
Microstructural evolution of NiFe2O4-10NiO powder prepared by
high temperature solid state reaction
ZHANG Lei(张 雷), ZHOU Ke-chao(周科朝), LI Zhi-you(李志友), YANG Wen-jie(杨文杰)
State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China
Received 29 November 2005; accepted 3 April 2006
Abstract: The NiFe2O4-10NiO powder for inert anode of aluminium electrolysis was prepared by high temperature solid state reaction. The microstructural evolution from the raw materials NiO and Fe2O3 to the NiFe2O4-10NiO powder was studied by SEM. The results show that the domain structure making up of the agglomerate particles of Fe2O3 remains after high temperature solid state reaction, and the diffusion of Ni2+ into Fe2O3 structure is the control step of the reaction process. A microstructure with compact structure and fine grain inside the particle results from the sintering of NiFe2O4-10NiO powder.
Key words: high temperature solid state reaction; NiFe2O4-NiO; microstructural evolution; inert anode
1 Introduction
The development of inert anode materials for aluminium production has gained considerable attention in recent years[1,2]. The significance of such anodes is the ability to produce environment-friendly O2 gas during electrolysis instead of greenhouse gases, and dramatically reduce the cost for the production of Al. Several materials have been investigated for developing inert anodes including oxides[3], such as CeO2, Cr2O3- NiO-CuO, NiFe2O4, CoFe2O4, SnO2 and ZnFe2O4; cermets[4,5] such as NiFe2O4+NiO+Cu, NiFe2O4+NiO+ Cu+Ag; and metals such as Al-Cu, Ni-Al-Fe-Cu-X, Ni-Fe. The cermets NiFe2O4+NiO+M, where M is metals (such as Ni, Cu, Fe) and their alloys, are the most promising candidate materials of inert anode[6]. The composite oxide NiFe2O4-NiO has high thermodynamic stability and favorable electro-catalytic activity for oxygen evolution and high corrosion stability in molten cryolite electrolyte. A number of methods can be used to produce this composite powder, such as coprecipitaion of hydroxides, carbonates and oxalates, freeze-drying of mixed carboxylates, and high temperature solid state reaction[7,8]. For processing a great amount of raw NiFe2O4-NiO powder for developing inert anode in commercial size, the solid state reaction, a conventional process in advanced ceramic industry, was used in this work. The solid state reaction can achieve high purity, high surface area, and good sintering activity powder with high efficiency[9, 10]. In this work, the synthesis of NiFe2O4-10NiO by high temperature solid state reaction process with NiO and Fe2O3 powder as raw materials was studied. The crystalline phases of the prepared powders were identified by powder X-ray diffractometry. The evolution of microstructure from the raw materials NiO and Fe2O3 to the products, NiFe2O4- 10NiO was studied by SEM.
2 Experimental
The initial materials used were: high purity (99.9%, mass fraction) NiO with mean particle size of 40 μm produced by Jinchuan Group Ltd; high purity (99.2%) Fe2O3 with mean particle size of 30 μm produced by Hecheng Ferrite Ltd. For the synthesis of NiFe2O4- 10NiO, the as-received raw materials were mixed and ground for 2 h in de-ionized water by ball milling, with the mass ratio of NiO to Fe2O3 being 61.8?38.6, and the mass ratio of ball to material 5?1. Then the slurry was removed and dried at 120 ℃ for 48 h in air.
20 kg of mixed oxides per batch were heated in a furnace for high temperature solid state reaction in air. According to the research work of ZHANG et al [11] and QIN et al[12], the reaction temperature was risen to 1 250 ℃ at a heating rate of 100 ℃/h, and held for 2 h, then was slowly cooled down to ambient temperature. After solid state reaction, the NiFe2O4- 10NiO powder was ball milled for 6 h again with the ball-to-powder mass ratio of 5?1 and water-to-material ratio 2?3.
The particle size distribution of the as-received raw materials and the ball milled NiFe2O4-10NiO powder was examined by MICRO-PLUS laser size analyzer. X-ray diffraction analysis was performed on Rigaku 3014 diffractometer with a Kα radiation of Fe anode to determine the phase present in the final samples. The evolution of microstructure from the as-received raw materials NiO and Fe2O3 to the NiFe2O4-10NiO powder was observed by a JEOL JSM-6400F scanning electron microscope(SEM).
3 Results and discussion
3.1 XRD analysis
The X-ray powder diffraction pattern of NiFe2O4- 10NiO synthesized by high temperature solid state reaction is shown in Fig.1. All the diffraction peaks of well crystallized NiO and NiFe2O4 are detected in the XRD patterns, which are indentifical with the results in Ref.[13]. It reveals that the sample has typical cubic spinel structure without impurity phases.
