
Removal of boron from metallurgical grade silicon by
electromagnetic induction slag melting
LUO Da-wei1, LIU Ning1, LU Yi-ping1, ZHANG Guo-liang1, LI Ting-ju1, 2
1. School of Materials Science and Engineering, Dalian University of Technology, Dalian 116024, China;
2. Key Laboratory of Materials Modification by Laser, Ion and Electron Beams, Ministry of Education,
Dalian University of Technology, Dalian 116024, China
Received 16 August 2010; accepted 5 January 2011
Abstract: A new purification process was developed to remove impurities in metallurgical grade silicon (MG-Si) by electromagnetic induction slag melting (EISM). Vacuum melting furnace was used to purify boron in different slag systems. The results show that the removal effect in SiO2-CaO-Al2O3 systems is better than that in other slag systems by EISM. The boron content in MG-Si is successfully reduced from 1.5×10-5 to 0.2×10-5 during EISM at 1 823 K for 2 h. Meanwhile, Al, Ca and Mg elements in MG-Si are also well removed and their removal efficiencies reach 85.0%, 50.2% and 66.7%, respectively, which indicates that EISM is very effective to remove boron and metal impurities in silicon.
Key words: metallurgical grade silicon; slag system; partition ratio
1 Introduction
The production of solar cells increased rapidly in recent years. The market which uses the scrap of semiconductor grade silicon (SEG-Si) is difficult to afford a steady supply of this material. Therefore, alternative production processes are needed to increase the feedstock. The refinement of metallurgical grade silicon (MG-Si) is always desirable[1-2]; however, the removal of boron is still a challenge during refining. It is very difficult to vaporize boron by vacuum process because its vapor pressure is very low compared with that of silicon (10-4 Pa for boron and 10-1 Pa for silicon)[3-4]. Another difficulty regarding boron extraction is that its segregation coefficient in silicon is 0.8 which results in the fact that there is no removal effect by unidirectional solidification process[5-7]. The process usually used for the boron removal is plasma melting in an oxidizing atmosphere (O2, CO2 or H2O), during which boron is transformed into oxide forms, increasing its vapor pressure[8-9]. However, the plasma equipment and operation require a large initial investment.
Slag refining is an option for the removal of boron from molten silicon, and effective removal may be obtained for a certain slag composition. The technical viability of the slag refining process is confirmed, but the result is based on laboratory scale, and the refining time in static theoretical research is very long. The value of partition ratio (LB) obtained from experiments varies between 1.5 and 2.5[10-15]. The partition ratios of boron between SiO2-CaO-Na2O or SiO2-CaO-Al2O3 slag systems and liquid Si melt at 1 823 K were simulated using an assessed thermochemical databank together with the FACT oxide thermodynamic database by BALE et al[16]. The calculated values are approximately two times higher than the experimental ones, which indicates that the reaction kinetic barriers might play important roles in the refining processes. In the present study, pilot scale and dynamic slag experiments were carried out by using electromagnetic induction slag melting (EISM). The LB value between the slag systems and silicon phases was measured and the metallic impurities removal effects were analyzed. The mechanism of boron removal from silicon under EISM was discussed. Additionally, a detailed analysis for the thermodynamics and kinetics of boron removal were carried out. Impurity contents and microanalysis were confirmed by inductively coupled plasma atomic emission spectrometer (ICP-AES) and electron probe microanalysis (EPMA).
2 Experimental
The schematic diagram of experimental devices is shown in Fig.1. An inductive furnace with 200 kW and 3 000 Hz was used for melting. The silicon was loaded in a quartz ceramic crucible with a size of d120 mm×220 mm. The crucible was surrounded by a graphite heater which was not a conductor at room temperature. The induction melting of silicon was carried out through water-cooled copper coil under protective gas. Three slag systems were chosen according to previous studies and the mass ratios of CaO/SiO2 in all slag systems were 1.21. Silicon material used in this study was treated by acid leaching. Metal impurities in silicon were reduced significantly, while non-metallic impurities such as B and P did not change after acid treatment. Silicon of 3 kg and slag of 0.3 kg were used in each experiment. The sample was withdrawn from the furnace after the test. Both the silicon phase and the slag phase were analyzed using ICP-AES to quantify boron and metal impurities contents. The partition ratio of boron between the slag and the silicon phase was defined as:
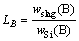 (1)
(1)
where wslag(B) and wSi(B) are the concentration of boron in slag and in silicon, respectively. The experimental conditions and parameters are listed in Table 1.
