
J. Cent. South Univ. (2020) 27: 1186-1196
DOI: https://doi.org/10.1007/s11771-020-4359-1
Sulfidation roasting of zinc leaching residue with pyrite for recovery of zinc and iron
MIN Xiao-bo(��С��)1, 2, JIANG Guang-hua(���⻪)1, WANG Yun-yan(������)1, 2,
ZHOU Bo-sheng(�ܲ���)1, XUE Ke(Ѧ��)1, KE Yong(����)1, 2, XU Qiu-jing(�����)1,
WANG Jing-wen(������)1, REN Hui-chuan(�λ۴�)1
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Chinese National Engineering Research Center for Control and Treatment of Heavy Metal Pollution, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: Zinc leaching residue (ZLR) contains high content of valuable metals such as zinc and iron. However, zinc and iron mainly exist in the form of zinc ferrite, which are difficult to separate and recover. This study proposed a new process involving sulfidation roasting, magnetic separation and flotation to recover zinc and iron in ZLR. Through sulfidation roasting of ZLR with pyrite, zinc and iron were converted into ZnS and Fe3O4. The effects of pyrite dosage, roasting temperature and roasting time on the sulfidation of zinc in ZLR were investigated. The results showed that the sulfidation percentage of zinc reached 91.8% under the optimum condition. Besides, it was found that ball-milling was favorable for the separation and recovery of zinc and iron in sulfidation products. After ball-milling pretreatment, iron and zinc were enriched from sulfidation products by magnetic separation and flotation. The grade of iron in magnetic concentrates was 52.3% and the grade of zinc in flotation concentrates was 31.7%, which realized the recovery of resources.
Key words: zinc leaching residue; sulfidation roasting; recovery; zinc; iron; magnetic separation; flotation
Cite this article as: MIN Xiao-bo, JIANG Guang-hua, WANG Yun-yan, ZHOU Bo-sheng, XUE Ke, KE Yong, XU Qiu-jing, WANG Jing-wen, REN Hui-chuan. Sulfidation roasting of zinc leaching residue with pyrite for recovery of zinc and iron [J]. Journal of Central South University, 2020, 27(4): 1186-1196. DOI: https://doi.org/10.1007/s11771- 020-4359-1.
1 Introduction
Zinc leaching residue (ZLR) is a typical residue generated after roasting and leaching of zinc concentrates in the zinc hydrometallurgy [1]. Hydrometallurgy is a predominate technique in zinc smelting process, accounting for more than 85% of worldwide zinc output and about 95% of the total zinc output in China [2]. Thus, this process produces a considerable amount of ZLR. Due to the high contents of Zn and Fe as well as a small amount of Pb [3], improper treatment of ZLR will not only threaten the surrounding environment, but also cause the waste of natural resource. Therefore, from the viewpoint of environment protection and the conservation of metal resources, ZLR should be served as a kind of the secondary resource rather than final wastes.
Zn and Fe are mainly in the form of zinc ferrite (ZnFe2O4 or ZnO��Fe2O3) in ZLR, which is generated inevitably during the oxidation and roasting process of zinc concentrates [4, 5]. However, the zinc ferrite in ZLR has a typical spinel-type structure with extremely stable chemistry properties, which makes the recovery of Zn and Fe difficult. Up to now, the recycling of zinc ferrite has been extensively investigated, developing a series of recovery technologies for zinc, iron and other valuable metals [6-9]. Although these methods have achieved the recovery of resources in the zinc ferrite to varying degrees, they still lack an energy-saving and environment- friendly technology that can fully recover the zinc and iron in ZLR and convert them into usable resources. In practice, the rotary kiln fuming process is widely used in the treatment of ZLR to recover valuable metals. However, it consumes huge energy to provide high operating temperature (~1200 ��C) [10]. In addition, the kiln coating in rotary kiln roasting also affects the yield of iron resource and its industrial application [11]. In hydrometallurgy treatment of ZLR, zinc and iron are leached firstly and then iron is precipitated [12, 13]. However, this process requires a high temperature and strong acid environment, which not only generates a large quantity of wastewater and residue containing heavy metals, but also leads to the loss of iron resources. Therefore, it is urgent to develop a new technology that can realize the resource recovery of zinc and iron in ZLR with high efficiency.
