POSS-�ۺ���������ӵ����Ӧ�õ��о���չ
��Դ�ڿ����й���ɫ����ѧ��2013���10��
�������ߣ��� �� ����ƽ ��˼� ������ ��ҵ��
����ҳ�룺2851 - 2863
�ؼ��ʣ�POSS-�ۺ������ӵ�أ��۵����
Key words��POSS-polymers; lithium-ion batteries; polyelectrolyte
ժ Ҫ���;����α��������(POSS)-�ۺ�������POSS�;ۺ�����ԵĽṹ�����ܵ����ƣ���ʾ�����صĵ绯ѧ���ܺ���ѧ���ܣ�������ӵ������õ�Խ��Խ���о��ߵĹ�ע���ۺ������ʵĸ����ӵ絼�ʡ����˵���ѧ���ܼ��ȶ��ĵ绯ѧ���ܶ���ߵ�ص������к���Ҫ��Ӱ�졣POSS����������ͨ�����ٵ�����������߾ۺ������ʵ����ȶ��Ժ���ѧ���ܣ���һ����߾ۺ������ʵ��������ӵ絼�ʣ���ø��õĵ�س�ŵ����ܡ�����POSS-�ۺ���ṹ�͵������֮��Ĺ�ϵ���������д����Ե�POSS-�ۺ���������ӵ�ص������Ӧ�õ��о���չ�����Ը��־ۺ�����﮵���е�Ӧ��ǰ������չ����
Abstract: Polyhedral oligomeric silsesquioxane (POSS) is a silicone compound with unique structure. POSS-based polymer (POSS-polymer) attracts much attention due to its unique electrochemical and mechanical properties. Polymer electrolyte materials with high ionic conductivity, suitable mechanical properties, and stable chemical and electrochemical properties play an important role in developing batteries with high qualities. POSS-polymer can improve the thermal stability and mechanical properties, further enhance the ionic conductivity of polymer electrolyte, and accordingly it is considered as one of the most promising candidates for the future electrolyte materials of lithium ion batteries. Additionally, with a little addition of POSS in the polymer electrolytes, better battery charge/discharge performance can be achieved. This review was intended to cover the more recent advances in structure-property relationships of POSS-polymers. An application prospect of POSS-polymers in lithium ion batteries was also proposed.
11���±�ţ�1004-0609(2013)10-2851-12
�� ��1, 2������ƽ1����˼�1��������2����ҵ��2
(1. ��ɳ������ѧ �������е����ѧԺ����ɳ 410114��
2. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083)
ժ Ҫ���;����α��������(POSS)-�ۺ�������POSS�;ۺ�����ԵĽṹ�����ܵ����ƣ���ʾ�����صĵ绯ѧ���ܺ���ѧ���ܣ�������ӵ������õ�Խ��Խ���о��ߵĹ�ע���ۺ������ʵĸ����ӵ絼�ʡ����˵���ѧ���ܼ��ȶ��ĵ绯ѧ���ܶ���ߵ�ص������к���Ҫ��Ӱ�졣POSS����������ͨ�����ٵ�����������߾ۺ������ʵ����ȶ��Ժ���ѧ���ܣ���һ����߾ۺ������ʵ��������ӵ絼�ʣ���ø��õĵ�س�ŵ����ܡ�����POSS-�ۺ���ṹ�͵������֮��Ĺ�ϵ���������д����Ե�POSS-�ۺ���������ӵ�ص������Ӧ�õ��о���չ�����Ը��־ۺ�����﮵���е�Ӧ��ǰ������չ����
�ؼ��ʣ�POSS-�ۺ������ӵ�أ��۵����
��ͼ����ţ�TB383��O634.4 �� �� ���ױ�־�룺A
LIANG Bo1, 2, LIU Yan-ping1, TANG Si-qi1, LAI Yan-qing2, LIU Ye-xiang2
(1. School of Automobile and Mechanical Engineering, Changsha University of Science and Technology, Changsha 410114, China;
2. School of Metallurgy and Environment, Central South University, Changsha 410083, China)
Abstract: Polyhedral oligomeric silsesquioxane (POSS) is a silicone compound with unique structure. POSS-based polymer (POSS-polymer) attracts much attention due to its unique electrochemical and mechanical properties. Polymer electrolyte materials with high ionic conductivity, suitable mechanical properties, and stable chemical and electrochemical properties play an important role in developing batteries with high qualities. POSS-polymer can improve the thermal stability and mechanical properties, further enhance the ionic conductivity of polymer electrolyte, and accordingly it is considered as one of the most promising candidates for the future electrolyte materials of lithium ion batteries. Additionally, with a little addition of POSS in the polymer electrolytes, better battery charge/discharge performance can be achieved. This review was intended to cover the more recent advances in structure-property relationships of POSS-polymers. An application prospect of POSS-polymers in lithium ion batteries was also proposed.
