Preparation of TiO2 photocatalyst loaded with V2O5 for O2 evolution
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2009���6��
�������ߣ������� ����Ԫ ͯ��ϼ ����Ƽ ���ǻ� �ܽ���
����ҳ�룺919 - 924
Key words��TiO2; V2O5; photocatalytic activity; O2 evolution
Abstract: TiO2 photocatalysts loaded with V2O5 were prepared via a modified hydrolysis process, and characterized by X-ray diffraction, transmission electron microscopy, Raman spectra and diffuse reflectance UV-Vis spectra measurements. The photocatalytic activity of V2O5/TiO2 was investigated by employing splitting of water for O2 evolution. The results indicate that V2O5 loading can pronouncedly improve the photocatalytic activity of TiO2 with Fe3+ as an electron acceptor under UV or visible light irradiation. The optimum mass fraction of the loaded V2O5 is 8%, and the largest speed of O2 evolution for 8%V2O5 (mass fraction) loaded TiO2 catalyst is 118.2 ��mol/(L��h) under UV irradiation, and 83.7 ��mol/(L��h) under visible light irradiation.
������Ϣ��supported by the Natural Science Foundation of Hunan Province, China
J. Cent. South Univ. Technol. (2009) 16: 0919-0925
DOI: 10.1007/s11771-009-0153-9
![]()
GAO You-liang(������)1,2, CHEN Qi-yuan(����Ԫ)1, TONG Hai-xia(ͯ��ϼ)1, HU Hui-ping(����Ƽ)1,
QIAN Dong(Ǯ ��)1, YANG Ya-hui(���ǻ�)1, ZHOU Jian-liang(�ܽ���)1
(1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. School of Materials Science and Engineering, Jingdezhen Ceramic Institute, Jingdezhen 333403, China)
Abstract: TiO2 photocatalysts loaded with V2O5 were prepared via a modified hydrolysis process, and characterized by X-ray diffraction, transmission electron microscopy, Raman spectra and diffuse reflectance UV-Vis spectra measurements. The photocatalytic activity of V2O5/TiO2 was investigated by employing splitting of water for O2 evolution. The results indicate that V2O5 loading can pronouncedly improve the photocatalytic activity of TiO2 with Fe3+ as an electron acceptor under UV or visible light irradiation. The optimum mass fraction of the loaded V2O5 is 8%, and the largest speed of O2 evolution for 8%V2O5 (mass fraction) loaded TiO2 catalyst is 118.2 ?mol/(L��h) under UV irradiation, and 83.7 ?mol/(L��h) under visible light irradiation.
Key words: TiO2; V2O5; photocatalytic activity; O2 evolution
1 Introduction
Nowadays, more and more attention is paid to the research on photocatalytic decomposition of water because of the high efficiency, cleanness and other advantages of hydrogen energy. FUJISHIMA and HONDA[1] used n-type semiconductor TiO2 to split water and produce H2 in the 20th century. As titania with large band gap is cheap, nontoxic, stable and reclaimable[2], it is a very promising kind of semiconductor material that can be photo-excited to generate electron-hole pairs on its surface and create a strong oxidation ability when it absorbs light with energy greater than its band gap, and the electrons in the valence band are excited to the conduction band. TiO2 is an effective photocatalyst under UV irradiation for decomposing volatile organic compounds[3], smart-windows[4] and water electrolysis for hydrogen production[5]. However, pure titania has low photo-activity and slow photolysis speed for photocatalysis of water and is very difficult to be practically used. So it is necessary to improve its photo-activity and enhance the photolysis speed for water splitting. Generally, the photocatalytic activity of semiconductor can be improved through doping or loading semiconductor, such as Ru3+/TiO2[6], TiO2-Al2O3, V2O5/TiO2-Al2O3[7], TiO2-La2O3[8], TiO2- WO3[9], V2O5-WO3-TiO2[10]. And most of these doped semiconductors were used to photo-decompose organic pollutants, and some of them were used to split water with low photocatalytic efficiency[6]. When TiO2 is combined with other oxides such as V2O5, the spectral range of absorption is increased due to the small band gap of V2O5 (2.2 eV, ��=564 nm) compared with that of TiO2 (3.0 eV, ��=413 nm). This allows light with �ˡ� 564 nm to be absorbed by the heterogeneous structure, and the photocatalytic activity can be extended to a larger spectral range. In addition, V2O5 can act as an electron scavenger and pull the photo-generated conduction band electrons of TiO2 to its own conduction band, which keeps the electron-hole pairs apart longer (charge separation), and improves the overall photocatalytic performance. Vanadium supported on titania is commonly used as a catalyst for a number of industrially important reactions, including the selective oxidation reactions of o-xylene[11], ammoxidation of hydrocarbons[11-12], as well as selective reduction of NOx with NH3 in the presence of O2[13]. JIANG et al [14] reported that the spreading of vanadium over TiO2 support leads to a modification of the chemical-physical peculiarities of the former and to an enhancement of its catalytic properties. However, there are few reports about V2O5/TiO2 used as a catalyst for splitting water. As you know, the reaction of photocatalytic splitting water is carried out via two half reactions, of which, one is the photo-reduction with H2 evolution, the other is the photo-oxidation with O2 evolution. And the latter is more difficult to achieve. In this work, by using Fe3+ as electron acceptors, V2O5-TiO2 catalysts prepared by a modified hydrolysis process were employed for the photocatalytic oxidation of water with O2 evolution.