Inspired by the flotation and enrichment of sulfide ores, sulfidation treatment as a potential method for valuable metals recovery has attracted a great deal of attention [14-18]. At present, the prevalent sulfidation methods include traditional solution sulfidation with Na2S, mechanochemical sulfidation, hydrothermal sulfidation and sulfidation roasting. Although traditional solution sulfidation, mechanochemical sulfidation and hydrothermal sulfidation are cost saving, they take less effect on the sulfidation of materials or wastes containing complex zinc phases. Many studies indicated that sulfidation roasting shows better results for the destruction and transformation of complex zinc phases in low grade ore and smelting slags [19, 20]. ZHENG et al [19] found that the mineralogical reconstruction of lead smelter slag can be realized after sulfidation roasting with pyrite, carbon and sodium carbonate. MIN et al [20] achieved phase transformation of hemimorphite by sulfidation roasting and enriched Zn and Pb in sulfide products by flotation. In our previous studies, the sulfidation behavior of pure zinc ferrite and zinc silicate roasted with pyrite has been investigated [21, 22]. Results revealed that the conversion of zinc ferrite and zinc silicate into zinc sulfide is a sulfur inducing and sulfur-oxygen interface exchange mechanism. However, the actual wastes containing zinc ferrite by sulfidation roasting have not been systematically investigated.
Therefore, the disposal of ZLR by the combination of sulfidation roasting with pyrite, magnetic separation and flotation technology was studied. The conditions such as roasting temperature, pyrite dosage and roasting time were optimized. The phase transformation, variation of particle size and microstructure changes during the roasting process were analyzed by XRD, laser particle size analysis (LPSA) and SEM-EDS. The separation of ZnS and Fe3O4 in sulfidation product was achieved by ball milling. Finally, the recovery of iron and zinc by magnetic separation and flotation was investigated. This work aims to provide a new technology for the separation and recovery of zinc and iron from zinc ferric- containing wastes.
2 Experimental methods
2.1 Materials
ZLR used in this study was obtained from a large smelter in Hunan, China. X-ray fluorescence analysis shows that the contents of zinc and iron were 19.5% and 24.3%, respectively (Table 1). X-ray diffraction pattern of ZLR in Figure 1 indicates that the main phase in ZLR was zinc ferrite (ZnFe2O4), while lead sulfate (PbSO4), zinc silicate (Zn2SiO4), calcium sulfate (CaSO4), zinc sulfate (ZnSO4) and zinc sulfide (ZnS) were the minor phases. The chemical phase analysis of zinc in ZLR was also performed. It can be seen from Table 2 that the content of Zn in the form of ferrite accounted for 79.7% of the total zinc in ZLR.
The sulfidizing agent used in this study was pyrite (FeS2), which was taken from a mine in Anhui Province, China. The contents of Fe and S in pyrite were 48.0% and 52.0%, respectively. The X-ray diffraction pattern of pyrite was shown in our previous study [20, 23].
Table 1 Main elemental composition of ZLR (mass fraction, %)
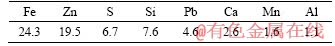
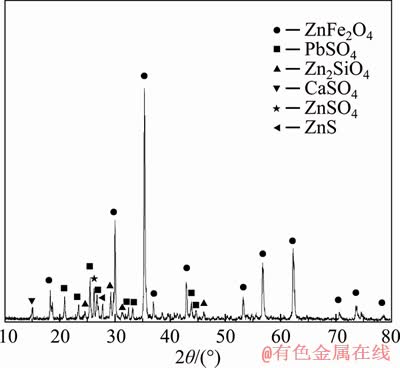
Figure 1 X-ray diffraction pattern of ZLR
Table 2 Chemical phase composition of zinc in ZLR

2.2 Experimental procedure
The process flow sheet of the experiments is shown in Figure 2. Sulfidation roasting was carried out in a tube furnace (Thermconcept, ROS 75/600/12). First, a certain amount of ZLR and pyrite were uniformly mixed according to the predetermined mass ratio. Then, 10 g of the mixture was transferred into a corundum crucible and roasted in the tube furnace with a heating rate of 5 ��C/min from 25 ��C to a setting temperature (800-1000 ��C) for 10-120 min under N2 flow (1.5 L/min). Next, the roasted sample was naturally cooled to room temperature under N2 flow (1.5 L/min). Finally, the cooled sulfidation products were removed from the tube furnace for following experiments.
To achieve the separation of zinc and iron,10 g of sulfidation products and 100 mL of water were mixed in a planetary ball mill (QM-3SP4, Nanjing, China) for 2-8 h under a ball-to-powder of 20:1 at a speed of 400 r/min. Then, the milled slurry was treated by a magnetic separation process. The magnetic separation was performed for 20 min with magnetic field intensity of 60 kA/m. The obtained concentrates from magnetic separation were magnetic separated again (magnetic field intensity of 40 kA/m) as shown in Figure 2.
