�Ӹ߹�����軯������ˮ��Ԥ����-��ѡ����
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2013���4��
�������ߣ������� �÷ ���Ƚ�
����ҳ�룺1165 - 1173
�ؼ��ʣ�ˮ�����軯��������Ʒλ���������ʣ��������
Key words��water leaching; cyanide tailings; grade of magnetic concentrate; recovery rate of iron; yield of magnetic concentrate
ժ Ҫ�����ա�ˮ����Ԥ��������ȥ���߹衢�����軯���е����ʣ����������о��˹������ˮ�������ж���ȡ����Ӱ�졣 ͨ����ѧ�ɷַ���������������Ա�ֱ�ӻ�ԭ���մŷ��뷽���ͻ�ԭ����������ŷ��뷽���� ���������ˮ�����ѡ��������Ʒλ���������ʷֱ�Ϊ10%��15%����ԭ����-ˮ��-��ѡ�������ʺ��軯����������ȡ���о��˷�Ӧ�������������ʵ�Ӱ�죬ȷ����ѷ�Ӧ����Ϊ���¶�60 ��C��ʱ��5 min��Һ�̱�15:1�������ٶ�20 r/min����ѡ����2 A������������£�������������ƷλΪ56.82%��������Ϊ79.39%������, ͨ��XRD��BEI��XRF��EDS�ȷ����о���ˮ�����������軯�����۽ṹ�������̡� �������, �ڱ��չ��������ɵĿ����Ի�����������ˮ�����̱�ϴ�������ܽ������ͨ��ˮ����������Ǵ������ʣ��Ӷ�ʵ�������ʺ�������Ч���롣
Abstract: Pretreatment of high content of Si- and Al-containing cyanide tailings by water leaching to remove some impurities, such as the major impurities minerals of Si and Al, as well as its effect on Fe extraction in the water leaching process was investigated. The effects of different parameters on iron recovery were studied, and the reaction parameters were proposed as follows: sodium carbonate content of 30%, water leaching at 60 ��C for 5 min, liquid/solid ratio of 15:1, and exciting current of 2 A. Under these optimal conditions, magnetic concentrate containing 59.11% total iron and a total iron recovery rate of 76.12% was obtained. In addition, the microstructure and phase transformation of the process of water leaching were studied by X-ray powder diffraction technique (XRD), Electronic image of backscattering (BEI), X-ray fluorescence (XRF), and energy dispersive spectrometry (EDS). The results indicate that the soluble compound impurities generated in the roasting process are washed out, and the dissoluble substances enter into nonmagnetic materials by water leaching, realizing the effective separation of impurities and Fe.
Trans. Nonferrous Met. Soc. China 23(2013) 1165-1173
Ya-li ZHANG, Huai-mei LI, Xian-jin YU
College of Chemical Engineering, Shandong University of Technology, Zibo 255081, China
Received 22 February 2012; accepted 10 April 2012
Abstract: Pretreatment of high content of Si- and Al-containing cyanide tailings by water leaching to remove some impurities, such as the major impurities minerals of Si and Al, as well as its effect on Fe extraction in the water leaching process was investigated. The effects of different parameters on iron recovery were studied, and the reaction parameters were proposed as follows: sodium carbonate content of 30%, water leaching at 60 ��C for 5 min, liquid/solid ratio of 15:1, and exciting current of 2 A. Under these optimal conditions, magnetic concentrate containing 59.11% total iron and a total iron recovery rate of 76.12% was obtained. In addition, the microstructure and phase transformation of the process of water leaching were studied by X-ray powder diffraction technique (XRD), Electronic image of backscattering (BEI), X-ray fluorescence (XRF), and energy dispersive spectrometry (EDS). The results indicate that the soluble compound impurities generated in the roasting process are washed out, and the dissoluble substances enter into nonmagnetic materials by water leaching, realizing the effective separation of impurities and Fe.
Key words: water leaching; cyanide tailings; grade of magnetic concentrate; recovery rate of iron; yield of magnetic concentrate
1 Introduction
Cyanide tailing is a kind of solid waste produced in the process of gold extraction from gold ore. With the increasing demand of gold, both in China and abroad, a continuous increase of hazardous industrial wastes like cyanide tailings generated by gold industry is becoming a serious problem, and there are approximately 20 million tons of cyanide tailings produced in China every year [1-3].
Cyanide tailings were characterized as high contents of hazardous materials, which could cause serious environmental problems. Huge areas of land are required for the storage of cyanide tailings, which may cause the reduction of farming lands; the construction and maintenance of cyanide tailings disposal sites also increase the production cost of gold making plants. However, cyanide tailings usually contain some valuable metals, such as Fe and Au, which can be used as secondary resources. Generally, how to solve environmental problems and realize comprehensive utilization of valuable metals simultaneously is needed to pay close attention by the fields of environment and resources.
Mineral compositions, physical and chemical properties of cyanide tailings differ from different gold plants. Cyanide tailings from Shandong gold plants contain high content of impurities, such as SiO2 and Al2O3, which exist together with Fe2O3 in a complex relationship, and can bring certain difficulties to the extraction of iron. Therefore, how to realize the effective separation of iron, aluminum and silicon is a problem needed to be solved urgently which constraints the improving of grade of magnetic concentrate and recovery rate of iron.
There are several techniques of iron recovery reported in literature. One such example is the use of magnetizing roasting-magnetic separation technique to recover iron from red mud [4-8], and iron ore tailings [9-11]. In other studies, sponge iron or reduced iron is produced from pyrite cinder [12], red mud [13,14], oily hot rolling mill sludge [15] and copper matte smelting slag [16] by a direct reduction-magnetic separation technique. It has also been reported traditional mineral processing techniques used for recovering iron from ore tailings and smelting slag [17], nickel metallurgical slag [18], blast furnace gas ash [19] and copper converter slag [20]. Magnetizing roasting or direct reduction followed by magnetic separation has been demonstrated to be effective for iron recovery from solid wastes [3,5,6,12-16]. However, these techniques mentioned above are not applicable to iron recovery from cyanide tailings, which have complex structures and were rarely seen as the research objects.
As the high iron content of cyanide tailings and their complex embedded relationship, some traditional researches were carried out to recover iron. However, only about 50% of grade of magnetic concentrate and about 55% of recovery rate of iron can be obtained, so they are not effective ways compared with the huge amount of the production of cyanide tailings. In this approach, the recovery of iron can be improved by water leaching pretreatment. It should be noted that after the water leaching pretreatment, the soluble materials could solve into the water solution, and the dissoluble materials entered into the magnetic tailings with the nonmagnetic materials by magnetic separation. By water leaching pretreatment, the grade of magnetic concentrate increased due to the removal of some impurities, such as Si and Al, during the process of pretreatment, which can realize effective separation of Fe, Si and Al. Therefore, the water leaching pretreatment is a profitable process, which could help the cyanide tailings win a higher economic value and environmental benefit. So, in this work, the recovery of iron from cyanide tailings using water leaching pretreatment was studied and the reaction process was analyzed.
2 Experimental
2.1 Raw materials
Cyanide tailings used in the study were the residual slags after extracting gold from gold-bearing ores. The chemical composition is given in Table1.
Table 1 Chemical composition of cyanide tailings

