the antimony, tin and zinc show an opposite leaching behavior to the arsenic. The leaching efficiencies of antimony, tin and zinc decrease from 26.88%, 49.51% and 58.18% to 5.62%, 14.18% and 15.16%, respectively, with the range of liquid-to-solid ratio from 3 to 10, which are attributed to the decrease of OH- concentration in leach liquor. However, the decrease of loss of valuable metals by increasing liquid-to-solid ratio is accompanied with the decrease of production capacity. Therefore, the optimum liquid-to-solid ratio is fixed at 5:1(mL/g).
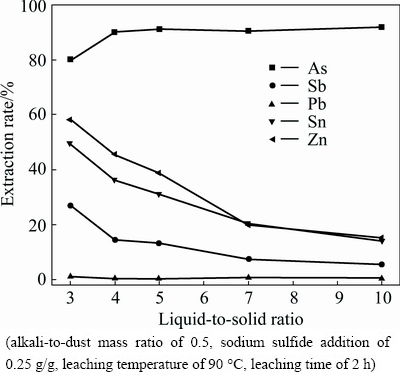
Fig. 6 Effect of liquid-to-solid ratio on metal extraction rate
3.7 Optimum conditions and residue characterization
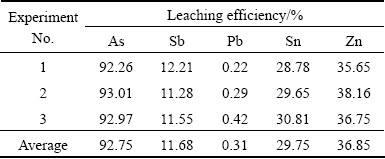
Base on the results of factorial experiments, the optimum conditions are determined as: alkali/dust mass ratio of 0.5, sodium sulfide addition of 0.25 g/g, leaching temperature of 90 °C, leaching time of 2 h, and liquid-to- solid ratio of 5:1 (mL/g). The optimum experiment was repeated three times, and the results are listed in Table 3. The average leaching efficiencies of arsenic, antimony, lead, tin and zinc are 92.75%, 11.68%, 0.31%, 29.75% and 36.85%, respectively.
Table 3 Results of optimum leaching experiments
The chemical composition and XRD analysis of the leach residue are listed in Table 4 and Fig. 7. The XRD pattern of leach residue shows that the main phases in the leach residue are galena (PbS), mopungite (NaSb(OH)6) and senarmontite (Sb2O3), and no phase containing arsenic is detected, which prove that the removal of arsenic from high-arsenic dust is good. The contents of lead and antimony in leach residue are 59.26% and 10.25%, respectively. Given consideration on a better economic value and lower arsenic content, the leach residue can be recycled as raw material to a lead smelting plant.
Table 4 Chemical composition of leach residue under optimum conditions (mass fraction, %)
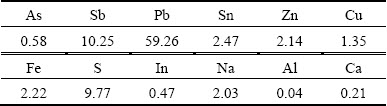
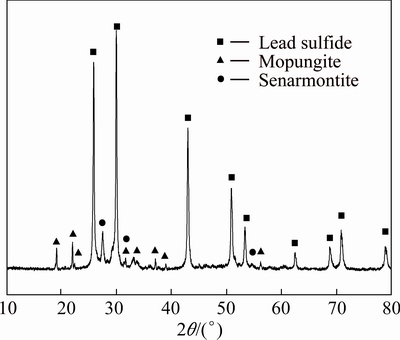
Fig. 7 XRD pattern of leach residue
4 Conclusions
1) Base on the results of factorial experiments, the optimum conditions for the extraction of arsenic from high-arsenic dust are determined as: alkali-to-dust mass ratio of 0.5, sodium sulfide addition of 0.25 g/g, leaching temperature of 90 °C, leaching time of 2 h, and liquid-to- solid ratio of 5:1 (mL/g).
2) Under these optimum conditions, the average leaching efficiencies of arsenic, antimony, lead, tin and zinc are 92.75%, 11.68%, 0.31%, 29.75% and 36.85%, respectively. And arsenic extraction rate is greatly influenced by the alkali-to-dust ratio, sodium sulfide addition, leaching temperature and liquid-to-solid ratio.
3) The main chemical compositions in leach residue are PbS, NaSb(OH)6 and Sb2O3, and the content of arsenic is below 1%. The leach residue could be applied as raw material to a lead smelting plant.
References
[1] CHOONG T S Y, CHUAH T G, ROBIAH Y, GREGORY F L, AZNI I. Arsenic toxicity, health hazards and removal techniques from water: An overview [J]. Desalination, 2007, 217: 139-166.
[2] LING Min, LIU Qi-zhan. Arsenic due to epigenetic changes and carcinogenesis studies progress [J]. Chinese Journal of Endemiology, 2012, 31(1): 107-110. (in Chinese)
[3] MIN Xiao-bo, LIAO Ying-ping, CHAI Li-yuan, YANG Zhi-hui, XIONG Shan, LIU Lin, LI Qing-zhu. Removal and stabilization of arsenic from anode slime by forming crystal scorodite [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(4): 1298-1306.
[4] YI Yu, SHI Jing, TIAN Qing-hua, GUO Xue-yi. Novel technology for preparation of sodium pyroantimonate from alkali leaching residue of high arsenic dust [J]. The Chinese Journal of Nonferrous Metals, 2015, 25(1): 241-249. (in Chinese)
[5] MONTENEGRO V, SANO H, FUJISAWA T. Recirculation of high arsenic content copper smelting dust to smelting and converting processes [J]. Minerals Engineering, 2010, 37: 29-33.
[6] CHEN Ya, LIAO Ting, LI Gai-bian, CHEN Bai-zhen, SHI Xi-chang. Recovery of bismuth and arsenic from copper smelter flue dusts after copper and zinc extraction [J]. Minerals Engineering, 2012, 39: 23-28.
