二硫代氨基甲酸盐络合分离钒铬及其在钒铬还原渣处理中的应用
来源期刊:中国有色金属学报(英文版)2019年第11期
论文作者:彭雪枫 张洋 范兵强 郑诗礼 王晓健 张盈 李平 刘丰强
文章页码:2400 - 2410
关键词:钒;铬;分离;络合;二硫代氨基甲酸盐
Key words:vanadium; chromium; separation; complexation; dithiocarbamate
摘 要:提出一种从酸性溶液中分离回收钒和铬的新方法。该分离方法的原理基于选择性络合,五价钒可以与二硫代氨基甲酸盐络合反应生成VO(R1R2NCS2)3沉淀,三价铬则留在酸性溶液中;然后通过解络合从VO(R1R2NCS2)3沉淀中回收钒以及再生络合剂,通过中和水解法从三价铬溶液中回收铬。研究络合反应的动力学并计算其表观活化能。工艺优化后钒的沉淀率达到97%,铬损失小于4%,络合剂再生率可达99.5%。该分离方法已成功应用于常规钒生产工业产生的钒铬还原渣的处理,钒以钒酸钙的形式回收,铬以Cr2O3的形式回收,Cr2O3产品纯度超过98%。
Abstract: A novel method for the separation and reclamation of vanadium and chromium from acidic solution was proposed. The principle for the separation is based on selective complexation. In specific, V(V) can be complexated by dithiocarbamate to form VO(R1R2NCS2)3 precipitate, leaving Cr(III) remained in the acidic solution. Then the reclamation of V and Cr from VO(R1R2NCS2)3 precipitate and Cr(III)-bearing solution can be reached by decomplexation and neutralization, respectively. The kinetics of the complexation reaction was studied. In addition, the precipitation ratio of vanadium reached 97% and the loss of chromium was less than 4% after process optimization. The complexing agent could be regenerated with a high ratio of 99.5%. The method was successfully applied to treating chromium-vanadium-bearing slag produced in conventional vanadium production industry. Vanadium and chromium could be extracted from the slag in the form of calcium vanadate and Cr2O3 product with a purity of over 98%.
Trans. Nonferrous Met. Soc. China 29(2019) 2400-2410
Xue-feng PENG1,2, Yang ZHANG1, Bing-qiang FAN1, Shi-li ZHENG1, Xiao-jian WANG1, Ying ZHANG1, Ping LI1, Feng-qiang LIU3
1. Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, China;
2. School of Chemical Engineering, University of Chinese Academy of Sciences, Beijing 100049, China;
3. Pangang Group Co., Ltd., Panzhihua 617067, China
Received 26 February 2019; accepted 24 June 2019
Abstract: A novel method for the separation and reclamation of vanadium and chromium from acidic solution was proposed. The principle for the separation is based on selective complexation. In specific, V(V) can be complexated by dithiocarbamate to form VO(R1R2NCS2)3 precipitate, leaving Cr(III) remained in the acidic solution. Then the reclamation of V and Cr from VO(R1R2NCS2)3 precipitate and Cr(III)-bearing solution can be reached by decomplexation and neutralization, respectively. The kinetics of the complexation reaction was studied. In addition, the precipitation ratio of vanadium reached 97% and the loss of chromium was less than 4% after process optimization. The complexing agent could be regenerated with a high ratio of 99.5%. The method was successfully applied to treating chromium-vanadium-bearing slag produced in conventional vanadium production industry. Vanadium and chromium could be extracted from the slag in the form of calcium vanadate and Cr2O3 product with a purity of over 98%.
Key words: vanadium; chromium; separation; complexation; dithiocarbamate
1 Introduction
Vanadium and chromium are important strategic metals and play an important role in the development of society. Vanadium is widely used in steel, chemical and battery industries due to its excellent mechanical, catalytic, electrochemical, and other physicochemical properties [1,2]. Chromium has wide application in alloy production, electroplating, leather tanning and refractory material preparation [3-5].
Vanadium and chromium are often associated in many minerals, such as vanadium titano-magnetite and chromite ore because of their similar physicochemical nature [6,7]. Vanadium titano-magnetite is the most important resource for vanadium extraction [8,9] through the blast furnace (BF) process. During BF smelting, titanium is concentrated in titanium slag, while iron, vanadium and chromium turn into vanadium-bearing pig iron. Then vanadium and chromium are separated from the pig iron in the form of vanadium slag by converter smelting [10]. Vanadium, as the most valuable element in the vanadium slag, is conventionally extracted by sodium salt roasting and water leaching process [11]. However, a large amount of wastewater containing Cr(VI) and V(V) is generated in this process, which has to be treated by reduction, neutralization, and filtration, generating chromium-vanadium-bearing slag. This sort of slag exhibits a complex composition containing amorphous chromium hydroxide, tetravalent vanadium hydroxide, ferric hydroxide, and soluble sulfate salts [12,13]. Furthermore, it is urgent to harmlessly treat this slag since heavy metals in it bring huge environmental risk.
To date, abundant of researches can be found in open literatures on the treatment of the chromium- vanadium-bearing slag. Using the property that vanadium is more prone to be oxidized than chromium in alkaline medium (for instance NaOH solution) by the oxidant of H2O2, KClO3, or O2 [14-17], vanadium alone can be extracted in the form of vanadate after oxidation.
The slag can also be oxidized via alkaline roasting to selectively extract vanadium [18,19]. But chromium is not able to be extracted by this type of method, and the remained residue after vanadium extraction generally contains highly toxic Cr(VI), which can cause even severer environmental issues. Thus, it is strongly required to extract vanadium and chromium simultaneously when dealing with the chromium- vanadium-bearing slag.
However, the challenge of separating vanadium and chromium with high efficiency will spring out during co-extraction of V and Cr. The chromium product of Cr2O3 requires vanadium content less than 0.15%, and the vanadium product of V2O5 requires chromium content less than 0.1%. Therefore, a strategy for deep separation of vanadium and chromium is of high demand. Using the frequently reported methods of the ion exchange [20], solvent extraction [21-23], chemical precipitation [24,25], membrane filtration [26], and adsorption [27] are difficult to meet the target because of the similarities of their properties. In addition, each method has its own disadvantages, such as poisoning of the resin in the ion exchange method, expensive extractants and pollution from organics in the solvent extraction method, low separation efficiency and high impurity content in the chemical precipitation method.