The total iron content was approximately 27.69%. But the contents of SiO2 and Al2O3 were relatively high in the tailings. X-ray powder diffraction (XRD) pattern of cyanide tailings is shown in Fig. 1.
The XRD pattern (Fig. 1) shows hematite, quartz and muscovite as the major components. The SEM images of cyanide tailings are shown in Fig. 2.

Fig. 1 XRD pattern of cyanide tailings
According to Fig. 2, Si, Al and Fe are embedded with each other in an extremely complex way, which caused the hematite not to be reduced effectively, so that the grade of magnetic concentrate and the recovery rate of iron were relatively low. So how to make Fe, Al and Si separate effectively and lessen the content of these main impurities seems to be an important problem needed to be solved urgently.
The roasted sample used in the study was obtained under the previous best reduction roasting conditions. These optimum conditions are proposed, i.e., ratio of activated carbon to cyanide tailings to sodium sulfate as 10:100:10, roasting at 750 ��C for 60 min.
2.2 Method
After the reduction roasting process, the roasted product was taken out rapidly and quenched with water quickly, then vacuum filtered, and kiln dried. The dried sample was ground and then put into glass beakers with water to form slurry with a certain liquid/solid ratio. The beaker was placed in an electrical heating oil-bath vessel equipped with a continuous agitating device, and leached with warm water at the required temperature. After that, the slurry was separated by buchcer funnel into filtrate rich in soluble materials and filter cake was rich in Fe3O4 mainly. The filter cake was dried and then matched into about 30% slurry, then the slurry was separated by a low intensity magnetic separator (model : XCGS-��50) with working electrical current of 2 A. The grade of magnetic concentrate was analyzed by a chemical method, and the recovery rate of iron was deduced according to mass balance in the magnetic separation process. The flow sheet in principle of novel process is shown in Fig. 3.
3 Results and discussion
3.1 Contrast of three different processes
Based on the optimal reduction roasting conditions, experiments of reduction roasting-acid leaching�C magnetic separation and reduction roasting-water leaching-magnetic separation were conducted under different conditions. Other optimum reduction roasting conditions are proposed, i.e., ratio of activated carbon to cyanide tailings to sodium sulfate as 10:100:10, roasting at 750 ��C for 60 min, and compared the results with each other to determine an appropriate process flow. The results are shown in Tables 2- 6.

