纯钛和镍包埋渗铝的热力学研究
来源期刊:中国有色金属学报(英文版)2013年第6期
论文作者:H. R. KARIMI ZARCHI M. SOLTANIEH M. R. ABOUTALEBI X. GUO
文章页码:1838 - 1846
关键词:包埋渗铝反应;钛;镍;热力学;气相渗铝
Key words:pack aluminizing reactions; titanium; nickel; thermodynamics; gas aluminizing
摘 要:分别在敞开和密闭系统中进行纯钛和纯镍的气相渗铝实验。渗铝实验在1100 °C下进行3 h,包埋粉末含10%Al、5%NH4Cl和85%Al2O3。在敞开条件下,渗铝后纯钛的表面会生成α(Al)固溶体,样品的质量损失达到32.37%。然而,在密闭条件下,纯钛样品的表面生成TiAl3层和α(Al)固溶体,没有质量损失。热力学分析表明,在敞开条件下对纯钛渗铝时,会生成钛的氯化物,导致其质量减少。而在密闭条件下,由于生成的氯化物会达到平衡状态而使生成反应停止。相反,无论是在敞开或密闭条件下,纯镍样品都可以进行表面渗铝,不会导致质量损失。在两种状态下,镍的表面都生成了AlNi和Ni3Al相。
Abstract: Gas aluminizing of pure titanium and nickel samples was carried out in open and closed systems. All the samples were aluminized at 1100 °C for 3 h using pack containing 10%Al+5%NH4Cl+85%Al2O3. Although an α solid solution of Al in Ti was formed on the surface of titanium samples treated in the open set-up, but the samples experienced a mass loss of 32.37%. However, the results of titanium samples treated in the closed set-up showed that a TiAl3 layer and some α solid solution were formed on the surface of these samples without any reduction in mass. Thermodynamic analysis demonstrated that the mass loss of treated titanium samples in the open set-up is due to the continuous formation of titanium chlorides during aluminizing process. Nevertheless, running the experiments in the closed system indicated that the corrosion of titanium samples vanished due to attaining the equilibrium condition. On the contrary, it was observed that nickel samples can be successfully pack aluminized without any mass loss in both open and closed systems. The presence of NiAl and Ni3Al phases on the surface of treated nickel samples was confirmed by XRD and EPMA results in both set-ups.
Trans. Nonferrous Met. Soc. China 23(2013) 1838-1846
H. R. KARIMI ZARCHI1, M. SOLTANIEH1, M. R. ABOUTALEBI1, X. GUO2
1. Centre of Excellence for High Strength Alloys Technology, School of Metallurgy and Materials Engineering, Iran University of Science and Technology, Narmak, Tehran 168463114, Iran;
2. State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, Xi’an 710072, China
Received 24 August 2012; accepted 30 October 2012
Abstract: Gas aluminizing of pure titanium and nickel samples was carried out in open and closed systems. All the samples were aluminized at 1100 °C for 3 h using pack containing 10%Al+5%NH4Cl+85%Al2O3. Although an α solid solution of Al in Ti was formed on the surface of titanium samples treated in the open set-up, but the samples experienced a mass loss of 32.37%. However, the results of titanium samples treated in the closed set-up showed that a TiAl3 layer and some α solid solution were formed on the surface of these samples without any reduction in mass. Thermodynamic analysis demonstrated that the mass loss of treated titanium samples in the open set-up is due to the continuous formation of titanium chlorides during aluminizing process. Nevertheless, running the experiments in the closed system indicated that the corrosion of titanium samples vanished due to attaining the equilibrium condition. On the contrary, it was observed that nickel samples can be successfully pack aluminized without any mass loss in both open and closed systems. The presence of NiAl and Ni3Al phases on the surface of treated nickel samples was confirmed by XRD and EPMA results in both set-ups.