[7] BAKHTIARI F, ATASH H, ZIVDAR M, SEYEDBAGHERI S, FAZAELIPOOR M S. Bioleaching kinetics of copper from copper smelters dust [J]. Journal of Industrial and Engineering Chemistry, 2011, 17: 29-35.
[8] SHAWABKEH R A. Hydrometallurgical extraction of zinc from Jordanian electric arc furnace dust [J]. Hydrometallurgy, 2010, 104: 61-65.
[9] JIANG Tao, HUANG Yan-fang, ZHANG Yuan-bo, HAN Gui-hong, LI Guang-hui, GUO Yu-feng. Behaviors of arsenic in arsenic-bearing iron concentrate pellets by pre-oxidizing weak reduction roasting process [J]. Journal of Central South University: Science and Technology, 2010, 41(1): 1-7. (in Chinese)
[10] ZHANG Shu-hui,  Qing, HU Xiao. Thermodynamics of arsenic removal from arsenic-bearing iron ores [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(7): 1705-1712. (in Chinese)
Qing, HU Xiao. Thermodynamics of arsenic removal from arsenic-bearing iron ores [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(7): 1705-1712. (in Chinese)
[11] SHIBAYAMA A, TAKASAKI Y, WILLIAM T, YAMATODANI A, HIGUCHI Y, SUNAGAWA S, ONO E. Treatment of smelting residue for arsenic removal and recovery of copper using Pyro-hydrometallurgical process [J]. Journal of Hazardous Materials, 2010, 181: 1016-1023.
[12] DRAHOTA P, FILIPPI M. Secondary arsenic minerals in the environment: A review [J]. Environment International, 2009, 35: 1243-1255.
[13] SHI Jing, YI Yu, GUO Xue-yi. Research progress of arsenic-bearing solid waste by hydrometallurgy [J]. Nonferrous Metals Science and Engineering, 2015, 6(2): 14-20. (in Chinese)
[14] YU Guo-lin, ZHANG Ying, ZHENG Shi-li, ZOU Xing, WANG Xiao-hui, ZHANG Yi. Extraction of arsenic from arsenic-containing cobalt and nickel slag and preparation of arsenic-bearing compounds [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(10): 1918-1927.
[15] PENG Ying-lin, ZHENG Ya-jie, ZHOU Wen-ke, CHEN Wen-mi. Separation and recovery of Cu and As during purification of copper electrolyte [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(9): 2268-2273.
[16] TONGAMP W, TAKASAKI Y, SHIBAYAMA A. Selective leaching of arsenic from enargite in NaHS-NaOH media [J]. Hydrometallurgy, 2010, 101: 64-68.
[17] LI Yu-hu, LIU Zhi-hong, LI Qi-hou, ZHAO Zhong-wei, LIU Zhi-yong, ZENG Li. Removal of arsenic from Waelz zinc oxide using a mixed NaOH-Na2S leach [J]. Hydrometallurgy, 2011, 108: 165-170.
[18] SULLIVAN C, TYRER M, CHEESEMAN, CHRISTOPHER R, GRAHM, NIGEL J D. Disposal of water treatment wastes containing arsenic―A review [J]. Science of the Total Environment, 2010, 408: 1770-1778.
[19] LI Yu-hu, LIU Zhi-hong, LI Qi-hou, ZHAO Zhong-wei, LIU Zhi-yong, ZENG Li, LI Li. Removal of arsenic from arsenate complex contained in secondary zinc oxide [J]. Hydrometallurgy, 2011, 109: 237-244.
[20] YI Yu, SHI Jing, TIAN Qing-hua, GUO Xue-yi. Arsenic removal from high-arsenic dust by NaOH-Na2S alkaline leaching [J]. The Chinese Journal of Nonferrous Metals, 2015, 25(3): 806-814. (in Chinese)
[21] FENG Shu-ping. Analytical chemistry of Arsenic [M]. Beijing: China Environmental Science Press, 1986: 178-180. (in Chinese)
[22] LEWIS A E. Review of metal sulphide precipitation [J]. Hydrometallurgy, 2010, 104: 222-234.
郭学益1,2,易 宇1,2,石 靖1,田庆华1,2
1. 中南大学 冶金与环境学院,长沙 410083;
2. 中南大学 中国有色金属工业清洁冶金工程研究中心,长沙 410083
摘 要:采用NaOH-Na2S碱浸方法实现了高砷烟尘中砷的选择性浸出。在砷浸出过程中,探讨了碱料比、硫化钠用量、浸出温度、浸出时间和液固比对各金属元素浸出率的影响。结果表明:在碱料质量比为0.5,硫化钠用量为0.25 g/g,浸出温度为90 °C,浸出时间为2 h,液固比为5:1的优化条件下实现了砷与其他金属的有效分离。在优化条件下,砷、锑、铅、锡和锌的平均浸出率为92.75%, 11.68%, 0.31%, 29.75%和36.85%。NaOH-Na2S碱性浸出过程提供了一种从含砷烟尘中脱除砷的简单有效的方法,同时产生具有利用价值的铅渣。
关键词:高砷烟尘;碱性浸出;浸出行为;砷
(Edited by Yun-bin HE)
Foundation item: Project (2012AA04022) supported by the Scientific Research and Technology Development Project of Guangxi, China
Corresponding author: Xue-yi GUO; Tel: +86-731-88877863; E-mail: xyguo@csu.edu.cn
DOI: 10.1016/S1003-6326(16)64118-3