Fig. 2 SEM image (a) of cyanide tailings and corresponding line-by-line scanning images of Si (b), Al (c) and Fe (d)

Fig. 3 Flow sheet in principle of novel process
By comparing the received grade of magnetic concentrate and recovery rate of iron under different additions with each other, it can be found that both the grade of magnetic concentrate and recovery rate of iron obtained by water leaching pretreatment are higher than those obtained by the other two processes. Especially, when the sodium carbonate content is 3%, and sodium sulfate is 10%, the grade of magnetic concentrate of 56.82% and the recovery rate of iron of 79.39% can be obtained. Therefore, water leaching pretreatment is an effective way for improving both the grade of magnetic concentrate and recovery rate of iron. So, in this research, the water leaching pretreatment was studied and experiments of different parameters were carried out.
Table 2 Results with 10% Na2SO4
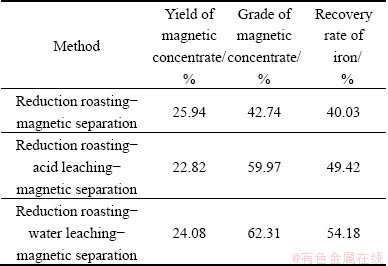
Table 3 Results without Na2SO4
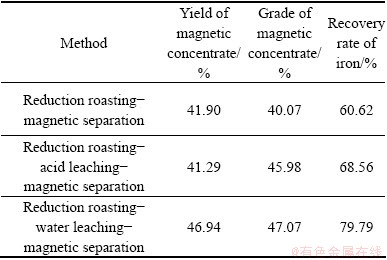
Table 4 Result with 10% Na2SO4 and 3% Na2CO3

Table 5 Results with 10% Na2CO3

Table 6 Results with 10% Na2SO4 and 6% Na2CO3

3.2 Water leaching pretreatment
3.2.1 Effect of sodium carbonate content on iron recovery rate
To optimize the amount of sodium carbonate addition, various sodium carbonate contents were tested. Other parameters were kept constant: the optimum reduction roasting conditions, particle size of roasted sample after rod mill at 85% less than 74 ��m, leaching temperature of 60 ��C, leaching time of 5 min, liquid/solid ratio of 5:1, exciting current of 2 A, and stirring rate of 20 r/min. The effect of sodium carbonate content on iron recovery is shown in Fig. 4.

Fig. 4 Effects of sodium carbonate content on iron recovery
As shown in Fig. 4, there was nonlinear relationship between parameters of iron recovery rate and sodium carbonate content, the joining of additives (sodium sulfate and sodium carbonate) can greatly improve the physical and chemical properties of the samples, and make the samples loose and porous, which is helpful to reduction reaction. When the sodium carbonate content was about 3%, each parameter of iron reduction achieved a peak, so the optimum sodium carbonate content was about 3%.
3.2.2 Effect of leaching time on iron recovery
By keeping the optimum reduction roasting conditions, sodium carbonate content of 3%, liquid/solid ratio of 5:1, the mixtures were leached at 60 ��C for different time; the roasted samples were ground to particle size at 85% less than 74 ��m, at exciting current of 2 A, and stirring rate of 20 r/min. The effect of leaching time on iron recovery is shown in Fig. 5.

