Trans. Nonferrous Met. Soc. China 23(2013) 420-425
Effect of hexagonal-BN on phase transformation of additive-free Si3N4/SiC nanocomposites prepared from amorphous precursor
Alfian NOVIYANTO1, Dang-Hyok YOON1, Kyungseok LEE2, Young Moon KIM3, Doo-In KIM2, Young-Keun JEONG2, Kwang Ho KIM2, Sehun KWON2, Young-Hwan HAN1
1. School of Materials Science and Engineering, Yeungnam University Gyeongsan 712-749, Korea;
2. National Core Research Center for Hybrid Materials Solution, Pusan National University, Busan, Korea;
3. Samsung Mobile Display, Korea
Received 2 May 2012; accepted 21 September 2012
Abstract: Si3N4/SiC nanocomposites are well known and attractive for advanced ceramic applications due to excellent mechanical and thermal properties, which make them suitable for use in turbine engines, heat exchangers, and other sophisticated applications. However, without the presence of additives, the fabrication of Si3N4/SiC composites is difficult. The additives form a liquid phase during sintering and facilitate the densification of the composite. However, the additives present a drawback at high temperatures since they decrease the mechanical properties of the composites. Recently, Si3N4/SiC composites were fabricated via the polymer precursor route without any additives, using electric field assisted sintering (EFAS). In this study, fully densified Si3N4/SiC nanocomposites incorporating hexagonal-BN were successfully fabricated by hot pressing without any additives at 1700 ��C for 2 h under vacuum at a pressure of 50 MPa (via the amorphous precursor route). Moreover, the incorporation of additives and h-BN is found to decrease the content of SiC. The phase transformation, densification, microstructure, and mechanical properties were discussed and presented.
Key words: Si3N4/SiC; hot pressing; hexagonal-BN; additives; sintering
1 Introduction
As advanced ceramics, Si3N4/SiC composites are attractive due to excellent mechanical and thermal properties [1,2], which make them suitable for use in turbine engines, heat exchangers, and other sophisticated applications. With the high covalence and low diffusivity of the Si-based ceramics, it is very difficult to sinter to high density without any additives. For example, pure SiC at near theoretical density could only be achieved by hot pressing at 2500 ��C and 50 MPa [3]. The addition of sintering additives is therefore necessary to obtain high-density Si-based ceramics at a lower temperature. The additives form a liquid phase during sintering and densified Si-based ceramic from the liquid phase sintering mechanism. The additive used for the enhancement of densification becomes a glassy phase in the final ceramic, which usually reduces its mechanical properties at high temperatures [4]. Moreover, the liquid at the grain boundary produces grain growth, which occurs easily due to high boundary mobility. Not only densification, but also grain growth, occurs significantly [5]. This has led to the omission or minimized use of sintering additives in Si-based ceramics.
Recently, a polymer precursor has been used to fabricate Si3N4/SiC nanocomposites [6-9] by two steps. First, the polymer precursor is pyrolyzed to obtain an amorphous powder. The second step is the sintering of the amorphous powder, prepared by hot pressing (HP) or electric field assisted sintering (EFAS). By adopting the EFAS route, a fine grain size can be achieved at a relatively low temperature without the addition of sintering additives [8,9].
Hexagonal BN (h-BN), known for its white graphite structure with low hardness, is an important additive to the field of advanced ceramics due to its excellent thermal shock resistance and machinability [10].
Si3N4-BN composites have exhibited exceptional corrosion resistance [11]. Additionally, SiC�CBN composites are expected to be structural material candidates used in high-temperature and thermal/ mechanical shock environments [10]. Unfortunately, thus far, there are no reports on the effects of h-BN on the phase transformation of Si3N4/SiC composite synthesized by the precursor route.
The goal of this study is to investigate the effects of h-BN addition in Si3N4/SiC on phase transformation via the polymer precursor route. The results on phase composition, densification, and microstructure are discussed.