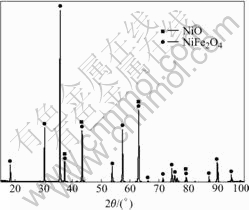
Fig.1 XRD pattern of NiFe2O4-10NiO powder calcined at 1 250 ℃ for 2 h in air
3.2 Evolution of microstructure
The as-received Fe2O3 powder is produced by the oxidation of iron scale, and granulated by spray dry process. The SEM photographs of the as-received Fe2O3 powder are shown in Fig.2. It can be seen that the Fe2O3 powder is made up of agglomerates, and the agglomerates are composed of small and high density packed subunit which can be called “domains”. The mean particle size of the as-received Fe2O3 is 30 μm, and the size of domains is 3-6 μm. Fig.3 shows the SEM photographs of the as-received NiO powder. The NiO powder has crumble and lumpy structure with high surface area (Fig.3(b)). After ball milling, both NiO powder and Fe2O3 agglomerates are broken up into fine particle size, and the mean size of the composite oxide powder is 4.36 μm. Figs.4(a) and (b) show the SEM images of the milled NiFe2O4-10NiO powder calcinated at 1 250 ℃ for 2 h in air. The NiFe2O4-10NiO powder is of fine and polyhedral structure with a mean particle size of 3.55 μm. Fig.4(c) shows the interior conformation of NiFe2O4-10NiO particles, where the “domains” structure can be seen once again, and its size is obviously smaller than that of Fe2O3.
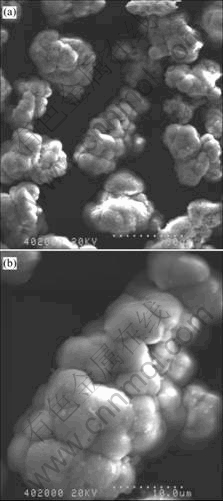
Fig.2 SEM images of as-received Fe2O3 powder

Fig.3 SEM images of as-received NiO powder
In Figs.4 (a) and (b), the polyhedral particles that look like the subunit particles of Fe2O3 agglomerates are observed, and the size of the particles is 1-3 μm, similar to the domains’ size of the as-received FeO3 powder. It can be deduced that the domain structure of Fe2O3 remains after high temperature solid state reaction. For the as-received NiO powder, the aggregative structure is constructed by subunit particles which has smaller particle size than the domains of Fe2O3. After ball milling and calcining, the structure of NiO particle is destroyed, but excessive NiO powder remains in the compound oxide powder.

Fig.4 SEM images of NiFe2O4-10NiO powder calcinated at 1 250 ℃ for 2 h
The special microstructure which has high density and fine grain inside the particle shown in Fig.4(c) is the result of sintering of the composite powder at high temperature.
3.3 Reaction mechanism
Fig.5 shows an illustrative and classical example for elucidating material transport mechanisms in solid-state reactions using the diffusion couple arrangements. The diffusion couple experiments are helpful for illustrating the complexity of reaction mechanisms in ionic compounds, especially ferrites[14].
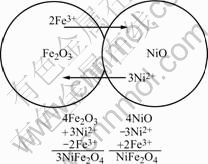
Fig.5 Schematic diagram of NiFe2O4 formation reaction
At high temperature, the products phase nucleates and grows, then separates from the reactants. For relatively simple reaction mechanisms, it is possible to deduce the nature of the diffusing species from the relative growth of the new phase.
The overall reaction of the formation of spinel NiFe2O4 is
NiO+Fe2O3→NiFe2O4 (1)
The reaction may occur in many mechanisms. The relative amount of the spinel phase formed at the original boundary is indicative of the diffusion mechanism of the reaction, the amount of spinel products formed is in a ratio of 1?3[15]. Due to the larger ionic radius, O2- ions are expected to have a considerably lower mobility than most cations. When cations are only the transporting species, the mechanism is known as the Wagner mechanism[10]. The formation of the spinel phase can be easily explained by the diffusion of Ni2+ and counter- diffusion of Fe3+. To maintain electroneutrality, the mechanism involves the counter-diffusion of three Ni2+ ions for every two Fe3+ ions. While three Ni2+ ions produce three NiFe2O4, the diffusion of two Fe3+ ions results in only one NiFe2O4. The results of the evolution of microstructure show that the domain structure which makes up of the agglomerative Fe2O3 particle remains after high temperature solid state reaction, indicating that the diffusion of Ni2+ to Fe2O3 structure controls the reaction process.
The existence of an extensive range of solid solubility can lead to a difference in the rate of consumption of reactants. Fe2O3 has a high solubility in the spinel phase through partial reduction of Fe3+ to Fe2+ to produce magnetite:
 →
→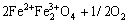 ↑ (2)
↑ (2)
Reaction (1) begins at 700 ℃, and gains a high reaction rate at 1 000-1 100 ℃[16]. While reaction(2) occurs at about 1 000 ℃ when heated in air[16]. Before the solid state reaction (1) is fully in action, the particles of Fe2O3 has been consumed by reaction (2), and the products Fe3O4 become the reactant of the solid state reaction (1). Then the formation reaction of nickel ferrite spinel is reaction (3) at higher temperature (>1 000℃). At high temperature, with the reoxidation of Fe2+ back to Fe3+, the diffusion of Ni2+ to the Fe3O4 particle becomes the control step of the reaction, and the NiFe2O4 particles are sintered. So the particle size of the composite oxide is smaller than the size of domains in Fe2O3.