Recently, a novel method for deep separation of vanadium and chromium in the acidic solution was proposed by the present authors of this research [28]. It was found that some sort of amines can react with V selectively to form V-containing complex precipitates but it will not act on Cr(III) in acidic solution. The V-containing complexes can be readily dissociated in alkaline medium to regenerate the complexing agent and reclaim the valuable vanadium metal. However, there are still a few questions to be answered in this new method, including the inherent principle and the kinetics of the complexation and decomplexation reactions, and the optimization of the process parameters, which will be the main contents for this study. The application of this technology to harmlessly treat chromium-vanadium- bearing slag will also be reported in this research.
2 Experimental
2.1 Materials
The chromium-vanadium-bearing slag used in this study was obtained from the Pangang Group Co., Ltd., China. After preprocessing by selective oxidation of V(IV) to V(V), the composition of its acid leaching solution is listed in Table 1. Therefore, the solutions containing 5 g/L V(V) and 10 g/L Cr(III) were used in the experiments and prepared in the laboratory using sodium metavanadate and basic chromic sulfate. A dithiocarbamate (R1R2NCS2-) complexing agent was used, where R1 and R2 can be the same or different alkyl. All reagents and materials used in the research were of analytical grade and without further purification. Deionized water was produced by a water purification system.
Table 1 Composition of acid leaching solution of chromium- vanadium-bearing slag (g/L)

2.2 Experimental procedure
The complexation separation process was carried out according to the flow sheet shown in Fig. 1. For better separation of vanadium and chromium, the solution was acidified using sulfuric acid to a pH ranging from 2.5 to 3.0 with constant stirring at room temperature. Vanadium reacts with the complexing agent added to form an insoluble V complex (a yellow solid precipitate), and the precipitate was filtered out to obtain V-free Cr(III)-bearing solution. The slurry was obtained by mixing the V complexes with deionized water, and then the slurry was alkalized by adding NaOH to a pH value of 11.0 to 12.0 with stirring at 40-50 °C. After the V complex was dissolved (also decomplexation), CaO was further added to precipitate vanadium in the form of calcium vanadate. Thus, the alkaline complexing agent solution was reused for the next batch of separation.
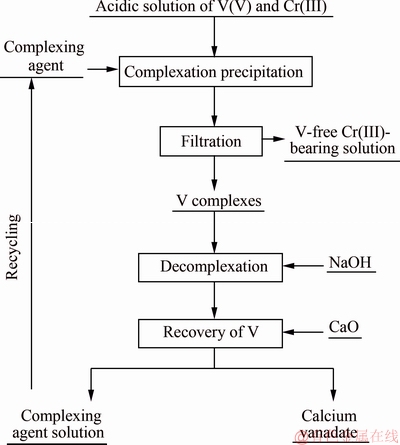
Fig. 1 Flow chart of complexation separation of V and Cr in acidic solution
2.3 Sample analysis
The vanadium and chromium concentrations in solutions were measured by inductively coupled plasma optical emission spectrometry (ICP-OES). Elemental analyses of vanadium complexes were performed on a Vario-EL cube Elementar to determine the contents of C, N, and H. Vanadium complexes and complexing agent were characterized by X-ray diffraction (XRD) at diffraction angles (2θ) ranging from 5° to 90° to determine the phases. The structure of the complexes was also resolved by Fourier Transform Infrared (FT-IR) analysis with a resolution of 4 cm-1 in the range of 400-4000 cm-1.
3 Results and discussion
3.1 Principle of complexation separation
3.1.1 Reaction between vanadium and complexing agent
It is accepted that V(V) exists in the form of VO3+, 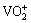 ,
, 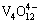 ,
,  or other polyvanadate anion in acidic solution with a pH value of 0-4.0 [29-31], thus it can be predicted that the complexation reaction may be as in reactions (1)-(5). During the complexation reaction the polyvanadate anion would be transformed into vanadate cation to react with complexing agent anion.
or other polyvanadate anion in acidic solution with a pH value of 0-4.0 [29-31], thus it can be predicted that the complexation reaction may be as in reactions (1)-(5). During the complexation reaction the polyvanadate anion would be transformed into vanadate cation to react with complexing agent anion.
VO3++3R1R2NCS2- →VO(R1R2NCS2)3- (1)
VO2+ + R1R2NCS2- →R1R2NCS2VO2- (2)
VO2+ +2H+ VO3++H2O (3)
VO3++H2O (3)
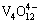 +8H+
+8H+ 4
4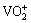 +4H2O (4)
+4H2O (4)
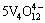 +8H+
+8H+ 2
2 +4H2O (5)
+4H2O (5)
WANG et al [32] reported a reliable method to determine the coordination number and complexing constant for complexation reaction. The experimental results in this research show that the coordination number of the V complexes is 3.18, which is consistent with reaction (1). Meanwhile, the complexing constant of the V complexes is 6.54×1013.
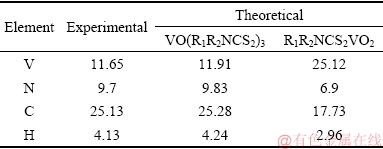
The elemental analysis results are given in Table 2. The analyzed data are quite fitted with the theoretical data of VO(R1R2NCS2)3, further demonstrating the empirical formula of the V complexes is VO(R1R2NCS2)3.
Table 2 Elemental analysis of V complexes (wt.%)
The XRD patterns of the V complexes and complexing agent are shown in Fig. 2. The patterns reveal that a new crystal phase was obtained but this could not be matched to any known crystal phases.