Key words: POSS-polymers; lithium-ion batteries; polyelectrolyte
�������ӻ�еϵͳ(MEMS)���������ܿ�����Ƶʶ��(RFID)�ȼ����ķ�չ�����нϸߵ������ܶȡ������İ�ȫ�Ժ�ѭ���Ե�����ӵ���ܵ�Խ��Խ��Ĺ�ע[1-7]����������ͨ�������˿[8]����Ϳ��ӡ[9]�ȹ����Ʊ��ۺ��ﱡĤ��ؼ����������ղ������˳ɱ���ʹ�ô��ģ������Ĥ��س�Ϊ�о��ȵ㡣
�ۺ��������Ƴɸ�����״������ص��������ܺͰ�ȫ��������MEMS�����ܿ��������㷺Ӧ���ھۺ��ﱡĤ����ӵ����[10]���۱���(PANI)[11-14]��������(PPy)[15-18]�������(PTP)[19-22]�Ⱦۺ�������Ϊ�缫����Ӧ���ڱ�Ĥ﮵���С��Ż��ۺ�������;����[23-27]�������ʶȵĽ��������ۻ죻���������ײ��ϣ����һ����ƽᾧ���Ӷ����Ͳ���̬ת���¶�(tg)��������ӵ絼��[28]�����Խṹת��Ϊ��֧���ṹ[29]��
�;������α��������(Polyhedral oligosilsesquioxane��POSS)-�ۺ������ʱ��ֳ��ϸߵ����ӵ絼�ʵ�ͬʱ�����˽ϸߵĻ�еǿ�ȡ�Si��O��Si�����ɵ�����״�ṹ�ĺ��ģ��������õ����ȶ��ԣ��ⲿ�ɻ��Ի�ǻ���R�������Ż��ƣ��л����ʹ�����кܺõľۺ��������ԡ����������Ժ�����һЩ���������ԡ��о�����POSS������ṹͨʽΪ(RSiO1.5)8����ṹʾ��ͼ��ͼ1��ʾ��
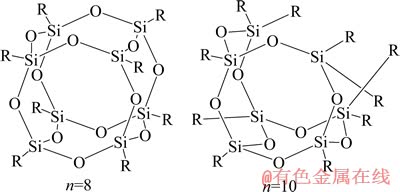
ͼ1 POSS�Ľṹʾ��ͼ
Fig. 1 Schematic diagram of structures of POSS
POSS�����ھۺ���������ܹ���������ѧ���ܺ������ܣ�ʹ���ܻ���Ӧ��������ҽѧ�����������İ뵼������У���غϳɺ�Ӧ�õ������Ѽ�����[30-32]����POSSӦ��������ӵ�ص�������δ���������������������˽������й�POSS�ۺ���������ӵ���е��о���չ���������ص��о���ϵ���ܽ��������POSS������ӵ�ؾۺ����о��е�Ӧ�á�
1 POSS-�ۺ���ṹ�����ܹ�ϵ
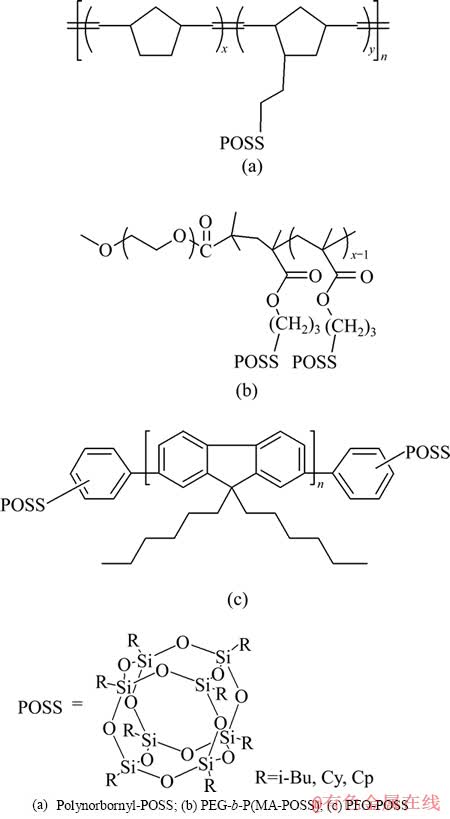
��POSS-�ۺ�����POSS��Ϊ�������ϱ��ֳ���������ܣ����絥һ��ɢ�ԡ����ܶȡ������ȶ��ԺͿɵ��ı������ܡ�POSS��Ҫͨ��3�ַ�ʽ��ۺ����ϣ�(a) POSS��Ϊ�˺ʹ��������������POSS������Ի�����ۺ������ۺ��Ƶ����δ���ӣ�(b) �����POSS��Ϊ�������ϻ������л������ӻ�ۺ���ۺ��Ƶ���������ϲ��ϣ�(c) ��������POSS��֦���ۺ����������γ����ҹ�������˹������ͼ2[33]��ͼ3��ʾΪ����ͼ2��(c)�����Ʊ���POSS-�ۺ��ͼ3(a)[34]��ʾ������Ҫ�����о�POSS-�ۺ���ṹ�����ܵĹ�ϵ��3(b)[35]��3(c)[36]��ʾ����Ŀǰ��ҪӦ���ڹ�������о�������POSS��˴ﵽ�����绯ѧ���ܵ�Ŀ�ġ�

ͼ2 �Ʊ�POSS�ۺ����3����Ҫ����[33]
Fig. 2 Three main types of POSS-based nanocompoiste[33]
�о�POSS-�۵���ʽṹ������֮���ϵ�ķ�����Ҫ�У�����ģ�ͺ�ģ�⣬�۲��ù���ѧ�ͺ�۲������ܲ��ԡ�MATHER��[34]���ù���ѧ�����ܲ��Եķ����о�POSS-�����ϲ��ϵĽṹ������֮��Ĺ�ϵ��ͨ���ı�Ƕ�κ���POSS-����Ƭϩ������(ͼ2(a))����Է��������ֲ���ѡ��ͬ��POSS�����о�POSS�Ծۺ����Ӱ�죬���о��˲�ͬȡ�����Ծۺ�������Ӱ�죬�绷���-POSS(Cp-POSS)ֱ��Ϊ6 nm������Ϊ36 nm��������-POSS(Cy-POSS)ֱ���ﵽ12 nm������Ϊ62nm��POSS��֦��Cy�ϵ���Ŀ�ǽ�֦��Cp�ϵ�7�������������R����Ӱ��Բ����ijߴ磬����ѧ���ܲ����ϴ�Ӱ�졣WANG��[37]����-���ǰ��������ϳ��˹��ۼ����ӵ�POSS�ۺ����ΪPOSS�������Ӹ߶Ȼ������νṹ�������ͬ�������Ĵ�����ϩ(PE)��POSS-PE������Ĺ����ȼ�����ȵؼ�С������ѧ�о�����: POSS������ϴ������˾ۺ�������ȶ��ԣ���������POSS����������tg����
ͨ�����ۼ���POSS�����ھۺ��������ϻ�����POSS��˾ۺ�������ͬ������߾ۺ���������ܺ���ѧ���ܡ�POSS���ӻ��ۺ������Է��Ӽ������ʽ��ɢ������߾ۺ�������ȶ��Ժͻ�еǿ�ȡ�POSS���ӽϴ������Ϳռ�λ�裬����ؽ�����ȡ����Ӧ�ľۺ϶ȡ�
����POSS-�۶���ϩ�;���ϩ������ϵ��ZHENG��[38]�����POSS�ۺ����һ�ֽṹģ�ͣ���ͼ4��ʾ��ģ��ͼ�����������(TEM)���Խ��һ�£����ֳ�POSS�;ۺ������ġ���״���ͽᾧ�ṹ�����ܴ��������ΪPOSS-�ۺ�������ά�ṹ�������ܵ��ۺ�������Լ����POSS�ۺ����������Ƕ�ά��״�ṹ�����ط���(TGA)������POSS����߾ۺ����������ȶ����нϴ����á�12%(��������)POSS-����ϩ�ķֽ��¶������70 �档ͨ���ȽϺ�5%(��������)������ʮ�����POSS�ľ۱���ϩ(PS)-POSS�ۺ��������ϲ��ϵ�td20(��ʧ20%����ʱ�ķֽ��¶�)ֵ���ֱ�Ϊ24.5 ���28.6 �棻�����������ͼ�����POSS��PMMA-POSS����td20ֵ�����64 �档
XU��[39]��ϸ�о���POSS-�ۺ�����POSS��tg��Ӱ����ơ�tgֵ�Ĵ�С��Ҫ��3�����ؾ�����1) POSS��ϡ��ЧӦ���;ۺ������ż����֮�������ã�2) POSS����;ۺ����л����ӵļ����ʻ�֮���ż�������ã�3) POSS-POSS�����ںͷ����ⲿ֮�������á���POSS�����ϵ�ʱ������1����Ҫ���ã��ܽ���tgֵ��POSS�����ϸ�ʱ������2��3����Ҫ���ã������tgֵ��
ͼ3 POSS��֦������[34-36]
Fig. 3 POSS tethered with polymer materials[34-36]

