J. Cent. South Univ. Technol. (2011) 18: 290-295
DOI: 10.1007/s11771-011-0693-7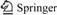
Microstructures, mechanical properties and compressive creep behaviors of as-cast Mg-5%Sn-(0-1.0)%Pb alloys
WANG Qing(����)1, CHEN Yun-gui(���ƹ�)1, XIAO Su-fen(Ф�ط�)1,
LIU Hong-mei(����÷)2, TANG Yong-bai(������)1, ZHAO Yuan-hua(��Դ��)1
1. School of Materials Science and Engineering, Sichuan University, Chengdu 610065, China;
2. Department of Chemical and Materials Engineering, The University of Auckland,
Private Bag 92019, Auckland, New Zealand
? Central South University Press and Springer-Verlag Berlin Heidelberg 2011
Abstract: The microstructures, tensile properties and compressive creep behaviors of Mg-5%Sn-(0-1.0)%Pb (mass fraction) alloys were studied. The microstructures of the Mg-Sn-Pb alloys consist of dendritic ��-Mg and Mg2Sn phase. The addition of Pb can refine the size of Mg2Sn phase and grain size, reduce the amount of Mg2Sn phase at grain or inter-dendrite boundaries and change the distribution of Mg2Sn phase. Pb exists in the Mg2Sn phase or dissolves in ��-Mg matrix. The mechanical properties of the tested alloys at room temperature are improved with the addition of Pb. When the Pb content is over 0.5%, the mechanical properties are decreased gradually. The Mg-5%Sn-0.5%Pb shows the best ultimate tensile strength and elongation, 174 MPa and 14.3%, respectively. However, the compressive creep resistance of the Mg-Sn-Pb alloys is much lower than that of the Mg-Sn binary alloy at 175 ��C with applied load of 55 MPa, which means that Pb has negative effects on the compressive creep resistance of the as-cast Mg-Sn alloys.
Key words: Mg-Sn-Pb alloy; microstructures; mechanical properties; compressive creep; Mg2Sn phase
1 Introduction
Magnesium-based alloys are increasingly used in many engineering areas, such as portable microelectronics, automobile and aerospace industries, due to their low density, high specific strength, high specific toughness and good damping properties [1]. Mg alloys can be broadly divided into Al-containing and Al-free alloy systems. They are designed for either enhancing room-temperature mechanical properties or elevated-temperature creep resistance.
The interest in the Mg-Sn based alloys started in the early 1930s. According to the Mg-Sn binary phase diagram [2], the maximum solidification temperature range is about 67 ��C. It is much narrower than that of the Mg-Al and Mg-Zn binary alloys, which is 136 ��C and 283 ��C, respectively. The fact that solid solubility of Sn in ��-Mg drops sharply from 14.48% (mass fraction) at the eutectic transformation temperature of 561 ��C to 0.45% (mass fraction) at 200 ��C shows that the system of Mg-Sn alloy is promising aging-hardenable alloy. Moreover, the melting point of 770 ��C of the Mg2Sn phase existing in the alloy, which is much higher than that of the Mg17Al12 phase (462 ��C) in the Mg-Al alloys, means that the alloy is likely to have superior high-temperature creep resistance. LIU et al [3] reported that the Mg-5%Sn (mass fraction) alloy offered good comprehensive properties. However, compared with some other magnesium alloys, the mechanical properties of the Mg-Sn alloy still need to be further improved. Many studies have shown that room and elevated temperature mechanical properties of the Mg-Sn alloys were improved by RE elements addition in the past few years [4-11]. In these literatures, there are some reports about the effect of Pb addition on the microstructure and mechanical properties of the magnesium alloys [12-15]. The result is very optimistic. However, the effect of Pb addition on the microstructures and mechanical properties of the Mg-Sn alloys has less been reported. So, in this work, the microstructures, mechanical properties and compressive creep behaviors of the as-cast Mg-Sn-Pb alloys are focused on.
2 Experimental
Five kinds of Mg-5%Sn (mass fraction) alloys with the Pb addition levels of 0, 0.25%, 0.5%, 0.75%, and 1.0% (mass fraction) were prepared. Pure magnesium (99.95%, mass fraction), pure tin (99.98%, mass fraction) and pure lead (99.9%, mass fraction) were melted in a iron crucible under the protection of N2+0.2%SF6 (volume fraction) mixed gas atmosphere. Tin and lead were added after magnesium was melted at 780 ��C. The melt is stirred to ensure the homogeneity. It was then held at 730 ��C for about 30 min, finally it was cast into an iron mould which had been preheated up to 300 ��C. The cavity dimension of the mould is 10 mm �� 40 mm �� 90 mm.