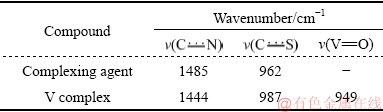
The IR absorption spectra of the complexing agent and V complexes are given in Fig. 3 and Table 3. All the dithiocarbamate complexes show bands assigned to  vibrations in the 1505-1450 cm-1 range, which lies between those of ν(C=N) and ν(C―N) at 1690-1640 cm-1 and 1350-1250 cm-1, respectively. The bands in the 995-950 cm-1 range can be attributed to the prevailing contribution of
vibrations in the 1505-1450 cm-1 range, which lies between those of ν(C=N) and ν(C―N) at 1690-1640 cm-1 and 1350-1250 cm-1, respectively. The bands in the 995-950 cm-1 range can be attributed to the prevailing contribution of  [29]. The strong or medium-strong bands observed at 985-945 cm-1 in the V(V) derivatives are characteristic of vanadyl compounds with terminal V=O groups [29,31].
[29]. The strong or medium-strong bands observed at 985-945 cm-1 in the V(V) derivatives are characteristic of vanadyl compounds with terminal V=O groups [29,31].

Fig. 2 XRD patterns of V complexes and complexing agent
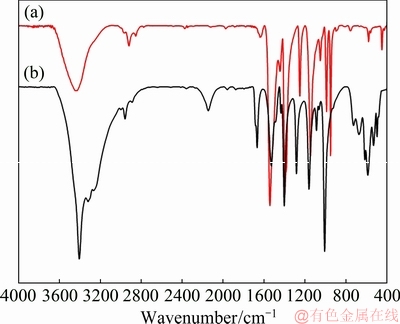
Fig. 3 IR absorption spectra of V complexes (a) and complexing agent (b)
Table 3 Most important IR bands for complexing agent and V complexes
Therefore, in the acidic solution containing V(V) and Cr(III), V can react with complexing agent and can be transformed to V complexes, VO(R1R2NCS2)3, for precipitation, thus to be separated from Cr.
3.1.2 Kinetics of complexation reaction between vanadium and complexing agent
According to the dissociation mechanism of the complex substitution reaction, when the concentration of the substitution ligand X is large, the rate equation of complexation reaction can be expressed as
MLn+X→MLn-1X+L (6)
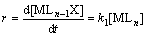 (7)
(7)
where MLn is the coordination ion formed by vanadium ion with water or a hydroxyl in aqueous solution, and X is the complexing agent. It shows that the reaction rate (r) is proportional to the concentration of MLn, but independent of the concentration of X [33].
Since the precipitation of vanadium ions with complexing agent will lead to the decrease of the total amount of ions in the solution, conductivity of the solution was used in this research as an indirect indicator of the concentration of vanadium ion during the reaction. Figure 4 shows the relationship between the conductivity (σ) and the initial V concentration (c(V)) in the solution, and a satisfactory linear relationship is exhibited.
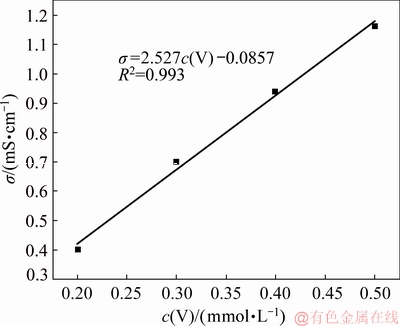
Fig. 4 Relationship between conductivity and V concentration
Figure 5 shows the conductivity of the solution as a function of time during precipitation. It can be seen that with different initial vanadium concentrations, the change on conductivity due to the complexation reaction is similar. It is obvious that the curves of conductivity with time can be divided into two stages. The stage with rapid decreasing of conductivity should be due to the nucleation, and the stage with slow decreasing should be attributed to the crystal growth [34]. Since the complexation reaction happens very fast, only two points were caught in the nucleation stage.
Kinetic calculations were performed for each set of data with different reaction orders, and the highest correlation coefficient was obtained for both nucleation and crystal growth stages with the first-order reaction. Figure 6 shows the calculated fitting results for nucleation and crystal growth. In addition, the fitted values of the reaction rate constants for the four vanadium-bearing solutions are listed in Table 4.
The reaction rate constants at 25, 30, 37, and 52 °C were determined in the same manner. Then the reaction rates of nucleation and crystal growth at different temperatures are fitted in Fig. 7, and the values of K1 and K2 are obtained, where K is the reaction rate constant corresponding to the slopes of the straight lines. Nucleation and crystal growth are the first-order reaction at all temperatures, with the rate constants listed in Table 5.
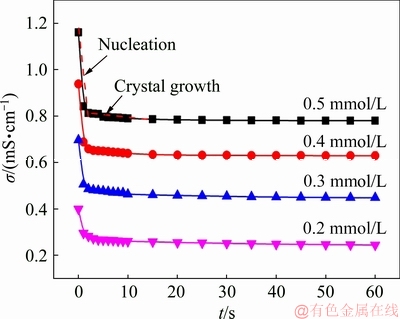
Fig. 5 Relationship between conductivity and time
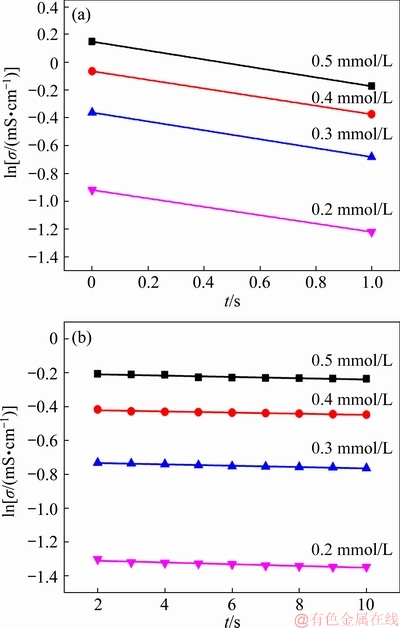
Fig. 6 Calculated fitting results for nucleation (a) and crystal growth (b) at different concentrations of V
Table 4 Reaction rate constant at different concentrations of V
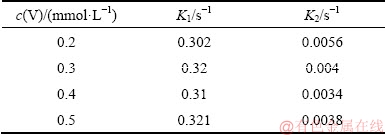
Table 5 Reaction rate constant at different temperatures
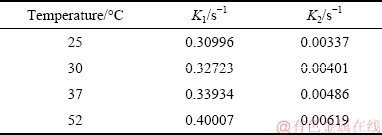
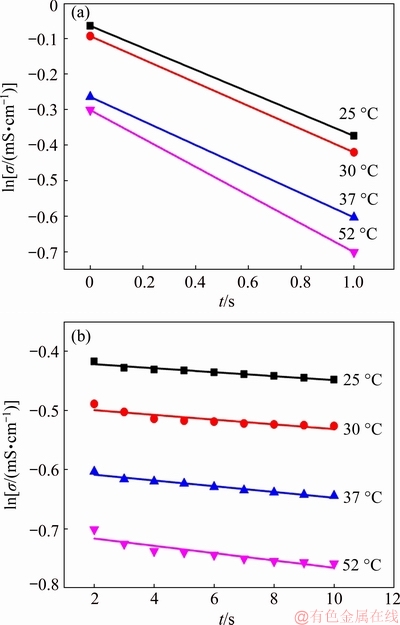
Fig. 7 Calculated fitting results for nucleation (a) and crystal growth (b) at different temperatures
Figure 8 shows the linear diagram of ln K vs T-1 of nucleation and crystal growth. Then the specific apparent activation energy could be calculated based on the Arrhenius equation, and the result is shown in the linear diagram. The apparent activation energy of the nucleation was calculated to be 7473.7 J/mol. And the apparent activation energy of the crystal growth was calculated to be 17748.8 J/mol.
