
Phase composition and structure of grain boundary of
oversintered Y3Al5O12 ceramics
LI Chang-qing(李长青), ZUO Hong-bo(左洪波), HAN Jie-cai(韩杰才),
ZHANG Ming-fu(张明福), MENG Song-he(孟松鹤), YAO Tai(姚 泰)
Center for Composite Materials, Harbin Institute of Technology, Harbin 150008, China
Received 28 July 2006; accepted 15 September 2006
Abstract: Phase composition and microstructures of grain boundary of oversintered yttrium aluminum garnet (Y3Al5O12, YAG) ceramics by vacuum sintering at 1 850 ℃ were investigated. For synthesizing YAG, grain boundary is a key factor for YAG ceramics. The morphology of grain boundary was observed by SEM, TEM and its composition was analyzed by EDS. It is identified that the grain boundary is composed of α-Al2O3 and yttrium aluminum perovskite (YAP, YAlO3) eutectics. At the edge of YAG crystal grain, YAG phase is decomposed into perovskite YAP and α-Al2O3 during high temperature sintering. Due to refractive indexes of YAP and α-Al2O3 phases in wide grain boundary are different from those of YAG, the transmittance of oversintered YAG ceramics is lower than that of YAG ceramics sintered at 1 750 ℃.
Key words: YAG; eutectics; YAP; grain boundary; α-Al2O3
1 Introduction
Many researchers have studied Y2O3-Al2O3 system for long time in high-temperature ceramic applications. MAH and PETRY[1] studied Y2O3-Al2O3 phase diagram, and found that three intermediate compounds such as Y3Al5O12, YAlO3 and Y4Al2O9 existed within the Y2O3- Al2O3 system. Crystalline Y4Al2O9 with a monoclinic structure is referred as YAM. Crystalline YAlO3, yttrium aluminum perovskite, or YAP has orthorhombic and hexagonal polymorphs[2]. MAH et al[3] reported the thermal stability of a series of compounds in Y2O3-Al2O3 system based on vacuum and air heat treatment results and YAlO3 phase is stable at 1 650 ℃ in air and under vacuum. Crystalline Y3Al5O12, commonly known as “Yttrium Aluminum Garnet” or YAG, is widely used in laser as host materials, or as a refractory coating, and normally exists in a cubic form with garnet structure. Single crystal YAG is highly creep resistant and is the best known oxide for creep resistance[4].
For growing YAG garnet crystals at high temperature, there exists one of the difficulties associated with the deviation of the melt composition from stoichiometry of the Y3Al5O12 garnet for different melting points of Al2O3 and Y2O3[5-6]. Transparent YAG ceramics is easy to make compared to single YAG crystal, so many scientists devoted to fabricate transparent YAG ceramics[7-11]. For ceramics, grain boundary is a sensitive factor as some papers report [12-14], but there is few reports[15] about oversintered YAG ceramics grain boundary microstructure and composition that affect the ceramics transmittance.
In this study, stoichiometric Y2O3-Al2O3 powders are mixed together and sintered into YAG ceramics under vacuum at 1 850 ℃. The ceramics was compared with YAG ceramics sintered under vacuum at 1 750 ℃.
2 Experimental
In the present work, aggregated Al2O3 and Y2O3 powders with average grain size of about 4 mm were used as starting materials. These starting powders were weighed so that the chemical one would become stoichiometric composition of designed compound. α(Al) and yttria powders were mixed with tetraethyl silicate (0.5% mixed powder in ethyl alcohol ) and the mixture was milled in analytical ethanol with milling balls (steel ball covered with nylon) for 12 h. The milled slurry was dried, and the dried powders were calcined at different temperatures in air for 4 h. The synthesized powder was analyzed by XRD. Then the calcined powder was uniaxially pressed into pellets of 13 mm in diameter at 250 MPa, and the pellets were sintered at 1 850 ℃ for 4 h under vacuum of 7×10-3Pa in the tungsten mesh furnace. Disk specimens were polished on both sides and used for measuring optical transmittance. Grain and grain boundary morphologies of oversintered YAG ceramics were observed by SEM and TEM, and its composition was analyzed by EDS.