The metallographic specimens were sliced from the same place of each casting. They were polished and etched with a solution of 4% nitric acid (volume fraction)+ethyl alcohol. The samples used for the grain size examination were preheated to 480 ��C for 2 h under the protection of the troilite, followed by water quenching. The linear-intercept method was employed. Fifty fields of view were selected and measured with digital optical phase conjugation (OLYMPUS-BH-2). The overall phase structures of the investigated alloys were analyzed by X-ray diffractometry (DX2600) with Cu K�� radiation. Scanning electron microscopy (JEOL-JSM-6490LV) and energy dispersion spectrometry (EDS) analyses were performed to identify the intermetallic phases in the alloys.
The tensile tests were conducted at room temperature on an electro-universal testing machine (Instron5569) with a crosshead speed of 5 mm/min. The specimens were cut into slices with an electrical discharge wire-cutting at the middle of each sample. The gage dimension of each specimen is 70.0 mm��3.5 mm�� 2.0 mm. Six samples of each alloy were employed to obtain one set of mechanical data.
The compressive creep tests were carried out on a self-made tester, which contains a constant-load equipment, a temperature controller and an experiment data collector. The creep specimens were cut by electric spark machining from the middle part of the ingots. The specimen size of creep test is d 8 mm �� 10 mm. Oil was used as protecting medium during the creep test. The compressive creep tests were performed under a load of 55 MPa and a fixed temperature of 175 ��C.
3 Results and discussion
3.1 Microstructure
Figs.1 and 2 show the microstructures and XRD patterns of the investigated alloys. The microstructure of the as-cast Mg-5%Sn alloy consists of ��-Mg matrix and Mg2Sn phase. The Mg2Sn phase exists in the form of (��-Mg+Mg2Sn) eutectic indicated by arrows in Fig.1(a). The result of XRD analysis also confirms the presence of two phases, ��-Mg and Mg2Sn, in the Mg-5%Sn alloy. With the addition of Pb, the amount of (��-Mg+Mg2Sn) eutectic distributed between dendritic arms and grain boundaries is obviously reduced. When the content of Pb is over 0.5%, (��-Mg+Mg2Sn) eutectics disappear. With the addition of Pb, Mg2Sn phase at grain or inter-dendrite boundaries also becomes fine and discontinuous. When the content of Pb is over 0.5%, the distribution of Mg2Sn phase becomes much even. The results of XRD analysis indicate that the addition of Pb does not form any new phase except for Mg2Sn, but the diffraction peak intensities of Mg2Sn is gradually weakened. The lattice parameters of Mg-5%Sn and Mg-5%Sn-0.5%Pb are listed in Table 1, which are taken from the Rietveld analysis result of the XRD patterns in Fig.2. It can be found that the addition of Pb increases the a and c parameters of hexagonal ��-Mg matrix and the a value of cubic Mg2Sn phase, which is possible due to the solid-solution of Pb in the ��-Mg matrix and Mg2Sn phase, because the atomic radius of Pb is larger (0.181 nm) than that of Mg and Sn (0.172 nm).
Table 2 lists the EDS analysis results of the Mg-5%Sn-(0-1.0)%Pb alloys. Positions A and B correspond to the analysis points in Fig.1. It can be found that the content of Pb in the ��-Mg matrix and Mg2Sn phase is increased, but the content of Sn is decreased in the Mg2Sn phase and increased in the ��-Mg matrix. Fig.3 shows the grain size of Mg-5%Sn-(0-1.0)%Pb alloys. It can be found that the grain size of the investigated alloys is gradually reduced with the increase of Pb content.
Based on the above results, it can be concluded that the addition of Pb can refine the size of Mg2Sn phase and grain size, reduce the amount of Mg2Sn phase at grain or inter-dendrite boundaries, change the distribution of Mg2Sn phase, and increase the content of Sn in the ��-Mg matrix. Pb exists in the Mg2Sn phase as well as in the ��-Mg solid-solution.
