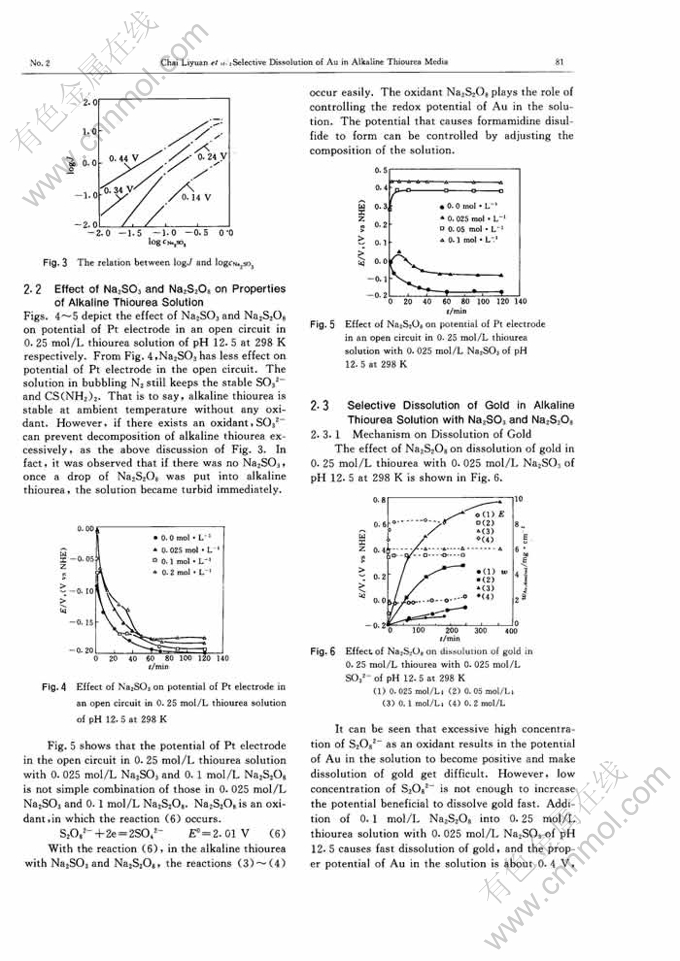
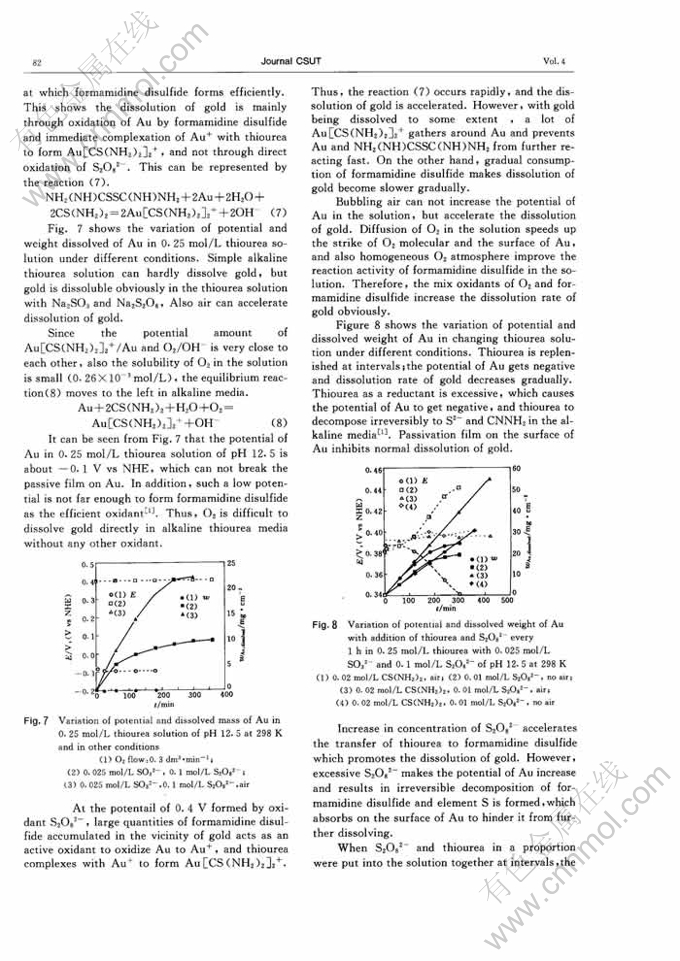
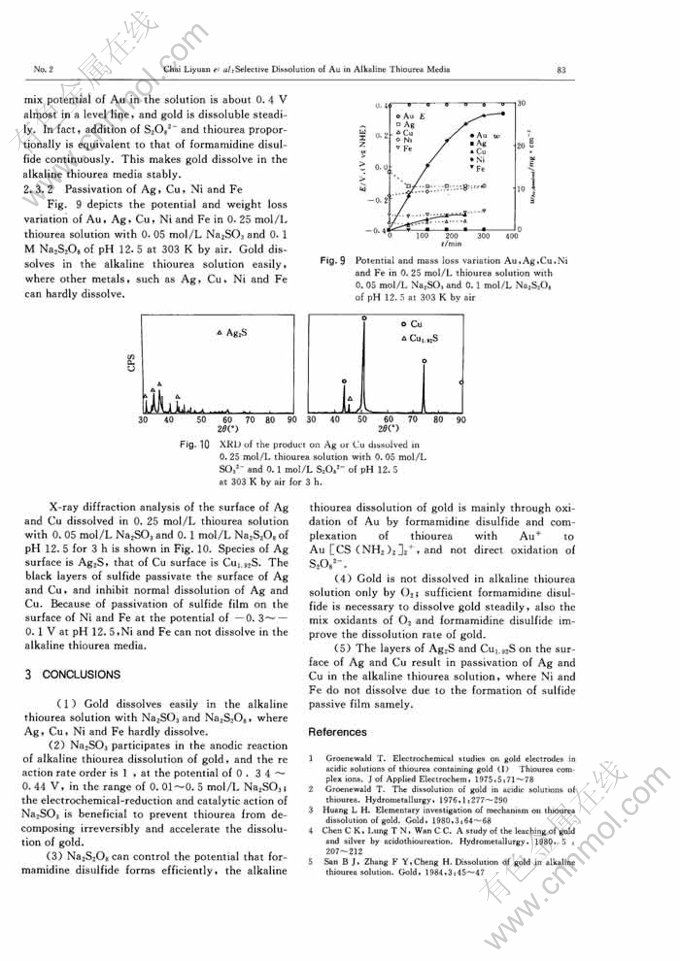
STUDY ON SELECTIVE DISSOLUTION OF GOLD IN ALKALINE THIOUREA MEDIA
来源期刊:中南大学学报(英文版)1997年第2期
论文作者:Chai Liyuan Zhong Haiyun Zhang Chuanfu Masazumi Okido Ryouichi Ichino
文章页码:79 - 83
Key words:mechanism; selective dissolution of gold; alkaline thiourea; electrochemical reduction and catalytic action; formamidine disulfide; mix potential
Abstract: Gold dissolves easily in an alkaline thiourea solution with Na2SO3 and Na2S2O8, where the accompanied elements of Au, such as Ag, Cu, Ni and Fe can hardly dissolve. It was considered that electrochemical reduction and catalytic action of SO32- prevent thiourea from decomposing irreversibly and accelerates dissolution of gold.The S2O82- as an oxidant can control the potential so that formamidine disulfide may form efficiently. The dissolution of gold is mainly through oxidation of Au by formamidine disulfide and complexation of thiourea with An+,and not direct oxidation of S2O82-. Thus,sufficient formamidine disulfide is necessary to accelerate the dissolution of gold. The reason for Ag, Cu hardly being dissoluble is that the black layers of Ag2S and Cu1.92S form on the surface of Ag and Cu. The metal Ni and Fe do not dissolve in the alkaline media due to their sulfide films passivation samely.