
Photocatalytic activity of CuO towards HER in catalyst from oxalic acid solution under simulated sunlight irradiation
YAO Mao-hai(Ҧï��)1, TANG You-gen(���и�)1, ZHANG Li(�� ��)2,
YANG Hai-hua(���)1, YAN Jian-hui(�ֽ���)1, 2
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. Department of Chemistry and Chemical Engineering,
Hunan Institute of Science and Technology, Yueyang 414000, China
Received 8 September 2009; accepted 14 January 2010
Abstract: CuO was synthesized by thermal decomposition of Cu(NO3)2?3H2O at various temperatures and characterized by powder X-ray diffractometry (XRD) as well as scanning electron microscopy (SEM). The effects of calcination temperature, category of sacrificial reagent, initial sacrificial reagent concentration, and Ag loading content on the photocatalytic activity of the as-obtained CuO sample were investigated. The results show that the as-obtained CuO exhibits high activity for photocatalysis of H2 evolution reaction (HER) in oxalic acid solution under simulated sunlight irradiation. The highest photocatalytic activity of the as-obtained CuO was achieved at the calcination temperature of 1 000 ��C, and oxalic acid was used as the sacrificial reagent with the concentration 0.05 mol/L. H2 evolution rate is as high as 2.98 mmol/(h��g) with 2% (mass fraction) loaded Ag. The possible photocatalytic reaction mechanism on the CuO photocatalyst for HER in oxalic acid solution was also discussed.
Key words: copper oxide (CuO); hydrogen evolution reaction (HER); photocatalytic activity; oxalic acid; simulated sunlight
1 Introduction
When considering the environmental pollution and energy crisis, H2 is deemed to be the best energy source to resolve this two problems in the future. For this purpose, searching new photocatalysts for photocatalytic H2 evolution, which can utilize solar energy from renewable resources and natural energy sources, has been attracted wide attention[1-2] since the Honda-Fujishima effect was reported in 1972[3]. Despite a considerable progress has been made, the conversion efficiency of solar energy to H2 energy is still undesirable. In this work, we introduced CuO as a photocatalyst for H2 evolution from the oxalic acid solution under simulated sunlight irradiation.
Copper oxide (CuO), a well-known p-type semiconductor with a narrow band gap (Eg = 1.2 eV), is an important industrial material and has been widely used, such as gas sensors[4], electrode materials[5], magnetic ceramics[6], hydrogen storage materials[7], solar cells[8], and photocatalysis[9]. At present, as for the photocatalysis, most of the investigations have been focused on the composite of CuO/TiO2. BANDARA and co-workers[10] investigated the photocatalytic H2 production activity of CuO/TiO2 in methanol solution under UV light irradiation. JIN et al[11] reported CuO/TiO2 photocatalytic H2 generation by eosin-sensitization under visible light irradiation. However, to our knowledge, there has been no report on pure CuO for photocatalytic H2 evolution under simulated sunlight irradiation. In order to utilize sunlight to produce H2 from water, the semiconductor photocatalyst materials should adsorb visible light irradiation to generate electron-hole pairs, and the bottom of the conduction band level is more negative than the redox potential of H+/H2 (0 eV vs NHE). The band gap of CuO is 1.2 eV, and thus CuO can efficiently absorb sunlight, but is not active enough to release H2 from pure water because of the position of the conduction band level. Therefore, selecting an excellent sacrificial reagent is a key factor to photocatalytic H2 evolution with pure CuO as photocatalyst under simulated sunlight irradiation.
Sacrificial reagents (electron donors or hole scavengers) play an important role in photocatalytic H2 evolution. It was reported[12] that sacrificial reagents could not only effectively restrain the recombination of the electrons and holes, but also prove whether photocatalysts suitable valence band position for oxidizing H2O to O2 or suitable conduction band position for reducing H2O to H2. There have been many reports on sacrificial reagents, among which some act as electron donors for water reduction (such as methanol, formic acid, oxalic acid, Na2SO3 and Na2S)[13-16], and some act as electron scavengers for water oxidation (such as AgNO3)[17]. Meanwhile, a few of redox couples have been used in Z-scheme photocatalysis system for overall water splitting, such as Ge3+/Ge4+, Fe2+/Fe3+ and IO3-/I-[18-20]. Here, we selected oxalic acid as the sacrificial reagent. Oxalic acid is not only a common pollutant in industrial wastewater[21], but also a strong reductive reagent as electron donors[22]. The photocatalytic decomposition of organic pollutants and photocatalytic H2 evolution take place simultaneously in this work.