Key words: pack aluminizing reactions; titanium; nickel; thermodynamics; gas aluminizing
1 Introduction
Ni-based and Ti-based alloys have been widely used in high temperature oxidation and hot corrosion environments. Since these alloys are relatively expensive, prolonging their life span has been one of the main goals of researchers in this field. The aluminizing processes have been applied to these alloys for further prolonging their life span in severe corrosive and oxidative conditions. Due to aluminizing treatment, a continuous and dense Al2O3 layer is formed on the surface of these alloys under high-temperature applications. Therefore, the oxygen penetration to the interior of such alloys is restricted. As a consequence, this results in decrease of oxide formation on the surface and hence hinders the high temperature failure [1]. Different methods have been used to produce an aluminide layer on the surface of these alloys. These methods include pack-aluminizing [2,3], magnetron sputtering [4,5], hot dipping [6,7], PVD [8,9], etc.
Pack aluminizing or halide-gas aluminizing is one of the most common aluminizing methods which have been industrially applied for coating of Ni-based [10] and Ti-based [11,12] alloys. Numerous researchers have investigated the effects of coating temperature, coating time, powder mixture compositions, different activators, and substrate compositions on the microstructure and mechanical properties of aluminide layers on these alloys. In addition, some of them have focused on the oxidation and corrosion behavior of the aluminized Ni-based and Ti-based alloys [12-17]. To the best of authors’ knowledge, insufficient data are available in the literature about the effect of open and closed aluminizing set-ups on the phases formed during the process as well as the effect of system types on the thermodynamics of the process. Thus, the present research work was undertaken to investigate the pack aluminizing of pure titanium and nickel samples in both open and closed set-ups. An open system is, hereinafter, assigned to a set-up which is capable of exchanging materials to or from the environment while a closed set-up is referred to one that does not allow any materials to be exchanged between the system and the environment.
2 Experimental
Nickel (99.99% purity, mass fraction) and titanium (99% purity) samples with the dimensions of 16 mm×16 mm×1.5 mm and 20 mm×20 mm×0.6 mm were prepared and used in this work, respectively. The samples were ground up to 1000 grit emery paper, washed with acetone, ultrasonically cleaned in ethanol for 15 min and dried. The dried specimens were then weighed by a balance with the precision of ± 0.001 g before the aluminizing process.
100 g of powder mixture containing 10% Al, 5% NH4Cl, 85% Al2O3 was used as a chloride-gas-generating source for each run in both the open and closed aluminizing set-ups. The powder mixture was used after being mixed up by tumbling in a ball mill for 4 h.
Two different systems were designed and built as illustrated schematically in Fig. 1. In the open set-up (Fig. 1(a)), the high purity argon was purged into the system for 15 min after placing the samples and powder mixture inside the furnace. The temperature of the tube furnace was raised to 1100 °C in 1 h and held for 3 h. The samples were then cooled to ambient temperature in the furnace after the aluminizing time was over. The open set-up was run under a slight positive pressure of argon to ensure a neutral medium inside the system during aluminizing process.
In the closed set-up (Fig. 1(b)), after placing the crucible (containing sample and powder mixture) inside the furnace, it was evacuated to the pressure of 3×10-2 Pa. Similar to the open set-up, the furnace temperature was raised to 1100 °C in 1 h and held in that temperature for 3 h and then cooled to room temperature. In order to decrease the corrosive effects of halide gases on furnace walls, high purity argon was injected to the furnace to increase the furnace pressure to 1.92×104 Pa when the temperature was 350 °C.
After the samples were cooled to ambient temperature, they were weighed by a precise balance. Then they were sectioned and observed with an optical microscope. The thicknesses of the coating layers were measured from optical images using Clemex Vision software. An X-ray diffractometer (Panalytical X’per Pro) with Cu Kα radiation was used to determine the phases in the coatings. Further microstructural and chemical analyses were done using a scanning electron microscope equipped with EDS (EDS Inca X-sight) and an electron probe micro analyzer (EPMA). In addition, thermodynamic analysis was carried out using the data from HSC Chemistry Software version 5.1118. This software contains the thermodynamic data for more than 25000 chemical compounds and is used for various kinds of chemical reactions and equilibria calculations as well as process simulations.