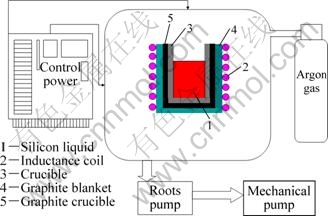
Fig.1 Schematic diagram of experimental devices
3 Results
3.1 Effect of slag system on boron removal
Four types of typical slag systems were chosen for comparative study according to previous research [11-14]. The photograph of silicon ingots after test is shown in Fig.2. From Fig.2(a) it could be found that most of slag freezes at the bottom of silicon ingots after test and a small amount covers the side of silicon ingot. Slag deposits to the bottom of the crucible and then solidifies because the density of slag is greater than that of silicon melt. Thus, the purified silicon and the slag could be separated very well. Sodium hydroxide solution with concentration of 20% (mass fraction) is used to remove the surface slag of ingot and then the silicon ingots was cut off from the center. From Fig.2(b) it could be found that the internal silicon ingot is very bright and there is no slag, which further illustrates that the slag and silicon melt could be separated very well.
Table l Experimental conditions in slag treatment

Fig.2 Photos of silicon ingot after electromagnetic induction slag melting
The LB values with different types of slag after melting at 1 773 K for 1 h are shown in Fig.3. It could be seen that boron removal is significantly improved by EISM compared to previous static test results[10-12]. Although CaO/SiO2 ratios are the same in different slag systems, the values of LB are very different by adding different oxides to the CaO/SiO2 slag system. The values of LB are significantly reduced by adding CaF2 but significantly enhanced by adding Na2O and Al2O3. Therefore, this work focuses on the removal of boron at different refining parameters in SiO2-CaO-Na2O and SiO2-CaO-Al2O3 slag systems.
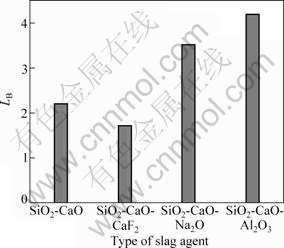
Fig.3 Partition ratio LB in different types of slag systems
3.2 Effect of refining process parameters on boron removal
3.2.1 Refining time
Figure 4 shows the change of boron removal with time during refining at 1 823 K in SiO2-CaO-Na2O and SiO2-CaO-Al2O3 slag systems. It is found that LB is significantly improved compared to previous results in which LB was between 1.5 and 2.5 by EISM, although the variation tendency of LB under the two kinds of slag systems is different. EISM could reduce the diffusion resistance of impurity elements near the boundary layer and enhance the diffusion by increasing the melt flow rate, which could greatly speed up the removal rate of boron. Therefore, the required refining time is substantially reduced. The removal effect in SiO2-CaO-Al2O3 slag systems is obviously better than that in SiO2-CaO-Na2O systems, and LB gradually increases with increasing time in SiO2-CaO-Al2O3 systems, which indicates that the increase of refining time is conducive to the removal of boron. But LB increases and then reduces with increasing time in SiO2-CaO system because the use of Na2O might increase the basicity and decrease the melting point of the slag. However, Na2O could be easily reduced to the silicon phase and becomes volatile at high temperature, which makes it difficult to work for a prolonged period [13].
