J. Cent. South Univ. Technol. (2007)06-0779-04
DOI: 10.1007/s11771-007-0148-3
Thermal stability and oil absorption of aluminum hydroxide treated by dry modification with phosphoric acid
ZHOU Xiang-yang(������)1, LI Chang-lin(�����)1, HUO Deng-wei(����ΰ)1,2,
LI Jie(�� ��)1, WU Shang-yuan(����Ԫ)1, LIU Ye-xiang(��ҵ��)1
(1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. Shandong Branch,China Aluminum Corporation, Zibo 255052, China)
Abstract: The dry modification of aluminum hydroxide powders with phosphoric acid and the effects of modification of technological conditions on thermal stability, morphology and oil absorption of aluminum hydroxide powders were investigated. The results show that the increase of mass ratio of phosphoric acid to aluminum hydroxide, the decrease of mass concentration of phosphoric acid and prolongation of mixing time are favorable to the improvement of thermal stability of aluminum hydroxide; when the mass ratio of phosphoric acid to aluminum hydroxide is 5?100, the mass concentration of phosphoric acid is 200 g/L and the mixing time is 10 min, the initial temperature of loss of crystal water in aluminum hydroxide rises from about 192.10 to 208.66 ��, but the dry modification results in the appearance of agglomeration and macro-aggregate in the modified powders, and the oil absorption of modified powders becomes higher than that of original aluminum hydroxide.
Key words: aluminum hydroxide; phosphoric acid; thermal stability; oil absorption; dry modification
1 Introduction
Aluminum hydroxide is a well known non-toxic and cheap flame retardant. However, its high concentration introduction and poor filling performance impair the properties of the organic high molecule materials[1-4], especially its lower decomposing temperature makes it only suitable for high polymers with lower shaped temperature[5]. Partial dehydration, ultra-micronization, high purification, combination with other flame retardants and surface modification are employed to improve the thermal stability of aluminum hydroxide, among which surface modification is an effective and simple method[1, 6-7]. Phosphoric acid modification can raise the thermal stability of aluminum hydroxide[8], which is mainly due to that phosphoric acid can react with aluminum hydroxide and generate phosphate aluminum such as Al(H2PO4)3 and AlPO4, which have higher thermal stability[9-12], so phosphoric acid is used as modification reagent to raise the thermal stability of aluminum hydroxide in this work.
Usually, the wet and dry processing are used to modify the surface of powder particles[13], and the dry processing has many advantages such as simpler operation and fewer working steps. Therefore, in this study, the effects of dry modification with phosphoric acid on the properties of aluminum hydroxide were investigated, and the influences of processing conditions on thermal stability, morphology and oil absorption of the modified aluminum hydroxide were also researched.
2 Experimental
2.1 Preparation of samples
The preparation of the modified aluminum hydroxide samples can be described as follows: a certain amount of aluminum hydroxide powder (industrial product from Shangdong Aluminum Co.) was added into a mixer (maximal rotary speed of 20 000 r/min, FW100, Tianjin Taisite Instrument Co. Ltd), and a certain amount of phosphoric acid(mass fraction of 85%, analytical grade) was introduced into moderate deionized water to form uniform solution. Then the phosphoric acid solution was dispersed into aluminum hydroxide powder in batches by an injector, followed by mixing for several minutes, here, the times of phosphoric acid solution injection was 5. At last, the aluminum hydroxide was taken into a drier (top temperature of 250 ��, 101-1AB, Tianjin Taisite Instrument Co. Ltd) and dried overnight at 120 ��.
2.2 Testing methods
2.2.1 Thermal stability analysis
The thermal stability of aluminum hydroxide samples was tested by the following two methods.
Method 1 The mass loss ratio (Rloss, %) at 200 ��
for 30 min was used as an indication of thermal stability of samples. This method can qualitatively compare the Rloss changes of samples in the same group. Rloss can be calculated by
Rloss��(m1��m2)/m1��100% (1)
where m1 and m2 are the mass of aluminum hydroxide before and after heating at 200 �� for 30 min, respectively.
Method 2 TGA-DSC was carried out to investigate the behaviors of samples during thermal decomposition and performed in the temperature range from 25 �� to 800 �� on a TA instrument SDT Q600 thermobalance. The diagrams were obtained at heating rate of 5 ��/min and linear gas flow rate of 100 mL/min in air.
2.2.2 Oil absorption test
Before testing, aluminum hydroxide powder was dried at 105 �� for 1 h. About 5 g dried sample was taken into the glass board, then castor oil was added into the sample with a burette. Meanwhile the oil and powders were mixed with glass rod. When the mixture turned to strips and bits, and could roll onto the glass rod with no oil trace on the board, stop the titration of castor oil. The castor oil absorption per 100 g aluminum hydroxide powders was denoted as the oil absorption.
2.2.3 SEM analysis
Scanning electron microscope (SEM, JEOL, JSM5600) was employed to investigate the grain morphology of samples.