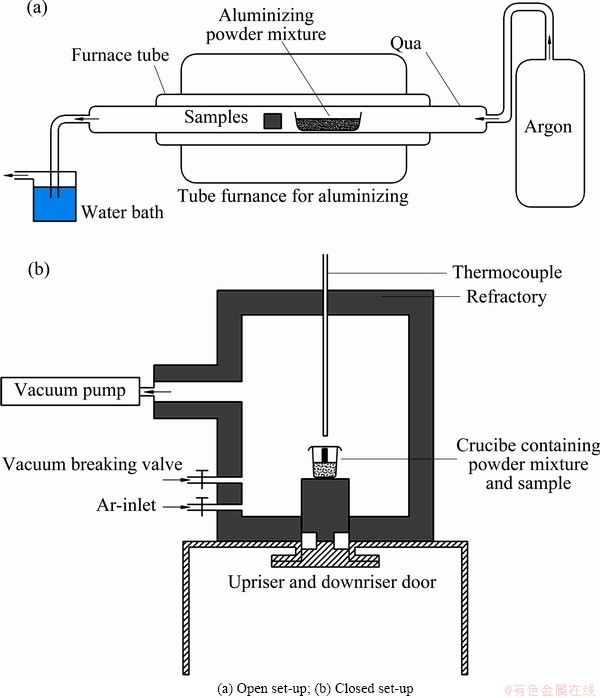
Fig. 1 Schematics of aluminizing systems
3 Results and discussion
3.1 Aluminizing results
The cross-sectional observations of nickel samples treated at 1100 °C for 3 h in both set-ups are shown in Fig. 2. As can be apparently seen, the coatings consist of two layers with different thicknesses. Gravimetric measurements confirmed that nickel samples were successfully aluminized and the mass increases of 1.37% and 1.77% (Table 1) for the samples treated in the open and closed set-ups were obtained, respectively. The mass increase of nickel samples is due to the Al take-up during aluminizing process. XRD patterns (Fig. 3) show that two layers of intermetallic phases, i.e. NiAl (48.2 μm) and Ni3Al (4.7 μm), are formed on the surfaces of nickel samples treated in the open set-up (Fig. 2(a)). The details of EPMA analyses of points 1 to 4 marked on the sample cross section given in Fig. 2 are presented in Table 2. These EPMA results are in accordance with those obtained from XRD patterns. Moreover, the results show that the surfaces of nickel samples treated in the closed set-up are also composed of NiAl (52.7 μm) and Ni3Al (5.2 μm) phases (Fig. 2(b)). Comparison of the EPMA analyses of point 1 with point 5 in Table 2 shows that the surface of nickel samples treated in the closed set-up has a slightly higher Al content than the surface of samples aluminized in the open set-up.
However, the different behavior was observed for aluminizing of titanium samples at 1100 °C for 3 h in the open and closed set-ups. The microstructures of the treated titanium samples are shown in Fig. 4. This figure shows a porous layer on the sample treated in the open system (Fig. 4(b)), while nearly dense layers can be seen on those treated in the closed set-up (Fig. 4(d)). Although XRD patterns (Fig. 5(a)) and EPMA results show that an α(Al) solid solution in Ti is formed on the titanium samples treated in the open set-up, but gravimetric measurements proved that the samples experienced a mass loss of about 32.37% (Table 1). This significant mass loss and the presence of porous layer (Figs. 4(a) and (b)) on the surface of these titanium samples are evidences of corrosion. On the contrary, the results of aluminizing of titanium samples at 1100 °C for 3 h in the closed set-up show a mass increase of almost 10% (Table 1). Further investigations by XRD technique (Fig. 5(b)) and EPMA demonstrate that two rather thick phases including a TiAl3 phase (39.7 μm) and an α(Al) solid solution in Ti (nearly 100 μm) are formed on titanium samples treated in the closed set-up (Fig. 4(c)).