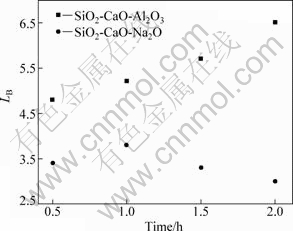
Fig.4 LB in slag systems at 1 823 K for different time
3.2.2 Refining temperature
Figure 5 shows the change of boron removal with temperature during refining for 1 h in SiO2-CaO-Na2O and SiO2-CaO-Al2O3 slag systems. The value of LB gradually increases with the increase of refining temperatures in SiO2-CaO-Al2O3 slag systems, which indicates that the increase of refining temperature is conducive to the removal of boron. Because the viscosity of slag reduces as the temperature increases, the mobility of slag and the silica activity in slag increase. All of these are conducive to the removal of boron. However, boron removal at low temperature is better than that at high temperature in the SiO2-CaO-Na2O system. This is because Na2O could be easily reduced to the silicon phase and become volatile at high temperature[13].

Fig.5 LB at different temperatures after refining for 1 h
3.2.3 Slag composition
Figure 6 shows the changes of boron removal with the content of Na2O and Al2O3 in slag. LB has the same change trend with the contents of Na2O and Al2O3 in SiO2-CaO-Na2O and SiO2-CaO-Al2O3 slag systems, it increases as the proportion of Na2O and Al2O3 in slag increases. It is apparent that if compounds with a basicity higher than that of CaO (or CaF2) is used, a better partition ratio and a higher value for capacity can be obtained. This is because not only the basicity of slags could be greatly improved but also the activity of silica in slag could also be greatly increased by adding alkaline oxide into slag[13].

Fig.6 LB with contents of Na2O and Al2O3 in slag after refining at 1 823 K for 1 h
3.3 Removal of other impurity elements
Not only boron but also other elements such as Ca, Al and Mg could be easily removed by EISM. Table 2 shows impurities changes in silicon before and after refining. Al, Ca and Mg elements were well removed by slag refining and their removal rates reached 85.0%, 50.2% and 66.7%, respectively. The EPMA analysis of metal impurity elements in silicon and slag is shown in Fig.7 after refining for 2 h at 1 823 K in SiO2-CaO-Al2O3 system. It could be clearly seen that the distribution of Ca, Al and Mg elements is all on the slag side between silicon and slag, and these elements all concentrated in slag phase. Although the slag contains much Ca and Al, they do not contaminate silicon melt because they exist in slag phase with a relatively stable state and they contact with silicon melt in the form of molten oxide, thus they would not cause silicon pollution. As the oxygen content in slag phase is relatively high, Al and Mg are more inclined to enrich in slag phase because the affinity of slag to these two elements is greater than that of silicon phase.
Table 2 Impurity content in silicon before and after slag refining at 1 823 K for 2 h (mass fraction, 10-6)
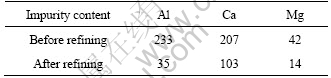
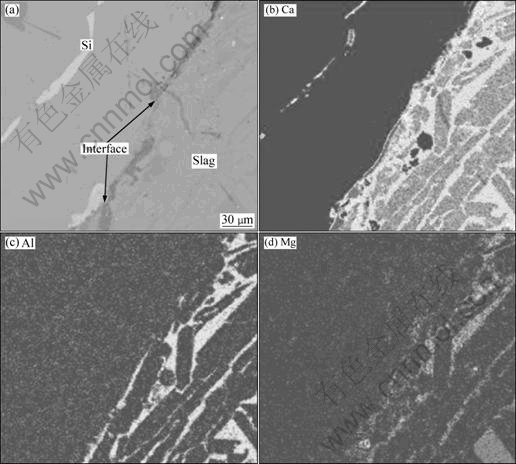
Fig.7 SEM image (a) and EPMA analysis (b-d) of metal impurity elements in silicon and slag after refining at 1 823 K for 2 h
4 Analysis and discussion
4.1 Thermodynamic analysis of boron removal
The reaction of removal of boron from Si by using basic fluxes could be expressed as:
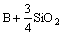 =
=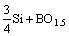 (2)
(2)
The boron dissolved in the liquid silicon reacts with silica in the slag. Boron is oxidized and enters the slag, while the silica is reduced and enters the liquid melt. The distribution coefficient of boron is given as the ratio between the boron content in the slag and the boron content in the silicon as:
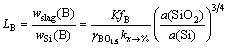 (3)
(3)
where K is the equilibrium constant of Eq.(2), kx→% is the coefficient for the transition from molar fraction to mass fraction, γ(BO1.5) and fB are the activity coefficients of BO1.5 and B in slag and silicon phases, respectively.