3 Results and discussion
3.1 Effect of mass fraction of phosphoric acid on Rloss
The mass fraction of phosphoric acid refers to mass ratio of phosphoric acid to aluminum hydroxide. Here, 50 g aluminum hydroxide was chosen to react with phosphoric acid, the mass fraction of phosphoric acid was 0, 1%, 3%, 5%, 7%, respectively. The mixing time after each injection was 10 min, the phosphoric acid was diluted to solution of 12.5 mL.
Fig.1 shows the changes of Rloss with the mass fraction of phosphoric acid. As can be seen from Fig.1 that Rloss decreases as the mass fraction of phosphoric acid increases. This may be due to that more phosphoric acid leads to the formation of thicker heat-resistant film on the surface of powder particles, thus the mass loss reduces and thermal stability rises. When the mass fraction of phosphoric acid exceeds 5%, the downtrend of Rloss becomes slower. Besides, agglomeration in modified powders is found, as can be easily seen in Fig.2. This may be attributed to a mass of viscous compounds formed on the surface of aluminum hydroxide particles.
3.2 Effect of mass concentration of phosphoric acid solution and mixing time on Rloss
Fig.3 reveals Rloss changes of samples generated through reaction of aluminum hydroxide with phosphoric acid solution of different mass concentrations. Here, 50 g aluminum hydroxide and 2.5 g phosphoric acid were chosen as initial reactants, and the mass concentrations of phosphoric acid were 500, 300, 250, 200, 150 g/L, respectively, the mixing time after each injection was 10 min. Fig.3 indicates that Rloss decreases while lowering the mass concentration of phosphoric acid, i.e. lowering the mass concentration of phosphoric acid is effective to reduce Rloss. This may be due to the facts that lowering the mass concentration of phosphoric acid is effective for phosphoric acid to contact much more aluminum hydroxide particles, i.e. much more aluminum hydroxide particles are coated by heat-resistant film. It can be seen that the downtrend of Rloss becomes slower, when the mass concentration of phosphoric acid excesses 200 g/L. Considering that more water and more energy are consumed during the drying process, the mass concentration of phosphoric acid of 200 g/L is better.
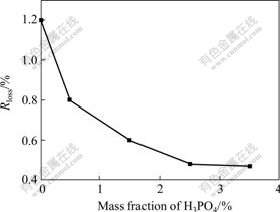
Fig.1 Effect of mass fraction of H3PO4 on mass loss ratio of samples

Fig.2 SEM images of Al(OH)3 powders before and after modification
(a) Before modification; (b) After modification(mass of phosphoric acid is 2.5 g)
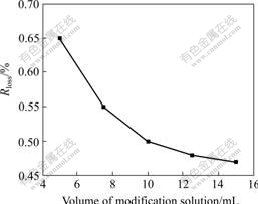
Fig.3 Effect of mass concentration of phosphoric acid on mass loss ratio of samples
Fig.4 shows the effect of mixing time on Rloss, where, the mass fraction of phosphoric acid is 5%, the mass concentrate of phosphoric acid solution is 200 g/L. Obviously, Rloss decreases as the mixing time prolongs, and the downtrend of Rloss becomes slower after the mixing time of 10 min.
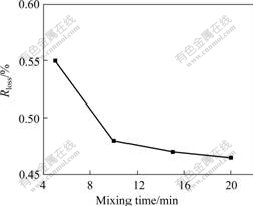
Fig.4 Effect of mixing time on mass loss ratio of samples
3.3 DSC-TGA testing results and oil absorption of samples
The samples used in this part were prepared under the following technological conditions: the mass of phosphoric acid is 2.5 g and the volume of phosphoric acid solution is 12.5 mL (i.e. the mass concentration of phosphoric acid solution is 200 g/L), the mass of aluminum hydroxide is 50 g and mixing time is 10 min.
The TGA-DSC curves of the samples before and after modification by phosphoric acid are shown in Fig.5. Apparently, it can be easily seen from the figure: 1) the shapes of all DSC-TGA curves are almost similar, a little mass loss below 100 �� can be found maybe due to the fact that the particles absorb moisture in air; 2) the total mass loss of the modified aluminum hydroxide between 100 �� and 600 �� is about 34%, which is less than that of the original particle. This results from the production of aluminum phosphate that cannot be decomposed at the testing temperature; 3) the initial temperature of loss of crystal water in the particles rises from 192.10 to 208.66 ��, which indicates that the dry modification described in this work can improve the thermal stability of aluminum hydroxide.