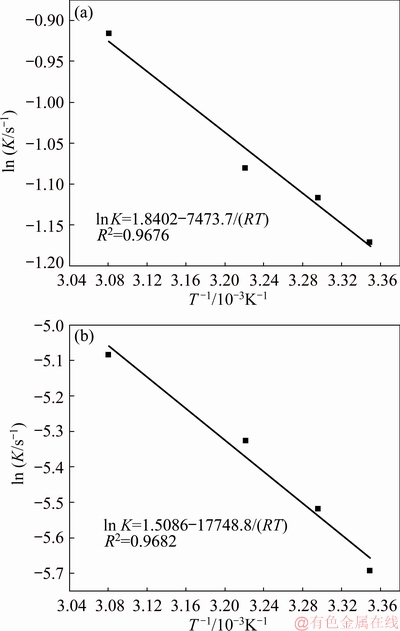
Fig. 8 Linear diagram of ln K vs T-1 of nucleation (a) and crystal growth (b)
3.2 Optimization of complexation separation efficiency of V and Cr
The effects of pH, reaction temperature, reaction time, and dosage of the complexing agent on the separation of vanadium and chromium were studied. The separation efficiency of vanadium and chromium was measured in terms of V precipitation (η) and Cr loss (x).
η=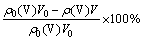 (8)
(8)
x=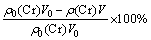 (9)
(9)
where ρ0(V) and ρ(V) are the V concentrations (g/L) in the solutions before and after precipitation, ρ0(Cr) and ρ(Cr) are the Cr concentrations (g/L) in the solutions before and after precipitation, and V0 and V are the volumes of the solutions before and after precipitation, respectively.
3.2.1 Effect of pH
Figure 9 shows the effect of pH on V precipitation and Cr loss. Other experimental parameters are a mass ratio of the complexing agent to V of 12:1, constant stirring for 20 min, and a temperature of 25 °C. As can be seen, the rate of precipitated V increased from 87.4% to the highest of 97.1% with the increased pH value from 1.5 to 2.8, followed by a gradual decrease of the V precipitation rate with further increased pH value to 4.0. Thus the optimized pH value for complexation reaction should be in the range of 2.5-3.0.
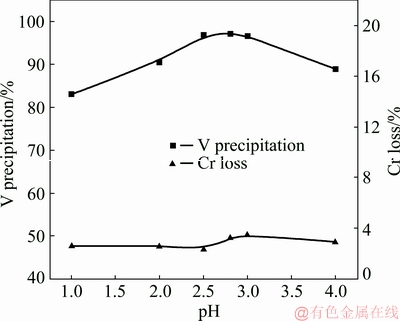
Fig. 9 Effect of pH on separation efficiency of V and Cr
The trend in Fig. 9 versus pH is related to the chemical state of vanadium ions in aqueous solution and the solubility of the V complexes. In low pH solution (pH<3.0), the pentavalent vanadium ion is mainly in the form of 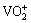 and VO3+. With increased pH,
and VO3+. With increased pH, 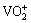 and VO3+ react with OH- to form
and VO3+ react with OH- to form  and
and 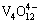 anions [35,36] which would be repelled by complexing agent because they have the same charge as
anions [35,36] which would be repelled by complexing agent because they have the same charge as 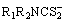 . The pH of the solution also affects the solubility of vanadium complexes thus the vanadium precipitation ratio. As shown in Fig. 10(a), the vanadium complex has the minimum solubility in the pH range of 3.0-7.0 at 25 °C, and the remarkably increased solubility in alkaline region is also the basis for the regeneration of the complexing agent by decomplexation reaction.
. The pH of the solution also affects the solubility of vanadium complexes thus the vanadium precipitation ratio. As shown in Fig. 10(a), the vanadium complex has the minimum solubility in the pH range of 3.0-7.0 at 25 °C, and the remarkably increased solubility in alkaline region is also the basis for the regeneration of the complexing agent by decomplexation reaction.
3.2.2 Effect of reaction temperature
Figure 11 shows the effect of temperature on V precipitation and Cr loss in a solution with a pH value of 2.8, a mass ratio of the complexing agent to V of 12:1 for 20 min. When the temperature was elevated from 25 to 70 °C, V precipitation was rapidly decreased from 96.9% to 50.3%. Thus the temperature for complexation reaction should be as low as possible. The reasons for the adverse impact of temperature on V precipitation lie in two aspects, the increased solubility of the V complexes with temperature, which is shown in Fig. 10(b), and the nature of exothermic reaction of driving the decomplexation reaction with temperature.