Microstructure of Mg-Sn alloy can be changed with the addition of Pb, mainly due to the fact that Pb changes the solidification process of the Mg-Sn alloy. With the addition of Pb, Sn and Pb are enriched in the liquid ahead of the solid/liquid interface. Compared with the Mg-Sn alloy, the concentration of Sn atoms in the liquid is reduced, so the eutectic reaction cannot occur and the amount of the (��-Mg+Mg2Sn) eutectics at grain or inter- dendrite boundaries is reduced. KASHYAP et al [16] reported that in the Mg-Al alloy, the addition of Pb could reduce the strain energy at the grain boundary of Mg-Al alloys and hence could reduce the amount of Mg17Al12 precipitates. Some other studies [12-14] also indicated that Pb in the form of solid solution might lower the diffusion dynamics of Al and Mg atoms in the Mg-Al alloys. They observed that the addition of Pb suppressed the formation and the growth of the intermetallics. Maybe, this is another reason for the decreased amount of (��-Mg+Mg2Sn) eutectic or Mg2Sn phase in the Mg-Sn-Pb alloys. Meanwhile, Pb is partitioned into the liquid ahead of the solidification front, causing a constitutional supercooling zone of liquid ahead of the interface. The constitutional supercooling caused by the Pb enrichment in the liquid ahead of the solid/liquid interface promotes the nucleation and hinders the fast growth of grains. Therefore, Pb induces a refinement effect in the Mg-Sn alloys.

Fig.1 SEM images of as-cast Mg-5%Sn- xPb alloys: (a) Mg-5%Sn; (b) Mg-5%Sn- 0.25%Pb; (c) Mg-5%Sn-0.5%Pb; (d) Mg- 5%Sn-0.75%Pb; (e) Mg-5%Sn-1.0%Pb
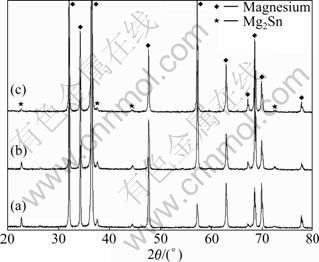
Fig.2 XRD patterns of Mg-5%Sn-x%Pb alloys: (a) Mg-5%Sn; (b) Mg-5%Sn-0.5%Pb; (c) Mg-5%Sn-1.0%Pb
Table 1 Lattice parameters of Mg-5%Sn-x%Pb (x=0, 0.5) alloys
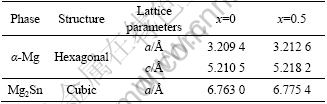
Table 2 EDS analysis results of Mg-5%Sn-x%Pb alloys
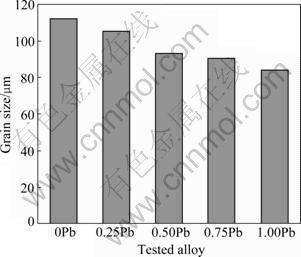
Fig.3 Grain size distribution of Mg-5%Sn-(0-1.0)%Pb alloys
Since the maximum solid solubility of Pb in ��-Mg is 41.73% (mass fraction), which is larger than that of Sn (14.48%, mass fraction). When both Pb and Sn elements are added into magnesium alloy at the same content, the amount of Pb dissolving in the ��-Mg matrix is greater than that of Sn. So, with increasing the Pb addition, the Pb content in the ��-Mg matrix increases. On the other hand, Pb atom also replace the Sn atom in the Mg2Sn phase, so the content of Pb in the Mg2Sn phase is increased with the addition of Pb, while Sn atoms in this part of Mg2Sn phase move into the ��-Mg matrix and reduce the amount of Mg2Sn phase. So, we can find discontinuous phases distributing along the grain boundaries with addition of Pb in Fig.1.
3.2 Mechanical properties
Fig.4 shows the ultimate tensile strength (UTS) and elongation of the as-cast Mg-5%Sn-(0-1.0)%Pb alloys. The UTS and elongation of the as-cast Mg-5%Sn can be improved with the addition of Pb. When the Pb content is less than 0.5%, the UTS and elongation of the alloys increase gradually with the addition of Pb. The optimum mechanical properties are attained at 0.5%Pb, where the UTS and elongation reach 174 MPa and 14.3%, respectively. But, when the Pb content is over 0.5%, the UTS and elongation are decreased gradually.
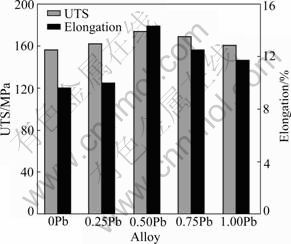
Fig.4 Relationship between mechanical properties and Pb content in Mg-5%Sn-(0-1.0)%Pb alloys
With the addition of Pb, the content of Pb in ��-Mg is increased, which leads to the grain refinement and the distribution change of Mg2Sn phase. So, the improvement of mechanical properties of the tested alloys mainly comes from the solid solution strengthening, refinement strengthening and dispersion strengthening.
