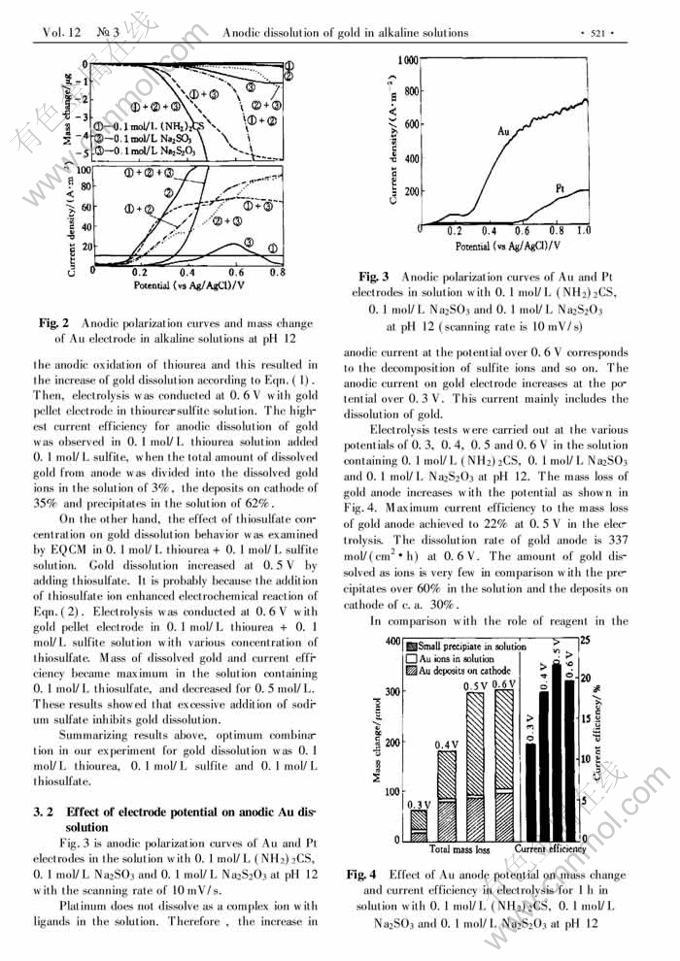
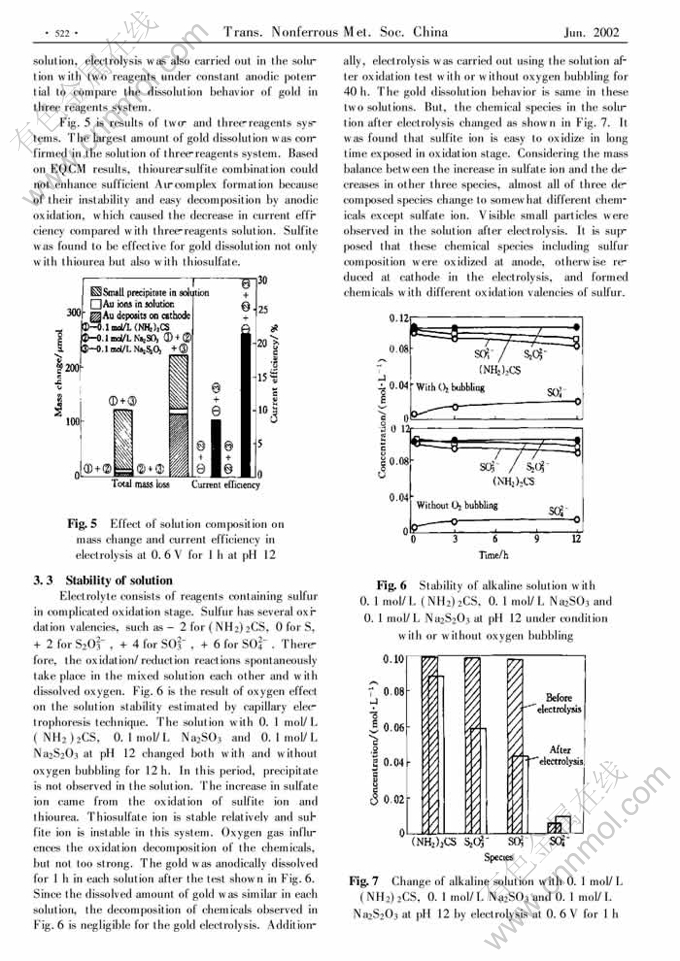
Anodic dissolution of gold in alkaline solutions containing thiourea, thiosulfate and sulfite ions
来源期刊:中国有色金属学报(英文版)2002年第3期
论文作者:M.Okido M.Ishikawa 柴立元
文章页码:519 - 523
Key words:gold selective dissolution; sulfite; alkaline thiourea solution
Abstract: Gold dissolves electrochemically in alkaline solutions containing ligands to form complex ions with gold ion. Therefore, selective leaching of noble metals is expected without dissolution of base metals such as steels, aluminum alloys in scrap treatment. Gold electrodes were investigated using linear sweep voltammetry, EQCM method and potentiostatic electrolysis in alkaline solutions containing thiourea, Na2SO3 and Na2S2O3. The solution composition, electrode potential affect gold dissolution rate and current efficiency. The gold dissolved from anode electrode forms complex ions, suspension particles as compound precipitates and deposits on cathode electrode as a metal. Anodic efficiency for gold dissolution is between 10% and 22%. This is caused by the oxidation decomposition of sulfite ions and thiourea. The stability of t he alkaline solution containing these elements was also estimated by capillary electrophoresis technique.
基金信息:the National Natural Science Foundation of China