2 Experimental
The polymer precursor used in this work was a commercially available polyureasilazane, Ceraset SN (Commodore Polymer Technologies, Columbus, OH). Hexagonal BN (Sonderkeramik) had an average particle size of 1 ��m. Al2O3 (Dm=150 nm, 99.9% purity, Baikowski, Japan) and Y2O3 (Dm=220 nm, 99.99% purity, Acros Organics, USA) were used as sintering additives in a mass ratio of 60:40, respectively. The details of sample composition and nomenclature are given in Table 1. Liquid polymer was cross-linked at 200 ��C for 90 min. A mortar and pestle were used to grind the cross-linked infusible polymer to a 75 ��m powder, which was then pyrolyzed under N2 atmosphere for 4 h. In order to reduce the particle size, the amorphous powder was milled in a high energy milling system (MiniCer, Netzch, Germany) for 1 h using 0.45 mm ZrO2 beads at 3000 r/min. The mean particle size after milling was about 1 ��m. The amorphous powder, h-BN, and sintering additive were mixed in ethanol and subjected to ball milling for 24 h using SiC balls (5 mm diameter) to minimize contamination, followed by drying in a rotary evaporator. Hot pressing was then carried out at 1700 ��C for 2 h under vacuum at a pressure of 50 MPa. For comparison, EFAS was performed at 1700 ��C for 10 min under vacuum at a pressure of 50 MPa.
Table 1 Samples and their composition
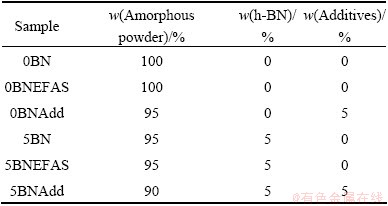
Densities of the sintered specimens were measured using the Archimedes method. The phases observed after sintering were later identified by X-ray diffraction (XRD, X��Pert-PRO MPD, PANalytical), using the Cu K�� line, 40 kV, and 30 mA. The microstructures of various specimens were examined by scanning electron microscopy (SEM, Hitachi S-4800), using 15 kV and 10 ��A. The hardness of the sintered samples was measured with a Vickers hardness indenter (MVK-H1, Akashi, Japan), with a 9.8N (1 kg) load for 10 s. Tabulated data of Gibbs free energies for thermodynamic calculations were obtained from BARIN [12] and CHASE [13].

Fig. 1 XRD patterns of pyrolyzed polymer powders at 1450 ��C and 1650 ��C
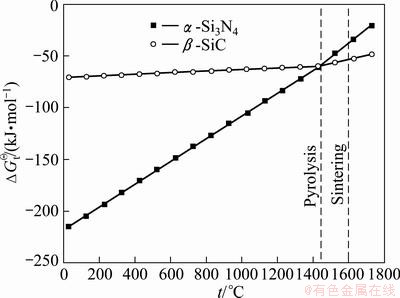
Fig. 2 Standard Gibbs formation free energy vs temperature for ��-SiC and ��-Si3N4 formation
3 Results and discussion
Figure 1 shows the XRD patterns of the pyrolyzed polymer precursor at different temperatures. Depending on the temperature, different phases were observed during pyrolysis. At 1450 ��C, only ��-Si3N4 and free C were observed in the XRD spectra, while at 1650 ��C, ��-Si3N4 and ��-SiC were observed. Phase formation during pyrolysis can be estimated by the Gibbs free energy analysis, as shown in Fig. 2, whereby ��-SiC is more stable than ��-Si3N4 at t>1427 ��C. However, ��-Si3N4 was formed during pyrolysis at both 1450 and 1650 ��C because pyrolysis under a nitrogen atmosphere prevented the decomposition of Si3N4 at high temperatures [14]. As proven by the Gibbs free energy analysis shown in Fig. 2, a small amount of free carbon can react with ��-Si3N4, which then forms SiC at 1650 ��C. Since an amorphous powder is much preferred in the sintering process, the powder was pyrolyzed at 1450 ��C rather than at 1650 ��C. Figure 3 presents the XRD pattern of the Si3N4/SiC composite sintered at 1700 ��C under a pressure of 50 MPa.
