
Characterization of rapid thermally processed LiMn2O4 thin films derived from solution deposition
MA Ming-you(麻明友)1, XIAO Zhuo-bing(肖卓炳)1, Li Xin-hai(李新海)2,
WU Xian-ming(吴显明)1, HE Ze-qiang(何则强)1, CHEN Shang(陈 上)1
1. College of Chemistry and Chemical Engineering, Jishou University, Jishou 416000, China;
2. School of Metallurgical Science and Engineering, Central South University,
Changsha 410083, China
Received 27 December 2005; accepted 27 March 2006
Abstract: Cathode material LiMn2O4 thin films were prepared through solution deposition followed by rapid thermal annealing. The phase identification and surface morphology were studied by X-ray diffraction and scanning electron microscopy. Electrical and electrochemical properties were examined by four-probe method, cyclic voltammetry and galvanostatic charge-discharge experiments. The results show that the film prepared by this method is homogeneous, dense and crack-free. As the annealing temperature and annealing time increase, the electronic resistivity decreases, while the capacity of the films increases generally. For the thin films annealed at different temperatures for 2 min, the thin film annealed at 800 ℃ has the best cycling behavior with the capacity loss of 0.021% per cycle. While for the thin films annealed at 750 ℃ for different times, the film annealed for 4 min possesses the best cycling performance with a capacity loss of 0.025% per cycle. For the lithium diffusion coefficient in LiMn2O4 thin film, its magnitude order is 10-11 cm2・s-1.
Key words: LiMn2O4; lithium ion; thin films; diffusion; electrochemical impedance spectroscopy
1 Introduction
Thin-film lithium-ion batteries have drawn a lot of attention due to their many possible applications, such as smart cards, CMOS-based integrated circuits and microdevices[1-8]. Among a lot of thin films that can be used as cathode material for thin-film lithium-ion batteries, LiMn2O4 is one of the most studied cathode materials for its relatively high voltage plateau, non-toxicity, and good rechargeability[9, 10].
LiMn2O4 thin films have been prepared by a few methods such as pulsed laser deposition[11], electrostatic spray deposition[12] and radio frequency magnetron sputtering[13, 14]. The heat treatment of the thin film is almost conventional furnace annealing. However, it is difficult to produce thin film by these methods with accurate stoichiometry.
Compared with the above-mentioned methods, solution deposition possesses several advantages of excellent control of stoichiometry, low cost and high deposition rate. While rapid thermal annealing has the advantages of short annealing time and relative simplicity. The short rise time to the desired annealing temperature minimizes the film/substrate interface reaction and results in dense and smooth film. Based on these considerations, LiMn2O4 were prepared thin films by solution deposition and rapid thermal annealing(RTA) and their characteristics were studied.
2 Experimental
Stoichiometric amount of lithium acetate and manganese acetate were dissolved in a small amount of deionized water. Then 2-methoxyethanol was added to adjust the viscosity and wetting property of the solution. Dust and other suspended impurities were removed from the solution by filtering through 0.2 mm syringe filters and thus formed LiMn2O4 precursor solution. The wet films were deposited by spin coating LiMn2O4 precursor solution onto substrates. Then the wet films were heated at 350 ℃ in air for 20 min to remove solvents and other organic substances at the heating rate of 10 ℃/min. The deposition and heat treatment procedure were repeated to prepare the desired thickness of films. The multilayered films were finally annealed by rapid thermal annealing.
Phase identification and surface morphology were studied by X-ray diffractometry(XRD) and scanning electron microscopy. The electronic resistivity of the thin films was measured by four probe method. For electrochemical measurements, LiMn2O4 thin films coated on Pt-coated substrate were placed in an open beaker cell which contained 1 mol/L LiPF6 dissolved in ethylene carbonate(EC) and dimethyl carbonate(DMC) (1:1, volumetric ratio). Lithium metal was used as both counter and reference electrodes. The entire cell was assembled in an argon-filled glove box. Electrochemical properties were investigated by cyclic voltammetry and galvanostatic charge-discharge experiments. The cyclic voltammetry were performed at the scan rate of 1 mV/s and the galvanostatic charge-discharge experiments were conducted between the cut-off voltages of 4.3 and 3.5 V under a constant current density of 100 mA/cm2.