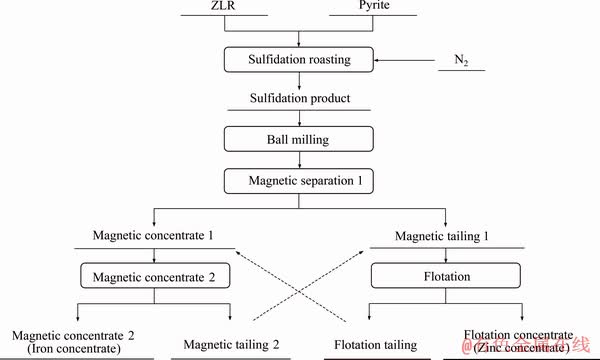
Figure 2 Diagram of experimental procedure
Meanwhile, the obtained tailings from magnetic separation were treated by a conventional rougher flotation process, which was made with laboratory scale floatation machines. The pulp density was 200 g/L and the pH value was adjusted to 10.0 by calcium hydroxide. Then, the dispersant (sodium hexametaphosphate, dosage of 2000 g/t) was added to the pulp, followed by conditioning for 3 min. Subsequently, the activator (copper sulfate, 200 g/t) was added, followed by conditioning for 3 min. After conditioning, the collectors (sodium butyl xanthogenate, 500 g/t) was added, followed by conditioning for 2 min. Finally, the frother (pine camphor oil, 30 g/t) was added to the pulp and followed by conditioning for 1 min. The air supply valve is opened to form a certain thickness of mineralized froth in the can and the froth layer was removed manually.
2.3 Analytical methods
The sulfidation percentage of zinc in ZLR was regarded as an evaluation index. The sulfidation percentage was determined by performing a procedure based on the chemical phase analysis of the sulfide ore [24]. Detailed procedures were shown in our previous studies [25].
The results of magnetic separation and flotation were evaluated by metal grade and recovery. The metal grades in concentrates and tailings are expressed as Gc (%) and Gt (%), respectively. The recovery of magnetic separation and flotation was calculated from Eq.(1):
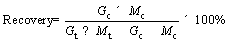 (1)
(1)
where Mc (g) and Mt (g) are the masses of concentrates and tailings, respectively.
The chemical composition of samples was analyzed using a Bruker S4-Pioneer X-ray fluorescence spectrometer. The crystallographic composition of samples was identified by X-ray diffraction (XRD, D/max2550 VB+18 KW) with the steps of 0.02�� at 10 (��)/min in 2�� ranging from 10�� to 80��. The micromorphology changes during sulfidation roasting process were analyzed by scanning electron microscopy (SEM, Nova Nano SEM 230). Particle size distribution was characterized using a laser diffraction particle size analyzer (Micro-plus, Malvern, USA).
3 Results and discussion
3.1 Sulfidation roasting of ZLR
3.1.1 Effect of pyrite dosage
It was confirmed that the sulfidation of zinc ferrite can be achieved by controlling the reaction conditions [22]. As the source of sulfur, the dosage of pyrite is an important factor in determining the sulfidation percentage of Zn in ZLR. Thus, the sulfidation roasting of ZLR was performed under different pyrite dosage varied from 10% to 40% (by mass) with a roasting temperature of 900 ��C and a roasting time of 60 min. As shown in Figure 3(a), the increase of pyrite dosage significantly promoted the sulfidation of Zn in ZLR. The sulfidation percentage of Zn markedly increased from 13.3% to 91.3% when the dosage of pyrite increased from 10% to 30%. However, when the dosage of pyrite further increased to 40%, the sulfidation percentage of Zn in ZLR remained at approximately 90%. Therefore, to ensure a high sulfidation percentage, the 30% pyrite dosage was selected as the condition for all the further experiments.
3.1.2 Effect of roasting temperature
As is known to all that the reaction rate is strongly related to the temperature. Hence, the effect of roasting temperature on the sulfidation percentage was investigated with the pyrite dosage of 30.0% and a roasting time of 60 min. Figure 3(b) shows that when the temperature varied from 800 ��C to 900 ��C, the sulfidation percentage increased from 68.4% to 83.0%. Beyond 900 ��C, the sulfidation percentage slightly decreased with the further increase of temperature. This is due to the formation of iron-zinc solid solution at high temperatures, which results in the decrease of ZnS content. Thus, 900 ��C was selected as the optimum roasting temperature and all of the subsequent experiments were conducted at this temperature.
3.1.3 Effect of roasting time
To seek for the best roasting time, the effect of roasting time on the sulfidation percentage was investigated when the amount of pyrite was 30% and the roasting temperature was 900 ��C. It can be seen from Figure 3(c) that the sulfidation percentage rapidly improved from 48.0% to 91.8% as the roasting time varied from 15 min to 60 min. However, there was no significant increase in sulfidation percentage with the further prolongation of the roasting time. It can be concluded that the sulfidation process of zinc is relatively slow. Proper prolongation of the roasting time can promote the improvement of the sulfidation percentage. Therefore, 60 min should be considered as the optimum roasting time.