2 Experimental
2.1 Synthesis of rutile TiO2 and V2O5-TiO2 catalysts
TiO2 colloid solution was prepared by the hydrolysis of tetrabutyl titanate [Ti(OC4H9)4]. All the chemical reagents were analytically pure and used without further purification. Ti(OC4H9)4 was dissolved in ethanol, and then the mixture was added to distilled water drop by drop under magnetic stirring. The mixture was stirred for 2 h and then deposited for 6-8 h before being dried at 373 K to obtain a white precursor. The white precursor was ground in a carnelian mortar and loaded in a navicular quartz vessel to be calcined at 1 223 K for 5 h. The white precursor was ground again to obtain rutile TiO2. Rutile TiO2 was dipped in ammonium matavanadate (NH4VO3) solution, then dried, and calcined at 723 K for 5 h. A series of catalysts were obtained with concentrations of 5%, 8%, 10%, 20% and 40% of loaded V2O5, respectively.
2.2 Characterization of V2O5-TiO2 catalysts
X-ray diffraction (XRD) analysis was used to check the coexistence of different crystal phases of the catalyst by a HATCHI D/max2250 powder X-ray diffractometer. The diffraction profiles were recorded with Cu Ka radiation (0.154 056 nm) over a 2�� range from 10? to 90?. A plumbaginous counter with monochromator was used. The X-ray tube was operated at 40 kV and 300 mA.
Diffuse reflectance UV-Vis spectra (UV-DRS) measurements were carried out on a Beijing Purkinje TU-1901 UV/Vis spectrophotometer equipped by a diffuse reflectance accessory with an IS19-1 integrating sphere, and BaSO4 powder was used as the reference.
Raman spectra were recorded on a RT1-30 spectrophotometer. The line at 4 888 nm from an argon ion laser was used as the excitation source. The laser power applied was 326 mW, and the raster width was 201 ��m. The scan was arranged in the wavenumber range of 100-1 100 cm-1.
The results were examined by a HATCHI SP-2305 gas chromatography equipped with a thermal conductivity detector. Argon was used as the carrier gas and the fixed phase was molecule sieve (0.5 nm).
2.3 Photo-activity tests
The photocatalytic reaction was performed using a closed gas-circulating system with an inner irradiation reactor. A light source (250 W high-pressure Hg lamp or 250 W Xe lamp), was covered with a glass jacket made of quartz (cut-off wavelength less than 200 nm). The reactor temperature was kept constant at 293 K using cooling water. A mixture of catalyst (2 g), distilled water (640 mL) and a required amount of Fe2(SO4)3 in the reactor was completely degassed, and then argon gas was introduced into the system. The catalyst powder was suspended by using a magnetic stirrer. The product gas oppressed the liquid in the reactor into the graduated cylinder through the outlet. So the volume of product gas can be read through the graduated cylinder indirectly. The evolution of O2 was detected by gas chromatography.