ͼ4 POSS������ṹ��Coughlinģ��[38]
Fig. 4 Coughlin model for structure of POSS-copolymer[38]
�ۼ���ϩ�����(PMMA)�;���ϩ��������ͪ(PVP)��Ŀǰ�о��϶�ľۺ���������ϵ֮һ��YEN��[40]�Ƚ���LiClO4/OP-POSS(8(����(4-�ǻ���))�����-POSS)/PMMA-PVP��LiClO4/PMMA-PVP�����ӵ絼�ʡ�������(PMMA-co-PVP)ͨ����POSS�������PMMA-co-PVP����������ܡ���Ϊ�ۺ�����֮����������OP-POSS��Ӱ�죬�����ӵ���������ۺ�������������ֱ����ϵ�����ԣ�OP-POSS�ɴٽ��ۺ������ʷ������������ԣ��Ӷ���һ����߾ۺ��������������µ����ӵ絼�ʣ�������OP-POSS��LiClO4/OP-POSS/PMMA-co-PVP����ۺ���������������ӵ絼�ʷ���������Ҫ���á�
�ɼ�����POSS-�ۺ���Ľṹ������﮵���е�Ӧ���нϴ��Ӱ�졣��ˣ�̽��POSS-�ۺ���ṹ�����ܵĹ�ϵ��������Ƴ�����������POSS-�ۺ�����ϡ�
2 POSS������ӵ���е�Ӧ��
2.1 ����ʲ���
2.1.1 ��̬�ۺ�������
�ۻ�������(PEO��Ҳ�о��Ҷ���(PEG))��ͨ�������ϵŶԵ������������ϣ��Ӷ��ܽ��������Ϊһ����Ҫ�ľۺ�������Ӧ��������ӵ����[41-43]��POSS-PEO(n)�ۺ������ʵĵͽᾧ���Լ���Խϵ͵�tgֵ��ʹ��������ӵ�ص����ܼ��о��кܺõ�Ӧ��DZ����ͨ��PEO�����Ľ�֦�γ���״�ۺ������״�ۺ����ͨ����Ϊ����֧�ܻ�������ѵı������ƽᾧ�����������PEO���ۺ������ʵĵ絼��[44]��
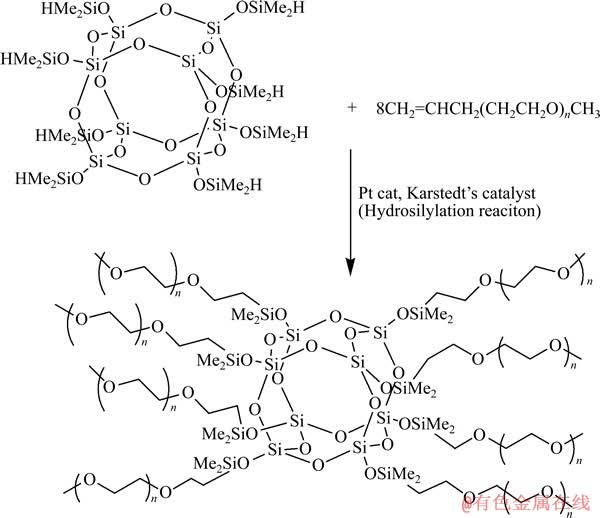
ͼ5 PEO(n)-POSS�ϳ�·��[47]
Fig. 5 Reaction scheme to prepare PEO(n)-POSS[47]
����Temple��ѧ��Wunder�������Ʊ��˵;���PEO����֦��Q8M8H�ϵĶ��PEO���������POSS���о��˽ṹ�Ծۺ�������ӵ絼���Լ��ᾧ�ȵȷ����Ӱ�졣2002��[45]���DZ�����POSS��֦��PEO�ڵ�غ�ҩ�﷽���Ӧ�á�PEO��POSS��֦������Ч����PEO�ᾧ�����統n=4ʱ��PEO����ȫ�ᾧ̬����POSS-PEO����Һ�壬����tg����-85 �档2004�꣬MAITRA��[46]������PEO(n)-POSS����λ�ϵõ���Ե������Һ���о������ӵ絼����ᾧ�Ⱥ�tg�Ĺ�ϵ��POSS-PEO(n=4)8��tg��-60~-70 ��֮�䡣ͼ5��ʾΪ�Ʊ�POSS-PEO(n)�ĺϳ�·��[47]��tg����n���������ߣ�������n�����ۺ������ᾧ�Ŀ�����Ҳ���ӡ�����(�ӽ�tg)ʱ��POSS-PEO(n)/LiClO4�����ӵ絼������PEO��ֵĽ��Ͷ���ߣ�����ʱ�����ӵ絼��������PEO��ֺ��������Ӷ���ߡ����¶�Ϊ90 ��ʱ��POSS-PEO (n=4)8/LiClO4(O/Li=32:1)�����ӵ絼��Ϊ2��10-4 S/cm��50 ��ʱ��POSS-PEO(n=x)/LiClO4�����ӵ絼�ʴ���1��10-3 S/cm���������ӵ絼�ʴ���1��10-4 S/cm��2006�꣬ZHANG��[47]�Ƚ����ڲ�ͬ�����»�����POSS-PEO(n=4)8/LiX�����ӵ����ʣ�X�ֱ�ȡClO4-��(CF3CF2SO2)2N-��(CF3SO2)2N-��CF3SO3-��PF6-��AsF6-��BF4-�����������tg�������Ũ�Ⱥ�PEO�����ȵ����Ӷ���ߣ����ӵ絼����tg�����߶����͡�����(t��tg�������¶�tm)������ʱ��������������(LiClO4��LiN(CF3CF2SO2)2��LiN(CF3SO2)2��LiPF6��LiAsF6)�Ƚ����ǿ�����(LiBF4��LiCF3SO3)�Ե��Ǩ���ʵĹ����������ӵ絼��Ҳ���ߡ��ڵ��¡�����ʱ����ε�tgԽ�ͣ������ӵ絼��Խ�ߡ���ͨ������л�������((CF3CF2SO2)2N-)�������ý�����ε�tgֵ������PEO�����Ӱ��tgֵ�������������θ�����PEO��ϣ����tgֵ��������30 ���£�POSS-PEO(n=4)8/LiN(CF3CF2SO2)2(n(O)/n(Li)=16:1)���������ӵ絼�ʴﵽ1.1��10-4 S/cm��10%����ά�غ�90% PEO-POSS/LiClO4�������ѧ���ܼ��õ�����װ��Ĥ����Ĥ�����µ絼��Ϊ10-5 S/cm������[47-48]��
2007�꣬ZHANG��[48]ͨ������POSS-PEO(n=4)8������Է�������PEO(Mw=600 000)�Լ�LiClO4�IJ�ͬ�����о����Һ̬�����(��̬�ۺ������ʵĻ���)�����ӵ絼������̬�Ĺ�ϵ����n(O)/n(Li)=8:1��12:1ʱ������������������һ�ָߵ�tg�Ǿ����� (��PEO����)��һ�ֵ͵�tg�Ǿ�����(��POSS-PEO(n=4)8����)�����ơ���n(O)/n(Li)=16:1���������зǽᾧ�����ȫ����PEO�Ľᾧ�ࡣ���ӵ絼��������������ʾ������ϵ�tg�ǽᾧ����������ӵ絼�ʽϸߣ��ᾧ̬������絼�ʽϵ͡����������������������ӵ絼��Ϊ8��10-6 S/cm������n(O)/n(Li)=12:1������PEO�����ӵ絼�ʽ�Ϊ2��10-6 S/cm��
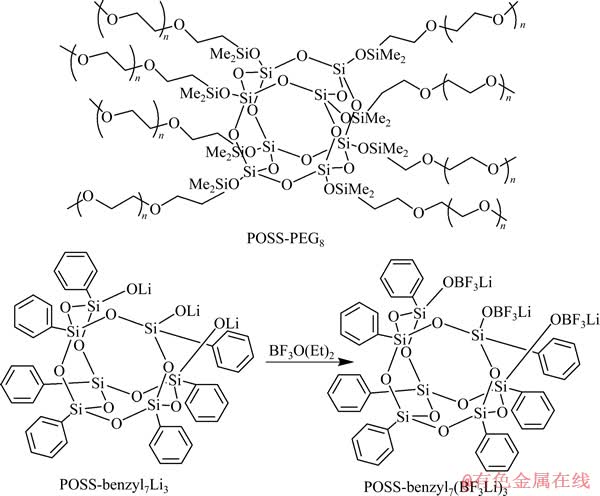
ͼ6 POSS-PEG8��POSS-benzyl7Li3��POSS-benzyl7(BF3Li)3�ṹʽ[49]
Fig. 6 Structures of POSS-PEG8, POSS-benzyl7Li3 and POSS-benzyl7 (BF3Li)3[49]

ͼ7 POSS-benzyl7(BF3Li)3/ POSS-PEG8������ṹʾ��ͼ[49]
Fig. 7 Schematic diagram of two-phase structure of POSS-benzyl7(BF3Li)3/POSS-PEG8[49]