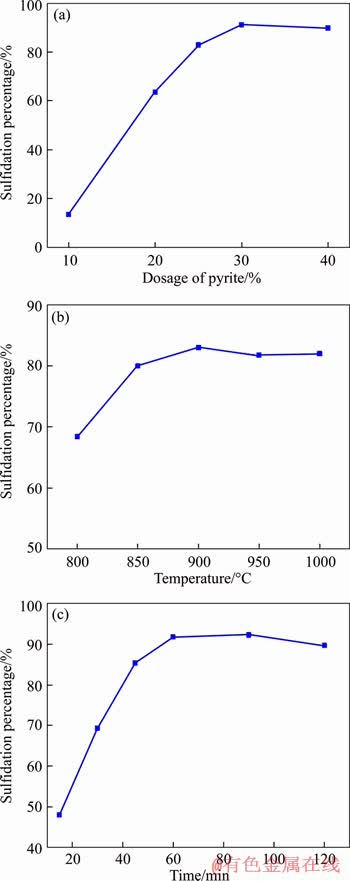
Figure 3 Effects of pyrite dosage (a), roasting temperature (b), and roasting time (c) on sulfidation percentage
Based on the results above, the optimal parameters of sulfidation roasting are determined to be pyrite dosage of 30%, roasting temperature of 900 ��C and roasting time of 60 min. Under these conditions, the sulfidation percentage of Zn in ZLR reached 91.8%. Unless otherwise specified, the sulfidation products used in subsequent experiments were obtained under the optimal conditions.
3.2 Characterization of sulfidation products
3.2.1 Phase composition
In order to study the phase transformation of ZLR during sulfidation roasting process, the sulfidation products obtained under different pyrite additions were analyzed with XRD. Figure 4 shows the XRD patterns and its enlarged patterns. It can be seen that besides the peaks of ZnFe2O4, Zn2SiO4 and PbSO4, weak ZnS and PbS peaks were also observed in the sulfidation products obtained at pyrite dosage of 10%. The results indicated that the sulfidation reaction had taken place, which was consistent with the results in Figure 3(a). When the dosage of pyrite was 20%, the characteristic peaks of ZnS can be clearly found in Figure 4, indicating that a considerable amount of ZnS had been generated. According to Figure 3(a), more than 60% of zinc was converted into ZnS with a pyrite dosage of 20%. Further elevating the pyrite dosage to 25%, the intensity of ZnS peaks increased. In addition, ZnFe2O4 peaks shifted to the right (Figure 4(b)), indicating the conversion of ZnFe2O4 into ferroferric oxide (Fe3O4). When the pyrite dosage varied from 25% to 40%, however, the intensity of ZnS and Fe3O4 peaks hardly changed. The results indicated that the sulfidation reaction almost completed, which agreed with the variation trends of sulfidation percentage shown in Figure 3.
To further quantify the chemical phase composition of Zn in sulfidation products and analyze the phase transformation of zinc during the sulfidation roasting, the chemical phase composition of Zn in ZLR and sulfidation products are presented in Figure 5. Compared with ZLR, the content of zinc in the form of sulfide in sulfidation products greatly increased, accounting for 91.8% of total zinc. The content of zinc in the form of ferrite significantly reduced after sulfidation roasting, which accounted for 79.65% of total zinc in ZLR but only occupied 8.0% of total zinc in sulfidation products. The results indicated that the zinc ferrite in ZLR can be efficiently converted into zinc sulfide by sulfidation roasting. In addition, the content of zinc in the form of sulfate, oxide and silicate also declined after sulfidation roasting, demonstrating that not only zinc in ferrite state but also zinc in sulfate, oxide and silicate states can be converted into zinc sulfide during the roasting.
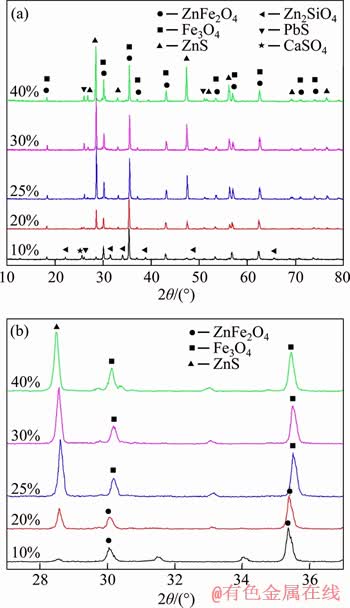
Figure 4 XRD patterns of sulfidation products obtained under different FeS2 dosages (a) and enlarged patterns (b)
3.2.2 Particle size
The particle size will affect the flotation of sulfidation products, thus the variation of particle size before and after sulfidation roasting was investigated. It can be seen from Figure 6 that both the mass fraction curve and the cumulative size distribution curve shifted to the right. Most of the particles in ZLR were distributed within the particle size range of 1-10 ��m, whereas the particle size of sulfidation products was concentrated in range of 3-90 ��m. The change of median diameter (D50) from 2.61 ��m to 17.03 ��m after sulfidation roasting also reflected the increase of particle size. The results show that the particle size of the sulfidation products obviously increased, which might affect the floatation recovery ZnS in sulfidation products [26].