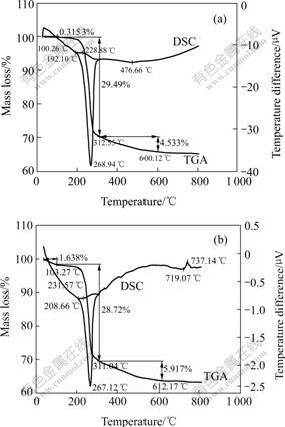
Fig.5 TGA-DSC curves of Al(OH)3 powders before and after modification
(a) Before modification; (b) After modification
Oil absorption is one of the main factors influencing the application of Al(OH)3 as flame retardant, the higher oil absorption results in not only higher viscosity in the high polymer slurry but also higher cost[14]. The testing results indicate that the oil absorption of the modified sample is 0.74 mL/g, whereas that of the original aluminum hydroxide is 0.44 mL/g. PARK et al[15] shows that the dimension of pores existing in powder congeries and surface morphology of powders are the decisive factors to oil absorption of powders. SEM images of samples (see Fig.2) show that agglomerations and macro-aggregates appear in modified sample, and the surface of macro-aggregates is very coarse and rugged, even some acuminate protuberances appear as well. As a result, the oil absorption in the modified sample becomes higher than that in original aluminum hydroxide.
4 Conclusions
1) The dry modification of aluminum hydroxide with phosphoric acid can raise its initial temperature of loss of crystal water. And the increase of mass fraction of phosphoric acid to aluminum hydroxide, the decrease of concentration of phosphoric acid solution and prolongation of milling time are favorable to the improvement of thermal stability of aluminum hydroxide.
2) The initial temperature of loss of crystal water in aluminum hydroxide rises from 192.10 to 208.66 �� when the mass fraction of phosphoric acid to aluminum hydroxide is 5%, the mass concentration of phosphoric acid is 200 g/L and mixing time is 10 min.
3) The dry modification with phosphoric acid results in the appearance of agglomeration and macro-aggregate, resulting in that the oil absorption of modified sample becomes higher than that of original aluminum hydroxide. The oil absorption of the modified sample is 0.74 mL/g, whereas that of the original aluminum hydroxide is 0.44 mL/g.
References
[1] HUANG Dong, NAN Hai, WU He. Flame retardancy property and application of Al(OH)3[J]. Materials Exploitation and Application, 2003, 19(3): 33-37. (in Chinese)
[2] LI Xue-feng, CHEN Xu-huang, ZHOU Mi. Applications of Al(OH)3 flame retardant in polymers[J]. China Plastics, 1999, 13(6): 80-85. (in Chinese)
[3] NACHTIGALL S M B, MIOTTO M, SCHNEIDER E E, et al. Macromolecular coupling agents for flame retardant materials[J]. European Polymer Journal, 2006, 42(5): 990-999.
[4] HIPPI U, MATTIL J, KORHONEN M, et al. Compatibilization of polyethylene/aluminum hydroxide (PE/ATH) and polyethylene/ magnesium hydroxide (PE/MH) composites with functionalized polyethylenes[J]. Polymer, 2003, 44(4): 1193-1201.
[5] LAURENT C, LAURENT F, ERIC L, et al. Influence of talc physical properties on the fire retarding behavior of (ethylenevinyl acetate copolymer/magnesium hydroxide/talc) composites[J]. Polymer Degradation and Stability, 2005, 88: 504-511.
[6] NEIL B, MICHAEL A. Nonhygroscopic thermally stable aluminum hydroxide: US, 6280839 B1[P]. 1999-08-28.
[7] SI Ji-chun, ZHENG Shui-lin, LU Mai-xi. Surface modifying and applied research of aluminum hydroxide[J]. China Powder Science and Technology, 2005, 11(2): 17-20. (in Chinese)
[8] ZHOU Xiang-yang, LI Chang-lin, HUO Deng-wei, et al. Thermal stability of aluminum hydrate modified by phosphoric acid[J]. The Chinese Journal of Nonferrous Metals, 2006, 16(12): 2115-2119. (in Chinese)
[9] WANG Yong-qiang. Flame Retardant Materials and Application Technology[M]. Beijing: Chemical Industry Press, 2003: 76-77. (in Chinese)
[10] TAN Wen-qing, LI Feng. Flame Retardant Technology of Material Surface Coating[M]. Beijing: Chemical Industry Press, 2004. (in Chinese)
[11] MIAO S. Polymer surface modification and characterization of particulate calcium carbonate fillers[J]. Applied Surface Science, 2003, 220(1/4): 359-366.
[12] YUAN Ai-qun. Preparation of AlH2P3O10?2H2O[J]. Sichuan Chemical Industry and Erosion Control, 2003, 3(3): 9-12. (in Chinese)
[13] WU Wei, CHEN Jiang-feng, LU Shou-ci. Ultramicro Particle Surface Modification[M]. Beijing: Chemical Industry Press, 2004: 7-8. (in Chinese)
[14] GAO Jian-yang. Physical and chemical properties of AH filler on its use and the products performance[J]. Chemical Engineer, 2006, 19(1): 62-64. (in Chinese)
[15] PARK J K, KIM J K, KIM H K. TiO2-SiO2 composite filler for thin paper[J]. Journal of Materials Processing Technology, 2007, 186: 367�C369.
(Edited by CHEN Wei-ping)
Foundation item: Project(06SK2011) supported by the Science and Technology Program of Hunan Province, China
Received date: 2007-05-13; Accepted date: 2007-06-28
Corresponding author: ZHOU Xiang-yang, PhD; Tel:+861-731-8836329; E-mail: xyzhou@mail.csu.edu.cn