3 Results and discussion3.1 Characterization of catalyst
Fig.1 shows XRD patterns for catalysts V2O5-TiO2. From the patterns some distinct peaks for TiO2 can be observed. From the left to right 2�� is 27.56?, 36.21?, 41.38?, 54.44?, 56.74? and 69.09? corresponding to the diffraction peak of rutile TiO2 crystal faces (110), (101), (111), (211), (220), and (311), respectively, and which are consistent with literature reports[15]. XRD analysis does not present any characteristic diffraction peak of V2O5 phase or other vanadium oxide until the loading concentration of V2O5 is more than 8%(mass fraction). The XRD patterns for the catalysts with 10%, 20% and 40% of V2O5-loaded TiO2 show the characteristic diffraction peak of V2O5 (2��=20.26?). Therefore, V2O5 will be well dispersed on the TiO2 phase when its concentration is small; and on the contrary, when the concentration of loading V2O5 is enhanced, V2O5 is gathered and crystallized.
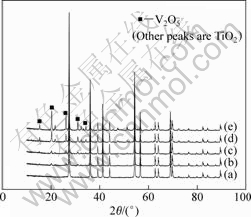
Fig.1 XRD patterns for V2O5-TiO2 catalysts: (a) 5%V2O5-TiO2; (b) 8%V2O5-TiO2; (c) 10%V2O5-TiO2; (d) 20%V2O5-TiO2; (e) 40%V2O5-TiO2
Raman spectra of V2O5-TiO2 catalysts synthesized under different dehydrated conditions, are presented in Fig.2. The rutile TiO2 possesses Raman bands at 440 and 612 cm-1[16], and V2O5 possesses Raman bands at 143, 280, 486, 695 and 995 cm-1[17]. Raman band at
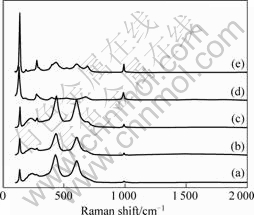
Fig.2 Raman spectra of V2O5-TiO2 catalysts: (a) 5%V2O5-TiO2; (b) 8%V2O5-TiO2; (c) 10%V2O5-TiO2; (d) 20%V2O5-TiO2; (e) 40%V2O5-TiO2
995 cm-1 is assigned to the stretching mode of V��O in the spectra of 10% V2O5-TiO2 catalysts from Fig.2(c), which indicates that V2O5 has dispersed on the surface of TiO2 in monolayer. When the concentration of V2O5 is beyond its dispersion ability, the microcrystallite of V2O5 is formed. With the quantity increase of loading V2O5, the intensity of the characteristic peak of V��O becomes more obvious. WACHS and WECKHUYEN[18] reported that when V2O5 dispersed on the surface of TiO2 in monolayer, the atomicity of vanadium would be 7-8 nm-2. In the spectra of 8%V2O5-TiO2 catalysts, there is no peak for V2O5 and the atomicity of vanadium on the surface of TiO2 is about 6.8 nm-2, which is close to the atomicity of the monolayer. However, when the coverage is beyond 8%, the peak for V��O appears, which is in a good agreement with XRD patterns.
Fig.3 shows an obvious absorption band in the wavelength range of 200-380 nm. It is the characteristic absorption of the charge transfer of O 2p��Ti 3d and the charge is from oxygen atom coordinated with titanium to the empty orbit of the center titanium atom[19]. As shown in Fig.3, the absorption band in the wavelength range of 200-600 nm results from electron transition of O 2p��V 3d, and the absorption band in the wavelength range of 300-350 nm results from electron transition of octahedron coordinate vanadium. But the absorption band in the wavelength range of 200-300 nm is from utterly isolated octahedron vanadium. So the absorption band in the wavelength range of 200-350 nm should be attributed to the electron transition interaction between O 2p��Ti 3d and O 2p��V 3d[20]. Meanwhile, the absorption of visible light for V2O5-TiO2 catalysts is enhanced obviously. So V2O5 loading can improve the absorption of visible light for TiO2 photocatalyst markedly.
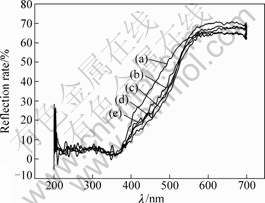
Fig.3 Diffuse reflectance UV-Vis spectra (UV-DRS) of catalysts: (a) 5%V2O5-TiO2; (b) 8%V2O5-TiO2; (c) 10%V2O5- TiO2; (d) 20%V2O5-TiO2; (e) 40%V2O5-TiO2
There are several existing forms for vanadium- oxygen species on the surface of TiO2 on account of the loading quantity of V2O5, such as isolated-VOx, poly-VOx and crystalloid-V2O5. So it is reasonable to consider that isolated-VOx is dominant when loading quantity is very small, which is likely to be concerned with TiO2 by V��O��Ti bond (shown in Fig.4(a)). With the increase of loading quantity of V2O5, vacancies near the isolated-VOx are gradually occupied and the proportion of poly-VOx that is formed by V��O��V chemistry bond connection increases gradually, leading to increasing the polymerization degree of VOx. The proportion of V��O��Ti bond declines with the proportion of V��O��V bond mounting up[21] (shown in Figs.4(b) and (c)).