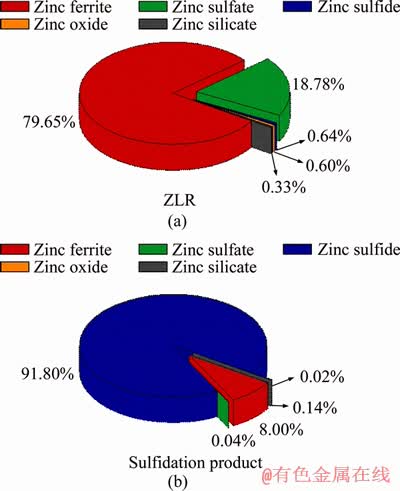
Figure 5 Chemical phase composition of Zn in ZLR (a) and sulfidation product (b)
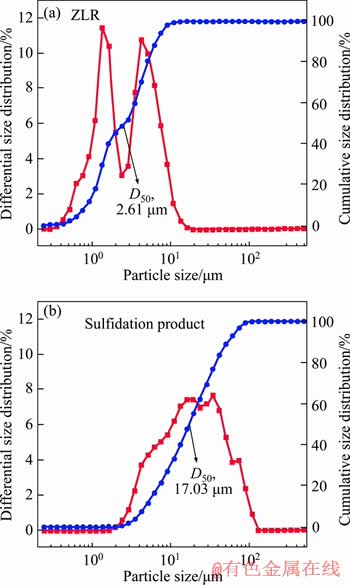
Figure 6 The particle size of ZLR (a) and sulfidation product (b)
3.2.3 Morphology
In order to study the morphological changes of ZLR during sulfidation roasting process, the sulfidation products obtained at different roasting time were analyzed. Many block crystals can be seen in ZLR from Figure 7(a). The crystals varied in size and irregularly distributed. After sulfidation roasting, the block crystals were converted into fine particles (Figure 7(b)). These particles seemed to be in an amorphous state with a rough surface and agglomerated together. With the increase of roasting time, some large particles with a smooth surface were found. Moreover, the size of these particles increased with the prolonging of the roasting time, which was consistent with the results of Figure 6. Finally, the sulfidation products were composed of large and fine particles, which adhered to each other closely (Figures 7(e) and 7(f)).
According to Figure 4, ZnS and Fe3O4 are the main minerals phased in sulfidation products. To ascertain the occurrence state of ZnS and Fe3O4, the sulfidation products with the roasting time of 60 min were characterized with SEM-EDS.Figure 8 shows that the fine particles in point A might be Fe3O4, and the large particles with a smooth surface in points B and C might be ZnS. However, these two kinds of particles embedded with each other rather than existed separately. In order to separate iron and zinc by magnetic separation and flotation, the sulfidation products should be crushed to achieve the dissociation of ZnS and Fe3O4 from each other.

Figure 7 SEM images of ZLR (a) and its sulfidation products obtained under different roasting time of 15 min (b),30 min (c), 45 min (d), 60 min (e) and 120 min (f)
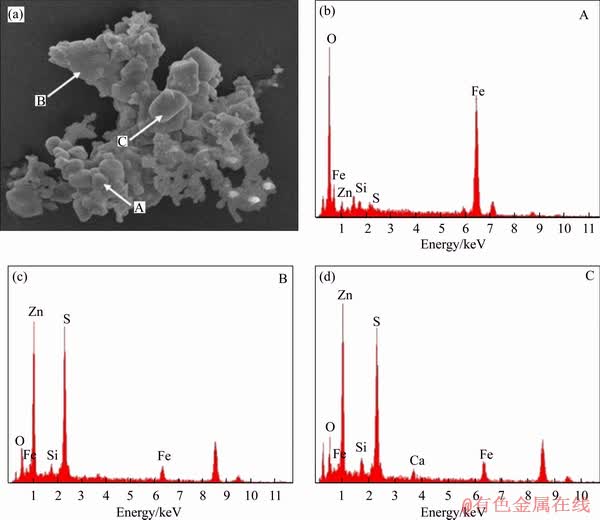
Figure 8 SEM image (a) and corresponding EDS patterns (b, c, d) of sulfidation products with roasting time of 60 min
3.3 Recovery of zinc and iron in sulfidation products
3.3.1 Dissociation of ZnS and Fe3O4 by ball milling
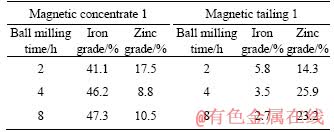
In order to facilitate the recovery of zinc and iron in subsequent processes, the sulfidation products were pretreated by ball milling to achieve the dissociation of ZnS and Fe3O4. According to Figure 2, the grades of zinc and iron after magnetic separation 1 were used to optimize the milling time, and the results were presented in Table 3. When the milling time varied from 2 h to 8 h, the grade of iron in concentrates increased from 41.1% to 47.3%, whereas the grade of zinc decreased and then increased. Table 3 also presents that with the increase of ball milling time, the grade of zinc in tailings increased firstly and then decreased, and the grade of iron decreased gradually. To achieve the separation of zinc and iron by magnetic separation, it is necessary to enrich iron in the concentrates and zinc in the tailings as much as possible. When the ball milling time was 4 h, iron and zinc were effectively enriched in concentrates and tailings, respectively. Therefore, the ball milling time of 4 h was selected.