������POSS���ײ��ϻ�Ͽɵõ������ϵ���ʡ�2011�꣬CHINNAM��[49]������������ƣ�����POSS-PEG8 /POSS-benzyl7(BF3Li)3���ϵ���ʲ��ϣ���ַ�����POSS-PEG8���õ������ܺ�POSS- benzyl7(BF3Li)3�ռ�Ǽܽṹ�����ԣ�����˸ø��ϵ���ʲ��ϵĵ绯ѧ��Ϊ��POSS-PEG8�ӷ��ԣ����нϸߵ����ȶ���(400 ��)������������κ����µ絼�ʴ�Լ1��10-4 S/cm��ͨ��POSS-benzyl7Li3��BF3(OC2H5)2��Ӧ�Ʊ��õ����������POSS- benzyl7(BF3Li)3���ṹʽ��ͼ6[49]��ʾ��POSS-benzyl7 (BF3Li)3������Janus���ܣ�������Ҫ����ˮ����ˣ����С�Si��O��BF3Li���ӻ��ŷ�ˡ�·��˹��BF3�ٽ���﮵缫���γɶۻ��㣬���ɱ�����ʹ�����С����ֹ﮺��ܼ�֮��ķ�Ӧ[50]������PEG/����Ȼ���ĵ绯ѧ�ȶ���[51]������POSS-PEG8�ٽ���POSS- benzyl7(BF3Li)3����ˮ�����ľۼ��ͽᾧ�����ϵ�����γ�������̬����������Ϊ�ṹ�࣬POSS-PEG8��Ϊ������(ͼ7[49])��POSS-benzyl7(BF3Li)3�ġ�Si��O��BF3��Li+���ŷ���ȡ���룬����Li+�ܽ���POSS-PEG8�С������ϵ������һ����Բ��ϣ������������²��ᷢ��������10 ��ʱ�������ϵ���ʵ����ӵ絼��Ϊ1.5��10-5 S/cm��30 ��ʱ��POSS-PEG8/POSS-benzyl7(BF3-Li)3(n(O)/n(Li)=16:1)�����ӵ絼��ԼΪ2.5��10-4 S/cm����POSS-PEG8/ POSS-LiBF4��ͬ���������ӵ絼�ʵ�17����90 ��ʱ�����ӵ絼��Ϊ1.6��10-3 S/cm���о����������ø��ϵ�����нϴ�������������˷������������ԣ��������Ǩ����Ϊ0.5 0.01����80 ����3 d�����ֳ�����ȶ��Ľ����迹������POSS�ڲ��������ӵ絼�ʵ�ǰ���¿ɸ��ƾۺ������ʵĽ����ȶ��ԡ�
0.01����80 ����3 d�����ֳ�����ȶ��Ľ����迹������POSS�ڲ��������ӵ絼�ʵ�ǰ���¿ɸ��ƾۺ������ʵĽ����ȶ��ԡ�
LEE��[52]ͨ������ϩ��-POSS��Ϊ�����������Ҷ���������(PEGDME)��Ϊ���ܼ����������ɻ��ۺϷ��Ʊ���̬�ۺ�������(SPE)����ϩ��-POSS���ṹ��ͼ8��ʾ��5% POSS��95% PEGDME(��������)ʱ��ϵ��������ӵ絼��Ϊ5.3��10-4 S/cm��������ʵ��Ӧ�õ�Ҫ�������£�Li/SPE/LiCoO2﮾ۺ�������0.1C�����·ŵ磬����20��ѭ�����Ա���ԭ�ŵ�������80%��
2.1.2 ����Һ�弰���������
����Һ��(Ionic liquids, ILs)���нϸߵ��������ӵ絼�ʡ����ӷ��ԺͲ�ȼ���ԣ���Ϊ���ƺ���ߵ�ذ�ȫ���ܵ���ѡ��[53-55]��ILs����ͨ���������������Ʊ�������������Һ�壬������Ի����Ѻõ�����Ҳ�ٽ�����Һ���Ϊ�����²����о���ǰ������ �㡣���ṹ�ۺ�����ϵĶ��ؽṹ����̬�������������ͨС���Ӷ��������Һ�����ܷ�����з�չDZ��[56-61]��POSS�����νṹ������ÿ������������ȡ�����ţ�������������Ӵ���������������POSS������Һ���ڵ����Ӧ�÷����ܹ����ֳ����������[62-63]��
ͼ8 ����ϩ��-POSS��ѧ�ṹ[52]
Fig. 8 Chemical structure of methacryl-POSS[52]
�ձ�������ѧ��TANAKA��[64-65]�о���POSS-����Һ��������ӵ���е�Ӧ�á������״γɹ��ϳ���POSS����������Һ��[64]�������Ȼ�-POSS- ([POSS-(COO-)8])Ϊ�����ӣ���������Һ�������f��([Bmim+])Ϊ�����ӵĴ�����������Һ�壬�ṹ��ͼ9(a)��ʾ��ͨ���ܽ�������(DGA)����ʾ��ɨ�����ȷ�(DSC)������ΪPOSS�ĸ��������ṹ�Խ��������¶�(25 ��)��������ȶ��Ծ�����Ҫ���á�
��������Ʊ���һϵ�и��Ե������f��-POSS�Ȼ��������Һ�壬�о��˽ṹ��������֮��Ĺ�ϵ(ͼ9(b))[65]��POSS�����ṹ�γɵ����νṹ����˾ۺ�������ȶ��Բ��������������¶ȡ�POSS-Im6��POSS-Im8���ȷֽ��¶�(Td)����POSS-(COOH)8��Arm-COOH�ε��ȷֽ��¶ȣ�����Ϊ���Թ��谭�˷��ӵ��ƶ����Ӷ������˲��ϵ��ȷֽ⡣��DSC�����ϣ�POSS-Im6��POSS-Im8�����ȷ�ֱ�Ϊ19��23 �档�෴�أ�POSS-Im2��POSS-Im4�����Arm-COOH�κ�POSS-(COOH)8���ֳ��ϵ͵�tdֵ��ͨ���ȶ�̬���������������Ϊ���ӶԵ����νṹӰ��������POSS���ӶԵ��������ܡ�
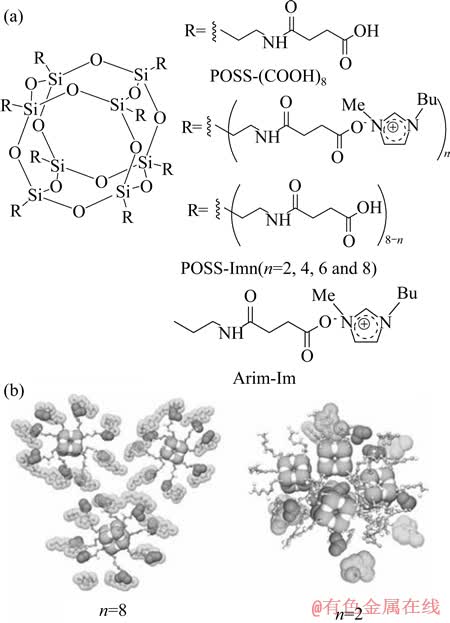
ͼ9 ���ӶԵĻ�ѧ�ṹ��POSS������Һ��ģ��[65]
Fig. 9 Chemical structures of ionic pairs (a) and proposed conformations of POSS-based ionic compounds (b)[65]
 ��[66]��POSSΪ�ṹ���Թ������õ������¶���150~200��֮���1, 3-��������f�ε⻯������Һ�塣�˴Ź���(NMR)�ͺ������(FTIR)������������POSS����ͨ�����������������f���ϣ�ͨ���ı�POSS���˵�����7��ȡ�����õ��˹�̬(PEOPrIm+I- IB7T8 POSS)��Һ̬(MePrIm+Ix- IO7T8 POSS)�⻯������Һ�塣���Ƿ��ֹ�̬����Һ��ĵ絼��ԼΪ10-7 S/cm�������Ŷ�⻯������γɣ����ӵ絼�������������������MePrIm+Ix-IO7 T8 POSS������ĵ絼��ȴû�����Ա仯��������Ϊ��⻯������γɲ�����Grotthus���Ӵ������(������Ծ)������������ͬ�Ľ����
��[66]��POSSΪ�ṹ���Թ������õ������¶���150~200��֮���1, 3-��������f�ε⻯������Һ�塣�˴Ź���(NMR)�ͺ������(FTIR)������������POSS����ͨ�����������������f���ϣ�ͨ���ı�POSS���˵�����7��ȡ�����õ��˹�̬(PEOPrIm+I- IB7T8 POSS)��Һ̬(MePrIm+Ix- IO7T8 POSS)�⻯������Һ�塣���Ƿ��ֹ�̬����Һ��ĵ絼��ԼΪ10-7 S/cm�������Ŷ�⻯������γɣ����ӵ絼�������������������MePrIm+Ix-IO7 T8 POSS������ĵ絼��ȴû�����Ա仯��������Ϊ��⻯������γɲ�����Grotthus���Ӵ������(������Ծ)������������ͬ�Ľ����
����Һ��ɽ��ͱ�Ĥ�ĵ���ģ���������ѧ���ܡ�CHANG��[67]�о���POSS�ڱ�Ĥ�е����ã�����POSS�ٽ�������ֺ;ۺ������֮������ã���Ĥ����ѧ������֮��ߡ�SUBIANTO��[63]�����˻ǻ�POSS(ͼ10(a))������Һ��[1-butyl-3-methylimid- dazolium bis(trifluoromethylsulfonyl)imide, BMI-BT-SI]��ѧ���ܵ�Ӱ�졣���ܾۺ���ֻ��������POSS��������ѧ���ܵ�Ӱ��ϴ�1% POSS�ɽϴ�̶ȵ����tgֵ�������ܡ����ö�̬�Ȼ�е����(DMA)�о���S-POSS������Һ���NafionĤ����ѧ���ܵ����á�ͼ10(b)�������Ӵ�ת���¶�(Ionic cluster transition�� tc��119 ����ߵ�127 �档tan �ķ����߱���S-POSSӰ����Nafion�����Ӵء����ң��ڸ���120 ��ʱ���ӻ���Ĥģ��Ϊ32 MPa�����������ֱ�Ĥģ����Ϊ23 MPa����ͼ10(c)��ʾ��ģ������߿�������ΪS-POSS����������������֮�������ã��Ӷ����NafionĤ�ڸ���tc�¶�ʱ����ѧ���ܡ�
��1975��FEUILLADE��[68]���������۵����(GPE)Ӧ����﮵�غܶ��о��߿�ʼ������GPE���о�����GPE�У������Ҫͨ���л��ܼ��ļ��Ի����ܽ⡣LI��[69]ͨ��������˵Ķ���ϩ-��ϩ��(ATBN)�ȹ̻��ڻ���������-POSS�������ϣ�����һ������Һ��������(LiClO4)�Ʊ���һ�����͵Ľ�������ṹ�������۵����(ͼ11(a))��LiClO4������Һ��(1-����-3-������������������(BMIMOTf))֮�������ã�������LiClO4�ķֽ⣬���GPE�����ӵ絼�ʣ�����������POSS��������ӵ絼�ʡ�����ϵ���ΪATBN-5% POSS-40 % LiClO4-50% ILʱ��GPE�����ӵ絼����30 ��ʱ��ߣ��ﵽ2.0��10-4 S/cm (ͼ11(b))���þۺ�����ϵ��30~70 �淶Χ���������ӵ絼�ʺ��¶�֮���Arrheniusģ�ͣ�����GPE������е������Ӽ�����ۺ�������Ƕ���˶��أ������ܼ�����̬�йء�������֬-POSS�ڸ�GPE��ϵ�г䵱�����������ã���������POSS���GPE�ľۺ��������ɶȺ������ԣ�������������ӵ絼�ʡ���������ܶȿ�������ӵ絼�ʣ���������GPE����ѧ���ܣ���ˣ��о����ӵ絼�ʺ���ѧ����֮��Ĺ�ϵ���о�GPE�Ĺؼ����⡣��ѭ���������߿��Կ�������GPE���ֳ��ߴ�4 V�ĵ绯ѧ�ȶ��ԡ���ĿǰΪֹ��POSS-GPE���Ͼۺ������ʵ�������δȡ�ýϴ��ͻ�ƣ��������POSS��ΪGPE���Ͼۺ������ʻ��ʲ��ϣ���չ�Ʊ��ۺ������ʷ���̽���Ե��о���ʮ�ֱ�Ҫ�ġ�
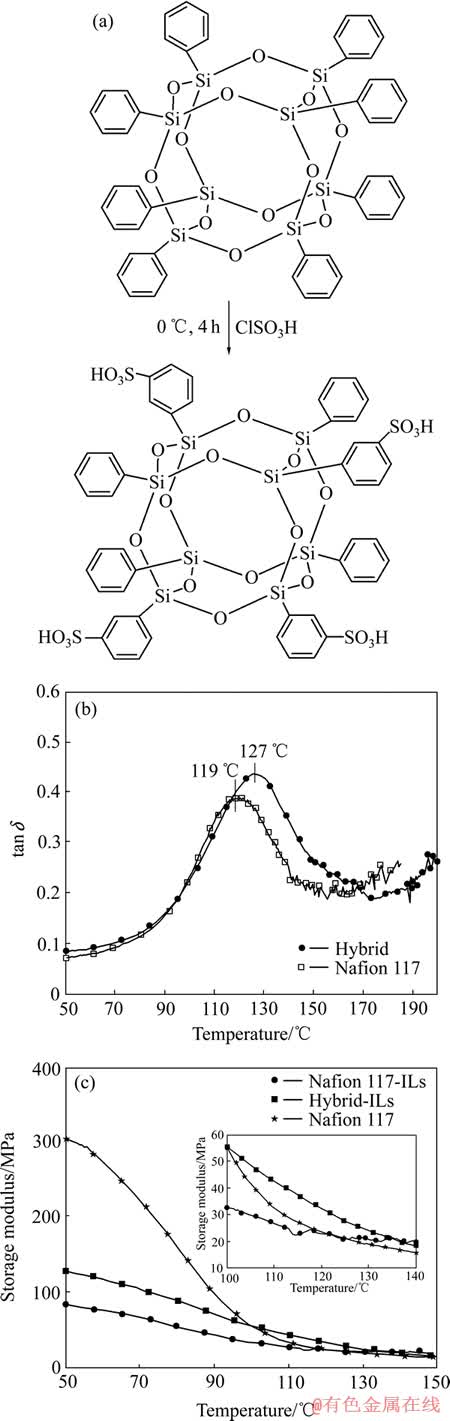
ͼ10 �ǻ�POSS, S-POSS�ӻ���Ĥ��Nafion��Ĥ��tan �����ߺ�S-POSS����Һ���ӻ���Ĥ������Һ�屡ĤDMA����ͼ[63]
Fig. 10 Sulfonation of POSS (a), DMA showing shift in tg in tan �� curves for Nafion S-POSS hybrid compared to Nafion (b) and DMA of Nafion S-POSS ILs composite membrane material and Nafion ILs membrane (c) (Inset: Enlargement of region between 100 and 140 ��)[63]
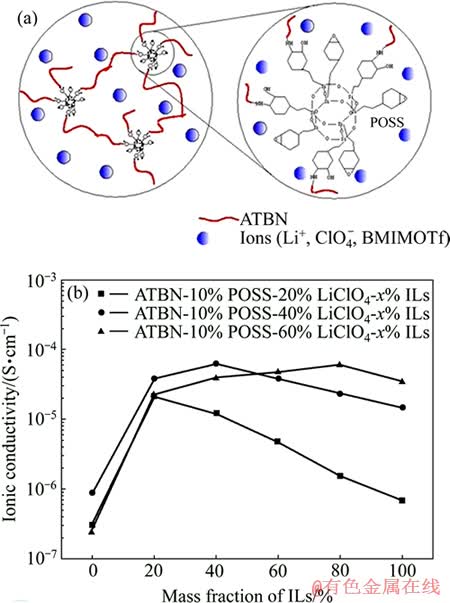
ͼ11 POSS��Ϊ��������GPEʾ��ͼ��GPE���ӵĵ絼��[69]
Fig. 11 Schematic diagram of GPE with POSS as cross- linking agent (a) and ionic conductivity of GPE based on ATBN with different LiClO4 and ILs contents (b)[69]
2.2 �缫��Ĥ����
POSS����﮵�ص����ܻ�������﮵�ص缫��Ĥ�����Ӧ�á�KIM��[70]��������⻯���α���������ս��Ӧ����﮵��������ר������صij�ŵ����ܵõ����ơ��״�ѭ���ŵ�������ߴﵽ905 mA��h/g��30��ѭ���������������Ϊ64.8%��ʯīϩ����ĵ�ѧ����ѧ�Լ���ѧ����ʹ���Ϊ��������Ӧ��ǰ��������ӵ�ص缫���ϡ�XUE��[71]�����˰���POSS���ܻ�������ʯīϩ(POSS-ʯīϩ)����ṹʾ��ͼ��ͼ12��ʾ��POSS���ܻ����ʯīϩ����������ò���۽ṹ��Ԥ������Ч���Ʋ��ϵĸ���绯ѧ���ܡ�POSS-ʯīϩ��Ϊһ�����͵��������Ͽ���߾ۺ�������tg��td��
HENNIGE��[72]���õ;۹����Ŵ����﮵�ض��Ĥ�Ļ�ѧ�����ȶ��ԡ���Ĥ�Զ�ǵ���ۺ���-Si8O12(OSiR3)8Ϊ�Ǽܣ��մ���ΪͿ�㡣���Ĥ������������������������ʹ�û����µ���ʿ�����Ĥ�����ױ���������ؾͻ����¶Ȼ��۳�����ʧ�ء�
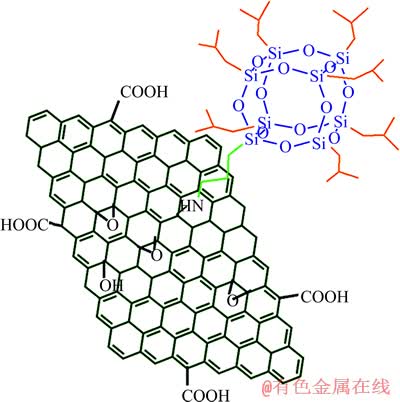
ͼ12 POSS���ܻ�ʯīϩ�ṹʾ��ͼ[71]
Fig. 12 Schematic diagram of POSS-functionalized grapheme nanosheets[71]
3 �ܽ���չ��
1) POSS-�ۺ�����һ����-�л��ӻ������ϲ��ϣ�����ۺ�����ϵ�У��������߾ۺ�����ȶ��ԡ��������ں���״�ṹ��������POSSͨ�����ۼ�����ʽ����ۺ������ۺ�������ӵ����ʺ���ѧ���ܾ��õ�������ߡ��ۺ�����ϵ������POSS����ϡ��ЧӦ��ʹ������������֮����ѻ�����������������̬ת���¶Ƚ��͡�POSS�ۺ������Щ�ص�������Ӿۺ������о��кܺõ�Ӧ��ǰ����
2) �����ľۺ������ʱ�Ӧ��������ӵ��ʱ���ӵ絼�ʺ������Ǩ����ƫ�ͣ�����Һ�嵥��ʹ�ÿ��ܳ���й©�¹ʣ���Ͱ�ȫ�ԡ���POSS����ۺ��������Һ���п��Ի�����Щ�ˡ�POSS����ϡ��ЧӦ�Լ����������е����������ܣ�ʹ�þۺ���IJ���̬ת���¶Ƚ��ͣ����ӵ����ʺͰ�ȫ����ߡ�
3) POSS�ھۺ����е�Ӧ�����ڱ�Ѹ���ؿ����Ժ���о��ص㽫��һ��̽��POSS�ھۺ����е�����ò�͵绯ѧ����֮��Ĺ�ϵ��Э��POSS-�ۺ�����ϵ����ӵ絼�ʺ���ѧ����֮��Ĺ�ϵ��ʹ������ʵ��Ӧ������
REFERENCES
[1] CHIANG Y M. Building a better battery[J]. Science, 2010, 330(6010): 1485-1486.