3 Results and discussion
The X-ray diffraction patterns of the prepared films are shown in Fig.1. As seen in the figure, three peaks at 2q=18.61?, 36.09? and 43.87? were observed, which correspond to the (111), (311), and (400) reflections of spinel, indicating that the prepared thin films are LiMn2O4. The intensities of the XRD peaks in Figs.1(a) and (b) increase with the increase of annealing temperature and annealing time, indicating that the crystallinity of thin film becomes better.
The scanning electron micrographs of LiMn2O4 thin films annealed under various conditions are shown in Figs.2 and 3. As observed in the figures, the grains of these films increase with the increase of annealing temperature and annealing time, and the films annealed under different conditions are homogeneous and crack-free. No significant defects can be observed for the thin films annealed at 700, 750 and 800 ℃ for 2 min, and the films annealed at 750 ℃ for 1, 2, and 4 min. The grain size is between 20 nm and 50 nm, suggesting that the thin film is very smooth, which is favorable for avoiding the short circuits of thin-film batteries.
The increase of the grain size of the films with the increase of annealing temperature and annealing time is associated with the increase of coalescence speed and coalescence time of grains, i.e., the film annealed at higher temperature or annealed for longer time has a higher coalescence speed or longer coalescence time than that annealed at lower temperature or annealed for a shorter time, leading to the result that the grain size of the former is larger than that of the latter. The growth mechanism can be easily verified by Fig.2 and Fig.3, where Fig.2(d) and Fig.3(d) (magnified by 20 000 times) is two and a half times the area of the films of Fig.2(a) and Fig.3(a) (magnified by 50 000 times). However, the grain number of the former is much less than that of the latter.
Fig.4 displays the resistivity of LiMn2O4 thin films annealed under different conditions. The resistivity of the films decreases from 3.84×103 W・cm to 2.71×103 W・cm as the annealing temperature increases from 700 ℃ to 850 ℃, and from 3.92×103 W・cm to 2.89×103 W・cm as the annealing time increases from 1 min to 8 min. The decrease of the electronic resistivity of the thin film is associated with the improvement of the crystallinity of the thin films.
Fig.5 shows the cyclic voltammograms of the LiMn2O4 thin film annealed at 750 ℃ for 2 min. As seen in the figure, all the cyclic voltammograms scanned at different rates display two couples of redox peaks, which represents the mixed phases and different intercalation stages of lithium ion, suggesting that both the intercalation and deintercalation of lithium ion are carried out in two steps, which can be written as the following two reversible reactions:
