
Corrosion of titanium in phosphoric acid at 250 ℃
LU Jian-shu(卢建树)
College of Chemical Engineering and Materials Science, Zhejiang University of Technology,
Hangzhou 310014, China
Received 25 February 2008; accepted 12 March 2008
Abstract: Corrosion studies of a commercially pure titanium in phosphoric acid solutions at 250 ℃ were carried out by immersion test in an autoclave. At lower phosphoric acid concentration (0.1 mol/L), the corrosion was mild. At higher phosphoric concentration (1.0 mol/L) corrosion, a 25 ?m-thick white corrosion products layer was formed on the samples after 24 h immersion. XRD analysis shows that the white layer consists mainly of titanium oxide phosphate hydrate (π-Ti2O(PO4)2?2H2O). The corrosion product shows the morphology of fiber bundles. A thermodynamic analysis of the formation of the corrosion product is presented.
Key words: titanium; corrosion; phosphoric acid; high temperature and high pressure; corrosion mechanism
1 Introduction
Previous work on the studies of corrosion resistance of metallic materials in supercritical water oxidation (SCWO) environments showed that titanium was corroded by phosphoric acid produced during the decomposition of a pesticide (chlorpyrifos, C9H11Cl3NO3- PS) containing phosphor in its molecule[1]. Work by BOUKIS et al[2] showed that the corrosion of grade 2 titanium in 0.2 mol/kg H3PO4 and 3.0 mol/kg O2 at 360 ℃ is 16 mg/cm2 in 100 h, severer than that in HCl of the same concentration. As titanium and its alloys are still the most promising materials for SCWO application and other special applications such as wet air oxidation process and flue gas desulfurization process, the further work on the corrosion and protection of titanium materials in elevated or high temperature aggressive environments is highly needed.
Early work on the corrosion resistance of titanium in phosphoric acid was done by TAYLOR in 1949. Corrosion rate of 0.006 75 inches per year was recorded by immersion test of titanium plate in 85% H3PO4 at 19-26 ℃[3]. Several authors[4-10] made electro- chemical investigations of titanium and its alloys in different concentrations of H3PO4 at room or elevated temperatures. The critical current density(Jc) and passive current density(Jp) of commercially pure titanium in 1 mol/L H3PO4 at 40 ℃ is 10 and 7 μA/cm2, respectively. The current densities increase with the increase of temperature[4].
As shown above, the previous studies on the corrosion of titanium in environments containing phosphoric acid or phosphate were very limited. In this work, the corrosion behavior of a commercially pure titanium in phosphoric acid at 250 ℃ is studied. This temperature is often experienced in many industrial processes such as power plant system, SCWO system, wet air oxidation system, and geothermal system. Recent corrosion study by OE et al[11] on a Ti-Pd alloy in 20% HCl at high temperature (<600 ℃) showed that the alloy suffered the most serious corrosion at 250 ℃.
2 Experimental
Titanium samples of 20 mm×10 mm×1 mm were cut from 1 mm-thick commercially pure titanium plate (TA3, hereafter called CP titanium or simply titanium) which was purchased from Baotai New Metals Ltd. (Baoji, China). Table 1 lists the chemical composition of the titanium sample. The chemical composition of titanium sample is similar to that of the ASTM grade 3 titanium.
Table 1 Chemical composition of commercially pure titanium TA3 (mass fraction, %)

The titanium samples were first ground using 1 200# grit SiC paper and then ultrasonically cleaned in ethanol. The samples were immersed in 0.1 mol/L and 1.0 mol/L phosphoric acid solutions at 250 ℃, respectively. Two samples were used in each test for reproducibility purpose. The solutions were prepared by diluting concentrated phosphoric acid (85%, analytical purity). The 0.1 mol/L concentration was adopted to simulate the acidic medium encountered in SCWO environments. The 1 mol/L H3PO4 at 250 ℃ represents a high concentration case in industrial processes. The samples were suspended in the solutions by PTFE tubes (Fig.1). A PTFE lined autoclave with a volume capacity of 250 mL was used to hold the acid solutions at the high temperature. The pressure of the autoclave is about 4 MPa. The test duration is 24 h.
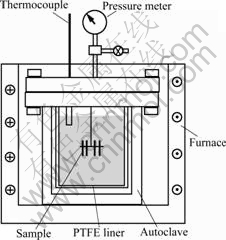
Fig.1 Corrosion test apparatus
Corrosion rates were calculated based on the mass changes of the samples. The mass change measurements were conducted by an analytical electron balance (0.1 mg in accuracy). Before weighting the samples, the corrosion product on the sample surfaces were carefully removed by a plastic brush and then treated ultrasonically in ethanol solution bath for 3 min.