[2] ARMAND M, TARASCON J M. Building better batteries[J]. Nature, 2008, 451(7179): 652-657.
[3]  I, ANDRZEJEWSKA E. Highly conductive ionic liquid based ternary polymer electrolytes obtained by in situ photopolymerisation[J]. Electrochimica Acta, 2009, 54(24): 5660-5665.
I, ANDRZEJEWSKA E. Highly conductive ionic liquid based ternary polymer electrolytes obtained by in situ photopolymerisation[J]. Electrochimica Acta, 2009, 54(24): 5660-5665.
[4] MENG C, LIU C, FAN S. Flexible carbon nanotube/polyaniline paper-like films and their enhanced electrochemical properties[J]. Electrochemistry Communications, 2009, 11(1): 186-189.
[5] WANG D W, LI F, ZHAO J, REN W, CHEN Z G, TAN J, WU Z S, GENTLE I, LU G Q, CHENG H M. Fabrication of graphene/polyaniline composite paper via in situ anodic electropolymerization for high-performance flexible electrode[J]. ACS Nano, 2009, 3(7): 1745-1752.
[6] PUSHPARAJ V L, SHAIJUMON M M, KUMAR A, MURUGESAN S, CI L, VAJTAI R, LINHARDT R J, NALAMASU O, AJAYAN P M. Flexible energy storage devices based on nanocomposite paper[J]. Proceedings of the National Academy of Sciences of the United States of America, 2007, 104(34): 13574-13577.
[7] ���ɹ�, ������, ����Ⱥ, ����ƽ. ����ӵ�ظ��µ��Һ[J]. ��ѧ��չ, 2013, 25(1): 54-59.
YIN Cheng-guo, MA Yu-lin, CHENG Xin-qun, YIN Ge-ping. Elevated-temperature electrolytes for Li-ion batteries[J]. Progress in Chemistry, 2013, 25(1): 54-59.
[8] LIU Y, GORGUTSA S, SANTATO C, SKOROBOGATIY M. Flexible, solid electrolyte-based lithium battery composed of LiFePO4 cathode and Li4Ti5O12 anode for applications in smart textiles[J]. Journal of the Electrochemical Society, 2012, 159(4): 349-356.
[9] SINGH N, GALANDE C, MIRANDA A, MATHKAR A, GAO W, REDDY A L, VLAD A, AJAYAN P M. Paintable battery[J]. Science Reporter, 2012, 2: 481-485.
[10] NYHOLM L, NYSTROM G, MIHRANYAN A, STROMME M. Toward flexible polymer and paper-based energy storage devices[J]. Advanced Materials, 2011, 23: 3751-3769.
[11] MANDIC Z, ROKOVIC M K, POKUPCIC T. Polyaniline as cathodic material for electrochemical energy sources: The role of morphology[J]. Electrochimica Acta, 2009, 54(10): 2941-2950.
[12] CAO Y, MALLOUK T E. Morphology of template-grown polyaniline nanowires and its effect on the electrochemical capacitance of nanowire arrays[J]. Chemical Reviews, 2008, 20: 5260-5265.
[13] ������, ������, ����. �۱�����LiCoO2��LiMn2O4�����е�˫�ع���[J]. �й���ɫ����ѧ��, 2011, 21(4): 796-803.
FAN Chang-ling, XU Zhong-yu, SU Yu-chang. Dual function of polyaniline in positive electrodes of LiCoO2 and LiMn2O4[J]. The Chinese Journal of Nonferrous Metals, 2011, 21(4): 796-803.
[14] ������, �ߴ���, �� ��, Ҧ��ޱ, ������. ʯīϩ/�۱������ϲ��ϵ��Ʊ�����绯ѧ����[J]. ������ѧѧ��, 2013, 29(1): 117-122.
WANG Hong-zhi, GAO Cui-xia, ZHANG Peng, YAO Su-wei, ZHANG Wei-guo. Synthesis and electrochemical performance of graphene/polyaniline[J]. Acta Physico-Chimica Sinica, 2013, 29(1): 117-122.
[15] XU C, SUN J, GAO L. Synthesis of novel hierarchical graphene/polypyrrole nanosheet composites and their superior electrochemical performance[J]. Journal of Materials Chemistry, 2011, 21(30): 11253-11258.
[16] WOO S W, DOKKO K, KANAMURA K. Composite electrode composed of bimodal porous carbon and polypyrrole for electrochemical capacitors[J]. Journal of Power Sources, 2008, 185(2): 1589-1593.
[17] JU Y W, CHOI G R, JUNG H R, LEE W J. Electrochemical properties of electrospun PAN/MWCNT carbon nanofibers electrodes coated with polypyrrole[J]. Electrochimica Acta, 2008, 53(19): 5796-5803.