2 Experimental
2.1 Photocatalyst preparation
All the chemicals used in experiments were in analytical grade and with no further treatment. The photocatalyst CuO was synthesized by thermal decomposition of Cu(NO3)2?3H2O at various temperatures. In a typical process, Cu(NO3)2?3H2O was milled for 10 min in an agate mortar, and calcined for 6 h in air with a heating rate of 10 ?C/min. The calcined sample was slowly cooled to room temperature and milled for 10 min again to get the final product.
The CuO powders with different Ag-loaded contents were prepared by photodeposition method. AgNO3 was used as Ag precursor. A typical preparation procedure was described as follows. CuO (3.0 g) powers were dispersed into AgNO3 solution containing MeOH (1%, mass fraction) in a Pyrex glass reaction vessel by ultrasonic treatment for 30 min. The as-obtained mixture was photoirradiated under a 250 W high-pressure Hg lamp for 2 h under continuous stirring. The radiative wavelength of the lamp was about 300-400 nm and the intensity of illumination was 15 �� 103 ��W/cm2, then the suspension was filtered, washed and dried at 373 K to get the Ag-loaded photocatalyst.
2.2 Photocatalyst characterization
The crystal structure of the as-prepared photocatalysts was identified by powder X-ray diffraction method (XRD, Bruker D8) using Cu K�� radiation (��= 1.5418 ?) with a scanning angle (2��) of 10��-85�� at a scan speed of 4 (��)/min and a voltage of 40 kV and current of 300 mA. The surface morphology of the samples was characterized with a JEOL JSM-5600LV scanning electron microscope (SEM) operated at 25 kV.
2.3 Photocatalytic activity measurement
The photocatalytic activity of the as-prepared photocatalyst was evaluated by measuring the amount of H2 evolution in the sacrificial reagent aqueous solution under simulated sunlight irradiation. The H2 production experiment was carried out in an inner irradiation-type reaction vessel made of Pyrex, and a Xe (250 W) lamp was used as the simulated sunlight source. The reaction setup was schemed in Fig.1. In all experiments, to keep the reaction temperature at room temperature, water was used as the external circulation cooling medium, and electric fan and vacuum pump were employed as the internal cooling method. Prior to irradiation, certain amount of the as-prepared photocatalyst (0.6 g) was dispersed in 100 mL of distilled water in an ultrasonic bath and was put in the reactor. Then, the reactor was left in dark and nitrogen flux was introduced into the system for 30 min to remove O2. After that, the photocatalytic reaction vessel was closed and exposed to the light irradiation. During the irradiation, the mixture was suspended by using a magnetic stirrer within the quartz cell. The produced H2 was collected in a water nanometer and analyzed by gas chromatography (TCD, N2 as gas carrier, zeolite NaX column). And an eliminator containing saturated NaOH solution was placed in front of the collector of H2 to remove CO2. The pure H2 was collected in the gas collector (Fig.1, D) by displacing the water in the collector, and the volume of the as-generated H2 was taken as the volume of the displaced water (Fig.1, H) at different intervals of irradiation time. Some
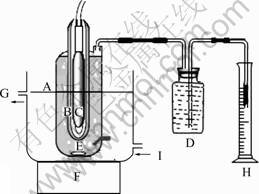
Fig.1 Device for photocatalytic H2 production: (A) Reactor; (B) Quartz jacket; (C) Xenon lamp; (D) Gas collector; (E) Magnetic bar; (F) Magnetic stirrer; (G) Outlet of cooling water; (H) Volumetric cylinder; (I) Entry of cooling water
processes were repeated three times and the results were reproducible within the experiment errors (��4%).
3 Results and discussion
3.1 Characterization
Fig.2 shows the XRD patterns of the as-obtained CuO samples at different calcination temperatures. The indexed results are in good agreement with the monoclinic lattice of CuO reported in the JCPDS database card (No. 80-1916). No peaks from any other phases of CuO or impurities were observed, indicating high purity of the as-obtained CuO catalysts. With increasing the calcination temperature, the width at half maximum of diffraction peaks becomes narrower and the diffraction intensity becomes stronger, indicating the improvement of crystallinity of the CuO samples.