The maximum solid solubility of Pb in ��-Mg is 41.73% (mass fraction), so its solid solution strengthening effect can be realized. The difference between the atomic radii of Pb and Mg causes the distortion of the Mg lattice and gives rise to solid solution strengthening. The resulting increase in the lattice parameters of ��-Mg is summarized in Table 1. With the addition of Pb, the grain size of investigated alloys is gradually reduced (see Fig.3). So, the number of grain boundaries is increased, which effectively restrains the movement of dislocations, resulting in a strengthening effect. The addition of Pb also makes the net-like Mg2Sn phase distributing at grain boundaries reduced and become discontinuous, and the distribution of Mg2Sn phase becomes more even. The alloy is expected to exhibit low ductility because the net-like Mg2Sn phase at grain boundaries easily breaks up and causes cracking during the plastic deformation. As a consequence, there is a resulting low elongation for the Mg-5%Sn alloy. But, with the addition of Pb, the net-like Mg2Sn phase gradually disappears at grain boundaries, and the discrete Mg2Sn phase precipitates may restrain the movement of dislocation, resulting in a dispersion strengthening effect. When the Pb addition is over 0.5%, the number of Mg2Sn phase is reduced, so the dispersion strengthening effect is weakened and the UTS and elongation of the alloys are decreased gradually.
3.3 Compressive creep behavior
Fig.5 shows the compressive strain-time curves of Mg-5%Sn, Mg-5%Sn-0.5%Pb and Mg-5%Sn-1.0%Pb alloys at a temperature of 175 ��C and a stress of 55 MPa. Table 3 summarizes the secondary creep rate data 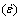 for the tested alloys at 175 ��C and 55 MPa. With the increase of Pb content, the compressive creep resistance of the alloys decreases, and much lower secondary creep rate
for the tested alloys at 175 ��C and 55 MPa. With the increase of Pb content, the compressive creep resistance of the alloys decreases, and much lower secondary creep rate 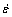 exists in the Mg-5%Sn alloy.
exists in the Mg-5%Sn alloy.
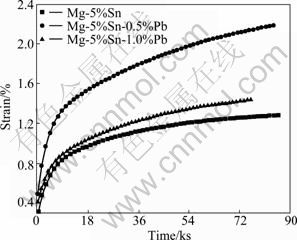
Fig.5 Compressive creep curves of as-cast Mg-5%Sn-xPb alloys at 175 ��C and 55 MPa
Table 3 Compressive creep rates of as-cast Mg-5%Sn-xPb alloys at 175 ��C and 55 MPa

For the Mg-5%Sn alloy, the high heat-resistance is mainly due to a large number of (��-Mg+Mg2Sn) eutectic which distributes at grain boundaries, and it is effective in inhibiting the grain boundary sliding at elevated temperatures. On the other hand, the Mg-5%Sn alloy has a lower volume fraction of grain boundaries due to its coarse-grained structure. The effect of grain boundary sliding on the deformation is considered to be much smaller than those of the Mg-Sn-Pb alloys with fine-grained structure. With the addition of Pb, the compressive creep resistance of the Mg-Sn-Pb alloys is weakened, especially the Mg-5%Sn-0.5%Pb alloy, which is mainly due to the fact that the amount of (��-Mg+Mg2Sn) eutectic at the grain boundaries is obviously reduced. Meanwhile, according to the Mg-Pb binary phase diagram, the melting point of Mg2Pb phase (549 ��C) is lower than that of Mg2Sn phase, so the Pb atom existing in Mg2Sn phase can weaken the heat-resistance of Mg2Sn phase. When the Pb content is over 0.5%, the compressive creep resistance of the Mg-5%Sn-1.0%Pb alloy is better than that of the Mg-5%Sn-0.5%Pb alloy. The reason is that a great number of Sn and Pb exist in the ��-Mg matrix, resulting in a solid solution strengthening effect; meanwhile, the uniform dispersion of precipitates can effectively impede the dislocation motions during the process of compressive creep at elevated temperatures.
4 Conclusions
1) The addition of Pb can refine the grain size, reduce the amount of Mg2Sn phase at grain or inter- dendrite boundaries, change the distribution of Mg2Sn phase, and increase the content of Sn in the ��-Mg matrix. Pb exists in Mg2Sn phase as well as in the ��-Mg solid-solution.
2) The ultimate tensile strength and elongation can be improved with the addition of Pb. The Mg-5%Sn- 0.5%Pb alloy shows the best mechanical properties at room temperature.
3) Pb has negative effects on the compressive creep resistance of the as-cast Mg-Sn alloy. The Mg-5%Sn alloy shows the best compressive creep behavior among these alloys.