An electron scanning microscope (Hitachi S570) was used to show the morphology of corrosion and corrosion product on the sample surfaces. The corrosion products were characterized by XRD spectrum and IR spectrum. A Siemens D5000 X-ray diffractometer with Cu Kα radiation (λ=0.154 06 nm) was used for XRD analysis. A Nicolet Avatar 360 FT-IR spectrometer was used for infrared spectrum analysis.
3 Results and discussion
3.1 Corrosion of titanium in 0.1 mol/L H3PO4
After immersion in 0.1 mol/L H3PO4 at 250 ℃ for 24 h, the titanium samples are covered with a thin layer of white corrosion products. Fig.2 shows the SEM micrographs of the corrosion products on the Ti surface. The corrosion product layer uniformly distributes on the surface, as shown in Fig.2(a). Higher magnification micrograph reveals that the corrosion products consist of well crystallized fine fibers, as shown in Fig.2(b).

Fig.2 Morphologies of corrosion products of CP titanium at 250 ℃ after 24 h immersion in 0.1 mol/L H3PO4
Table 2 shows the data of the corrosion test results. Both the samples gain mass when the surfaces are cleaned with a plastic brush. The average mass gain is 1.2 mg. The corrosion rate, according to this mass change, of the CP titanium is 0.01 mg/(cm2?h). Because the samples gain mass, the corrosion products of the titanium must deposit and attach on the Ti surface. The corrosion rate in this case is not meaningful but it shows that the reaction between titanium and phosphoric acid takes place in this condition.
Table 2 Data of corrosion test of CP titanium in phosphoric acid solution at 250 ℃

3.2 Corrosion of titanium in 1.0 mol/L H3PO4
In 1.0 mol/L H3PO4 at 250 ℃, the titanium suffers severer corrosion. A 25 μm-thick coating of white corrosion product covers the titanium sample surfaces after 24 h immersion. From Fig.3, the morphology of the corrosion product shows fiber bundle like or spaghetti like which grows outward from the titanium surface. The diameter of the fibers is less than 0.5 ?m. The length of the fibers is about 30 ?m. The fibrous morphology indicates that the corrosion product is a substance with strong anisotropical crystal structure.

Fig.3 Morphology of corrosion product of Ti at 250 ℃ after 24 h immersion in 1.0 mol/L H3PO4
As shown in Table 2, the titanium samples lose mass after the corrosion test(after removing the corrosion products). The average corrosion rate of the CP titanium in 1.0 mol/L phosphoric acid is 0.10 mg/(cm2?h). This equals the corrosion rate of 1.9 mm/a by assumption of a linear extrapolation over time. This corrosion rate is high for engineering application. Nevertheless, as mentioned in previous section, the corrosion products firmly adhere to the titanium surface. So, the thick corrosion product layer should have some degree of protection. Therefore, the long time corrosion rate may be lower than 1.9 mm/a.
In comparison of the two concentrations, the effect of concentration on the corrosion of titanium is obvious, that is, higher concentration causes higher corrosion rate. This means that phosphoric acid takes part in the corrosion reactions. In other words, the main corrosion products must be phosphates.
Fig.4(a) shows the XRD pattern of the corrosion products on Ti sample immersed in 0.1 mol/L H3PO4 solution. Fig.4(b) shows the XRD pattern of the corrosion products of Ti sample immersed in 1.0 mol/L H3PO4. Both patterns show the presence of a substance that has five clear reflections at 2θ=12.235?, 14.725?, 20.344?, 24.612? and 29.449?. By checking XRD data base it is identified that this substance is titanium oxide phosphate hydrate (π-Ti2O(PO4)2・2H2O) and its JCPDS number is 00-052-1529. This substance was first synthesized and characterized in 1996 by BORTUN et al [12] by hydrothermal method with TiCl4 and H3PO4 as starting compounds. The first five XRD peaks in Fig.4 match well with their reported data. The fibrous morphology of the corrosion products is also in accordance with their report. The first five peaks in Fig.4(b) are stronger than those in Fig.4(a). This is because the corrosion product layer produced in 1.0 mol/L H3PO4 is thicker than that produced in 0.1 mol/L H3PO4. In addition to reflections from the titanium oxide phosphate hydrate, Fig.4(a) shows also strong Ti reflections. This is because the corrosion product layer is very thin.