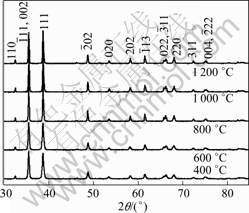
Fig.2 XRD patterns of CuO photocatalysts at different calcination temperatures
Fig.3 illustrates the SEM images of the CuO photocatalysts calcined at different temperatures. The morphologies of all the samples synthesized by thermal decomposition are in irregular, and the particle sizes increase gradually with increasing calcination temperature because of aggregation.
3.2 Photocatalytic activity of CuO towards HER
3.2.1 Effect of calcination temperature
Fig.4 depicts the photocatalytic H2 evolution activity of CuO as a function of the calcination temperature. Obviously, in the range of the calcination temperature from 400 to 1 000 ��C, the photocatalytic activity of CuO is improved with increasing calcination temperature and achieves maximum value at 1 000 ��C. With further increasing the calcination temperature, however, the photocatalytic activity decreases drastically. As mentioned above, the improvement of CuO crystallinity can be evidenced by the narrowing of the half-width of diffraction peaks and the increase of

Fig.3 SEM images of CuO photocatalyst calcined at different temperatures: (a) 400 ��C; (b) 1 000 ��C; (c) 1 200 ��C
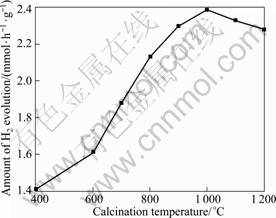
Fig.4 Effect of calcination temperature on H2 evolution activity over CuO photocatalyst (Reaction conditions: photocatalyst concentration, 1 g/L; initial oxalic acid concentration, 0.05 mol/L; distilled water volume, 600 mL)
the diffraction intensity. Higher crystallinity is a positive factor for improving the photocatalytic activity because the higher the crystalline quality is, the smaller the number of defects is. The defects act as trapping and recombination centers for photogenerated electrons and holes, resulting in a decrease in the photocatalytic activity. On the other hand, as shown in Fig.3, with increasing the calcination temperature, the particle sizes increase because of aggregation. The particle size determines the migration distance for photogenerated electrons or holes to reaction sites at the particle surface, and bigger size results in the increase of the recombination probability, which is a negative factor to the photocatalytic activity. The resultant photocatalytic activity is dominated by the balance between these two factors. However, higher crystallinity is often required rather than bigger size for water splitting because the recombination between photogenerated electrons and holes is especially a serious problem for uphill reactions[3].
3.2.2 Effect of sacrificial reagent
Fig.5 describes the photocatalytic activity of CuO in different kinds of sacrificial reagents under simulated sunlight irradiation. The results prove that not all the selected sacrificial reagents as electron donors are appropriate for H2 evolution using the CuO photocatalyst. Especially, methanol is a good electron donor in the process of reducing H2O to H2 for many photocatalysts[23-24], but for CuO photocatalyst, it is not efficient. The maximum values for H2 evolution in Na2S, CH3COOH, and H2C2O4 solutions are 0.30, 1.82, and 2.46 mmol/(h��g), respectively. It has demonstrated that oxalic acid is more efficient than other sacrificial reagents, and acts as the sacrificial reagent to consume the photogenerated holes so as to resist the
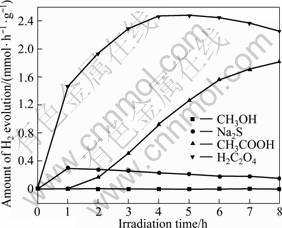
Fig.5 Effect of category of sacrificial reagent on H2 evolution activity over CuO photocatalyst (Reaction conditions: photocatalyst concentration, 1 g/L; initial sacrificial reagent concentration, 0.05 mol/L; distilled water volume, 600 mL)
recombination of the photogenerated electrons-holes pairs in the photocatalytic reaction procedure. This is probably due to the influence of H+ ions, which act as the hydrated electron scavenger, converting to H atoms and further combining to molecular hydrogen. The reaction conditions in the present experiments differ from the acidity of sacrificial reagent, and the acidic ranking of sacrificial reagents is H2C2O4 > CH3COOH > CH3OH > Na2S, basically, in accordance with the law of the increasing amount of H2 evolution. However, CuO is unstable in the oxalic acid solution. In the end of photoirradiation, few NaOH solutions have been dripped into the reaction vessel and some blue deposition appeared. This reveals that Cu2+ exists in the solution through photoirradiating reaction.