3.2.3 Effect of dosage of complexing agent
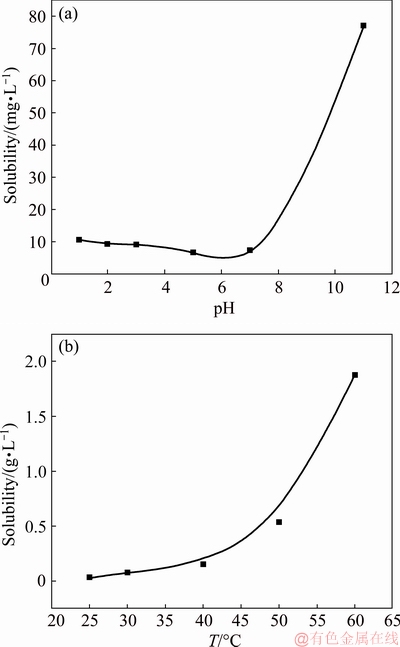
Fig. 10 Solubility of V complex at different pH (a) and temperatures (b)
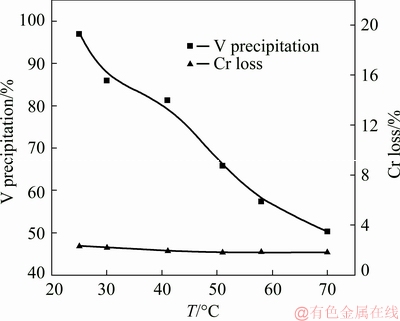
Fig. 11 Effect of temperature on separation efficiency of V and Cr
Figure 12 shows the effects of the dosage of the complexing agent on V precipitation and Cr loss. The mass ratio of the complexing agent to V changed from 4:1 to 22:1 at 25 °C under pH of 2.8 for 20 min. It can be seen that there is a turning point at a mass ratio of 12:1. Lower than the ratio, the V precipitation rate increased almost linearly from 50% to 97%; higher than that ratio of 12:1, it reached a plateau. Thus the optimized mass ratio of the complexing agent to V was 12:1.
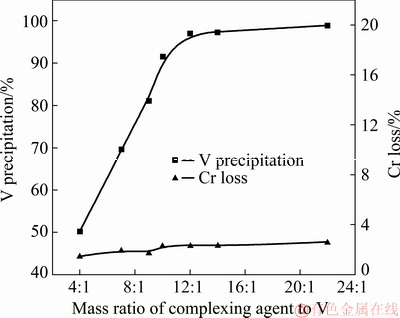
Fig. 12 Effect of mass ratio of complexing agent to V on separation efficiency of V and Cr
3.2.4 Effect of reaction time
The effects of reaction time on V precipitation and Cr loss are shown in Fig. 13. Other parameters were a pH value of 2.8, a mass ratio of the complexing agent to V of 12:1 and at 25 °C. It could be seen that a slight decreasing of the V precipitation rate and Cr loss was observed with prolonged time from 5 to 60 min, but the effect was negligible. Consistent with the results indicated by the conductivity before, the complexation reaction can reach equilibrium very quickly. A reaction time of 10 min was chosen for the complexation precipitation with a V precipitation rate of 97.1%.
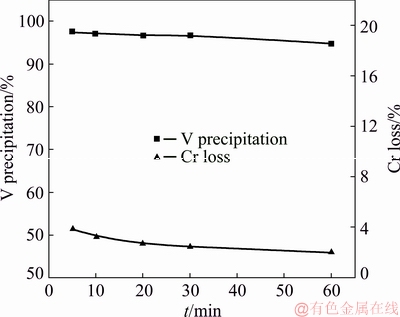
Fig. 13 Effect of reaction time on separation efficiency of V and Cr
3.2.5 Effect of mass concentration ratio of chromium to vanadium
Figure 14 shows the effects of the initial concentration of vanadium on V precipitation and the V concentration in the filtrate. The initial vanadium concentration changed from 0.5 to 10 g/L at 25 °C under pH of 2.8 for 20 min, a mass ratio of the complexing agent to V of 12:1 and the initial chromium concentration is constant at 10 g/L. It can be seen that the V precipitation rate peaked at 98.7% when the mass concentration ratio of Cr to V is 5:1 and slightly decreasing with the decrease of the V concentration. The vanadium concentration in the filtrate is less than 100 mg/L when the mass concentration ratio of Cr to V is greater than 2:1. Therefore, the mass concentration ratio of Cr to V should be controlled to be more than 2:1, and the mass concentration ratio of Cr to V of the acid leaching solution of chromium-vanadium-bearing slag is actually greater than 2:1.
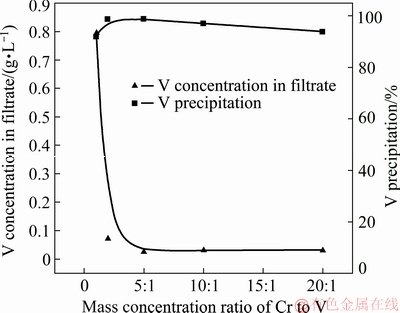
Fig. 14 Effect of mass concentration ratio of Cr to V on separation efficiency of V and Cr
3.3 Decomplexation of V complexes and regeneration of complexing agent
3.3.1 Principle for decomplexation of V complexes
As indicated earlier, there is a chance that the vanadium complexes can be dissociated under alkaline circumstance. The dissociation reaction can be expressed by following reactions (10) and (11). In alkaline solution, reaction (10) happens due to the increased solubility of V complexes, and the transformation from VO3+ into pervanadyl anion by reaction (10) further promotes the reaction (11) to shift to the right direction of dissociation.
VO(R1R2NCS2)3(s) VO3++
VO3++ (10)
(10)
2VO3+ +10OH-→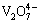 +5H2O (11)
+5H2O (11)
After V complexes are dissociated in alkaline solution to be near complete, CaO can be added to precipitate 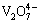 in the form of Ca2V2O7 solid via reaction (12), then vanadium and complexing agent are separated from each other. Meanwhile, the alkaline complexing agent solution can be regenerated and recycled for complexation separation of V and Cr for the next batch.
in the form of Ca2V2O7 solid via reaction (12), then vanadium and complexing agent are separated from each other. Meanwhile, the alkaline complexing agent solution can be regenerated and recycled for complexation separation of V and Cr for the next batch.