Fig.4 XRD patterns of Ti corrosion products in 0.1 mol/L H3PO4 (a) and 1.0 mol/L H3PO4 (b) at 250 ℃
The XRD pattern in Fig.4(b) shows that there are other substances in the corrosion product layer. One could be titanium oxide di-phosphate ((TiO)2P2O7, JCPDS number: 00-039-0207). Another could be titanium oxide (Ti1.87O3, JCPDS number: 01-071-0 151). The formation of titanium oxide di-phosphate is possible because phosphoric acid can condense to form pyrophosphoric acid (H4P2O7) at high temperature. The formation of non-stoichiometric oxide Ti1.87O3 is also possible because the corrosion environment is not aerated and thus lower valance titanium ions may exist in the oxide.
Fig.5 shows the IR spectrum of the corrosion products. The bands at 1 153, 1 068, 1 002 and 967 cm-1 are due to the stretching of bonds in PO4 tetrahedron. The bands at 629, 512 and 466 cm-1 are due to mixed δ(PO4) and δ(Ti―O) vibrations[11]. The 512 and 466 cm-1 bands are not indicated in this work. So, it is obvious that the corrosion products contain PO4 tetrahedron.

Fig.5 IR spectrum of corrosion products
3.3 Discussion on corrosion mechanism
The above result of forming titanium oxide phosphate on titanium surface in phosphoric acid is supported by a number of experimental results in literatures. By nuclear reaction analysis, FERDJANI et al[8] found phosphate incorporation in titanium oxide was formed by anodization in 0.3-3.0 mol/L H3PO4 solution. By qualitative EDS analysis, KRASICKA- CYDZIK[9] found titanium phosphate deposit on amorphous thin layer was formed by anodizing commercially pure titanium in 0.5 mol/L phosphoric acid. By FT-IR study, NARAYANAN et al[13] showed the presence of PO43- in the titanium surface anodized in 0.3 mol/L phosphoric acid. By EDX analysis, BOUKIS et al[2] gave a Ti:P:O molar ratio of 1:1.5:10 of the corrosion products on grade 2 titanium tested in 0.1 mol/kg H3PO4 and 1.5 mol/kg O2 solution at 300 ℃ for 200 h.
To explain the formation of the titanium oxide phosphate in this Ti-H3PO4 system, a thermodynamic analysis is attempted. Table 3 lists the standard state thermodynamic data of selected species in Ti-H3PO4 aqueous solution system. The selection takes into account of the stability and other physical chemistry natures of species in this system. For example, PO43- is not included because the third dissociation constant K3 of H3PO4 is quite small (4.7×10-13 at 25 ℃). Ti2+, Ti3+ and Ti4+ are also not included because of either low stability or high charge.
Table 3 Thermodynamic data of selected species in Ti-H3PO4 aqueous solution system
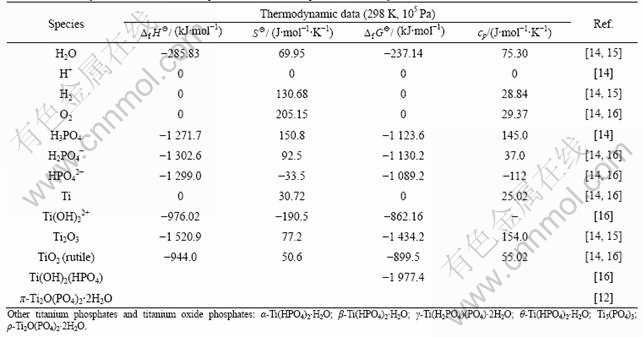
With these data, thermodynamic calculations of some reactions can be carried out. For example, one of the complexion reactions of phosphate ions with titanium ion can be as follows:
Ti(OH)22++HPO42-=Ti(OH)2(HPO4) (1)
The standard Gibbs free energy of this reaction is -26.04 kJ/mol. So, this reaction is thermodynamically favorable.
Based on above experimental results and the thermodynamic analysis, a mechanism for the titanium oxide phosphate formation in Ti-H3PO4 system solution is proposed:
Ti-3e=Ti3+ (2)
Ti3+-e=Ti4+ (3)
Ti4++2H2O=Ti(OH)22++2H+ (4)
Ti(OH)22++HPO42-=Ti(OH)2(HPO4) (5)
2Ti(OH)2(HPO4)=Ti2O(PO4)2?2H2O+H2O (6)
Reactions (2) and (3) are according to FRAYRET [10]. Reaction (4) is the hydrolysis of the highly charged Ti4+ ion. Reaction (5) is the complex of HPO42- with Ti(OH)2+. Reaction (6) is the dehydration of the complexes, leading to the formation of the titanium oxide phosphate.