[18] �콣��, ������, �� ��, ����ƽ. �ɿ��ٳ�ŵ������/̼���ܸ��ϲ��ϵ绯ѧ�ۺ������[J]. ������ѧѧ��, 2012, 28(2): 373-380.
ZHU Jian-bo, XU You-long, WANG Jie, WANG Jing-ping. Electropolymerization and characterization of fast charge-discharge PPy/F-SWNTs composite materials[J]. Acta Physico-Chimica Sinica, 2012, 28 (2): 373-380.
[19] CHO S I, LEE S B. Fast electrochemistry of conductive polymer nanotubes: Synthesis, mechanism, and application[J]. Accounts of Chemical Research, 2008, 41(6): 699-707.
[20] LAFORGUE A, ROBITAILLE L. Production of conductive PEDOT nanofibers by the combination of electrospinning and vapor-phase polymerization[J]. Macromolecules, 2010, 43(9): 4194-4200.
[21] ������, ��ʥ��, �ƹ�ǿ, ���Ʒ�, �� ��, ��ѧ��. �����/���̼���ܸ��ϲ��ϵĵ�������[J]. ��ѧѧ��, 2007, 65(24): 2923-2928.
WANG Hong-min, JIN Sheng-song, TANG Guo-qiang, HAN Fei-fei, LIANG Dan, XU Xue-cheng. Conductive properties of polythiophene/multi-walled carbon nanotubes composites[J]. Acta Chimica Sinica, 2007, 65(24): 2923-2928.
[22] �߷��, ����. �����/����̿���ϲ�����Ϊ�����������缫���ϵĵ�����[J]. �߷��Ӳ��Ͽ�ѧ�빤��, 2011, 27(2): 152-155.
GAO Feng-ge, TIAN Yan-hong. The electrochemical performances of polythionphene/activated carbon composites as electrode materials in supercapacitors[J]. Polymer Materials Science and Engineering, 2011, 27(2): 152-155.
[23] CROCE F, APPETECCHI G, PERSI L, SCROSATI B. Nanocomposite polymer electrolytes for lithium batteries[J]. Nature, 1998, 394(6692): 456-458.
[24] WANG S, MIN K. Solid polymer electrolytes of blends of polyurethane and polyether modified polysiloxane and their ionic conductivity[J]. Polymer, 2010, 51(12): 2621-2628.
[25] DEKA M, NATH A, KUMAR A. Effect of dedoped (insulating) polyaniline nanofibers on the ionic transport and interfacial stability of poly (vinylidene fluoride-hexafluoropropylene) based composite polymer electrolyte membranes[J]. Journal of Membrane Science, 2009, 327(1/2): 188-194.
[26] NEVES S, CANOBRE S C, OLIVEIRA R S, FONSECA C P. Electrochemical performance evaluation of polyaniline/lithium manganese nickel oxide composites synthesized using surfactant agents[J]. Journal of Power Sources, 2009, 189(2): 1167-1173.
[27] PAN L J, YU G H, ZHAI D Y, LEE H R, ZHAO W T, LIU N, WANG H L, TEE B C K, SHI Y, CUI Y, BAO Z N. Hierarchical nanostructured conducting polymer hydrogel with high electrochemical activity[J]. Proceedings of the National Academy of Sciences of the United States of America, 2012, 109(24): 9287-9292.
[28] LICOCCIA S, TRAVERSA E. The nano-micro interface[M]. Weinheim: Wiley-VCH Verlag, 2004: 289-301.
[29] HAWKER C J, CHU F, POMERY P J, HILL D J T. Hyperbranched poly (ethylene glycol)s: A new class of ion-conducting materials[J]. Macromolecules, 1996, 29(11): 3831-3838.
[30] TANAKA K,CHUJO Y. Advanced functional materials based on polyhedral oligomeric silsesquioxane (POSS)[J]. Journal of Materials Chemistry, 2012, 22(5): 1733-1738.
[31] WU J, MATHER P T. POSS polymers: Physical properties and biomaterials applications[J]. Polymer Reviews, 2009, 49(1): 25-63.
[32] KUO S W, CHANG F C. POSS related polymer nanocomposites[J]. Progress in Polymer Science, 2011, 36(12): 1649-1696.
[33] WANG F, LU X, HE C. Some recent developments of polyhedral oligomeric silsesquioxane (POSS)-based polymeric materials[J]. Journal of Materials Chemistry, 2011, 21(9): 2775-2780.
[34] MATHER P T, JEON H G, ROMO-URIBE A, HADDADD T S, LICHTENHAN J D. Mechanical relaxation and microstructure of poly(norbornyl-poss) copolymers[J]. Macromolecules, 1999, 32(4): 1194-1203.
[35] HUSSAIN H, TAN B H, SEAH G L, LIU Y, HE C B, DAVIS T P. Micelle formation and gelation of (PEG-P(MA-POSS)) amphiphilic block copolymers via associative hydrophobic effects[J]. Langmuir, 2010, 26(14): 11763-11773.
[36] XIAO S, NGUYEN M, GONG X, CAO Y, WU H B, MOSES D, HEEGER A J. Stabilization of semiconducting polymers with silsesquioxane[J]. Advanced Functional Materials, 2003, 13(1): 25-29.
[37] WANG J, YE Z, JOLY H. Synthesis and characterization of hyperbranched polyethylenes tethered with polyhedral oligomeric silsesquioxane (POSS) nanoparticles by chain walking ethylene copolymerization with acryloisobutyl-POSS[J]. Macromolecules, 2007, 40(17): 6150-6163.
[38] ZHENG L, HONG S, CARDOEN G, BURGAZ E, GIDO S P, COUGHLIN E B. Polymer nanocomposites through controlled self-assembly of cubic silsesquioxane scaffolds[J]. Macromolecules, 2004, 37(23): 8606-8611.
[39] XU H, KUO S W, LEE J S, CHANG F C. Preparations, thermal properties, and Tg increase mechanism of inorganic/organic hybrid polymers based on polyhedral oligomeric silsesquioxanes[J]. Macromolecules, 2002, 35(23): 8788-8793.
[40] YEN Y C, KUO S W, HUANG C F, CHEN J K, CHANG F C. Miscibility and hydrogen-bonding behavior in organic/inorganic polymer hybrids containing octaphenol polyhedral oligomeric silsesquioxane[J]. The Journal of Physical Chemistry B, 2008, 112(35): 10821-10829.
[41] YEN Y C, CHENG C C, KUO S W, CHANG F C. A new poly (amide urethane) solid state electrolyte containing supramolecular structure[J]. Macromolecules, 2010, 43(5): 2634-2637.
[42] CHIU C Y, CHEN H W, KUO S W, HUANG C F, CHANG F C. Investigating the effect of miscibility on the ionic conductivity of LiClO4/PEO/PCL ternary blends[J]. Macromolecules, 2004, 37(22): 8424-8430.
[43] CHIU C Y, HSU W H, YEN Y J, KUO S W, CHANG F C. Miscibility behavior and interaction mechanism of polymer electrolytes comprising LiClO4 and MPEG-block-PCL copolymers[J]. Macromolecules, 2005, 38(15): 6640-6647.
[44] �� ��, Ǯ����, ������, ���ܿ�. ���ӵ���ۺ������ʵ��о�[J]. ��ѧ��չ, 2002, 14(5): 374-374.
ZHAO Feng, QIAN Xin-ming, WANG Er-kang, DONG Shao-jun. Advanced in ionic conductive polymer electrolytes[J]. Progress in Chemistry, 2002, 14(5): 374-374.
[45] MAITRA P, WUNDER S L. Oligomeric poly (ethylene oxide)-functionalized silsesquioxanes: Interfacial effects on Tg, Tm, and ��Hm[J]. Chemistry of Materials, 2002, 14(11): 4494-4497.
[46] MAITRA P, WUNDER S L. POSS based electrolytes for rechargeable lithium batteries[J]. Electrochemical and Solid-State Letters, 2004, 7(4): 88-92.