Table 3 Results of ball milling time on zinc and iron grade from magnetic separation 1
3.3.2 Recovery of zinc and iron
After ball milling pretreatment and magnetic separation 1, the recovery of zinc and iron was performed by flotation and magnetic separation 2, respectively. The results are presented in Table 4.
After magnetic separation 2, the iron was further enriched in magnetic concentrates, whose grade and recovery reached 52.3% and 90.5%, respectively. The grade of zinc decreased in magnetic concentrates 2 but increased in magnetic tailings 2. The enrichment of zinc was achieved by the flotation of magnetic tailings 1. Zinc grade in flotation concentrates reached 31.7%, and the recovery of zinc was 76.6%. Based on these results, it can be seen that the separation and recovery of zinc and iron have been achieved.
Table 4 Results of magnetic separation 2 and flotation
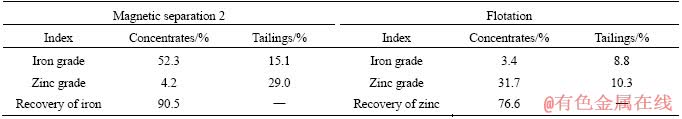
Figure 9 shows the XRD patterns of magnetic concentrates 2 and flotation concentrates. Strong Fe3O4 peaks were obviously found in Figure 9(a), indicating that Fe3O4 was the major mineral phase and ZnS, PbS and silicon dioxide (SiO2) were the minor mineral phases in magnetic concentrates 2. In contrast, strong ZnS and PbS peaks and weak Fe3O4 peaks were observed in Figure 9(b). These results reveal that Fe3O4 can be concentrated in magnetic concentrates 2, and ZnS is mainly enriched in flotation concentrates, which agrees with the results shown in Table 3.
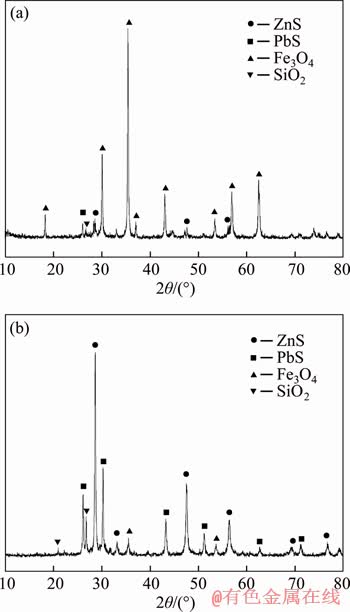
Figure 9 XRD patterns of magnetic concentrates (a) and flotation concentrates (b)
4 Conclusions
In this study, the sulfidation roasting of ZLR with pyrite was investigated for the recovery of zinc and iron. Pyrite dosage, roasting temperature and roasting time had a significant influence on the sulfidation of zinc in ZLR. The optimal conditions of sulfidation roasting were determined to be pyrite dosage of 30%, roasting temperature of 900 ��C and roasting time of 60 min. Under these conditions, the sulfidation percentage of zinc in ZLR reached 91.8%. XRD analysis showed that zinc in ZLR was converted into ZnS, whereas iron was transformed into Fe3O4 after sulfidation roasting. However, sulfides and Fe3O4 embedded with each other in sulfidation products. Ball milling pretreatment is beneficial to the dissociation of ZnS and Fe3O4, and the ball milling time was determined to be 4 h. By ball milling pretreatment, magnetic separation and flotation, iron and zinc can be concentrated in magnetic concentrates and flotation concentrates, respectively. Results showed that the grades of iron and zinc were 52.3% and 31.7% and the recoveries of iron and zinc were 90.5% and 76.6%. Further XRD analysis confirmed that Fe3O4, ZnS and PbS were the major mineral phases of magnetic concentrates and floatation concentrates, respectively. In summary, this study provides a feasible method for the recovery of zinc and iron in zinc leaching residue.
References
[1] OZVERDI A, ERDEM M. Environmental risk assessment and stabilization/solidification of zinc extraction residue: I Environmental risk assessment [J]. Hydrometallurgy, 2010, 100(3, 4): 103-109. DOI: 10.1016/j.hydromet.2009.10.011.
[2] EJTEMAEI M, GHARABAGHI M, IRANNAJAD M. A review of zinc oxide mineral beneficiation using flotation method [J]. Adv Colloid Interface Sci, 2014, 206: 68-78. DOI: 10.1016/j.cis.2013.02.003.