Fig. 2 Cross sectional microstructures of aluminized nickel samples at 1100 °C for 3 h
Table 1 Summary of results of aluminizing samples at 1100 °C for 3 h
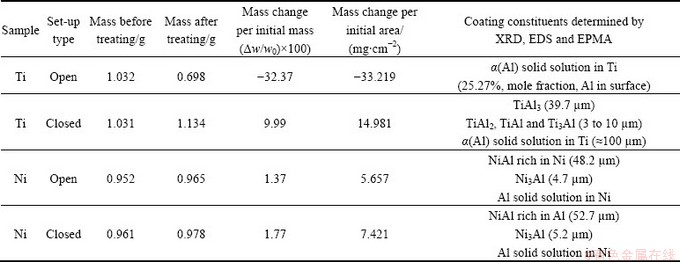
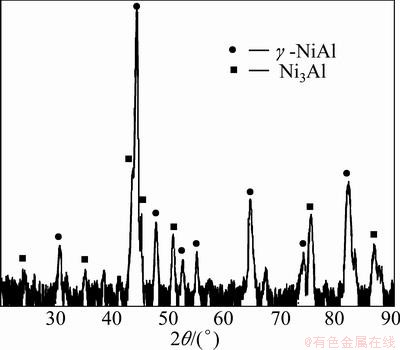
Fig. 3 XRD pattern of nickel sample aluminized at 1100 °C for 3 h in open set-up
Moreover, it is observed that three very thin and uneven layers of TiAl2, γ-TiAl and α2-Ti3Al (altogether 3 to 10 μm) are formed between the TiAl3 phase and α(Al) solid solution layer on these samples (Fig. 4(d)).
3.2 Evaluation of corrosion behavior
The possible independent reactions taking place in the pack aluminizing process of titanium and nickel samples are listed in Table 3 using HSC 5.11 software [18]. The NH4Cl activator is totally decomposed into HCl and NH3 gases (Reaction 1 in Table 3) in the heating step during aluminizing process as pointed out earlier by other researchers [19,20].

Fig. 4 Cross-sectional microstructures of aluminized titanium samples at 1100 °C for 3 h
Table 2 Results of EPMA analysis from points marked in Fig. 2
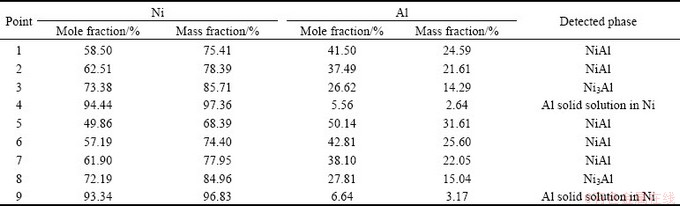
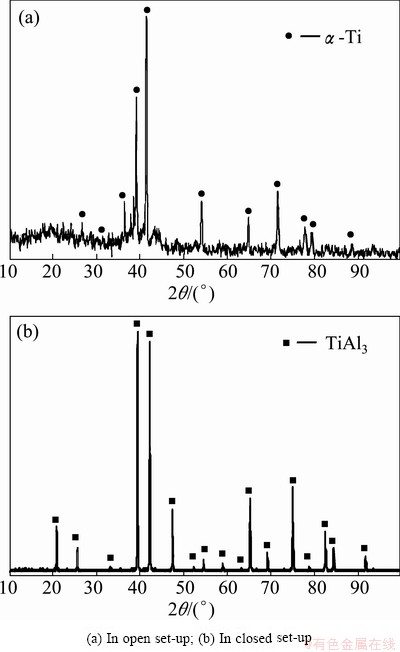
Fig. 5 XRD patterns from surface of titanium samples aluminized at 1100 °C for 3 h
The released HCl gas reacts with aluminum powders in the pack to form various aluminum chlorides (Reactions 7-10 in Table 3), which play the main role in aluminum transfer during the aluminizing process [10,19]. Since the samples are also in contact with the released HCl gas in the aluminizing systems, the reactions between the samples and HCl gas can also occur. These reactions for nickel (Reactions 11-13) and titanium samples (Reactions 14-17) are listed in Table 3. Comparing the standard Gibbs free energy changes for the above mentioned reactions at 1100 °C, it can be concluded that the reactions between HCl and aluminum (Reactions 7-10 in Table 3) are more preferable than those between HCl and nickel (Reactions 11-13 in Table 3). This is due to the positive standard Gibbs free energy changes for the latter reactions. As a result, when nickel samples and aluminum powders are exposed to HCl gas in the aluminizing set-up at 1100 °C, the HCl gas does not have any thermodynamic tendency to react with nickel samples. Consequently, no corrosion and no mass loss are observed in the nickel samples treated at 1100 °C in both the open and closed aluminizing set-ups.