From Fig.3 it is known that the removal effect of boron in SiO2-CaO-Na2O and SiO2-CaO-Al2O3 slag systems is better than that in SiO2-CaO-CaF2 system. The activity coefficient of boron oxide calculated for CaO/SiO2 based slag at 1 723-1 873 K could be approximately expressed as:
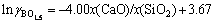 (4)
(4)
When CaF2 is added in the slag, CaO could be generated after CaF2 reacts with silica, thereby increases the basicity of slags. Although the addition of CaF2 provides a broader basicity range compared to the binary system, the content of SiO2 in slag reduces and the oxygen partial pressure (a(SiO2)/a(Si))3/4 also significantly reduces. Meanwhile, the increased ratio of CaO/SiO2 in Eq.(4) results in the decrease of γ(BO1.5). But the change degree of γ(BO1.5) is smaller than that of (a(SiO2)/a(Si))3/4. Therefore, LB would be reduced according to Eq.(3)[13]. It is apparent that when Na2O and Al2O3 are added in slag, a better partition ratio could be obtained, because not only the basicity of slag but also the oxygen partial pressure of slag could be greatly increased by adding alkaline oxide into slag. In this case, the oxygen partial pressure (a(SiO2)/a(Si))3/4 and the ratio of x(CaO)/x(SiO2) in Eq.(4) also significantly increase, which results in the reduction of γ(BO1.5). So LB would increase according to Eq.(3). In addition, the use of Al2O3 might decrease the melting point of the slag and the mobility and affinity of slag are also improved markedly[14]. LB value gradually increases with temperature during EISM. The activity coefficient of boron (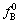 ) in dilute solution between 1 723 and 1 923 K silicon can be expressed as[11]:
) in dilute solution between 1 723 and 1 923 K silicon can be expressed as[11]:
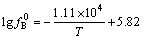 (5)
(5)
 and the equilibrium constant K increase with temperature. The effect of temperature on γ(BO1.5) is very small, so it could be ignored. The activity coefficient of boron oxide is affected by the incorporation between boron and silicate network. Further decrease of γ(BO1.5) with increasing silica content is not necessary for the amount of boron incorporated in the network might be related to the degree of polymerization of the slag structure. So LB increases according to Eq.(3) in SiO2-CaO-Al2O3 slag system, but it decreases with increasing temperature in the SiO2-CaO-Na2O slag system because Na2O could easily become volatile at high temperature[13]. Because boron is in micro scale in the silicon, it takes a certain time to diffuse from silicon into slag phase.
and the equilibrium constant K increase with temperature. The effect of temperature on γ(BO1.5) is very small, so it could be ignored. The activity coefficient of boron oxide is affected by the incorporation between boron and silicate network. Further decrease of γ(BO1.5) with increasing silica content is not necessary for the amount of boron incorporated in the network might be related to the degree of polymerization of the slag structure. So LB increases according to Eq.(3) in SiO2-CaO-Al2O3 slag system, but it decreases with increasing temperature in the SiO2-CaO-Na2O slag system because Na2O could easily become volatile at high temperature[13]. Because boron is in micro scale in the silicon, it takes a certain time to diffuse from silicon into slag phase.
4.2 Kinetic analysis of boron removal
When a dissolved element is refined by slag treatment, it goes through the following five steps: 1) the impurity element transfers from the bulk metallic phase to the metal boundary layer; 2) the impurity element diffuses through the metal boundary layer; 3) the metal is oxidized at the interphase between metal and slag; 4) the impurity element diffuses through the slag boundary layer; 5) the impurity elements transfer from the slag boundary layer to slag bulk phase. Steps 1) and 5) depend on the stirring and mixing in silicon fluid and slag. Stirring is often done by gas bubbling or mechanical devices to increase the mass transfer in the bulk phases. Hence, slag properties such as viscosity are important. A high-viscosity would lead to low velocities in the slag and hence a low mass transfer of the impurity element. High-viscosity also lowers the diffusivity of the impurity element. Steps 2) and 4) depend on the mass transfer coefficients in the metal (β) and in the slag (βs), respectively. A serious difficulty with refining silicon by extraction to a second (slag) phase is the problem of mixing in the slag phase. Often, the slag phase is relatively viscous, so it is difficult to mix the impurity element throughout the slag.