3 Results and discussion
Fig.1(a) shows X-ray diffraction pattern of the mixed powder as-milled. No reaction occurs between the two components in mixture, yttria peaks are much stronger than those of α-alumina, and α-alumina in the mixture remains crystalline. The stability of the crystal structure of the materials depends on their inherent nature. Yttria acts as soft materials, while α-Al2O3 acts as hard materials during milling, so yttria is not easy to grind. Yttria peaks are relatively weak than those of α- Al2O3[16], but both of them do not transform into amorphous phase.
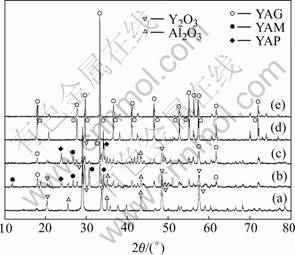
Fig.1 X-ray diffraction patterns of mixed powders calcined at different temperatures: (a) As-milled; (b) 1 300 ℃; (c) 1 400 ℃; (d) 1 500 ℃; (e) 1 850 ℃
In the case of the conventional solid state synthesis, YAG phase is generated by a series of reactions as follows: 1) 2Y2O3+Al2O3→Y4A12O9(YAM); 2) Y4Al2O9+Al2O3→ 4YAlO3(YAP) and 3) 3YA1O3+Al2O3→Y3Al5O12(YAG). The yttrium-rich YAM phase is initially generated in spite of the aluminum-rich stoichiometry, indicating that the reaction is proceeded by the diffusion of Al into Y2O3 grains. The diffusion rate of Al in Y2O3 seems to be higher than that of Y in Al2O3. The phase generation reflects the local composition of the heterogeneous mixture[17].
As Fig.1 shows, there exist YAG, YAlO3(YAP), Y4Al2O9(YAM), Y2O3 and α-Al2O3 phases in the mixed powder calcined at 1 300 ℃. And other X-ray diffraction pattern indicates that powders calcined at 1 500 ℃ are composed of YAG phase. The ceramic specimen calcined at 1 850 ℃ is almost YAG, which indicates that polycrystalline YAG at this temperature remains stable.
Fig.2 shows SEM images of as-milled mixture powder and powder calcined at 1 400 ℃. As Fig.2(a) shows, the powders consist of submicrometer particles under the effect of high energy milling. Most of large particles are Y2O3 phase and small grains are Al2O3 phase and they agglomerate weakly together, maybe due to highly defective energy for high energy milling. This phenomenon is further confirmed by XRD in Fig.1. Fig.2(b) shows that the calcined powder grows much larger in dimension and more regular in shape than as-milled powder. The calcined powder particle size distribution is suitable that is demonstrated to a material with a high green density easily[18].

Fig.2 SEM micrographs of powders: (a) As-milled mixture powder; (b) Calcined at 1 400 ℃
For synthesis of YAG ceramics, Y2O3 and α-Al2O3 contents in YAG should not deviate stoichiometric composition by 2%[19]. But during high temperature sintering, for different melting points of Y2O3 and α-Al2O3, optimum amounts of these two contents will be different, and it is very difficult to control precise stoichiometric Y2O3, α-Al2O3 in YAG. Fig.3 shows specimen A sintered at 1 750 ℃ and specimen D sintered at 1 850 ℃. Fig.4 indicates the transmittance curves of specimen A and specimen D ranging from 190 nm to 900 nm, and it clearly shows the transmittance of specimen D is much lower than that of specimen A.

Fig.3 Appearance of specimens
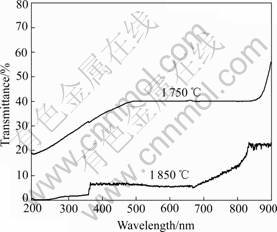
Fig.4 Transmittance curves of YAG ceramics
Fig.5 shows the grain boundary of oversintered YAG ceramics observed by SEM. As can be seen from Fig.5(a), the grain size of specimen D is so larger even more than 150 mm, and some grain boundary is wider over 100 mm. Fig.5(b) shows that grain boundary is melted during high temperature stage for its melting point is lower than that of crystal grain. Fig.6 shows TEM image of submicrometer-sized domains of YAG grain boundary.