On the contrary, the standard Gibbs free energy changes for reactions between HCl gas and titanium (Reactions 15-17 in Table 3, reaction 14 is an exception) are negative and in the same order of those for reactions between HCl gas and aluminum (Reactions 7-10 in Table 3). Therefore, when pure titanium samples and aluminum powders are exposed to HCl gas in an aluminizing system at 1100 °C, the HCl gas has the same thermodynamic tendency to react with both the titanium samples and aluminum powders to form titanium and aluminum chlorides. It is believed that titanium chloride gases, i.e. TiCl2, TiCl3 and TiCl4, are continuously formed and exhausted from the open set-up during aluminizing process and hence causes the samples to be corroded.
However, in the case of aluminizing in the closed set-up at 1100 °C no corrosion was detected in the titanium samples and nearly 10% mass gain was obtained. To address this phenomenon, it has to be considered that since the set-up is closed and the temperature is high enough, the equilibrium condition is readily established in the closed system. It means that the reactions between HCl gas and titanium samples (Reactions 14-17 in Table 3) will reach equilibrium because the pressures of titanium chlorides are increased gradually due to non-removal from the closed aluminizing set-up. Therefore, it can be implied that, when a closed aluminizing set-up is used, very small amounts of titanium samples are consumed by Reactions 14-17 prior to equilibrium conditions. Thereafter, further corrosion of titanium samples is stopped. A model (section 3.3), which is based on the thermodynamic principles and mass balance calculations, was used to explain this situation. The results obtained from this model showed that the initial mass loss is very small and in the order of 10-6 to 10-9 g depending on several factors such as process temperature, system volume and initial activity of Ti in titanium samples. Clearly, this mass loss of titanium samples is negligible and eventually compensated by the Al take-up during the aluminizing process.
3.3 Mass loss prediction in closed aluminizing set-up
As mentioned in the previous section, a thermodynamic model was developed to predict the mass loss of titanium samples during aluminizing in the closed set-up. At the first stage, this model calculates the partial pressures of gases by using the equations derived from equilibrium conditions of reactions occurring in the system. The reactions and the resulting equations are listed in Table 3. In order to find the equilibrium partial pressures of gases, all the equations have to be solved simultaneously. For solving this set of equations with 15 unknown partial pressures, 15 equations are required. As a consequence, two more equations were derived on the basis of mass balances from the decomposition of NH4Cl activator. The derived equations as well as the details for deriving them are presented in Appendix A.