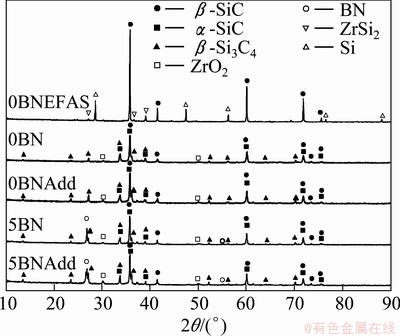
Fig. 3 XRD pattern of Si3N4/SiC composites after sintering at 1700 ��C under pressure of 50 MPa in vacuum using hot press and EFAS
The amorphous powder has already been crystallized into the main phases: ��- and ��-SiC, ��-Si3N4, ZrO2, ZrSi2, Si and h-BN. Although the sintering additives were added in the samples (i.e., 0BNAdd and 5BNAdd), peaks for additives were not detected in the XRD patterns due to the small amount of addition. The other peaks observed in the XRD were from ZrO2, presenting as an impurity from the high energy ball milling with ZrO2 media. ��- and ��-SiC were observed, even though ��-SiC was initially formed, as shown in Fig. 1. However, at the sintering temperature, a portion of ��-SiC was transformed into ��-SiC, where ��- to ��-SiC transformation is common at high temperatures [15]. ��-Si3N4 peaks were also observed without any trace of ��- Si3N4. According to TANAKA [16], ��- to ��-Si3N4 transformation occurs at 1750 ��C. However, ��-Si3N4 was already completely transformed to ��-Si3N4 at 1700 ��C in this study, which is lower than TANAKA��s result [16]. This may be due to the different sintering conditions. In contrast, for the EFAS sample, 0BNEFAS, a highly crystalline ��-SiC peak was observed along with free Si and ZrSi2, without peaks for Si3N4. It appears that under the high temperature EFAS conditions, all the Si3N4 had already transformed to SiC, leaving the excess Si due to the insufficient amount of carbon in the amorphous powder.
This indicates that the kinetics of phase transformation in the EFAS is faster than that in the hot pressing. This phenomenon is also observed in the sintering of Ta [17], where the formation of TaC in the EFAS is higher than that in the hot pressing. Moreover, no ��- to ��-SiC transformation was observed in the hot pressed samples, since the holding time for EFAS was only 10 min. ZrSi2 was observed in the EFAS samples instead of ZrO2. The formation of ZrSi2 is due to the reaction between ZrO2 and free Si in the EFAS samples, as previously observed in the deposition of ZrO2 on a silicon wafer [18].
The phase composition of Si3N4/SiC composites after hot pressing based on the Rietveld refinement method is shown in Table 2. The amount of SiC decreased with the addition of additives and h-BN. At 1700 ��C, SiC is more stable than Si3N4 (Fig. 2), and therefore, the amount of SiC is higher than that of Si3N4, as shown in the 0BN sample. The amount of SiC decreased with the incorporation of additives, as shown in the 0BNAdd sample. The additives form a liquid phase at high temperatures, acting as a mass transport medium to help densification, and locate finally in the grain boundary. Since Si3N4 is formed initially, the additives at the grain boundary of the Si3N4 probably hinder the transformation of Si3N4 to SiC. In the case of h-BN, chemical inertness hinders the transformation of Si3N4 to SiC. The addition of h-BN was also found to restrain the growth of ��- Si3N4 [19].
Table 2 Phase composition of Si3N4/SiC composites after hot pressing based on Rietveld refinement method

Figure 4 shows the bulk density and relative density of the Si3N4/SiC composites after sintering. The density of the sintered sample was not found to significantly increase in the presence of additives. In other words, the amorphous powders were easily densified and formed SiC and Si3N4 without any additives. This result is different from the conventional crystalline powder because at least 8% of additive was needed to aid the densification of SiC [20-26]. The addition of h-BN decreased the bulk density of the sintered samples, as shown in Fig. 4. The sintering temperature of h-BN was found to be higher than 1800 ��C [19]. Hence, the porosity increased with the addition of h-BN, which decreased the relative density of the sintered sample. The same behavior was also observed in the EFAS samples: the addition of BN simply decreased the density. However, the overall density of the EFAS samples was lower than that of the hot-pressed samples due to the formation of free Si, which has a lower density than SiC or Si3N4.