3.2.3 Effect of initial concentration of oxalic acid
Fig.6 illustrates the effect of initial oxalic acid concentration on the photocatalytic activity of CuO. No H2 is detected without any oxalic acid in the aqueous solution. However, the presence of oxalic acid can markedly improve the amount of H2 evolution. The results demonstrate that oxalic acid is a strong reductive reagent and can effectively consume the photogenerated holes. With the increase of the initial oxalic acid concentration, the photocatalytic H2 evolution activity is enhanced obviously and the optimized value is achieved at 0.05 mol/L. But with further increasing the concentration of oxalic acid, the photocatalytic activity decreases pronouncedly. It is possible that oxalic acid has three forms in the aqueous solution, and the reactivity decreases in the order of HC2O4- > C2O42- >H2C2O4[25]. In the solution of an appropriate concentration with the HC2O4- form as the prevailing species of oxalic acid, the reactivity is the best and the consumption of the photogenerated holes is more
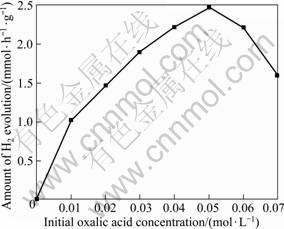
Fig.6 Effect of initial concentration of oxalic acid on photocatalytic H2 evolution activity of CuO photocatalyst (Reaction conditions: photocatalyst concentration, 1 g/L; distilled water volume, 600 mL)
effective than other two forms. H2C2O4 is dominant when the addition of oxalic acid is excessive, leading to a decrease of the photocatalytic activity of CuO.
3.2.4 Effect of Ag loading content
Fig.7 illustrates the photocatalytic activity of CuO samples with different contents of Ag loading under simulated sunlight irradiation. The results clearly reveal that the photocatalytic activity increases with increasing Ag contents to a maximum of 2% (mass fraction), and the highest H2 evolution rate is 2.98 mmol/(h��g). While the Ag loading content exceeds the optimum value, the photocatalytic activity decreases markedly. As a noble metal, Ag is a good co-catalyst on the surface of photocatalysts. Generally, co-catalyst is used to introduce active sites[3], which behaves as electron traps to promote the separation of the electrons and holes in/on the photocatalyst and decline the probability of photogenerated electron�Chole pairs recombination, resulting in the increase of photocatalytic activity. When the Ag loading content is too low, the size of Ag particles covered on the surface of catalyst is small and Ag is uniformly dispersed. The small amount of the Ag loading may cause the failure of effective separation of the electrons and holes, and thus leads to the low photocatalytic activity. When the Ag loading content is higher than the optimum value of 2% (mass fraction), Ag particles covered on the surface of catalyst are excess, causing the aggregation of Ag particles and the formation of larger size Ag clusters, which can scatter the visible light, leading to the failure to trigger the catalyst and the decrease of photocatalytic activity[13, 15].