The chemical composition of the grain boundary is composed of eutectic type of structure such as YAP phase and α- Al2O3 phase tested by energy-dispersive spectrometry(EDS) analysis in Fig.7. Although hexagonal YAP is not obtained in the solid-state reaction [17], where YAP phase must be perovskite structure. In spot B, Cu peaks originate from the sample holder and the grid. The result of grain boundary in this specimen is similar with COCKAYNE et al[20], WANG et al[21], LIN et al[22] and PRADHAN et al[23] who have confirmed some pores in grain boundary.
YAG grain boundary energy is much higher than that of crystal grain, and it can make YAG phase in the grain boundary decompose into YAP and α- Al2O3, as expressed by the following reaction, Y3Al5O12→3YAlO3+Al2O3. From Fig.5, grain boundary is melted during high temperature stage. Since YAG crystal unit cell contains 160 atoms and YAP unit cell contains only 20 atoms, YAG crystal is suggested to possess a higher nucleation energy than that of perovskite structure of YAP and creation of YAP structure as a nucleus is energetically more favorable in the melt, which indicates by above experimental results that metastable phase YAP can be precipitated from the melt with temperature much lower than the melting point of YAG[21]. The interface characteristics of the specimen shown in Fig.6 may imply that the eutectic nucleation of YAG occurs at the solid/liquid interfaces during the slow solidification process.

Fig.5 SEM micrographs of grain boundary in specimen D: (a) Grain boundary morphology; (b) Magnified grain boundary
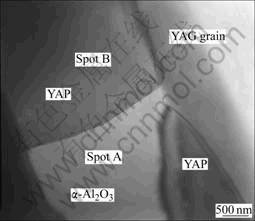
Fig.6 TEM micrograph of grain boundary
Perhaps even YAG crystal grains near the grain boundary are decomposed and grain boundary is much wider than normal. Since Al2O3 refractive index (ne=1.760, no=1.768) and YAP refractive index (1.96) are different from those of YAG(refractive index is 1.815), grain boundary width is larger than normal. This would make the transmittance of the high temperature sintered ceramics such as specimen D lower than that of specimen A. There exist a few second phase particles in crystal grain which would act as scattering centers and make the transmittance of the specimen D lower.
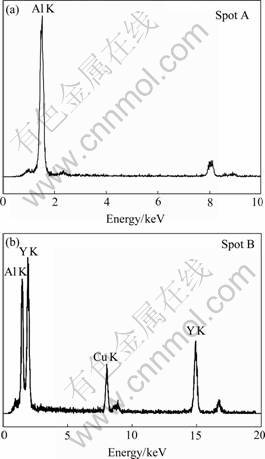
Fig.7 EDS patterns of different spots of YAG grain boundary
4 Conclusions
At higher sintering temperature such as 1 850 ℃, the grain size of YAG ceramics grows unusually large and grain boundary grows much wide. At the edge of the boundaries, YAG decomposes into perovskite structure YAP and α-Al2O3, which results in decreased ceramics transmittance.
References
[1] MAH T I, PETRY M D. Eutectic composition in the pseudobinary of Y4Al2O9 and Y2O3 [J]. J Am Ceram Soc, 1992, 75(7): 2006-2009.
[2] HESS N J, MAUPIN G D, CHICK L A, SUNBERG D S, MCCREEDY D E, ARMSTRONG T R. Synthesis and crystallization of yttrium-aluminum garnet and related compounds [J]. Journal of Materials Science, 1994, 29(7): 1873-1878.
[3] MAH T I, KELLER K A, SAMBASIVAN S, KERANS R J. High-temperature environmental stability of the compounds in Al2O3-Y2O3 system [J]. J Am Ceram Soc, 1997, 80(4): 874-878.
[4] PARTHASARATHY T A, MAH T I, KELLER K. Creep mechanism of polycrystalline yttrium aluminum garnet [J]. J Am Ceram Soc, 1992, 73(7): 756-759.
[5] SOBOLEVA L V, CHIRKIN A P.Y2O3-Al2O3-Nd2O3 Phase diagram and the growth of (Y, Nd)3Al5O12 single crystals [J]. Crystallography Reports, 2003, 48(5): 883-887.
[6] KAZAKOVA Y L I, KUZ’MICHEVA G M, SUCHKOVA E M. Growth of Y3Al5O12 crystals for jewelry [J]. Inorganic Materials, 2003, 39(9): 959-970.