Fig. 5 Effects of leaching time on iron recovery
As shown in Fig. 5, in the initial stage, the yield of magnetic concentrate, grade of magnetic concentrate and recovery rate of iron decrease rapidly as the leaching time increases from 5 min to 10 min, then the yield of magnetic concentrate and the recovery rate of iron decrease slowly but continually, but the grade of magnetic concentrate increases slowly before 10 min. It is maybe because that the content of iron increases gradually with the extension of leaching time, and soluble compounds produced in the roasting process enter into the solution. Therefore, at the initial stage of reaction, both the grade of magnetic concentrate and recovery rate of iron acquired are the highest. The optimum leaching time is 5 min.
3.2.3 Effect of leaching temperature on iron recovery
In order to investigate the optimum leaching temperature, different leaching temperatures were studied respectively, and other experiment parameters were kept as follows: sodium carbonate content of 3%, particle size of roasted sample after rod mill at 85% less than 74 ��m, the liquid/solid ratio of 5:1, leaching time of 5 min, exciting current of 2 A, stirring rate of 20 r/min. The results are shown in Fig. 6.
Figure 6 indicates that while the leaching temperature is less than 60 ��C, the grade of magnetic concentrate increases remarkably with the increase of leaching temperature. This indicates that the higher temperature promotes the molecular motion and reduces solution viscosity, which is more conductive to leaching. However, it can be seen that the grade of magnetic concentrate decreases slowly when the leaching temperature is higher than 60 ��C. It is maybe because magnetite produced in roasting process is easy to form complex iron compounds with some impurities. This causes the loss of iron concentrate, and the decrease of both the grade of magnetic concentrate and recovery rate of iron. This may be because the burning loss of water-leached ore keeps increasing with the increase of leaching temperature, so the recovery rate of iron tends to decrease gradually. The optimum leaching temperature, therefore, is 60 ��C.

Fig. 6 Effects of leaching temperature on iron recovery
3.2.4 Effect of liquid to solid ratio on iron recovery
Samples were leached at 60 ��C for 5 min under different liquid/solid ratios, particle size of roasted sample after rod mill at 85% less than 74 ��m, sodium carbonate content of 3%, exciting current of 2 A, and stirring rate of 20 r/min. The effect of liquid/solid ratio on iron recovery is shown in Fig. 7.