3.3 Mechanism of H2 production in oxalic acid solution
The reaction is initiated by the photoexcitation of
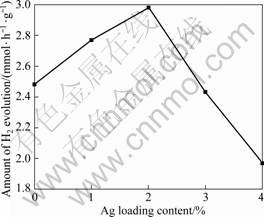
Fig.7 Effect of different contents of Ag deposited CuO on photocatalytic H2 evolution activity (Reaction conditions: photocatalyst concentration, 1 g/L; initial oxalic acid concentration, 0.05 mol/L; distilled water volume, 600 mL)
CuO particles to form photogenerated electron-hole pairs. Then the photogenerated electron (e-) and hole (h+) can be transferred to electron acceptor H+ and electron donors C2O42-, HC2O4-, H2C2O4 and surface hydroxyl groups on CuO, respectively. The formed hydroxyl radical can continuously oxidize oxalic acid species. Meanwhile, CuO is unstable in the oxalic acid solution. According to Refs.[21, 25], the reaction mechanism is summarized as
CuO+hv��CuO(e-+h+) (1)
H++e-��H?��1/2H2 (2)
OH-+h+��OH? (3)
HC2O4-+h+��HC2O4? (4)
HC2O4-+OH?��HC2O4?+OH- (5)
HC2O4?��?CO2-+CO2+H+ (6)
?CO2-+H+��CO2+1/2H2 (7)
CuO+HC2O4-��CuC2O4+OH- (8)
CuC2O4+2h+��Cu2++?C2O4 (9)
?C2O4+H+��2CO2+1/2H2 (10)
4 Conclusions
1) No H2 was detected for CuO photocatalyst in pure water under simulated sunlight irradiation. Oxalic acid, used as an excellent sacrificial reagent, can greatly improve the photocatalytic activity of CuO touards HER, and the optimal initial concentration is 0.05 mol/L. However, CuO is unstable in the oxalic acid solution.
2) Calcination temperature also affects the photocatalytic activity. When the calcination temperature is 1 000 ��C, the optimized photocatalytic H2 evolution activity under simulated sunlight irradiation can be achieved.
3) As a co-catalyst depositing on the surface of CuO, Ag also can greatly improve the photocatalytic activity, and the highest H2 evolution amount is 2.98 mmol/(h��g) when the Ag loading content is 2% (mass fraction).
References
[1] OSTERLOH F E. Inorganic materials as catalysts for photochemical splitting of water [J]. Chem Mater, 2008, 20: 35-54.
[2] KUDO A, MISEKI Y. Heterogeneous photocatalyst materials for water splitting [J]. Chem Soc Rev, 2009, 38: 253-278.
[3] FUJISHIMA A, HONDA K. Electrochemical photolysis of water at a semiconductor electrode [J]. Nature, 1972, 238: 37-38.
[4] LI Yue-ming, LIANG Jing, TAO Zhan-liang, CHEN Jun. CuO particles and plates: Synthesis and gas-sensor application [J]. Materials Research Bulletin, 2008, 43: 2380-2385.
[5] ZHANG Hong-xia, ZHANG Mi-lin. Synthesis of CuO nanocrystalline and their application as electrode materials for capacitors [J]. Materials Chemistry and Physics, 2008, 108: 184-187.
[6] ARBUZOVA T I, GIZHEVSKII B A, NAUMOV S V, KOROLEV A V, ARBUZOV V L, SHAL��NOV K V, DRUZHKOV A P. Temporal changes in magnetic properties of high-density CuO nanoceramics [J]. Journal of Magnetism and Magnetic Materials, 2003, 258/259: 342-344.
[7] GAO Peng, CHEN Yu-jin, LU Hai-jiao, LI Xue-fei, WANG Ying, ZHANG Qin. Synthesis of CuO nanoribbon arrays with noticeable electrochemical hydrogen storage ability by a simple precursor dehydration route at lower temperature [J]. International Journal of Hydrogen Energy, 2009, 34: 3065-3069.
[8] MARUYAMA T. Copper oxide thin films prepared by chemical vapor deposition from copper dipivaloylmethanate [J]. Solar Energy Materials and Solar Cells, 1998, 56: 85-92.
[9] BARRECA D, FORNASIERO P, GASPAROTTO A, GOMBAC V, MACCATO C, MONTINI T, TONDELLO E. The potential of supported Cu2O and CuO nanosystems in photocatalytic H2 production [J]. Chem Sus Chem, 2009, 2(3): 230-233.
[10] BANDARA J, UDAWATTA C P K, RAJAPAKSE C S K. Highly stable CuO incorporated TiO2 catalyst for photocatalytic hydrogen production from H2O [J]. Photochem Photobiol Sci, 2005, 4: 857-861.
[11] JIN Zhi-liang, ZHANG Xiao-jie, LI Yue-xiang, LI Shu-ben, LU Gong-xuan. 5.1% apparent quantum efficiency for stable hydrogen generation over eosin-sensitized CuO/TiO2 photocatalyst under visible light irradiation [J]. Catalysis Communications, 2007, 8: 1267-1273.