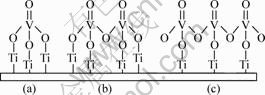
Fig.4 Bond graphs of V2O5 and TiO2 on surface of TiO2
Fig.5 shows the transmittance spectra of the suspending liquid of TiO2 powder with different V2O5 loadings. It is clear from the transmittance curves that with the increase of the concentration of loading V2O5, the optical transmittance gradually decreases. The average transmittance in the visible region is larger than that in UV-light region for all samples, and the transmission in the UV-light region is less than 5%. This means that all catalysts can absorb almost all the UV-light in the photocatalytic reactions according to UV-DRS in Fig.3 and the transmitted spectra of the suspending liquid of V2O5-TiO2 catalysts in Fig.5. In the wavelength region of 400-580 nm, for all the samples the refraction and the transmittance increase gradually from Fig.3 and Fig.5. This indicates that the catalysts can absorb visible light. But obviously when the red shift of the wavelength occurs the absorption will decrease. That is to say, the catalysts have a good absorption to UV-light and can also absorb visible light. But the transmittance does not change obviously with the increase of loading V2O5.
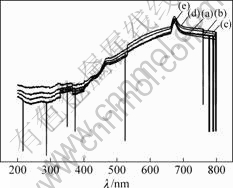
Fig.5 UV-Vis transmittance spectra of V2O5-TiO2 catalysts: (a) 5%V2O5-TiO2; (b) 8%V2O5-TiO2; (c) 10%V2O5-TiO2; (d) 20%V2O5-TiO2; (e) 40%V2O5-TiO2
Under the illumination, the transition for electrons absorbing photons must keep conservation of momentum besides conservation of energy, which satisfies the selection rule. It is assumed that the primary wave vector of the electron is K, which will transit to the state with wave vector of K��. As for electrons in the energy band, hK is similar to the property of momentum, so during the process of transition K and K must satisfy the conditions as follows[22]:
hK��-hK=M (1)
where M denotes the photons�� momentum.
Generally, as photons�� momentum resulting from semiconductor absorbing is less than the electrons�� momentum, the photons�� momentum can be ignored. So Eq.(1) can be written as follows:
K�� = K (2)
This shows that the wave vector of electrons keeps equal after absorbing photon and the electrons�� energy increases, which is the electrons�� selection rule. In order to satisfy the selection rule and keep the wave vector constant during the transition, electrons in valence band in excited state A can only transit to state B in the conduction band. Because of states A and B on the same droop line in one dimension energy curve this kind of transition is called direct transition. The intrinsic absorption of semiconductor forms a continuous absorption band, and has a long wave absorption edge ��0=Eg/h. So the band gap of semiconductor (Eg) can be extracted from the measurement of absorption. The common semiconductors such as GaAs and InSb in ��-�� and ��-�� subgroups are called direct band semiconductors because their minimum conduct band and maximum valence band are corresponding to the same wave vector. Theoretical calculation of electrons of this kind of semiconductors from their intrinsic absorption shows that if the transition for any K is permitted in the direct transition the relationship between the absorption coefficient and photon energy can be denoted as follows:

where �� is the absorption coefficient, and the relationship between �� and transmissivity T is ��=lnD- lnT (D is the thickness of solution); hv is the energy of photon with 1 240/��; and A is a constant.