Table 3 Possible reactions occurring in pack aluminizing of titanium and nickel samples at 1100 °C as well as derived equations on basis of equilibrium conditions

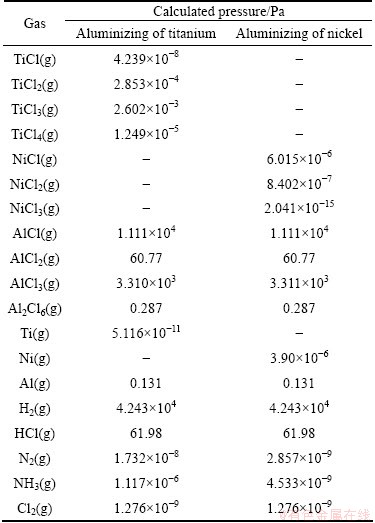
After attaining the required numbers of equations, the set was solved using a semi-analytical method. Table 4 presents the answer for the case of pack aluminizing of titanium samples at temperature 1100 °C. After calculating the partial gaseous pressures, the corroded amounts of titanium samples were determined by using the calculated partial pressures of titanium-containing gases according to Eq. (1):
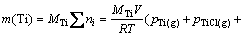
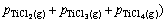 (1)
(1)
In the above equation, m(Ti) is the mass loss of titanium samples (g), MTi the molar mass of titanium (47.87 g), ni the number of titanium moles in different titanium-containing gases, V the set-up volume (50 L), R the gas constant, T the treating temperature (K) and pi is the partial pressure of gas i. The mass loss of titanium samples treated in the closed set-up at 1100 °C was computed from the calculated partial pressures of titanium-containing gases presented in Table 4. The computed value is equal to 6.08×10-7 g. The same procedure was also used for the case of pack aluminizing of nickel samples at 1100 °C in a closed set-up. The calculated partial pressures of gases are also shown in Table 4. The mass loss of nickel samples was computed to equal 2.26×10-9 g based on the calculated nickel-containing gases. It is clearly observed that this value is nearly 270 times smaller than the titanium mass loss at 1100 °C.
Table 4 Calculated gas pressures inside a closed set-up in the case of pack aluminizing of titanium and nickel samples at 1100 °C
Although only the results of aluminizing at 1100 °C were presented and discussed up to now, but the obtained experimental and thermodynamic model results show that the same situation also exists in the temperature range of 800 to 1200 °C (this range is a common temperature range for pack aluminizing of titanium and nickel alloys). Figure 6(a) compares the standard Gibbs free energy changes for reactions between HCl gas, nickel and aluminum in the mentioned temperature range. It is apparent that the reactions between HCl gas and nickel cannot cause corrosion of nickel samples during aluminizing process at this temperature range due to higher and positive standard Gibbs free energy changes. As a result, it can be concluded that for pack aluminizing of pure nickel samples by using NH4Cl activator, only a neutral medium is required in order to prevent the nickel samples and other constituents from oxidation. The type of aluminizing set-up is not a critical and problematic point in pack aluminizing of pure nickel samples.
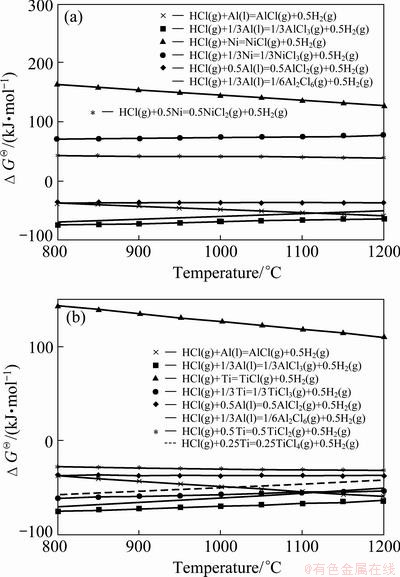
Fig. 6 Comparison of standard Gibbs free energy changes for reactions between HCl gas and nickel (a) and HCl gas and titanium (b) in temperature range of 800-1200 °C
In addition, Fig. 6(b) compares the standard Gibbs free energy changes for reactions between HCl gas, titanium and aluminum in the temperature range of 800 to 1200 °C. It is clear that the reactions between HCl gas and titanium samples can take place simultaneously with reactions between HCl gas and aluminum during aluminizing process. As mentioned earlier, this is because the standard Gibbs free energy changes for reactions between HCl gas and titanium (except for reaction 14 in Table 3) are negative and in the order of those for reactions between HCl gas and aluminum in the temperature range 800 to 1200 °C. This can cause a severe corrosion of pure titanium samples, if they are pack aluminized in an open set-up. As a result, for successful pack aluminizing of pure titanium samples, a closed system must be used or the gases produced during aluminizing process must be circulated in the set-up in the temperature range of 800 to 1200 °C.
4 Conclusions
1) Pure nickel samples can be successfully pack aluminized under neutral atmosphere using either an open or a closed set-up.