1/2Li++1/2e+2λ-MnO2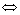 Li0.5Mn2O4 (1)
Li0.5Mn2O4 (1)
1/2Li++1/2e+2λ-MnO2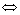 Li0.5Mn2O4 (2)
Li0.5Mn2O4 (2)
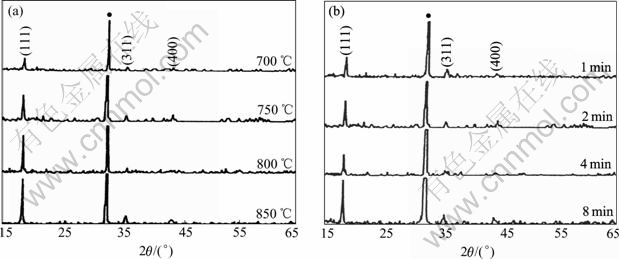
Fig.1 XRD patterns of prepared films annealed under different conditions: (a) Annealed at different temperatures for 2 min; (b) Annealed at 750 ℃ for different times (peak marked with black solid circle belongs to substrate)
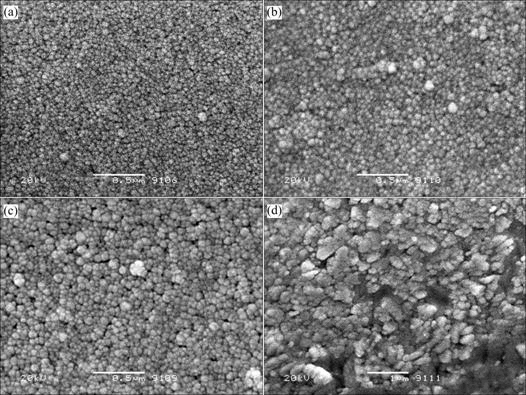
Fig.2 Scanning electron micrographs of LiMn2O4 thin films annealed at different temperatures for 2 min: (a) 700 ℃; (b) 750 ℃; (c) 800 ℃; (d) 850 ℃
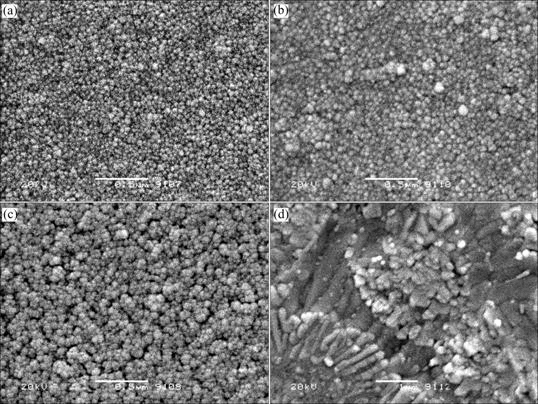
Fig.3 Scanning electron micrographs of LiMn2O4 thin films annealed at 750 ℃ for different time: (a) 1 min; (b) 2 min; (c) 4 min; (d) 8 min
The peak current as a function of n1/2 is shown in Fig.6. The figure shows the peak current has a linear relationship with n1/2, indicating that the thin film possesses good rechargeability. From the following
equation[15], the chemical diffusion coefficient DLi in the solid phase of LiMn2O4 thin film can be determined.
ip=0.446 3n3/2F3/2Ac0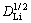 (1/RT)1/2n1/2 (3)
(1/RT)1/2n1/2 (3)
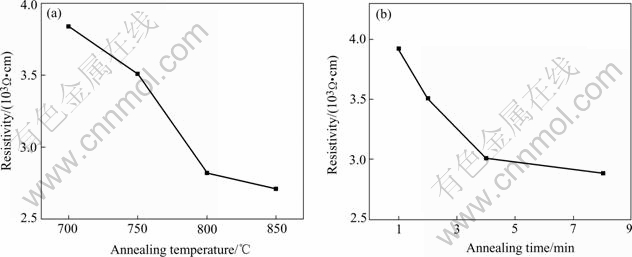
Fig.4 Electronic resistivity of LiMn2O4 thin films annealed under different conditions: (a) Annealed at different temperatures for 2 min; (b) Annealed at 750 ℃ for different time
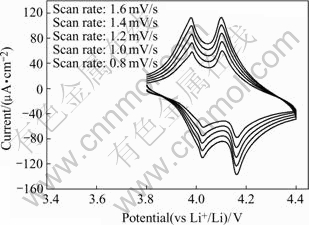
Fig.5 Cyclic volatmmograms of LiMn2O4 thin film scanned at different rates
where n is the number of redox reaction, F is the Faraday constant, A is the surface area of electrode (cm2), c0 is the lithium ion concentration in the solid thin-film electrode (mol?cm-3), DLi is the diffusion coefficient, and n is the scan rate.
Table 1 presents the diffusion coefficients of lithium ion in LiMn2O4 thin films obtained by cyclic voltammetry technique. The table shows that the film annealed under different conditions has different diffusion coefficient. However, the difference is not significant. These values are in agreement with Ref.[16].
Table 2 displays the discharge capacity of LiMn2O4 thin films prepared under different conditions. As observed in the table, as the annealing temperature increases from 700 ℃ to 850 ℃, the capacity of thin film increases from 34 mAh/(cm2?mm) to 40 mAh/(cm2?mm). While for the thin films annealed at 750 ℃ for different times, as the annealing time increases from 1 min to 4 min, the capacity of the film increases from 36 mAh/(cm2?mm) to 41 mAh/(cm2?mm), while as the annealing time increases to 8 min, the capacity of film decreases to 39.5 mAh/(cm2?mm), indicating that long annealing time does not necessarily result in a high capacity for the thin film.