But both theory and experiment prove that the conduct bands and valence bands for many semiconductors such as Ge and Si are not corresponding to the same wave vector, which are called indirect semiconductors. For indirect semiconductors the relationship between the absorption coefficient and photon energy is shown as follows:
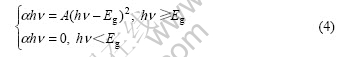
The curves of (��hv)1/2 vs hv for V2O5-TiO2 catalysts are shown in Fig.6. The linearity parts of the curves are extended to the point of ��=0, and the intercept on abscissa is the band gap (Eg). V2O5 is an indirect gap semiconductor[23]. According to Eq.(4) and Figs.6(a)- (e), the optical energy band gap of the catalysts of 5%V2O5-TiO2, 8%V2O5-TiO2, 10%V2O5-TiO2, 20% V2O5-TiO2 and 40%V2O5-TiO2 are 2.12, 2.14, 2.21, 2.25 and 2.26 eV, respectively. Rutile TiO2 is a direct gap semiconductor, and according to Eq.(3) and Fig.6(f), the optical energy band gap of rutile TiO2 is about 3.04 eV.
3.2 Photocatalytic activity of catalysts
The photocatalytic activities of V2O5-TiO2 and pure rutile TiO2 were studied through the splitting of water for O2 evolution under UV-light irradiation. The results are presented in Fig.7, which shows that the rates of O2 evolution of TiO2 loaded with V2O5 are larger than that of pure rutile TiO2 after irradiation for 12 h. With the increase of the quantity of loaded V2O5, the rate of O2 evolution also increases, which reaches the maximum rate of 118.2 ?mol/(L��h) when the concentration of loaded V2O5 is 8%. The rate of O2 evolution declines when the concentration of loaded V2O5 is more than 10%. Therefore, TiO2 loaded with V2O5 in a suitable amount can improve the photocatalytic activity of TiO2.
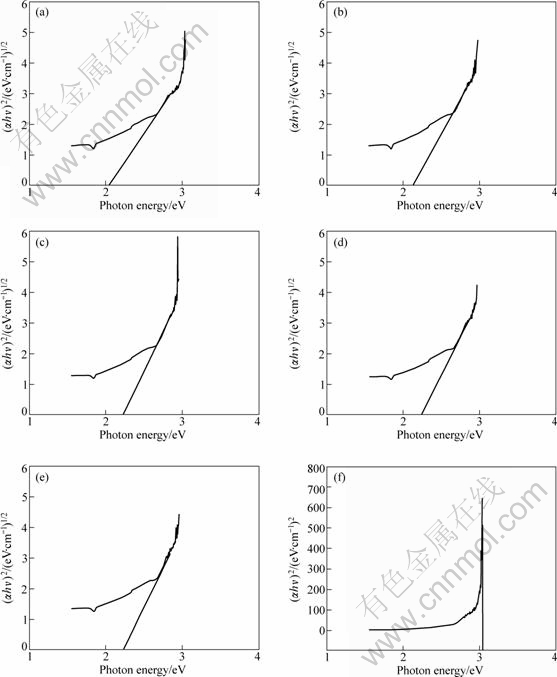
Fig.6 Absorption coefficient as function of incident photon energy of V2O5-TiO2 catalysts: (a) 5%V2O5-TiO2; (b) 8%V2O5-TiO2; (c) 10%V2O5-TiO2; (d) 20%V2O5-TiO2; (e) 40%V2O5-TiO2; (f) Rutle TiO2
The photocatalytic activities of V2O5-TiO2 were investigated through the splitting of water for O2 evolution under visible light irradiation. The results are shown in Fig.8. The lamp source is 250 W Xe, which is basically consistent with the characteristic spectrum of sun shine. Compared with the photocatalytic activity of V2O5-TiO2 catalysts under UV-light irradiation, the order of O2 evolution speed does not change. But the rates of O2 evolution change, and the maximum rate is 83.7 ?mol/(L��h) for 8%V2O5-TiO2. The band gap of pure TiO2 is 3.0 eV, and the largest response wavelength is 413 nm according to formula Eg=1 240/��. V2O5 loading makes TiO2 response to visible light because the band gap of V2O5 is about 2.2 eV.
Generally, the photocatalytic activity of photocatalyst is determined by its ability of light absorbing, the efficiency of separation between photoelectrons and holes, and the increase of the transfer rate of charge carriers[24].