[12] LIU Hui, YUAN Jian, SHANGGUAN Wen-feng. Photochemical reduction and oxidation of water including sacrificial reagents and Pt/TiO2 catalyst [J]. Energy and Fuels, 2006, 20: 2289-2292.
[13] SREETHAWONG T, LAEHSALEE S, CHAVADEJ S. Use of Pt/N-doped mesoporous assembled nanocrystalline TiO2 for photocatalytic H2 production under visible light irradiation [J]. Catalysis Communications, 2009, 10: 538-543.
[14] ZHANG Yao-jun, ZHANG Li. Preparation of Ru-loaded CdS/Al-HMS nanocomposites and production of hydrogen by photocatalytic degradation of formic acid [J]. Applied Surface Science, 2009, 255: 4863-4866.
[15] YAN Jian-hui, ZHU Yi-rong, TANG You-gen, ZHENG Shu-qin. Nitrogen-doped SrTiO3/TiO2 composite photocatalysts for hydrogen production under visible light irradiation [J]. Journal of Alloys and Compounds, 2009, 472: 429-433.
[16] XING Chan-juan, JING Deng-wei, LIU Mao-chang, GUO Lie-jin. Photocatalytic hydrogen production over Na2Ti2O4(OH)2 nanotube sensitized by CdS nanoparticles [J]. Materials Research Bulletin, 2009, 44: 442-445.
[17] KIM H G, HWANG D W, LEE J S. An undope, single-phase oxide photocatalyst working under visible light [J]. Am Chem Soc, 2004, 126: 8912-8913.
[18] KOZLOVA E A, KOROBKINA T P, VORONTSOV A V. Overall water splitting over Pt/TiO2 catalyst with Ge3+/Ge 4+ shuttle charge transfer system [J]. International Journal of Hydrogen Energy, 2009, 34: 138-146.
[19] KATO A, HORI M, KONTA R, SHIMODAIRA Y, KUDO A. Construction of Z-scheme type heterogeneous photocatalysis systems for water splitting into H2 and O2 under visible light irradiation [J]. Chem Lett, 2004, 33: 1348-1349.
[20] KAZUHIRO S, KAZUAKI M, RYU A, YOSHIMOTO A, HIRONORI A. A new photocatalytic water splitting system under visible light irradiation mimicking a Z-scheme mechanism in photosynthesis [J]. Journal of Photochemistry and Photobiology A: Chemistry, 2002, 148: 71-77.
[21] SONG Shuang, TU Jin-jun, XU Le-jin, XU Xing, HE Zhi-qiao, QIU Jian-ping, NI Jian-guo, CHEN Jian-meng. Preparation of a titanium dioxide photocatalyst codoped with cerium and iodine and its performance in the degradation of oxalic acid [J]. Chemosphere, 2008, 73: 1401-1406.
[22] YANG Hai-hua, YAN Jian-hui, LU Zhou-guang, CHENG Xiang, TANG You-gen. Photocatalytic activity evolution of tetragonal CuFe2O4 nanoparticles for the H2 evolution under visible light irradiation [J]. Journal of Alloys and Compounds, 2009, 476: 715-719.
[23] YOSHIDA H, KAZUHISA H, NISHIMOTO J, KATSUYA S, KATO S, ITOH H, HATTORI T. Hydrogen production from methane and water on platinum loaded titanium oxide photocatalysts [J]. Phys Chem C, 2008, 112: 5542-5551.
[24] LIU J W, CHEN G, LI Z H, ZHANG Z G. Hydrothermal synthesis and photocatalytic properties of ATaO3 and ANbO3 (A=Na and K) [J]. International Journal of Hydrogen Energy, 2007, 32: 2269-2272.
[25] LI Yue-xiang, LU Gong-xuan, LI Shu-ben. Photocatalytic hydrogen generation and decomposition of oxalic acid over platinized TiO2 [J]. Applied Catalysis A: General, 2001, 214: 179-185.
(Edited by LI Xiang-qun)
Foundation item: Project(20876039) supported by the National Natural Science Foundation of China; Project(09JJ3023) supported by Natural Science Foundation of Hunan Province, China
Corresponding author: TANG You-gen; Tel: + 86-731-88830886; E-mail: ygtang@263.com
DOI: 10.1016/S1003-6326(09)60399-X