[47] ZHANG H, KULKARNI S,WUNDER S L. Polyethylene glycol functionalized polyoctahedral silsesquioxanes as electrolytes for lithium batteries[J]. Journal of the Electrochemical Society, 2006, 153(2): A239.
[48] ZHANG H, KULKARNI S, WUNDER S L. Blends of POSS-PEO (n=4)8 and high molecular weight poly (ethylene oxide) as solid polymer electrolytes for lithium batteries[J]. The Journal of Physical Chemistry B, 2007, 111(14): 3583-3590.
[49] CHINNAM P R, WUNDER S L. Polyoctahedral silsesquioxane-nanoparticle electrolytes for lithium batteries: POSS-lithium salts and POSS-PEGs[J]. Chemistry of Materials, 2011, 23(23): 5111-5121.
[50]  BZDUCHA W, LANGWALD N, DYGAS J R, KROK F, MISZTAL-FARAJ B. Lithium gel polyelectrolytes based on crosslinked maleic anhydride-styrene copolymer[J]. Electrochim Acta, 2000, 45(21): 3563-3571.
BZDUCHA W, LANGWALD N, DYGAS J R, KROK F, MISZTAL-FARAJ B. Lithium gel polyelectrolytes based on crosslinked maleic anhydride-styrene copolymer[J]. Electrochim Acta, 2000, 45(21): 3563-3571.
[51] ITOH T, YOSHIKAWA M, UNO T, KUBO M. Solid polymer electrolytes based on poly(lithium carboxylate) salts[J]. Ionics, 2009, 15(1): 27-33.
[52] LEE J Y, LEE Y M, BHATTACHARYA B, NHO Y C, PARK J K. Solid polymer electrolytes based on crosslinkable polyoctahedral silsesquioxanes (POSS) for room temperature lithium polymer batteries[J]. Journal of Solid State Electrochemistry, 2009, 14(8): 1445-1449.
[53] MICHEL A, FRANK E, DOUGLAS R M, HIROYUKI O, BRUNO S. Ionic-liquid materials for the electrochemical challenges of the future[J]. Nature Materials, 2009, 8(8): 621-629.
[54] FERRARI S, QUARTARONE E, MUSTARELLI P, MAGISTRIS A, PROTTI S, LAZZARONI S, FAGNONI M, ALBINI A. A binary ionic liquid system composed of n-methoxyethyl-n-methylpyrrolidinium bis (trifluoro- methanesulfonyl)-imide and lithium bis (trifluoro- methanesulfonyl) imide: A new promising electrolyte for lithium batteries[J]. Journal of Power Sources, 2009, 194(1): 45-50.
[55] OLIVIER-BOURBIGOU H, MAGNA L, MORVAN D. Ionic liquids and catalysis: Recent progress from knowledge to applications[J]. Applied Catalysis A: General, 2010, 373(1/2): 1-56.
[56] OHNO H. Molten salt type polymer electrolytes[J]. Electrochimica Acta, 2001, 46(10/11): 1407-1411.
[57] TAKANORI F, TAKUZO A. Ionic liquids for soft functional materials with carbon nanotubes[J]. Chemistry A: European Journal, 2007, 13(18): 5048-5058.
[58] FERNICOLA A, PANERO S, SCROSATI B, TAMADA M, OHNO H. New types of  acid-base ionic liquids-based membranes for applications in PEMFCS[J]. Chem Phys Chem, 2007, 8(7): 1103�C1107.
acid-base ionic liquids-based membranes for applications in PEMFCS[J]. Chem Phys Chem, 2007, 8(7): 1103�C1107.
[59] KEN M, KINJI A, TAKUSHI S, KENJI K, ICHIROH T, NAOHIRO T, DON N F, KENJI H, TAKANORI F, TAKUZO A. Highly conductive sheets from millimeter-long single-walled carbon nanotubes and ionic liquids: Application to fast-moving, low-voltage electromechanical actuators operable in air[J]. Advanced Materials, 2009, 21(16): 1582-1585.
[60] MORI H, YAHAGI M, ENDO T. Raft polymerization of n-vinylimidazolium salts and synthesis of thermoresponsive ionic liquid block copolymers[J]. Macromolecules, 2009, 42(21): 8082-8092.
[61] MATSUMI N, KAGATA A, AOI K. Synthesis of supramolecular solid polymer electrolytes via self-assembly of diborylated ionic liquid[J]. Journal of Power Sources, 2010, 195(18): 6182-6186.
[62] VURK S A, JOVANOVSKI V, POLLET-VILLARD A, JERMAN I, OREL B. Imidazolium-based ionic liquid derivatives for application in electrochromic devices[J]. Solar Energy Material and Solar Cells, 2008, 92(2): 126-135.
[63] SUBIANTO S, MISTRY M K, CHOUDHURY N R, DUTTA N K, KNOTT R. Composite polymer electrolyte containing ionic liquid and functionalized polyhedral oligomeric silsesquioxanes for anhydrous PEM applications[J]. ACS Applied Materials & Interfaces, 2009, 1(6): 1173-1182.
[64] TANAKA K, ISHIGURO F, CHUJO Y. POSS ionic liquid[J]. Journal of the American Chemical Society, 2010, 132(50): 17649-17651.
[65] TANAKA K, ISHIGURO F, CHUJO Y. Thermodynamic study of poss-based ionic liquids with various numbers of ion pairs[J]. Polymer Journal, 2011, 43: 708-713.
[66]  M, JERMAN I,
M, JERMAN I,  OREL B. POSS based ionic liquid as an electrolyte for hybrid electrochromic devices[J]. Solar Energy Materials and Solar Cells, 2011, 95(12): 3472-3481.
OREL B. POSS based ionic liquid as an electrolyte for hybrid electrochromic devices[J]. Solar Energy Materials and Solar Cells, 2011, 95(12): 3472-3481.
[67] CHANG Y W, WANG E, SHIN G, HAN J E , MATHER P T. Poly(vinyl alcohol) (PVA)/sulfonated polyhedral oligosilsesquioxane (SPOSS) hybrid membranes for direct methanol fuel cell applications[J]. Polymers for Advanced Technologies, 2007, 18(7): 535-543.
[68] FEUILLADE G, PERCHE P. Ion-conductive macromolecular gels and membrances for solid lithium cells[J]. Journal of Applied Electrochemistry, 1975, 5: 63-69.
[69] LI M, REN W, ZHANG Y, ZHANG Y. Study on properties of gel polymer electrolytes based on ionic liquid and amine-terminated butadiene-acrylonitrile copolymer chemically crosslinked by polyhedral oligomeric silsesquioxane[J]. Journal of Applied Polymer Science, 2012, 126(1): 273-279.
[70] KIM H S, MAH S K. Anode active material, method of preparing the same, and anode and lithium battery containing the material: United States, 0166634 A1[P]. 2008-08-30.
[71] XUE Y H, LIU Y, LU F, QU J, CHEN H, DAI L M. Functionalization of graphene oxide with polyhedral oligomeric silsesquioxane (POSS) for multifunctional applications[J]. The Journal of Physical Chemistry Letters, 2012, 3: 1607-1612.
[72] HENNIGE V, HYING C, HOERPEL G, JOST C, KUEHNLE A. Separator comprising non-conductive polymer fibers with porous inorganic coating, useful in electrochemical cells, especially lithium batteries, where the polymer indudes silicon-oxygen clusters: Europe, DE 10304734 A1[P]. 2004-07-08.
(�༭ ����Ƽ)
������Ŀ��������Ȼ��ѧ����������Ŀ(50803008)���й���ʿ���ر�������Ŀ(201104508)������ʡ�Ƽ�����ʿ���ѧ����������Ŀ(2011RS4067)������ʡ�����������������������Ŀ(11B001)����������Դ������������ʡ�ص�ʵ���ҿ��Ż���������Ŀ(2011KFJJ006)
�ո����ڣ�2012-11-19�������ڣ�2013-08-16
ͨ�����ߣ������壬���ڣ���ʿ���绰��0731-88876454��E-mail: 13975808172@126.com