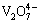 +2CaO(s)+2H2O→Ca2V2O7(s)+4OH- (12)
+2CaO(s)+2H2O→Ca2V2O7(s)+4OH- (12)
3.3.2 Effect of key parameters on recycling of complexing agent
As shown by reactions (10)-(12), pH value should be the critical factor influencing the regeneration of the complexing agent. In addition, other factors including temperature, time and the dosage of CaO have to be considered too. The effects of these factors were evaluated in terms of the vanadium removal (y) and the complexing agent regeneration (z), which were calculated by the following formula:
y=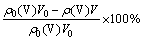 (13)
(13)
z=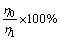 (14)
(14)
where η0 is the initial V precipitation, and η1 is V precipitation using the regenerated complexing agent solution as the new complexing agent. The V precipitation was calculated as shown in Eq. (8).
The experimental results for the effects of pH, temperature, dosage of CaO and time on dissociation are shown in Fig. 15. Other parameters for each single factor experiment are also listed out in the figure captions. By comprehensively analyzing the data, it was demonstrated that the optimized conditions for the removal of vanadium and the regeneration of the complexing agent were at a temperature of 60 °C, under a pH circumstance of 11.5, a mass ratio of CaO to V of 6:1 and a time of 1.5 h. The regeneration rate of the complexing agent can reach 99.5% under the optimized conditions.
3.4 Application in harmless treatment of chromium- vanadium-bearing slag
The method of complexation separation of vanadium and chromium has been successfully applied industrially to treat chromium-vanadium-bearing slag. The flow sheet of the entire process can be seen in Fig. 16.
A pilot plant with a scale of processing 18000 t of chromium-vanadium-bearing slag was first built in Liaoning province, China, and the project is being operated successfully and stably. Later on, another similar pilot plant was also built in Panzhihua city, China. Some of the operating data for the second project can be found in Table 6, Table 7 and Fig. 17.
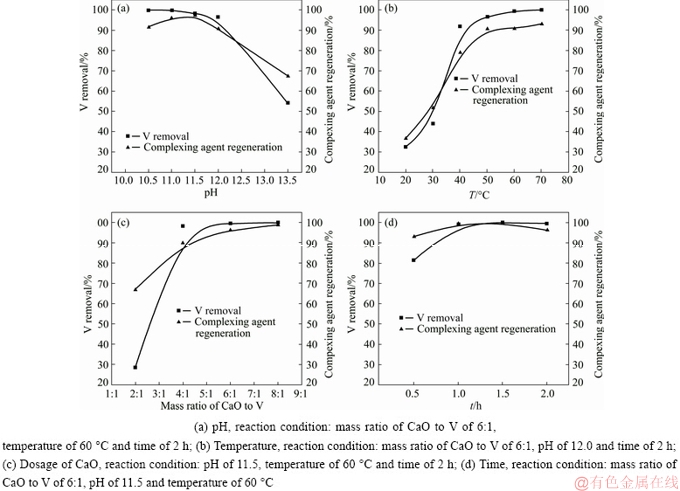
Fig. 15 Effect of key parameters on regeneration of complexing agent
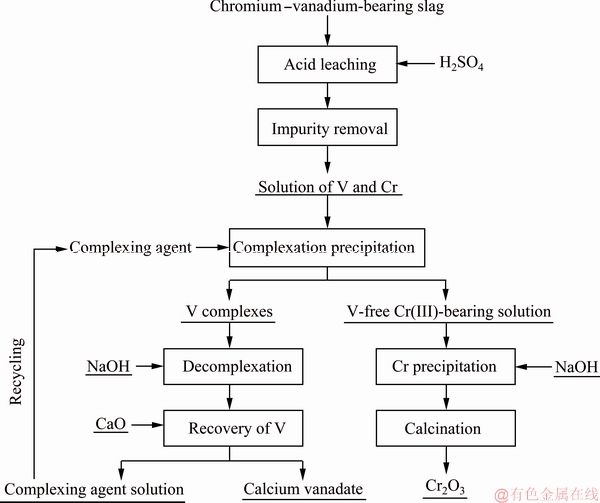
Fig. 16 Flow sheet for treatment of chromium-vanadium-bearing sla
Table 6 Concentrations of V and Cr in initial and vanadium precipitated solution (g/L)

Table 7 Compositions of Cr2O3 product (wt.%)
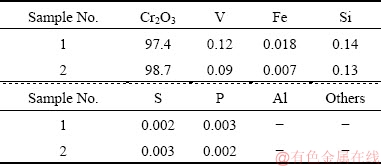
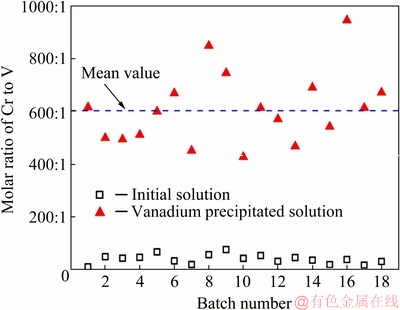
Fig. 17 Results after 72 h operation for the second project in Panzhihua city
The results show that the complexation separation can work well under high Cr concentration and low V concentration in the initial solution. The remaining concentration of V in solution is less 50 mg/L, whereasthe concentration of Cr is still at a high level. The molar ratio of Cr to V can increase from initially about 40:1 to an average level of 610:1 after selectively precipitating vanadium by the complexing agent. The V-free Cr(III)-bearing solution was further neutralized to get Cr2O3 product, and its purity was around 98% which meets the requirement for the producing of chromium metal.