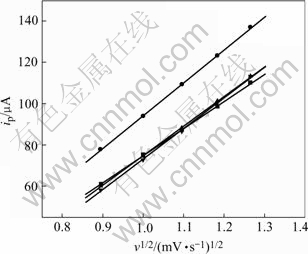
Fig.6 Peak current ip as function of v1/2
Table 1 Diffusion coefficients of Li+ in LiMn2O4 thin films annealed under different conditions
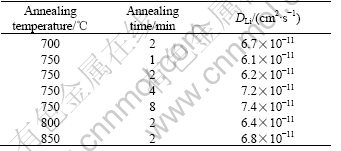
Fig.7 shows the capacity as a function of cycle number for the films annealed under different conditions. For the thin films annealed at different temperature for 2 min, the thin film annealed at 800 ℃ has the best cycling behavior with a capacity loss of 0.021% per cycle. While for the thin films annealed at 750 ℃ for different times, the film annealed for 4 min possesses the best cycling performance with a capacity loss of 0.025% per cycle. The relatively poorer rechargeability of thefilm annealed at 700 ℃ may be caused by its relatively poor crystallinity. While the relatively poorer cycling behavior of the film annealed at 850 ℃ might result from its relatively higher Mn3+ content, which is more stable than Mn4+ at higher heating temperature. The higher Mn3+ content (when Mn3+/(Mn3++ Mn4+)>50%) can deteriorate the rechargeability of LiMn2O4.
Table 2 Capacity of LiMn2O4 thin films prepared under different conditions
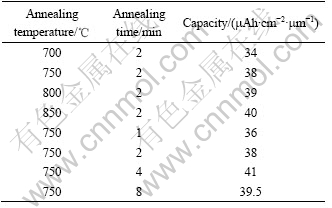
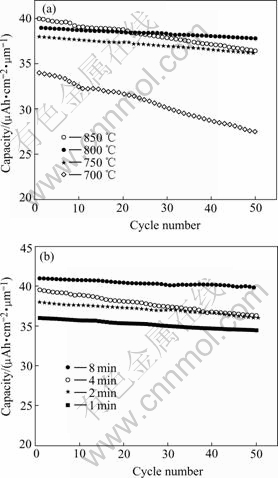
Fig.7 Capacity as function of cycle number for films: (a) Annealed at different temperatures for 2 min; (b) Annealed at 750 ℃ for different times
Compared with LiMn2O4 powders, the capacity loss of LiMn2O4 thin film is very small, and shows very good cycling behavior. This is caused by two factors. The first is that the grains of thin film is much smaller than the powders, these small grains greatly increase the effective surface area of electrode, which makes the intercalation and deintercalation of lithium ion into and out of LiMn2O4 much easier, resulting in good cycling behavior. The second is that the thickness of thin film is very small, i.e., the diffusion route of lithium ion inside the thin-film electrode is very short, which can avoid the cathode entering JAHN-TELLER effect zone during deep discharge. Taking the discharge process as an example, during the intercalation of lithium ion, if the electrode is thick, the lithium concentration at the interfaces near electrolyte and current collector will be different due to the small diffusion rate of lithium ion inside the electrode. In this case, although the whole electrode may have not entered the JAHN-TELLER effect zone during deep discharge, the surface of the electrode have been already in the JAHN-TELLER effect zone, resulting in bad cycling behavior. While for the thin-film electrode, the electrode is very thin, which can greatly decrease the lithium concentration difference throughout the electrode and avoid the electrode entering the JAHN-TELLER effect zone during deep discharge, leading to good cycling behavior.
4 Conclusions
LiMn2O4 thin films prepared by solution deposition and rapid thermal annealing are homogeneous, dense and crack-free. The resistivity of the films decreases from 3.84×103 W・cm to 2.71×103 W・cm as the annealing temperature increases from 700 ℃ to 850 ℃, and from 3.92×103 W・cm to 2.89×103 W・cm as the annealing time increases from 1 min to 8 min. The capacity of thin film generally increases with the increase of the annealing temperature and the annealing time. For the thin films annealed at different temperatures for 2 min, the thin film annealed at 800 ℃ has the best cycling behavior with the capacity loss of 0.021% per cycle after being cycled 50 times. While for the thin films annealed at 750 ℃ for different times, the film annealed for 4 min possesses the best cycling performance with a capacity loss of 0.025% per cycle. For the lithium diffusion coefficient in LiMn2O4 thin film, its magnitude order is 10-11 cm2・s-1.