2) Pack aluminizing of pure nickel samples using a powder mixture of 5%NH4Cl+10%Al+85%Al2O3 at 1100 °C for 3 h resulted in the formation of a coating layer containing NiAl and Ni3Al intermetallic phases.
3) In order to successfully aluminize pure titanium samples, the aluminizing system has to be closed.
4) Pack aluminizing of pure titanium samples using 5%NH4Cl+10%Al+85%Al2O3 powder mixture at 1100 °C for 3 h in a closed set-up resulted in the formation of a coating layer mainly composed of TiAl3 intermetallic phase and an α(Al) solid solution in titanium. Three very thin layers of TiAl2, TiAl and Ti3Al were observed between the TiAl3 and solid solution phases.
Appendix A
Two equations were derived on the basis of mass balance for activator decomposition. It is clear that the hydrogen resulting from NH4Cl activator decomposition would be consumed by hydrogen-containing materials such as NH3, H2 and HCl gases (Eq. (A1)):
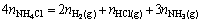 (A1)
(A1)
where ni is the mole number of species i. Since the equations derived from equilibrium conditions contain partial pressures of gases, Eq. (A1) should be converted to an equation containing partial gaseous pressures. This can be done by assuming the ideal behavior for gaseous species.
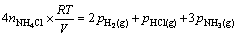 (A2)
(A2)
where V is the set-up volume (50 L), R is the gas constant, T is the treating temperature (K) and pi is the partial pressure of gas i. The same logic is also used to derive the equation for chlorine mass balance, i.e. the chlorine resulting from NH4Cl activator decomposition would be consumed in chlorine-containing materials.

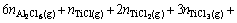
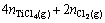 (A3)
(A3)
Similar to the previous case, the above equation can be converted to Eq. (A4) by assuming the ideal behavior for gaseous species:
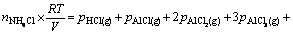
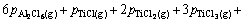
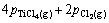 (A4)
(A4)
The equations (A2) and (A4) were used for completing the set of equations.
References
[1] ASM Handbook. Properties and selection: Nonferrous alloys and special-purpose materials [M]. ASM International, 2004.
[2] GORAL M, SWADZBA L, MOSKAL G, JARCZYK G, AGUILAR J. Diffusion aluminide coatings for TiAl intermetallic turbine blades [J]. Intermetallics, 2011, 19(5): 744-747.
[3] LU T, YAO D Z, ZHOU C G. Low-temperature formation of aluminide coatings on Ni-base superalloys by pack cementation process [J]. Chinese Journal of Aeronautics, 2010, 23(3): 381-385.
[4] GARBACZ H, WIECINSKI P, ADAMCZYK-CIESLAK B, MIZERA J, KURZYDLOWSKI K J. Studies of aluminium coatings deposited by vacuum evaporation and magnetron sputtering [J]. Journal of Microscopy, 2010, 237(3): 475-480.
[5] RAMOS A S, VIEIRA M T. Kinetics of the thin films transformation Ti-Al multilayer→γ-TiAl [J]. Surface and Coatings Technology, 2005, 200(1-4): 326-329.
[6] CAMMAROTA G P, CASAGRANDE A, SAMBOGNA G. Effect of Ni, Si and Cr in the structural formation of diffusion aluminide coatings on commercial-purity titanium [J]. Surface and Coatings Technology, 2006, 201(1-2): 230-242.
[7] ZHANG Z G, TENG X, MAO Y L, CAO C X, WANG S J, WANG L. Improvement of oxidation resistance of γ-TiAl at 900 and 1000 °C through hot-dip aluminizing [J]. Oxidation of Metals, 2010, 73(3-4): 455-466.
[8] WU F, MURAKAMI H, SUZUKI A. Development of an iridium-tantalum modified aluminide coating as a diffusion barrier on nickel-base single crystal superalloy TMS-75 [J]. Surface and Coatings Technology, 2003, 168(1): 62-69.