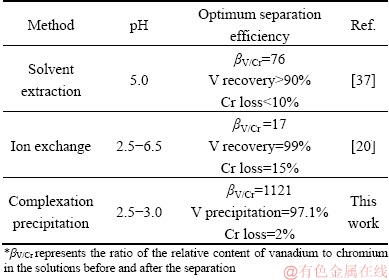
Table 8 further compares the vanadium and chromium separation results by different methods from the open literature. It shows that both the solvent extraction and ion exchange have a high recovery of vanadium, but the separation efficiency for vanadium and chromium is low in acidic conditions. Furthermore, the Cr loss rate is high because of the loss of chromium in the resins and extractants are unavoidable. It is showing that the method proposed in this work is with the highest separation coefficient and lowest Cr loss rate.
Table 8 Separation of vanadium and chromium by various methods under acidic conditions
4 Conclusions
(1) The complexing agent of dithiocarbamate (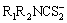 ) can react selectively with V(V) to form V complexes of VO(R1R2NCS2)3, which is a precipitate. And the target of separating vanadium from chromium can be reached.
) can react selectively with V(V) to form V complexes of VO(R1R2NCS2)3, which is a precipitate. And the target of separating vanadium from chromium can be reached.
(2) The kinetics of nucleation and crystal growth of the complexation reaction between complexing agent and V(V) both belong to the first-order reactions. The apparent activation energies of nucleation and the crystal growth are 7473.7 and 17748.8 J/mol, respectively.
(3) The optimized conditions for complexation separation are under pH 2.8, a temperature of 25 °C, a mass ratio of the complexing agent to vanadium of 12:1, a time of 10 min, and a mass ratio of chromium to vanadium of greater than 2:1. The vanadium precipitation rate can reach 97.1% with a low loss rate of Cr less than 4% under the optimized operation parameters.
(4) The optimized conditions for the regeneration of the complexing agent are under pH of 11.5, a temperature of 60 °C, a mass ratio of CaO to vanadium of 6:1 and a time of 1.5 h, and the regeneration rate can reach 99.5%.
(5) The technology of complexation separation of vanadium and chromium has been successfully applied to treating chromium-vanadium-bearing slag in pilot scale plants. The industrial operation data show that vanadium and chromium can be separated with very high separation coefficients, and qualified Cr2O3 product can be produced.
References
[1] MASE A, SUGITA T, MORI M, IWAMOTO S, TOKUTOME T, KATAYAMA K, ITABASHI H. Study of vanadium-modified N/Si co-doped TiO2 in aqueous solution and its photocatalytic activity [J]. Chemical Engineering Journal, 2013, 225: 440-446.
[2] NICHOLAS N J, SILVA G D, KENTISH S, STEVENS G W. Use of vanadium(V) oxide as a catalyst for CO2 hydration in potassium carbonate systems [J]. Industrial & Engineering Chemistry Research, 2015, 53(8): 3029-3039.
[3] MOSKALYK R R, ALFANTAZI A M. Processing of vanadium: A review [J]. Minerals Engineering, 2003, 16: 793-805.
[4] ZHANG Y M, BAO S X, LIU T, CHEN T J, HUANG J. The technology of extracting vanadium from stone coal in China: History, current status and future prospects [J]. Hydrometallurgy, 2011, 109: 116-124.
[5] ERDEM M, TUMEN F. Chromium removal from aqueous solution by the ferrite process [J]. Journal of Hazardous Materials, 2004, 109(1): 71-77.
[6] VITOLO S, SEGGIANI M, FILIPPI S, BROCCHINI C. Recovery of vanadium from heavy oil and Orimulsion fly ashes [J]. Hydrometallurgy, 2000, 57: 141-149.
[7] WANG Zhong-hang, ZHENG Shi-li, WANG Shao-na, LIU Biao, WANG Da-wei, DU Hao, ZHAN Yi. Research and prospect on extraction of vanadium from vanadium slag by liquid oxidation technologies [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(5): 1273-1288.
[8] LI H Y, LI C, ZHANG M, WANG K, XIE B. Removal of V(V) from aqueous Cr(VI)-bearing solution using anion exchange resin: Equilibrium and kinetics in batch studies [J]. Hydrometallurgy, 2016, 165: 381-389.
[9] QIU H, ZHANG H, ZHAO B, ZHU J, LIU D. Dynamics study on vanadium extraction technology from chloride leaching steel slag [J]. Rare Metal Material Engineering, 2013, 42: 696-699.
[10] DU He-gui. Theory of smelting V and Ti-Magnetite by blast furnace [M]. Beijing, China: Science Press, 1996. (in Chinese)
[11] LIU B, DU H, WANG S N, ZHANG Y, ZHENG S L, LI L J, CHEN D H. A novel method to extract vanadium and chromium from vanadium slag using molten NaOH-NaNO3 binary system [J]. Aiche Journal, 2013, 59: 541-552.
[12] ZHU Shou-chuan. Engineering application of deoxidation- neutralization and evaporation concentration process to the treatment of wastewater with sedimentated vanadium [J]. Industrial Water Treatment, 2009, 29 (9): 84-87. (in Chinese)
[13] WANG Ying. Current situation of the vanadium precipitating waste water treatment technology [J]. Ferro-Alloys, 2012, 6: 41-45. (in Chinese)
[14] WEN Shi-zu, YANG Ming-liang. Extraction of vanadium from reduced V-Cr slag [J]. Shanghai Metals (Non-ferrous Fascicule), 1988, 9: 38-43. (in Chinese)
[15] GE Bing-li. Technological study on vanadium extraction from neutralized sludge by alkaline leaching and smelting of Cr-V alloy using its residues [J]. Ferro-Alloys, 1990, 6: 39-42. (in Chinese)
[16] YANG K, TIAN X D, YANG Y L, ZHONG R H, CHEN Y B, LIU H. Leaching of vanadium from vanadium-bearing chromium residues by alkaline process [J]. Mining & Metallurgical Engineering, 2010, 27(3): 70-73.
[17] YANG K, ZHANG X, TIAN X, YANG Y, CHEN Y. Leaching of vanadium from chromium residue [J]. Hydrometallurgy, 2010, 103: 7-11.
[18] YANG H, MAO L, XUE X. Separation and recovery of chromium and vanadium from reduced vanadium-chromium precipitate by calcinations-alkaline leaching [J]. CIESC Journal, 2014, 65(3): 948-953.