References
[1] MORDIKE B L, EBERT T. Magnesium: Properties��application��potential [J]. Materials Science and Engineering A, 2001, 302: 37- 45.
[2] MASSALSKI T B, OKAMOTO H. Binary alloy phase diagrams [M]. Ohio: ASM International, Materials Park, 1990.
[3] LIU Hong-mei, CHEN Yun-gui, TANG Yong-bai, WEI Shang-hai, NIU Gao. The microstructure, tensile properties, and creep behavior of as-cast Mg-(1�C10)%Sn alloys [J]. Journal of Alloys and Compounds, 2007, 440: 122-126.
[4] WU Wen-hua, XIA Chang-qing. Microstructures and mechanical properties of Mg-Ce-Zn-Zr wrought alloy [J]. Journal of Central South University of Technology, 2004, 11(4): 19-23.
[5] LIU Hong-mei, CHEN Yun-gui, TANG Yong-bai, HUANG De-ming, NIU Gao. The microstructure and mechanical properties of permanent-mould cast Mg-5wt%Sn-(0-2.6)wt%Di alloys [J]. Materials Science and Engineering A, 2006, 437: 348-355.
[6] ZHANG Xiao-ping, CHEN Yun-gui, XIAO Su-fen, TANG Yong-bai, NIU Gao, WEI Shang-hai, ZHAO Yuan-hua. Microstructure, tensile properties and compressive creep resistance of Mg-(5-8.5)%Sn- 2%La alloys [J]. Transactions of Nonferrous Metals Society of China, 2008, 18: s229-s305.
[7] LIU Hong-mei, CHEN Yun-gui, TANG Yong-bai, WEI Shang-hai, NIU Gao. Tensile and indentation creep behavior of Mg-5%Sn and Mg-5%Sn-2%Di alloys [J]. Materials Science and Engineering A, 2007, 464: 124-128.
[8] WEI Shang-hai, CHEN Yun-gui, TANG Yong-bai, ZHANG Xiao-ping, LIU Ming, XIAO Su-fen, ZHAO Yuan-hua. Compressive creep behavior of Mg-Sn-La alloys [J]. Materials Science and Engineering A, 2009, 508: 59-63.
[9] JIAN Wei-wei, KANG Zhi-xin, LI Yuan-yuan. Effect of hot plastic deformation on microstructure and mechanical property of Mg-Mn-Ce magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2007, 17: 1158-1163.
[10] LIM H K, KIM D H, LEE J Y, KIM W T, KIM D H. Effects of alloying elements on microstructures and mechanical properties of wrought Mg-MM-Sn alloy [J]. Journal of Alloys and Compounds, 2009, 468: 308-314.
[11] YU Kun, LI Wen-xian, WANG Ri-chu. Mechanical properties and microstructure of as-cast and extruded Mg-(Ce, Nd)-Zn-Zr alloys [J]. Journal of Central South University of Technology, 2005, 12(5): 36-40.
[12] CANDAN S, UNAL M, TURKMEN M, KOC E, TUREN Y, CANDAN E. Improvement of mechanical and corrosion properties of magnesium alloy by lead addition [J]. Materials Science and Engineering A, 2009, 501: 115-118.
[13] BALASUBRAMANI N, SRINIVASAN A, PILLAI U T S, PAI B C. Effect of Pb and Sb additions on the precipitation kinetics of AZ91 magnesium alloy [J]. Materials Science and Engineering A, 2007, 457: 275-281.
[14] SRINIVASAN A, PILLAI U T S, PAI B C. Effect of Pb addition on ageing behavior of AZ91 magnesium alloy [J]. Materials Science and Engineering A, 2007, 452/453: 87-92.
[15] LUO A. Understanding the solidification of magnesium alloys [C]// Proceedings of the Third International Magnesium Conference, Manchester, UK, 1996: 449-464.
[16] KASHYAP K T, RAMACHANDRA C, SUJATHA M, CHATTERJI B. Role of diffusional coherency strain theory in the discontinuous precipitation in Mg-Al alloy [J]. Bulletin of Materials Science, 2000, 23: 39-45.
(Edited by YANG Bing)
Foundation item: Project(03GG009-007-4) supported by the Key Program of Science and Technology Bureau of Sichuan Province, China
Received date: 2010-03-26; Accepted date: 2010-07-15
Corresponding author: CHEN Yun-gui, Professor, PhD; Tel: +86-28-85405670; E-mail: ygchen60@yahoo.com.cn