Fig. 7 Effects of liquid/solid ratio on iron recovery
It indicates that when the liquid-to-solid ratio is small, pulp viscosity is high, and solution is easy to reach saturation at leaching interface, preventing the leaching reaction of bounding material. When the liquid/solid ratio keeps increasing, mass transfer speed is improved correspondingly and the solution material at the liquid-to-solid interface keeps speeding outside, which makes the reactions go well with the best speed. But when the liquid/solid ratio goes on increasing, the concentration of untreated and residual sodium carbonate in solution drops, which is detrimental to the dissolution of the impurities. So, considering the grade of magnetic concentrate and recovery rate of iron simultaneously, the liquid/solid ratio is chosen to be 15:1, and magnetic concentrate containing 59.11% total iron and a total iron recovery rate of 76.12% was obtained.
3.2.5 Effect of exciting current on iron recovery
By keeping liquid/solid ratio of 15:1, the mixtures were roasted at the temperature of 60 ��C for 5 min, sodium carbonate content of 3%, particle size of roasted sample after rod mill at 85% less than 74 ��m, stirring rate of 20 r/min. The effects of exciting current on iron recovery are shown in Fig. 8
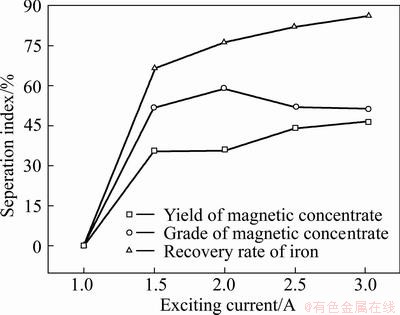
Fig. 8 Effects of exciting current on iron recovery
It can be seen from Fig. 8 that no magnetic concentrate can be separated with the magnetic separator when the exciting current is 1 A. While the exciting current is over 1 A, the recovery rate of iron increases remarkably from 1 A to 1.5 A, and then increases slowly from 1.5 A to 3 A. When exciting current is about 2 A, the grade of magnetic concentrate achieves a peak. It is probably because magnetic concentrate is hard to be chosen completely when the exciting current is smaller, namely, magnetic field intensity is weaker. When the exciting current is larger, impurity mineral mingles with the magnetic concentrate, producing magnetic reunion phenomenon and causing the yield of magnetic concentrate to increase, but the grade decreases due to the existence of nonmagnetic impurities mineral. Based on the results of the experiment, the best exciting current is 2 A.
3.3 Analysis of chemical composition and crystalline phase
3.3.1 X-ray diffraction analysis
The XRD analysis results of the roasted sample, water leached sample, magnetic concentrate and magnetic tailings under optimal conditions are shown in Fig. 9, respectively.
The diffraction peaks of the roasted sample in comparison with the water leached sample for the mineral phases can be seen obviously. This indicates that the diffraction peaks of hydrobiotite disappear after water leaching, magnetite is the major mineral that composes the water leached samples, and magnesioferrite the minor ones. Other diffraction peaks also indicate the presence the quartz, gismondine and sidorenkite minerals. This indicates that the impurities minerals are washed out by the water leaching pretreatment, which makes Fe more available for benefication, and the improving of the grade of magnetic concentrate and the recovery rate of iron. After magnetic separation, it can be seen from the XRD pattern of magnetic concentrate, that magnetite was the main mineral phase in the magnetic concentrate, and the diffraction peaks also indicated the small amount presence of magnesioferrite and quartz which were removed incompletely. This was the reason that the grade of magnetic concentrate reached only 59.11%, and was not very high. Meanwhile, in comparison with the XRD pattern of water leached sample, the peaks of gismondine and sidorenkite disappeared completely after magnetic separation. Moreover, the peaks of quartz, sidorenkite, gismondine and magnesioferrite appeared in the magnetic tailings. Therefore, it can be concluded that impurities of gismondine and sidorenkite were nonmagnetic and dissoluble, and entered into the magnetic tailings with the major impurities of quartz by magnetic separation. The possible chemical reactions of the whole process are as follows:
3Fe2O3+C=2Fe3O4+CO (1)
C+CO2=2CO (2)
3Fe2O3+CO=2Fe3O4+CO2 (3)
MgO+Fe2O3=MgFe2O4 (4)
Na2CO3+2MnO2+P2O5+2Na2O=2Na3Mn(PO4)(CO3) (5)
CaO+Al2O3+2SiO2+4H2O=CaAl2Si2O8��4H2O (6)
3.3.2 Energy dispersive spectrometry analysis
The roasted sample and the water leached sample were prepared under optimized conditions as follows: roasting temperature of 750 ��C, roasting time of 60 min, mass ratio of cyanide tailings to activated carbonate to sodium sulfate to sodium carbonate at 100:10:10:3, particle size of 85% less than 74 ��m in the roasted sample, leaching temperature of 60 ��C, leaching time of 5 min, liquid/solid ratio of 15:1, exciting current of 2 A, and stirring speed of 20 r/min. SEM-EDS was used to understand the experimental results discussed above. The SEM-EDS images are shown in Fig. 10.
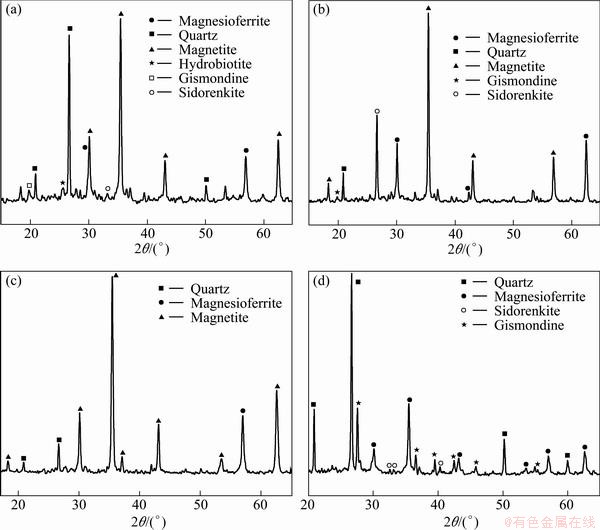
Fig. 9 XRD patterns of roasted sample (a), water-leached sample (b), magnetic concentrate (c) and magnetic tailings (d)
It can be seen from the SEM-EDS images that metallic iron particles occupied almost all of the image of water leached sample; a small amount of slag with fine particle sizes, containing Si, Al, Na, Ca, etc, was observed in the water leached sample for intergrowth with metallic iron particles, and was hard to be removed completely, as supported by XRD pattern shown in Fig. 6. Meanwhile, by comparing EDS images of roasted sample and water leached sample, it can be found that the diffraction peaks of K and Mg disappeared completely, and the diffraction peaks of Si and Al, etc, get lower, the diffraction peaks of Fe get higher, which was also supported by the mass fraction of main elements in roasted sample and water leached sample shown in Table 7. Si content dropped from 14.83% to 7.88%, Al content dropped from 4.43% to 1.10%, and Fe content rose from 30.09% to 42.38%. Therefore, it can be concluded that the soluble compounds can be washed out by the water leaching pretreatment, meanwhile the dissoluble and nonmagnetic materials entered into the magnetic tailings after magnetic separation and then the magnetic concentrate was in beneficiation, so the grade of magnetic concentrate and recovery rate of iron were improved.
3.3.3 X-ray fluorescence analysis
By comparing the mass fractions of main elements in cyanide tailings and water-leached sample, it can be seen from Table 7 that the contents of Si and Al in water leached sample reduced from 11.50% to 7.88%, and from 3.74% to 1.10%, respectively, and Fe content rose from 27.69% to 42.38%, but the content of Na also increased from 0.96% to 1.40% which was in accordance with XRD patterns in Fig. 9 and the SEM-EDS images in Fig. 10. Therefore, it can be concluded that effective separation of Fe, Si and Al can be realized by the water leaching pretreatment, and both the grade of magnetic concentrate and recovery rate of iron were improved.
4 Conclusions
1) Major chemical compositions of cyanide tailings are Fe2O3, SiO2 and Al2O3. Hematite, quartz and magnesioferrite exist in cyanide tailings as main phases, and most of iron and quartz are fine-disseminated together, which is hard to be separated.
2) Sodium sulfate content, leaching temperature, leaching time, ratio of liquid to solid, and exciting current are five main factors which have effects on recovery of iron. Through corresponding five groups of experiments, optimum conditions are obtained as follows: sodium carbonate content of 30%, leaching temperature at 60 ��C for 5 min, ratio of liquid to solid as 15:1, exciting current of 2 A. Under the optimum conditions, the magnetic concentrate grade of 59.11% Fe and the recovery rate of iron of 76.12% are obtained.
3) The soluble substances formed in the roasting process are washed out by water leaching pretreatment, and the dissoluble and nonmagnetic substances enter into nonmagnetic tailings after magnetic separation. But a small amount of SiO2 still exists in magnetic concentrate, making the grade of magnetic concentrate and the recovery rate of iron not very high, which needs to be studied further.