[7] DE WITH G, VAN DIJK H J A. Translucent Y3Al5O12 ceramics [J]. Mater Res Bull, 1984, 29(12): 1669-1674.
[8] AKIO I, ISAO F, KIICHIRO K. Fabrication of polycrystalline, transparent YAG ceramics by a solid-state reaction method [J]. J Am Ceram Soc, 1995, 78(1): 225-228.
[9] WANG Hong-zhi, GAO Lian, HIROKAZU K, TOHRU S, KOICHI N. Preparation and mechanical properties of polycrystalline yttrium aluminum garnet [J]. Journal of the Chinese Ceramics Society, 2001, 29(1): 35-38.
[10] LI J G, IKEGAMI V, LEE J H. Low-temperature fabrication of transparent yttrium aluminum garnet (YAG) ceramics without additives [J]. J Am Ceram Soc, 2000, 83(4): 961-963.
[11] WEN Lei, SUN Xu-dong, XIU Zhi-meng, CHEN Shao-wei, TSAI C T. Synthesis of nanocrystalline yttria powder and fabrication of transparent YAG ceramics [J]. Journal of the European Ceramics Society, 2004, 24(9): 2681-2688.
[12] SEKITA M, HANEDA H, SHIRASAKI S, YANAGITANI T. Optical spectra of undoped and rare-earth-(Pr, Nd, Eu, and Er) doped transparent ceramics Y3Al5O12 [J]. J Appl Phys, 1991, 69(6): 3709-3718.
[13] SAKAGUCHI I, HANEDA H, TANAKA J, YANAGITANI T. Effect of Composition on the Oxygen tracer diffusion in transparent yttrium aluminium garnet(YAG) ceramics [J]. J Am Ceram Soc, 1996, 79(6): 1627-1632.
[14] IKESUE A, YOSHIDA K, YAMAMOTO T, YAMAGA I. Optical Scattering centers in polycrystalline Nd:YAG laser [J]. J Am Ceram Soc, 1997, 80(6): 1517-1522.
[15] YASUDA H, OHNAKA I, MIZUTANI Y, WAKU Y. Selection of eutectic systems in Al2O3-Y2O3 ceramics [J]. Science and Technology of Advanced Materials, 2001, 2(1): 67-71.
[16] IKESUE A, KAMATA K. Microstructure and optical properties of hot isostatically pressed Nd:YAG ceramics [J]. J Am Cera Soc, 1996, 79(7): 1927-1933.
[17] MATSUBARA I, PARANTHAMAN M, ALLISON S W, CATES M R, BESHEARS D L, HOLCOMB D E. Preparation of Cr-doped Y3Al5O12 phosphors by heterogeneous precipitation methods and their luminescent properties [J]. Materials Research Bulletin, 2000, 35(2): 217-224.
[18] AYRAL A, PHALIPPOU J. Submicrometer alumina powders [J]. Advanced Ceramic Materials, 1988, 3(6): 575-579.
[19] KUKLJA M M. Defects in yttrium aluminium perovskite and garnet crystals: atomistic study [J]. J Phys: Condens Matter, 2000, 12(13): 2953-2967.
[20] COCKYNE B, LENT B. A complexity in the solidification behaviour of molten Y3Al5O12 [J]. Journal of Crystal Growth, 1979, 46(3): 371-378.
[21] WANG S, YAMAMOTO F, AKATSU T, TANABE Y, YASUDA E. Metastable precipitation of YAlO3 in isothermally solidified YAG/(Al2O3-rich) spinel composites [J]. Journal of Materials Science, 1998, 33(21): 5157-5162.
[22] LIN I C, NAVROTSKY A, WEBER J K R, NORDINE P C. Thermodynamics of glass formation and metastable solidification of molten Y3Al5O12 [J]. Journal of Non-Crystalline Solids, 1999, 243: 273-276.
[23] PRADHAN A K, ZHANG Kai, LOUTS G B. Synthesis of neodymium-doped yttrium aluminum garnet (YAG) nanocrystalline powders leading to transparent ceramics [J]. Material Research Bulletin, 2004, 39(9): 1291-1298.
(Edited by YANG Bing)
Foundation item: Project(41312040404) supported by the National Defence “15” Pre-Research Foundation of China
Corresponding author: LI Chang-qing; Tel: +86-451-86402310; E-mail: plumevergreen@163.com