[3] KE Yong, CHAI Li-yuan, MIN Xiao-bo, TANG Chong-jian, CHEN Jie, WANG Yan, LIANG Yan-jie. Sulfidation of heavy-metal-containing neutralization sludge using zinc leaching residue as the sulfur source for metal recovery and stabilization [J]. Minerals Engineering, 2014, 61: 105-112. DOI: 10.1016/j.mineng.2014.03.022.
[4] CHEN T T, DUTRIZAC J E. Mineralogical changes occurring during the fluid-bed roasting of zinc sulfide concentrates [J]. The Journal of The Minerals, Metals & Materials Society, 2004, 56(12): 46-51. DOI: 10.1007/ s11837-004-0235-y.
[5] YU Gang, PENG Ning, ZHOU Lan, LIANG Yan-jie, ZHOU Xiao-yuan, PENG Bing, CHAI Li-yuan, YANG Zhi-hui. Selective reduction process of zinc ferrite and its application in treatment of zinc leaching residues [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(8): 2744-2752. DOI: 10.1016/S1003-6326(15)63899-7.
[6] LECLERC N, MEUX E, LECUIRE J M. Hydrometallurgical extraction of zinc from zinc ferrites [J]. Hydrometallurgy, 2003, 70(1-3): 175-183. DOI: 10.1016/s0304-386x(03) 00079-3.
[7] TURAN M D, S ALTUNDOGAN H S, TUMEN F. Recovery of zinc and lead from zinc plant residue [J]. Hydrometallurgy, 2004, 75(1-4): 169-176. DOI: 10.1016/j.hydromet.2004.07. 008.
[8] HOLLOWAY P C, ETSELL T H, MURLAND A L. Roasting of La Oroya zinc ferrite with Na2CO3 [J]. Metallurgical and Materials Transactions B, 2007, 38(5): 781-791. DOI: 10.1007/s11663-007-9082-x.
[9] ZHANG Ya-li, YU Xian-jin, LI Xiao-bin. Zinc recovery from franklinite by sulphation roasting [J]. Hydrometallurgy, 2011, 109(3, 4): 211-214. DOI: 10.1016/j.hydromet.2011. 07.002.
[10] LI Chao, SUN Heng-hu, BAI Jing, LI Long-tu. Innovative methodology for comprehensive utilization of iron ore tailings: Part 2: The residues after iron recovery from iron ore tailings to prepare cementitious material [J]. Journal of Hazardous Materials, 2010, 174(1-3): 78-83. DOI: 10.1016/j.jhazmat.2009.09.019.
[11] YU Jian-wen, HAN Yue-xin, LI Yan-jun, GAO Peng. Recent advances in magnetization roasting of refractory iron ores: A technological review in the past decade [J]. Mineral Processing and Extractive Metallurgy Review, 2019. DOI: 10.1080/08827508.2019.1634565.
[12] DENG Jin-chuan, FENG Xin, QIU Xin-hong. Extraction of heavy metal from sewage sludge using ultrasound-assisted nitric acid [J]. Chemical Engineering Journal, 2009, 152(1): 177-182. DOI: 10.1016/j.cej.2009.04.031.
[13] SADEGH S M, BAFGHI M S, MORADKHANI D, ILKHCHI M O. A review on hydrometallurgical extraction and recovery of cadmium from various resources [J]. Minerals Engineering, 2007, 20(3): 211-220. DOI: 10.1016/j.mineng.2006.07.001.
[14] LIANG Yan-jie, CHAI Li-yuan, MIN Xiao-bo, TANG Chong-jian, ZHANG Hai-jing, KE Yong, XIE Xian-de. Hydrothermal sulfidation and floatation treatment of heavy-metal-containing sludge for recovery and stabilization [J]. Journal of Hazardous Materials, 2012, 217-218: 307-314. DOI: 10.1016/j.jhazmat.2012.03.025.
[15] WANG Jun, LU Jin-feng, ZHANG Qi-wu, SAITO F. Mechanochemical sulfidization of nonferrous metal oxides by grinding with sulfur and Iron [J]. Industrial & Engineering Chemistry Research, 2003, 42(23): 5813-5818. DOI: 10.1021/ie030046b.
[16] KUCHAR D, FUKUTA T, ONYANGO M S, MATSUDA H. Sulfidation of zinc plating sludge with Na2S for zinc resource recovery [J]. Journal of Hazardous Materials, 2006, 137(1): 185-191. DOI: 10.1016/j.jhazmat.2006.05.037.
[17] LI Yong, WANG Ji-kun, WEI Chang, LIU Chun-xia, JIANG Ji-bo, WANG Fan. Sulfidation roasting of low grade lead�Czinc oxide ore with elemental sulfur [J]. Minerals Engineering, 2010, 23(7): 563-566. DOI: 10.1016/j.mineng. 2010.01.004.