Fig. 10 SEM images (a, c) and EDS patterns (b, d) of roasted sample (a, b) and water-leached sample (c, d)
Table 7 Mass fraction of main elements in original slag, roasted sample and leached sample

References
[1] ZHANG Fu-yuan, ZHANG Yu-hua. Study on the recovery of gold and silver from cyanide residue [J]. Rare Metal Materials and Engineering, 2007, 36(3): 335-337.
[2] ZHANG S, XUE X, LIU X, DUAN P, YANG H, JIANG T, WANG D, LIU R. Current situation and comprehensive utilization of iron ore tailing resources [J]. Journal of Mineral Science, 2006, 42(4): 403-408.
[3] LI Chao, SUN Heng-hu, BAI Jing, LI Long-tu. Innovative methodology for comprehensive utilization of iron ore tailings: Part 1. The recovery of iron from iron ore tailings using magnetic separation after magnetizing roasting [J]. Journal of Hazardous Materials, 2010, 174: 71-77.
[4] YANG Hui-fen, JING Li-li, ZHANG Bao-gang. Recovery of iron from vanadium tailings with coal-based direct reduction followed by magnetic separation [J]. Journal of Hazardous Materials, 2011, 185: 1405-1411.
[5] SUN Yong-feng, DONG Feng-zhi, LIU Jiong-tian, WANG Shu-hong. Technology for recovering iron from red mud by Bayer process [J]. Metal Mine, 2009, 9: 176-178. (in Chinese)
[6] LIU Wan-chao, YANG Jia-kuan, XIAO Bo. Application of Bayer red mud for iron recovery and building material production from alumosilicate residues [J]. Journal of Hazardous Materials, 2009, 161: 474-478.
[7] WASMUTH H D, UNKELBATH, K H. Recent developments in magnetic separation of feebly magnetic minerals [J]. Minerals Engineering, 1991, 4: 825-837.
[8] WATSON J H P. Status of superconducting magnetic separation in the mineral industry [J]. Minerals Engineering, 1994, 7: 737-746.
[9] CUI Z, LIU Q, ETSELL T H. Magnetic properties of ilmenite, hematite and oilsand minerals after roasting [J]. Minerals Engineering, 2002, 15: 1121-1129.
[10] SONG S, LU S, LOPEZ-VALDIVIESO S. Magnetic separation of hematite and limonite fines as hydrophobic flocs from iron ores [J]. Minerals Engineering, 2002, 15: 415-422.
[11] SVOBODA J, FUJITA T. Recent developments in magnetic methods of material separation [J]. Minerals Engineering, 2003, 16: 785-792.
[12] GONG Zhu-qing, GONG Sheng ZHOU Bo, CHEN Bai-zhen. A study on techniques of producing sponge iron from pyrite cinder [J]. Mining and Metallurgical Engineering, 2006, 26(1): 45-48. (in Chinese)
[13] LI L Y. A study of iron mineral transformation to reduce red mud tailing [J]. Waste Manage, 2001, 21: 525-534.
[14] HUANG Zhu-cheng, CAI Ling-bo, ZHANG Yuan-bo, YANG Yong-bin, JIANG Tao. Study on the sponge iron preparation by direct reduction of high iron red mud by Bayer process [J]. Metal Mine, 2009, 39(3): 173-177. (in Chinese)
[15] MAWEJA K, MUKONGO T, MUTOMBO I. Cleaning of a copper matte smelting slag from a water-jacket furnace by direct reduction of heavy metals [J]. Journal of Hazardous Materials, 2009, 164: 856-862.
[16] JIN W P, JOONG C A, SONG H. Reduction characteristics of oily hot rolling mill sludge by direct reduced iron method [J]. Resources, Conservation and Recycling, 2002, 34(2): 129-140.
[17] CHEN Z Y, CHEN Q H. The recovery of iron from iron ore tailings and slag using strong magnetic separation and gravity separation [J]. Metal Mine, 2009, 39(9): 182-184. (in Chinese)
[18] DONG Hai-gang, GUO Yu-feng, JIANG Tao, LI Guang-hui, YANG Yong-bin. Study on recovering magnetite from nickel metallurgical residue with iron [J]. Mining and Metallurgical Engineering, 2008, 28(1): 37-39. (in Chinese)
[19] XU Bai-hui, WANG Er-jun, YANG Ju-wen. Research on recovery of iron and carbon from blast furnace gas ash [J]. Conservation and Utilization of Mineral Resources, 2007, 3: 51-54. (in Chinese)
[20] WANG Heng. Recovery of copper and iron in the converter slag from copper smelter [J]. Journal of Guangdong Non-ferrous Metals, 2003, 13(2): 83-88. (in Chinese).
�������÷�����Ƚ�
ɽ��������ѧ ��ѧ����ѧԺ���Ͳ� 255081
ժ Ҫ�����ա�ˮ����Ԥ��������ȥ���߹衢�����軯���е����ʣ����������о��˹������ˮ�������ж���ȡ����Ӱ�졣 ͨ����ѧ�ɷַ���������������Ա�ֱ�ӻ�ԭ���մŷ��뷽���ͻ�ԭ����������ŷ��뷽���� ���������ˮ�����ѡ��������Ʒλ���������ʷֱ�Ϊ10%��15%����ԭ����-ˮ��-��ѡ�������ʺ��軯����������ȡ���о��˷�Ӧ�������������ʵ�Ӱ�죬ȷ����ѷ�Ӧ����Ϊ���¶�60 ��C��ʱ��5 min��Һ�̱�15:1�������ٶ�20 r/min����ѡ����2 A������������£�������������ƷλΪ56.82%��������Ϊ79.39%������, ͨ��XRD��BEI��XRF��EDS�ȷ����о���ˮ�����������軯�����۽ṹ�������̡� �������, �ڱ��չ��������ɵĿ����Ի�����������ˮ�����̱�ϴ�������ܽ������ͨ��ˮ����������Ǵ������ʣ��Ӷ�ʵ�������ʺ�������Ч���롣
�ؼ��ʣ�ˮ�����軯��������Ʒλ���������ʣ��������
(Edited by Xiang-qun LI)
Foundation item: Projects (ZR2010EL006, Y2007F60) supported by the National Science Foundation of Shandong Province of China; Project (J12LA04) supported by High Education Science Technology Program of Shandong Province, China
Corresponding author: Xian-jin YU; Tel: +86-533-2781681; E-mail: xjy@sdut.edu.cn;
DOI: 10.1016/S1003-6326(13)62579-0