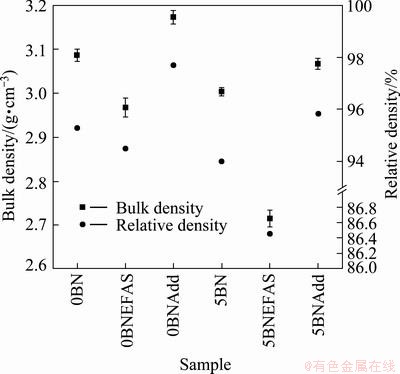
Fig. 4 Bulk density and relative density of Si3N4/SiC composites after sintering
Figure 5 shows SEM images of the polished surfaces of Si3N4/SiC after sintering. Three different colors were observed in the SEM images of samples 0BN, 0BNAdd, 5BN, and 5BNAdd��black, grey, and white.
Electron dispersive spectroscopy (EDS) revealed that the black, grey, and white phases were ��-Si3N4, SiC, and ZrO2, respectively. The ZrO2 impurity was located at the grain boundary of SiC. With irregular shape, the grain size of SiC varied between 50 and 300 nm. Moreover, the ��-Si3N4 content increased in the presence of additives and h-BN, as shown in Fig. 5. The EFAS samples show that grain sizes are three times larger (approximately >1 ��m) than the hot-pressed samples. The EFAS samples contain two different forms of SiC: stoichiometric SiC with a bright color, and non- stoichiometric one with a dark color. In the EFAS samples, EDS also revealed the ZrSi2 impurity and a free Si phase.

Fig. 5 SEM images of polished surfaces of Si3N4/SiC composites after sintering
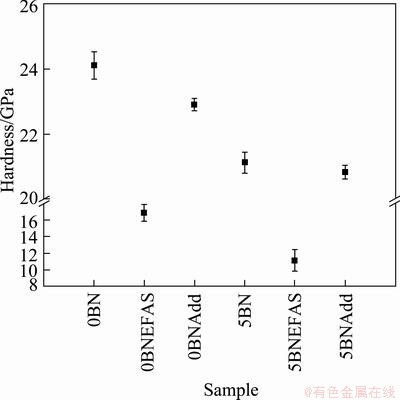
Fig. 6 Hardness of Si3N4/SiC composites after sintering
Figure 6 shows the hardness of the Si3N4/SiC composites after sintering. The highest hardness (24.1 GPa) was achieved in 0BN sample, and the hardness decreased with the incorporation of additives, as shown in the 0BNAdd sample. The decreasing hardness is a function of the SiC content, since the amount of SiC decreased with the incorporation of additives and h-BN. The hardness of the EFAS samples was lower than that of the hot-pressed samples because of the formation of the ductile Si phase. Moreover, the hardness of the EFAS samples was also low due to the large grain size of the samples.
4 Conclusions
The influence of h-BN addition on the phase transformation, microstructure, and mechanical properties of Si3N4/SiC ceramics were investigated. The incorporation of additives and h-BN simply decreases the densification. Furthermore, both the additives and h-BN hinder the transformation of Si3N4 to SiC, due to the liquid phase of the additives and the chemical inertness of h-BN. From these results, it is possible to fabricate h-BN Si3N4/SiC nanocomposites using the polymer precursor without any additives. The bulk density of the sintered samples reaches 95.5%, consisting of SiC grains of 50-300 nm. The SiC in the sample appears to play a significant role with respect to hardness.
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012000858).
References
[1]  DUSZA J, WARBICHLER P, HOFER F, RIEDEL R, LECOMTE E, HOFFMAN M J. SiC/Si3N4 nano/micro-composite��Processing, RT and HT mechanical properties [J]. J Eur Ceram Soc, 2000, 20(4): 453-462.