Electromagnetic induction slag refining method is used in this experiment. Figure 8 illustrates the movement of melt in the crucible under electromagnetic force which is formed by the interaction between the magnetic field in the medium frequency induction coil and the induction eddy current in melt. Because the electrical conductivity of silicon fluid and slag is different, slag would follow the silicon fluid movement and they could contact fully in the crucible, which is conducive to the removal of impurities. And electromagnetic induction melting is superior to gas mixing and mechanical agitation, since the mixing process would not contact with the silicon fluid and would not introduce new impurities.

Fig.8 Flow lines of silicon melt under electromagnetic induction melting
According to boundary layer theory, the effective boundary layer thickness (units) could be expressed as:
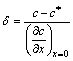 (6)
(6)
where c* is the concentration at the interface; c is the concentration of the liquid bulk concentration outside the boundary layer, and c*>c. In addition, it is also known by the surface renewal theory that the mass transfer coefficient of impurity elements could be expressed as:
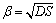 (7)
(7)
where D is the diffusion coefficient of impurity; S is the surface renewal rate.
It is known by Eq.(6) that the larger the concentration gradient near the interface, the thinner the thickness of boundary layer. The concentration gradient here could become larger by increasing fluid flow, thereby reduces the thickness of boundary layer. When the flow rate continues to increase until the boundary layer thickness approaches zero, diffusion resistance disappears, and the flow at this time is called critical velocity. Therefore, it could reduce the diffusion resistance of impurity elements near the boundary layer and enhance the diffusion of them by increasing the melt flow rate under electromagnetic stirring. Meantime, the diffusion coefficients of impurity elements are enhanced as the flow rate of melt is improved by the electromagnetic stirring. It could be inferred from Eq.(7) that the mass transfer coefficient in the metal (β) and in the slag (βs) both enhance, so it could improve and speed up the removal of boron. This is the reason that the EISM could significantly reduce boron impurities in silicon.
5 Conclusions
1) MG-Si with 1.5×10-5 B was successfully purified to 0.2×10-5 B after EISM at 1 823 K for 2 h in SiO2-CaO-Al2O3 slag systems. Al, Ca and Mg elements in MG-Si were also well removed and their removal efficiency reached 85.0%, 50.2% and 66.7%, respectively.
2) The LB value increased with temperature and refining time because the viscosity of slag reduced and the mobility of slag enhanced, and the silica activity in slag increased as temperature increased, which is conducive to the removal of boron.
3) The boron content in silicon was significantly improved compared to previous experimental results and it achieved the level required by EISM. Because EISM could reduce the diffusion resistance of impurity elements near the boundary layer and enhance the diffusion of them by increasing the melt flow rate. The mass transfer coefficient in the metal (β) and in the slag (βs) were both enhanced, so it could improve and speed up the removal of boron. This indicates that EISM is a very effective method for removal the boron impurities in silicon.
References
[1] TAKASHI I, MASAFUMI M. Purification of metallurgical silicon for solar-grade silicon by electron beam button melting [J]. ISIJ International, 1992, 32(5): 635-642.
[2] BRAGA A F B, MOREIRA S P, ZAMPIERI P R, BACCHIN J M G, MEI P R. New processes for the production of solar-grade polycrystalline silicon: A review [J]. Solar Energy Materials and Solar Cells, 2008, 92(4): 418-424.
[3] PIRES J C S, BRAGA A F B, MEI P R. Pro?le of impurities in polycrystalline silicon samples puri?ed in an electron beam melting furnace [J]. Solar Energy Materials & Solar Cell, 2003, 79(3): 347-355.
[4] KAZUHIRO H, NORIYOSHI Y, YOSHIEI K. Evaporation of phosphorus in molten silicon by an electron beam irradiation method [J]. Materials Transactions, 2004, 45(3): 844-849.