When the surface of the semiconductor is irradiated
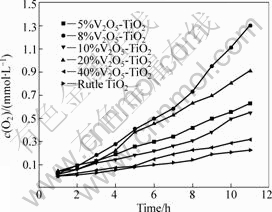
Fig.7 Dependence of photocatalytic O2 evolution on concentration of loaded V2O5 under UV-light irradiation
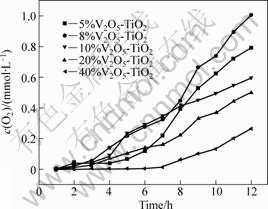
Fig8.Dependence of photocatalytic O2 evolution on concentration of loaded V2O5 under visible light irradiation.
by light whose energy is higher than the band gap of this semiconductor, electrons will obtain much energy to jump onto the conduction band and become free electrons named photoelectrons. Thus, holes are left in the valence band[25]. Because the conduction band of V2O5 is about 0.17 eV lower than that of rutile TiO2, electrons can be gathered in the conduction band of V2O5 and holes are left in the valence band of TiO2 after the photo-irradiation of the catalyst. The produced photoelectrons and holes can be separated efficiently. During the process of photocatalytic reactions, photo-electrons in conduction band of V2O5 can be captured by electron acceptor Fe3+, and anions OH�C in water can release their electrons in the valence band of TiO2 with O2 evolution.
4 Conclusions
(1) Rutile TiO2 powder photocatalysts are synthesized by hydrolysis process, then rutile TiO2 is dipped in the ammonium matavanadate solution, dried, and calcined, at last V2O5-TiO2 powders are obtained.
(2) Raman spectra of V2O5-TiO2 catalysts and XRD results show that 8%V2O5-TiO2 is near to monolayer. The diffuse reflectance UV-Vis spectra show that V2O5 loading can improve the absorption of visible light for TiO2 photocatalyst markedly. The optical energy band gaps of the catalysts of 5%V2O5-TiO2, 8%V2O5-TiO2, 10%V2O5- TiO2, 20%V2O5-TiO2 and 40 %V2O5-TiO2 are 2.12, 2.14, 2.21, 2.25 and 2.26 eV, respectively.
(3) V2O5 loading can improve the photocatalytic activity of TiO2. The maximum rate of water splitting for V2O5-TiO2 photocatalysts is 118.2 ?mol/(L��h) when the concentration of loaded V2O5 is 8% using Fe3+ as an electron acceptor under UV irradiation and 83.7 ?mol/(L��h) under visible light irradiation.
References[1] FUJISHIMA A, HONDA K. Electrochemical photolysis of water at a semiconductor electrode[J]. Nature, 1972, 238: 37-38.
[2] HOFFMAN M R, MARTIN S T, CHOI W, BAHNEMANN D W. Environmental applications of semiconductor photocatalysis[J]. Chem Rev, 1995, 95(1): 69-96.
[3] LIU Tong-xu, LI Fang-bo, LI Xiang-zhen. TiO2 hydrosols with high activity for photocatalytic degradation of formaldehyde in a gaseous phase[J]. J Hazard Mater, 2008, 152(1): 347-355.
[4] HOUMARD M, RIASSETTO D, ROUSSEL F, BOURGEOIS A, BERTHOME G, JOUD J C, LANGLET M. Morphology and natural wettability properties of sol-gel derived TiO2-SiO2 composite thin films[J]. Appl Surf Sci, 2007, 254(5): 1357-1544.
[5] PARK M S, KANG M. The preparation of the anatase and rutile forms of Ag-TiO2 and hydrogen production from methanol/water decomposition[J]. Mater Lett, 2008, 62(2): 183-187.
[6] OHNO T, TANIGAWA F, FUJIHARA K, IZUMI S, MATSUMURA M. Photocatalytic oxidation of water by visible light using ruthenium-doped titanium dioxide powder[J]. Photochemistry and Photobiology A: Chem, 1999, 127: 107-110.
[7] BENJARAM M R, BISWAJIT C. X-ray photoelectron spectroscopy study of V2O5 dispersion on a nanosized Al2O3-TiO2 mixed oxide[J]. Langmuir, 2001, 17(4): 1132-1137.
[8] BENJARAM M R, PAVANI M S, ETTIREDDY P R. Surface characterization of La2O3-TiO2 and V2O5/L A2O3-TiO2 catalysts[J]. Journal of Physical Chemistry, 2002, 106(22): 5695-5700.
[9] LI Fang-bo, GU Guo-bang, LI Xin-jun. Preparation, characterization and photo-catalytic behavior of WO3/TiO2 nanopowder[J]. Phys Chem Trans, 2000, 16(11): 997-1002. (in Chinese)
[10] ZHENG Y J, JENSEN A D, JOHNSSON J E, THOGEREN J R. Deactivation of V2O5-WO3-TiO2 SCR catalyst at biomass red power plants: Elucidation of mechanisms by lab-andpilot-scale experiments[J]. Applied Catalysis B: Environmental, 2008, 83: 186-194.