[18] KUCHAR D, FUKUTA T, ONYANGO M S, MATSUDA H. Sulfidation treatment of copper-containing plating sludge towards copper resource recovery [J]. Journal of Hazardous Materials, 2006, 138(1): 86-94. DOI: 10.1016/j.jhazmat. 2006.01.052.
[19] ZHENG Yong-xin, LIU Wei, QIN Wen-qin, HAN Jun-wei, YANG Kang, LUO Hong-lin, WANG Da-wei. Improvement for sulphidation roasting and its application to treat lead smelter slag and zinc recovery [J]. Canadian Metallurgical Quarterly, 2015, 54(1): 92-100. DOI: 10.1179/1879139514y. 0000000155.
[20] MIN Xiao-bo, XUE Ke, KE Yong, ZHOU Bo-sheng, LI-YANG Wen-jun, WANG Qing-wei. Sulfidation roasting of hemimorphite with pyrite for the enrichment of Zn and Pb [J]. The Journal of The Minerals, Metals & Materials Society, 2016, 68(9): 2435-2442. DOI: 10.1007/s11837-016- 1986-y.
[21] KE Yong, PENG Ning, XUE Ke, MIN Xiao-bo, CHAI Li-yuan, PAN Qing-lin, LIANG Yan-jie, XIAO Rui-yang, WANG Yun-yan, TANG Chong-jian, LIU Hui. Sulfidation behavior and mechanism of zinc silicate roasted with pyrite [J]. Applied Surface Science, 2018, 435: 1011-1019. DOI: 10.1016/j.apsusc.2017.11.202.
[22] MIN Xiao-bo, ZHOU Bo-sheng, KE Yong, CHAI Li-yuan, XUE Ke, ZHANG Chun, ZHAO Zong-wen, SHEN Chen. Sulfidation behavior of ZnFe2O4 roasted with pyrite: Sulfur inducing and sulfur-oxygen interface exchange mechanism [J]. Applied Surface Science, 2016, 371: 67-73. DOI: 10.1016/j.apsusc.2016.02.229.
[23] MIN Xiao-bo, LI-YANG Wen-jun, KE Yong, SHI Mei-qing, CHAI Li-yuan, XUE Ke. Fe-FeS2 adsorbent prepared with iron powder and pyrite by facile ball milling and its application for arsenic removal [J]. Water Science & Technology, 2017, 76(1): 192-200. DOI: 10.2166/wst.2017. 204.
[24] ZHANG Hui-bin. Chemical phase analysis of ore and industrial product [M]. Beijing: Metallurgical Industry Press, 1992. (in Chinese)
[25] KE Yong, CHAI Li-yuan, LIANG Yan-jie, MIN Xiao-bo, YANG Zhi-hui, CHEN Jie, YUAN Sheng. Sulfidation of heavy-metal-containing metallurgical residue in wet-milling processing [J]. Minerals Engineering, 2014, 53: 136-143. DOI: 10.1016/j.mineng.2013.07.013.
[26] FENG D, ALDRICH C. Effect of particle size on flotation performance of complex sulphide ores [J]. Minerals Engineering, 1999, 7(12): 721-731. DOI: 10.1016/s0892- 6875(99)00059-x.
(Edited by HE Yun-bin)
���ĵ���
п�����ͻ��������ջ���п��
ժҪ��п�����к��д���п�������м۽�����������п������Ҫ������п�ȸ���������ʽ���棬���Է���ͻ��ա����о�������漰���ա���ѡ��ѡ��������п������п�������¹��ա��Ի�����Ϊ����п�����������գ���п�����ֱ�ת��Ϊ��п���������������о��˱����¶ȡ�����ʱ�估��������������п������п����ת����Ӱ�졣�������������ѹ��������£�п������п�����ʿɴ�91.8%�����⣬�о�������ĥ������������п�����ķ���ͻ��ա���ĥԤ������ͨ����ѡ��ѡʵ������������п�ĸ�������������Ʒλ��52.3%��п����пƷλ��31.7%���ɼ����÷�����Ч��ʵ����п������п�����ķ�������ա�
�ؼ��ʣ�п���������գ����գ�п��������ѡ����ѡ
Foundation item: Project(2018YFC1900305) supported by the National Key R&D Program of China; Project(51825403) supported by the National Science Foundation for Distinguished Young Scholars, China; Projects(51634010, 51474247, 51904354) supported by the National Natural Science Foundation of China; Project(2019SK2291) supported by the Key Research and Development Program of Hunan Province, China
Received date: 2019-11-15; Accepted date: 2020-02-29
Corresponding author: KE Yong, PhD, Associate Professor; Tel: +86-731-88830511; E-mail: keyong000ke@csu.edu.cn; ORCID: 0000- 0001-7985-5906