[5] NORIYOSHI Y, KAZUHIRO H, YOSHIEI K. Removal of metal impurities in molten silicon by directional solidification with electron beam heating [J]. Materials Transactions, 2004, 45(3): 850-857.
[6] CISZEK T F, SCHWUTTKE G H, YANG K H. Solar-grade silicon by directional solidification in carbon crucibles [J]. IBM Journal of Research and Develop, 1979, 23(3): 270-277.
[7] LUO Da-wei, ZHANG Guo-liang, ZHANG Jian, LI jun, LI Ting-ju. Principle and research progress on preparation solar grade (SoG) silicon by metallurgical route [J]. Foundry Technology, 2008, 29(12): 1721-1726. (in Chinese)
[8] TAKASHI I, MASAFUMI M. Elimination of boron in molten silicon by reactive rotating plasma arc melting [J]. Mater Trans JIM, 1996, 37(5): 983-987.
[9] WU Ji-jun, MA Wen-hui, YANG Bin, DAI Yong-nian, MORITA K. Boron removal from metallurgical grade silicon by oxidizing refining [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(2): 463-467.
[10] LIAW H M, SECCO F. Purification of metallurgical grade silicon by slagging and impurity redistribution [J]. Solar Cells, 1983, 10(2): 109-118.
[11] RYOUJI N, KICHIYA S, FUMITAKA T, NOBUO S. Thermodynamics of boron in silicon melt [J]. Metallurgical and Materials Transactions B, 1994, 25(6): 903-907.
[12] TORSTEN W, KLAUS S. Chemical equilibria between silicon and slag melt [J]. Metallurgical and Materials Transactions B, 1994, 25(4): 497-504.
[13] LEANDRO A, VIANA T, KAZUKI M. Removal of boron from molten silicon using CaO-SiO2 based slags [J]. ISIJ International, 2009, 49(6): 783-787.
[14] LEANDRO A, VIANA T, YOMEI T, TOSHINOBU Y, KAZUKI M. Behavior and state of boron in CaO-SiO2 slags during refining of solar grade silicon [J]. ISIJ International, 2009, 49(6): 777-782.
[15] WANG Xin-guo, DING Wei-zhong, SHEN Hong, ZHANG Jing-jiang. Oxidizing refining process of metallurgical grade silicon [J]. The Chinese Journal of Nonferrous Metals, 2002, 12(4): 827-831. (in Chinese)
[16] BALE C W, CHARTRAND P, DEGTEROV S A, ERIKSSON G, HACK K, MAHFOUD R B, MELAN?ON J, PELTON A D, PETERSEN S. FactSage thermochemical software and databases [J]. Calphad, 2002, 26(2): 189-228.
应用电磁感应造渣法去除冶金级硅中杂质硼
罗大伟1,刘宁1,卢一平1,张国良1,李廷举1, 2
1. 大连理工大学 材料科学与工程学院,大连 116024;
2. 大连理工大学 三束材料改性教育部重点实验室,大连 116024
摘 要:提出一种新的提纯工艺即通过造渣与电磁感应熔炼相结合的方法来去除冶金级硅中的杂质硼。采用自行设计的真空感应熔炼炉进行不同造渣系条件下除硼的研究。结果表明,SiO2-CaO-Al2O3渣系的除硼效果优于其它渣系,在1 823 K经过2 h精炼后,冶金级硅中的硼含量由原来的1.5×10-5降到0.2×10-5。同时冶金级硅中的Al,Ca和Mg杂质得到了精炼,去除率分别达到85.0%, 50.2% 和66.7%。表明电磁感应造渣法是一种非常有效的去除冶金级硅中的硼和其他一些金属元素杂质途径。
关键词:冶金级硅;渣系;分配比
(Edited by FANG Jing-hua)
Foundation item: Project (50674018) supported by the National Natural Science Foundation of China
Corresponding author: LI Ting-ju; Tel: +86-411-84708940; Fax: +86-411-84709284; E-mail: tjuli@dlut.edu.cn
DOI: 10.1016/S1003-6326(11)60840-6