[11] MONGKHONSI T, KERSHENBAUN L. The effect of deactivation of a V2O5/TiO2(anatase) industrial catalyst on reactor behaviour during the partial oxidation of o-xylene to phthalic anhydride[J]. Appl Catal A, 1998, 170(1):33-48.
[12] AMIRIDIS M D, WACHS I E, DEO G, JEHNG J M, KIM D S. Reactivity of V2O5 catalysts for the selective catalytic reduction of NO by NH3: Influence of vanadia loading, H2O, and SO2[J]. Journal of Catalysis, 1996, 161(1): 247-253.
[13] CASAGRAND L, LIETTI L, NOVA I, FORZATTI P, BAIBER A. SCR of NO by NH3 over TiO2-supported V2O5-MoO3 catalysts: Reactivity and redox behavior[J]. Appl Catal B, 1999, 22(1): 63-77.
[14] JIANG Yin-hua, SUN Yue-ming, ZHAO Chen-xuan, WU Min, YIN Heng-bo, CHEN Kang-min. Effects of polyatomic acids as modifiers on hydrothermal synthesize of rutile TiO2 nanorods[J]. New Chemical Materials, 2007, 4: 28-30. (in Chinese)
[15] DIAS C R, PORTELA M F, BOND G C. Oxidation of o-xylene to phthalic anhydride over V2O5/TiO2 catalysts: I. Influence of catalyst composition, preparation method and operating conditions on conversion and product selectivities[J]. Journal of Catalysis, 1995, 157(2): 344-352.
[16] YANG Nan-ru. Testing methods of inorganic non-metallic materials[M]. Wuhan: Wuhan University of Technology Press, 1993: 146. (in Chinese)
[17] LIU Xue-jun, GU Xiao-dong, SHEN Jian-yi. Structure, surface acidity/basicity and redox properties of V2O5/TiO2 catalysts[J]. Chinese Journal Catalysis, 2003, 24(9): 674-680. (in Chinese)
[18] WACHS I E, WECKHUYEN B M. Structure and reactivity of surface vanadium oxide species on oxide supports[J]. Appl Catal A, 1997, 157(1/2): 67-90.
[19] BENGOA J F, GALLEGOS N G, MARCHETTI S G, ALVAREZ A M, CAGNOLI M V. Influence of TS-1 structural properties and operation conditions on benzene catalytic oxidation with H2O2[J]. Microporous and Mesoporous Mater, 1998, 24(4/6): 163-172.
[20] HU Rong-rong, ZHONG Shun-he. Mutual modification of V2O5 and TiO2 on the surface of supported coupled-semiconductor V2O5-TiO2/SiO2[J]. Chinese Journal Catalysis, 2005, 26(1): 32-36. (in Chinese)
[21] XU Lian-bo, FAN Yi-ning, LIU Liu, LIN Ming, CHEN Yi. The dispersion state and catalytic performance of vanadium-oxygen species on the surface of V2O5/TiO2 catalyst[J]. Science in China (Series B), 2002, 32(3): 235-242. (in Chinese)
[22] LIU En-ke, ZHU Bing-sheng, LUO Jin-sheng. Semiconductor physics[M]. Beijing: Electronic Industry Press, 2003: 326-327. (in Chinese)
[23] XU Min, QIU Jia-weng, HE De-yan. Microstructures and electrical and optical properties of vanadium pentoxide films[J]. Vacuum Science and Technology, 2003, 23(6): 373-376. (in Chinese)
[24] ZHANG Qi, LI Xin-jun, LI Fang-bo, CHANG Jie. Investigation on visible-light activity of WOx/TiO2 photocatalyst[J]. Acta Physico- Chimica Sinica, 2004, 20(5): 507-511. (in Chinese)
[25] HAN Wei-ping. Catalytic chemistry introduction[M]. Beijing: Science Press, 2003: 272-310. (in Chinese)
(Edited by CHEN Wei-ping)
Foundation item: Project(08JJ3022) supported by the Natural Science Foundation of Hunan Province, China
Received date: 2009-03-26; Accepted date: 2009-06-06
Corresponding author: GAO You-liang, Professor; Tel: +86-798-8499697; E-mail: gaocsu@163.com