References
[1] West W C, Whitacre J F, Lim J R. Chemical stability enhancement of lithium conducting solid electrolyte plates using sputtered LiPON thin films[J]. J Power Sources, 2004, 126: 134-138.
[2] Lee S J, Bae J H, Lee H W, Baik H K, Lee S M. Electrical conductivity in Li-Si-P-O-N oxynitride thin-films[J]. J Power Sources, 2003, 123: 61-64.
[3] Moon H S, Park J. Improvement of cyclability of LiMn2O4 thin films by transition-metal substitution[J]. J Power Sources, 2003, 119-121: 717-720.
[4] Yoon Y S, Kim J S, Choi S H. Structural and electrochemical properties of vanadium oxide thin films grown by d.c. and r.f. reactive sputtering at room temperature[J]. Thin Solid Films, 2004, 460: 41-47.
[5] Kim H K, Seong T Y, Yoon Y S. Characteristics of rapid-thermal-annealed LiNi1-xCoxO2 cathode films for all-solid-state rechargeable thin film microbatteries[J]. Thin Solid Films, 2004, 447-448: 619-625.
[6] Choi W G, Yoon S G. Structural and electrical properties of LiCoO2 thin-film cathodes deposited on planar and trench structures by liquid-delivery metalorganic chemical vapour deposition[J]. J Power Sources, 2004, 125: 236-241.
[7] Liao C L, Lee Y H, Yu H C, Fung K Z. Structure characterization and electrochemical properties of RF sputtered lithium nickel cobalt oxide thin films[J]. Electrochim Acta, 2004, 50: 461-466.
[8] Rho Y, Kanamura T. Fabrication of thin film electrodes for all solid state rechargeable lithium batteries[J]. J Electroanal Chem, 2005, 559: 69-75.
[9] Matsuda K, Taniguchi I. Relationship between the electrochemical and particle properties of LiMn2O4 prepared by ultrasonic spray pyrolysis[J]. J Power Sources, 2004, 132: 156-160.
[10] Eftekhari A. Aluminum oxide as a multi-function agent for improving battery performance of LiMn2O4 cathode[J]. Solid State Ionics, 2004, 167: 237-242.
[11] Liu H R, Chu Y Q, Fu Z W, Qin Q Z. Characterization and preparation of NiO-V2O5 composite film cathodes[J]. J Power Sources, 2003, 124: 163-169.
[12] Shui J L, Jiang G S, Xie S, Chen C H. Thin films of lithium manganese oxide spinel as cathode materials for secondary lithium batteries[J]. Electrochim Acta, 2004, 49: 2209-2213.
[13] Moon H S, Park J W. Improvement of cyclability of LiMn2O4 thin films by transition-metal substitution[J]. J Power Sources, 2003, 119-121: 717-720.
[14] Lee K L Jung J Y, Lee S W, Moon H S, Park J W. Electrochemical characteristics and cycle performance of LiMn2O4/a-Si microbattery[J]. J Power Sources, 2004, 130: 241-246.
[15] Bard A J, Faulkner L R. Electrochemical Methods [M]. New York: Wiley Press, 1980. 218.
[16] Dokko K, Nishizawa F, Mohamedim M, Umeda M, Uchida I, Akimoto J, Takahashi Y, Gotoh Y, Mizuta S. Electrochemical studies of Li-ion extraction and insertion of LiMn2O4 single crystal[J]. Electrochem Solid-State Lett, 2001, 4: A151-153.
Foundation item: Projects(04JJ40038; 04C475; 05C140) supported by Hunan Provincial Natural Science Foundation and Hunan Provincial Education Department
Corresponding author: MA Ming-you; Tel: +86-743-8563911; E-mail: mamy_9589@163.com
(Edited by PENG Chao-qun)