DUSZA J, WARBICHLER P, HOFER F, RIEDEL R, LECOMTE E, HOFFMAN M J. SiC/Si3N4 nano/micro-composite��Processing, RT and HT mechanical properties [J]. J Eur Ceram Soc, 2000, 20(4): 453-462.
[2] HIRANO T, NIIHARA K. Microstructure and mechanical properties of Si3N4/SiC composites [J]. Mater Lett, 1995, 22(5-6): 249-254.
[3] NADEAU J S. Very high pressure hot pressing of silicon carbide [J]. Am Ceram Soc Bull, 1973, 52: 170-174.
[4] HUANG Z H, JIA D C, ZHOU Y, LIU Y G. A new sintering additive for silicon carbide ceramic [J]. Ceram Int, 2003, 29(1): 13-17.
[5] LEE Y I, KIM Y W, MITOMO M, KIM D H. Fabrication of dense nanostructured silicon carbide ceramics through two-step sintering [J]. J Am Ceram Soc, 2003, 86(10): 1803-1805.
[6] WAN J, GASCH M J, MUKHERJEE A K. Silicon carbonitride ceramics produced by pyrolysis of polymer ceramic precursor [J] J Mater Res, 2000, 15(8): 1657-1660.
[7]  RIEDEL R. Polysilazane derived micro/nano Si3N4/SiC composites [J]. J Eur Ceram Soc, 2002, 22(16): 2963-2968.
RIEDEL R. Polysilazane derived micro/nano Si3N4/SiC composites [J]. J Eur Ceram Soc, 2002, 22(16): 2963-2968.
[8] WAN J, GASCH M J, MUKHERJEE A K. Silicon nitride�Csilicon carbide nanocomposites fabricated by electric-field-assisted sintering [J]. J Am Ceram Soc, 2003, 86(3): 526-528.
[9] WAN J, DUAN R G, GASCH M J, MUKHERJEE A K. Methods of processing Si3N4/SiC nano-nano composites from polymer precursor [J]. Mater Sci Eng A, 2006, 424(1-2): 105-116.
[10] ZHANG G J, YANG J F, ANDO M, OHJI T. Nonoxide�Cboron nitride composites: In situ synthesis, microstructure and properties [J]. J Eur Ceram Soc, 2002, 22(14-15): 2551-2554.
[11]  F, DOCHE C, MONGEOT H, GUILHON F, MIELE P, BONNETOT B. Si3N4 BN composites obtained from aminoboranes as BN precursors and sintering aids [J]. J Eur Ceram Soc, 1997, 17(15-16): 1911-1915.
F, DOCHE C, MONGEOT H, GUILHON F, MIELE P, BONNETOT B. Si3N4 BN composites obtained from aminoboranes as BN precursors and sintering aids [J]. J Eur Ceram Soc, 1997, 17(15-16): 1911-1915.
[12] BARIN I. Thermochemical data of pure substances [M]. New York: VCH, 1989.
[13] CHASE M W Jr. NIST-JANAF thermochemical tables [M]. 4th ed. New York: AIP, 1998.
[14] ZHANG G J, FANG J F, OHJI T. In situ Si3N4�CSiC�CBN composites: Preparation, microstructures and properties [J]. Mater Sci Eng A, 2002, 328(1-2): 201-205.
[15] LEE J S, LEE S H, NISHIMURA T, HIROSAKI N, TANAKA H. A ternary compound additive for vacuum densification of ��-silicon carbide at low temperature [J]. J Eur Ceram Soc, 2009, 29(16): 3419-3423.
[16] TANAKA I. Hot isostatic press sintering and properties of silicon nitride without additives [J]. J Am Ceram Soc, 1989, 72(9): 1656-1660.
[17] ANGERER P, NEUBAEUR E, YU L G, KHOR K A. Texture and structure evolution of tantalum powder samples during spark-plasma-sintering (SPS) and conventional hot-pressing [J]. Int J Refract Met H, 2007, 25(4): 280-285.