[19] JIANG Lin, WU Zhen-xiu. Extraction of vanadium from reduced vanadium-chromium precipitate by oxygen-enriched calcination and alkaline leaching [J]. Modern Chemical Industry, 2015, 35 (3): 87-89. (in Chinese)
[20] FAN Y, WANG X, WANG M. Separation and recovery of chromium and vanadium from vanadium-containing chromate solution by ion exchange [J]. Hydrometallurgy, 2013, 136(4): 31-35.
[21] SUN P, HUANG K, WANG X, SONG W, ZHENG H, LIU H. Separation of V from alkaline solution containing Cr using acidified primary amine N1923 with the addition of trisodium citrate [J]. Separation & Purification Technology, 2017, 179: 504-512.
[22] HU Q, ZHAO J, WANG F, HUO F, LIU H. Selective extraction of vanadium from chromium by pure (C8mim)(PF6): An anion exchange process [J]. Separation & Purification Technology, 2014, 131(1): 94-101.
[23] ZHAO J, HU Q, LI Y, LIU H. Efficient separation of vanadium from chromium by a novel ionic liquid-based synergistic extraction strategy [J]. Chemical Engineering Journal, 2015, 264(3): 487-496.
[24] WANG M, CHEN B, HUANG S, LIU B, GE Q, XIE S. A novel technology for vanadium and chromium recovery from V-Cr-bearing reducing slag [J]. Hydrometallurgy, 2017, 171: 116-122.
[25] CHEN B, HUANG S, LIU B, GE Q, WANG M, WANG X. Separation and recovery of vanadium and chromium from acidic leach solution of V-Cr-bearing reducing slag [J]. Journal of Environmental Chemical Engineering, 2017, 5(5): 4702-4706.
[26] SHANG G, ZHANG G, GAO C, FU W, ZENG L. A novel nanofiltration process for the recovery of vanadium from acid leach solution [J]. Hydrometallurgy, 2014, 142(2): 94-97.
[27] HU Q, PAUDYAL H, ZHAO J, HUO F, INOUE K, LIU H. Adsorptive recovery of vanadium (V) from chromium (VI)- containing effluent by Zr(IV)-loaded orange juice residue [J]. Chemical Engineering Journal, 2014, 248(1): 79-88.
[28] ZHANG Y, ZHENG S L, CHEN X F, QIAO S, WANG X J, FAN B Q. Method for complex separation of chromium and vanadium from chromium-containing vanadium acidic liquid: China, CN107964592 A [P]. 2016-10-19.
[29] FORGHIERI F, GRAZIOSI G, PRETI C, TOSI G. Cyclic substituted dithiocarbamates as ligands: Vanadium (III) and oxovanadium (IV, V) complexes [J]. Transition Metal Chemistry, 1983, 8(6): 372-376.
[30] SAKURAI H, WATANABE H, TAMURA H, YASUI H, MATSUSHITA R, TAKADA J. Insulin-mimetic vanadyl- dithiocarbamate complexes [J]. Inorganic Chimica Acta, 1998, 283(1): 175-183.
[31] SUTRADHAR M, POMBEIRO A J L. Coordination chemistry of non-oxido, oxido and dioxidovanadium (IV/V) complexes with azine fragment ligands [J]. Coordination Chemistry Reviews, 2014, 265(23): 89-124.
[32] WANG Xiao-hua, NIE Ming, TAN Shi-yu. To determine the complexing constants of several alginates, Food Science, 2005, 26(10): 194-195. (in Chinese)
[33] ZHANG Hui. Coordination chemistry: Principles and applications [M]. Beijing, China: Chemical Industry Press, 2009. (in Chinese)
[34] WANG L K, VACCARI D A, LI Y, SHAMMAS N K. Chemical Precipitation [J]. Water Encyclopedia, 2005: 141-155.
[35] CHEN Bian-fang, HUANG Sheng, LIU Biao, GE Qi, XIE Shu-shan, WANG Ming-yu, WANG Xue-wen. Thermodynamic analysis for separation of vanadium and chromium in V(IV)-Cr(III)-H2O system [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(3): 567-573.
[36] ZHOU X, WEI C, LI M, QIU S, LI X. Thermodynamics of vanadium-sulfur-water systems at 298K [J]. Hydrometallurgy, 2011, 106: 104-112.
[37] NING P, LIN X, CAO H, ZHANG Y. Selective extraction and deep separation of V(V) and Cr(VI) in the leaching solution of chromium-bearing vanadium slag with primary amine LK-N21 [J]. Separation & Purification Technology, 2014, 137(11): 109-115.
彭雪枫1,2,张 洋1,范兵强1,郑诗礼1,王晓健1,张 盈1,李 平1,刘丰强3
1. 中国科学院 过程工程研究所,北京 100190;
2. 中国科学院大学 化学工程学院,北京 100049;
3. 攀钢集团有限公司,攀枝花 617067
摘 要:提出一种从酸性溶液中分离回收钒和铬的新方法。该分离方法的原理基于选择性络合,五价钒可以与 二硫代氨基甲酸盐络合反应生成VO(R1R2NCS2)3沉淀,三价铬则留在酸性溶液中;然后通过解络合从VO(R1R2NCS2)3沉淀中回收钒以及再生络合剂,通过中和水解法从三价铬溶液中回收铬。研究络合反应的动力学并计算其表观活化能。工艺优化后钒的沉淀率达到97%,铬损失小于4%,络合剂再生率可达99.5%。该分离方法已成功应用于常规钒生产工业产生的钒铬还原渣的处理,钒以钒酸钙的形式回收,铬以Cr2O3的形式回收,Cr2O3产品纯度超过98%。
关键词:钒;铬;分离;络合;二硫代氨基甲酸盐
(Edited by Xiang-qun LI)
Foundation item: Project (51674230) supported by the National Natural Science Foundation of China; Project (2018YFC1900505) supported by the National Key R&D Program of China
Corresponding author: Yang ZHANG; Tel: +86-10-82544856; E-mail: yangzhang@ipe.ac.cn
DOI: 10.1016/S1003-6326(19)65146-0