[9] GUO C, ZHOU J, ZHAO J, WANG L, YU Y, CHEN J, ZHOU H. Improvement of the oxidation and wear resistance of pure Ti by laser-cladding Ti3Al coating at elevated temperature [J]. Tribology Letters, 2011, 42(2): 151-159.
[10] SQUILLACE A, BONETTI R, ARCHER N J, YEATMAN J A. The control of the composition and structure of aluminide layers formed by vapour aluminising [J]. Surface and Coatings Technology, 1999, 120-121: 118-123.
[11] ZHOU C, XU H, GONG S, KIM K Y. A study of aluminide coatings on TiAl alloys by the pack cementation method [J]. Materials Science and Engineering A, 2003, 341(1-2): 169-173.
[12] SUBRAHMANYAM J. Cyclic oxidation of aluminized Ti-14Al-24Nb alloy [J]. Journal of Materials Science, 1988, 23(6): 1906-1910.
[13] YUAN B, YU L, LU G. Oxidation resistance of low-temperature pack aluminizing coatings on Ni-base superalloy [J]. International Journal of Modern Physics B, 2010, 24(15-16): 3185-3189.
[14] ZHAN Z, LIU Z, LIU J, LI L, LI Z, LIAO P. Microstructure and high-temperature corrosion behaviors of aluminide coatings by low-temperature pack aluminizing process [J]. Applied Surface Science, 2010, 256(12): 3874-3879.
[15] LEE J W, KUO Y C. A study on the microstructure and cyclic oxidation behavior of the pack aluminized Hastelloy X at 1100 °C [J]. Surface and Coatings Technology, 2006, 201(7): 3867-3871.
[16] JUNG H G, JUNG D J, KIM K Y. Effect of Cr addition on the properties of aluminide coating layers formed on TiAl alloys [J]. Surface and Coatings Technology, 2002, 154(1): 75-81.
[17] GURRAPPA I. Effect of aluminizing on the oxidation behavior of the titanium alloy, IMI 834 [J]. Oxidation of Metals, 2001, 56(1-2): 73-87.
[18] Outotec Research Oy Center: HSC Chemistry Software Version 5.11 [M]. Finland, 2002.
[19] KUNG S C, RAPP R A. Analyses of the gaseous species in halide-activated cementation coating packs [J]. Oxidation of Metals, 1989, 32(1-2): 89-109.
[20] GEIB F D, RAPP R A. Simultaneous chromizing-Aluminizing coating of low-alloy steels by a halide-activated, pack-cementation process [J]. Oxidation of Metals, 1993, 40(3-4): 213-228.
H. R. KARIMI ZARCHI1, M. SOLTANIEH1, M. R. ABOUTALEBI1, X. GUO2
1. Centre of Excellence for High Strength Alloys Technology, School of Metallurgy and Materials Engineering, Iran University of Science and Technology, Narmak, Tehran 168463114, Iran;
2. State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, Xi’an 710072, China
摘 要:分别在敞开和密闭系统中进行纯钛和纯镍的气相渗铝实验。渗铝实验在1100 °C下进行3 h,包埋粉末含10%Al、5%NH4Cl和85%Al2O3。在敞开条件下,渗铝后纯钛的表面会生成α(Al)固溶体,样品的质量损失达到32.37%。然而,在密闭条件下,纯钛样品的表面生成TiAl3层和α(Al)固溶体,没有质量损失。热力学分析表明,在敞开条件下对纯钛渗铝时,会生成钛的氯化物,导致其质量减少。而在密闭条件下,由于生成的氯化物会达到平衡状态而使生成反应停止。相反,无论是在敞开或密闭条件下,纯镍样品都可以进行表面渗铝,不会导致质量损失。在两种状态下,镍的表面都生成了AlNi和Ni3Al相。
关键词:包埋渗铝反应;钛;镍;热力学;气相渗铝
(Edited by Sai-qian YUAN)
Corresponding author: H. R. KARIMI ZARCHI; Tel: +98-21-77240450; Fax: +98-21-77240480; E-mail: karimizarchi@iust.ac.ir
DOI: 10.1016/S1003-6326(13)62668-0