[18] SCHONOHM F, FLUCHER C R, WEIER D, LUHR T, BERGES U, DORING S, WESTPHAL C. Thermal stability of ultrathin ZrO2 films and structure determination of ZrSi2 islands on Si(100) [J]. Phys Rev B, 2009, 80(16): 165323.
[19] SUN Y, MENG Q, JIA D, GUAN C. Effect of hexagonal BN on the microstructure and mechanical properties of Si3N4 ceramics [J]. J Mater Process Tech, 2007, 182(1-3): 134-138.
[20] YONATHAN P, LEE J H, YOON D H, KIM W J, PARK J Y. Improvement of SiCf/SiC density by slurry infiltration and tape stacking [J]. Mater Res Bull, 2009, 44(11): 2116-2122.
[21] OMORI M, TAKEI H. Pressureless sintering of SiC [J]. J Am Ceram Soc, 1982, 65(6): c92.
[22] LEE S G, SHIM W H, KIM J Y, KIM Y W, KWON W T. Effect of sintering-additive composition on fracture toughness of liquid-phase-sintered SiC ceramics [J]. J Mater Sci Lett, 2001, 20(2): 143-146.
[23] LEE S P, SHIN Y S, BAE D S, MIN B H, PARK J S, KOHYAMA A. Fabrication of liquid phase sintered SiC materials and their characterization [J]. Fus Eng Des, 2006, 81(8-14): 963-967.
[24] SHE J, UENO H K. Effect of additive content on liquid-phase sintering on silicon carbide ceramics [J]. Mater Res Bull, 1999, 34(10-11): 1629-1636.
[25] LIN B W, IMAI M, YANO ISEKI T. Hot-pressing of ��-SiC powder with Al-B-C additives [J]. J Am Ceram Soc, 1986, 69(4): C67-C68.
[26] DATTA M S, BANDYOPADHYAY A K, CHAUDHURI B. Sintering of nano crystalline a silicon carbide by doping with boron carbide [J]. Bull Mater Sci, 2002, 25: 181-189.
����-BN�ԷǾ�ǰ�����Ʊ��������Ӽ���Si3N4/SiC���ϲ�������Ӱ��
Alfian NOVIYANTO1, Dang-Hyok YOON1, Kyungseok LEE2, Young Moon KIM3, Doo-In KIM2, Young-Keun JEONG2, Kwang Ho KIM2, Sehun KWON2, Young-Hwan HAN1
1. School of Materials Science and Engineering, Yeungnam University Gyeongsan 712-749, Korea;
2. National Core Research Center for Hybrid Materials Solution, Pusan National University, Busan, Korea;
3. Samsung Mobile Display, Korea
ժ Ҫ��Si3N4/SiC�����ϲ������ھ�����������ѧ��������,�㷺Ӧ�������ַ��������Ƚ�������������������С�Ȼ��,���������Ӽ������Ʊ���Si3N4/SiC���ϲ��ϡ����Ӽ����ս�����γ�Һ��Ӷ��ٽ����ϲ��ϵ����ܻ���Ȼ��,���Ӽ��Ĵ��ڽ����˸��ϲ��ϵĸ�����ѧ���ܡ�ͨ���ڲ��������Ӽ�������£����õ糡�����սᣬ���þۺ���ǰ��·���Ʊ�Si3N4/SiC���ϲ��ϡ����о��У��������Ӽ����¶�1700 ��C�����50 MPa�����£���ѹ�ս�2 h, ���÷Ǿ�ǰ��·�߳ɹ��Ʊ�������-BN���ܻ���Si3N4/SiC���ϲ��ϡ��ۺ���ǰ�����BN�����ü����˵�SiC������������䡢���ܻ�������֯����ѧ���ܽ��������ۡ�
�ؼ��ʣ�Si3N4/SiC����ѹ�ս����-BN�����Ӽ����ս�
(Edited by Xiang-qun LI)
Corresponding author: Young-Hwan HAN; Tel: +82-53-810-2539; E-mail: yhhanyu@yu.ac.kr
DOI: 10.1016/S1003-6326(13)62479-6