2Fe3O4+3NiO+1/2O2→3NiFe2O4 (3)
4 Conclusions
The NiFe2O4-10NiO powder with classic spinel phase, fine particle size, polyhedral structure and high surface area is synthesized by high temperature solid state reaction. The evolution of microstructure from raw materials NiO and Fe2O3 to the reaction resultant NiFe2O4-10NiO shows that the domain structure which makes up of the agglomerative Fe2O3 particle remains after heating process. The analysis results of micro- structural evolution show that the main reaction mechanism is the diffusion of Ni2+ to the Fe3O4 particles. High temperature has a sintering effect on the composite powder, and the microstructure with high density and fine grain inside the particle is gained.
References
[1] PAWLEK R P. Inert anodes: an update[A]. SCHNEIDER W. Light Metals 2002[C]. Warrendale PA, USA: TMS, 2002: 449-456.
[2] SADOWAY D R. Inert anodes for the Hall-Héroult cell: the ultimate materials challenge [J]. JOM, 2001, 53(5): 34-35.
[3] RAY S P, WOODS R W, Inert Anode Containing Metal Oxides, Copper and Nobel metal [P]. US Patent: 6126799, 2000-10-03.
[4] MCLEOD A D, HAGGERTY J S. Inert anode materials for Hall cells [A]. MILLER R E. Light Metals 1986 [C]. Warrendale, PA: TMS, 1986: 269-273.
[5] SEKHAR J S, LIU J, DENG H, DURUZ J J, DE NORA V. Graded non-consumable anode materials [A]. WELCH B. Light Metals 1998 [C]. Warrendale PA: TMS, 1998: 597-603.
[6] OLSEN E, THONSTAD J. The behaviour of nickel ferrite cermet materials as inert anodes [A]. Hale W. Light Metals 1996 [C]. Warrendale, PA: TMS, 1996: 249-257.
[7] BONSDORF G, SCHAFER K, TESKE K, LANGBEIN H, ULLMANN H. Stability region and oxygen stoichiometry of manganese ferrite [J]. Solid State Ionics, 1998, 110(1/2): 73-82.
[8] HASMONAY E, DEPEYROT J, SOUSA M H, TOURINHO F. Optical properties of nickel ferrite ferrofluids [J]. Journal of Magnetism and Magnetic Materials, 1999, 201(1): 195-199.
[9] ELWELL D, PARKER R, TINSLEY C J. The formation of nickel ferrite [J]. Solid State Communications, 1966, 4(1): 69-71.
[10] NOVELO F, VALENZUELA R. On the reaction kinetics of nickel ferrite from iron and nickel oxides [J]. Materials Research Bulletin, 1995, 30(3): 335-340.
[11] ZHANG Lei, ZHOU Ke-chao, LI Zi-you, ZHANG Xiao-yong. Effect of atmosphere on densification in sintering nickel ferrite ceramic for aluminium electrolysis [J]. The Chinese Journal of Nonferrous Metals, 2004, 14(6): 1002-1006.(in Chinese)
[12] QIN Qing-wei, LAI Yan-qing, ZHANG Gang, TIAN Zhong-liang, ZHOU Xin-lin, LI Jie, LIU Ye-xiang, HE Yue-hui. Solid state reaction synthesis of Ni(1-x)ZnxFe2O4 spinel used as matrix of inert anodes in aluminium electrolysis [J]. The Chinese Journal of Nonferrous Metals, 2003, 13(3): 967-733.(in Chinese)
[13] MITTAL V K, SANTANU BERA, NITHYA R, SRINIVASAN M P, VELMURUGAN S, NARASIMHAN S V. Solid state synthesis of Mg-Ni ferrite and characterization by XRD and XPS [J]. Journal of Nuclear Materials, 2004, 335(3): 302-310.
[14] VALENZUELA R. Magnetic Ceramics [M]. London: Cambridge University Press, 1994: 62-75.
[15] REIJNEN P L J. Solid-state reactions. Concise Encyclopedia of Advanced Ceramic Materials [M]. BROOK R J. Oxford: Pergamon Press, 1991: 445-454.
[16] LIN Qi-ren, Ferrite Technology [M]. Shanghai: Shanghai Technology Press, 1987: 50-69.
Foundation item: Project(2005CB623703) supported by the National Basic Research Program of China
Corresponding author: ZHANG Lei; Tel: +86-731-8836264; E-mail: fgmzhang@163.com
(Edited by YUAN Sai-qian)