Beside, the direct reaction of H3PO4 with TiO2 to form titanium oxide phosphate is also thermo- dynamically feasible. Assuming the following reaction:
TiO2+H3PO4=Ti(OH)2(HPO4) (7)
The Gibbs free energy of this reaction at room temperature is -225.74 kJ/mol. The negative value means that titanium oxide is thermodynamically unstable in phosphoric acid solution even at room temperature.
4 Conclusions
1) The corrosion of the commercially pure titanium in phosphoric acid solution at 250 ℃ depends on the acid concentration. At 0.1 mol/L concentration the corrosion is mild while at 1.0 mol/L the corrosion is much sever.
2) The main corrosion product is titanium oxide phosphate hydrate (π-Ti2O(PO4)2?2H2O). The corrosion products grow from titanium surface in the morphology of fiber bundles.
References
[1] LU J, ZHANG J, MAO Z. Corrosion of titanium in supercritical water oxidation environment [J]. Trans Nonferrous Met Soc China, 2002, 12(6): 1091-1094.
[2] BOUKIS N, FRIEDRICH C, DINJUS E. Titanium as reactor material for SCWO applications―First experimental results [C]// Proceedings of the 1998 NACE Conference. Houston, 1998.
[3] TAYLOR D F. Acid corrosion resistance of tantalum, columbium, zirconium and titanium [J]. Industrial and Engineering Chemistry, 1950, 42(4): 639.
[4] SINGH V B, HOSSEINI S M A. The electrochemical and corrosion behavior of titanium and its alloy (VT-9) in phosphoric acid [J]. Corrosion Science, 1993, 34(10): 1723-1732.
[5] SINGH V B, HOSSEINI S M A. Corrosion behavior of Ti-6Al-4V in phosphoric acid [J]. Journal of Applied Electrochemistry, 1994, 24: 250-255.
[6] ROBIN A, ROSA J L, SANDIM H R Z. Corrosion behavior of Ti-4Al-4V alloy in nitric, phosphoric, and sulfuric acid solutions at room temperature [J]. Journal of Applied Electrochemistry, 2001, 31: 455-460.
[7] SINGH V B, GUPTA A. Microstructure and corrosion studies of titanium alloy (IMI834) in acid solutions [J]. Journal of Applied Electrochemistry, 2002, 32: 795-803.
[8] FERDJANI S, DAVID D, BERANGER G. Anodic oxidation of titanium in phosphoric acid baths: Phosphorus incorporation into the oxide [J]. Journal of Alloys and Compounds, 1993, 200: 191-194.
[9] KRASICKA-CYDZIK E. Gel-like layer development during formation of thin anodic films on titanium in phosphoric acid solutions [J]. Corrosion Science, 2004, 46: 2487-2502.
[10] FRAYRET C, BOTELLA P, JASZAY T, DELVILLE M H. Titanium dissolution-passivation in highly chloridic and oxygenated aqueous solutions―Reaction mechanism extended to supercritical water conditions [J]. Journal of the Electrochemistry Society, 2004, 151: B543-B550.
[11] OE T, IWAMORI T, KAWASAKI S, SUZUKI A, DAIMON H, FUJIE K. Corrosion resistance in highly concentrated hydrochloric acid and creep rupture strength of a Ti-Pd alloy for a supercritical water oxidation reactor [J]. Corrosion, 2007, 63: 793-798.
[12] BORTUN A I, KHAINAKOV S A, BORTUN L N, POOJARY D M, RODRIGUEZ J, GARCIA J R, CLEARFIELD A. Synthesis and characterization of two novel fibrous titanium phosphates π-Ti2O- (PO4)2?2H2O [J]. Chemistry of Materials, 1997, 9: 1805-1811.
[13] NARAYANAN R, SESHADERI S K. Phosphoric acid anodization of Ti-6Al-4V: Structural and corrosion aspects [J]. Corrosion Science, 2007, 49: 542-558.
[14] LIDE D R. CRC handbook of chemistry and physics [M]. Boca Raton: CRC Press, 1996.
[15] KUBASCHEWSKI O, ALCOCK C B, SPENCER P J. Materials thermochemistry [M]. Oxford: Pergamon Press, 1993.
[16] ZIEMNIAK S E, JONES M E, COMS K E S. Solubility behavior of titanium oxide in alkaline media at elevated temperatures [J]. Journal of Solution Chemistry, 1993, 22: 601-623.
Foundation item: Project(50371078) supported by the National Natural Science Foundation of China
Corresponding author: LU Jian-shu; Tel: +86-571-88320202; E-mail: jianshul@zjut.edu.cn
DOI: 10.1016/S1003-6326(08)60311-8
(Edited by YANG Bing)