
J. Cent. South Univ. (2019) 26: 2011-2018
DOI: https://doi.org/10.1007/s11771-019-4148-x
Influence of Ag on photocatalytic performance of Ag/ZnO nanosheet photocatalysts
TAN Xu(̷��)1, ZHOU Shan(��ɺ)1, TAO Hui-jin(�ջԽ�)1, 2,
WANG Wei-yang(��ΰ��)1, WAN Qiang-wei(��ǿΰ)1, ZHANG Ke-chen(�ſƳ�)1
1. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
2. Key Lab of Nonferrous Materials Science and Engineering (Ministry of Education), Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: Ag/ZnO nanosheet composites were prepared by zinc nitrate, sodium hydroxide and silver nitrate via a simple hydrothermal method. The crystal structure, morphology, optical property and photocatalytic performance were studied by means of XRD, SEM, HRTEM, XPS, and PL methods. It is found that both the pure ZnO and composite Ag/ZnO samples have the same morphology of nanosheet. The interaction of spherical Ag particles with ZnO matrix in the Ag/ZnO sample leads to an increase in photocatalytic efficiency for the possible increase of concentration of surface hydroxyl and the photo-induced electrons and holes. The addition of Ag can reduce the recombination rate of photo-generated carriers and the sample with addition of 3 at% Ag to ZnO exhibits the best photocatalytic activity with the degradation rate up to 95% within 15 min.
Key words: Ag/ZnO nanosheet photocatalyst; hydrothermal method; photocatalytic activity
Cite this article as: TAN Xu, ZHOU Shan, TAO Hui-jin, WANG Wei-yang, WAN Qiang-wei, ZHANG Ke-chen. Influence of Ag on photocatalytic performance of Ag/ZnO nanosheet photocatalysts [J]. Journal of Central South University, 2019, 26(7): 2011-2018. DOI: https://doi.org/10.1007/s11771-019-4148-x.
1 Introduction
Zinc oxide is a semiconductor material with excellent photoelectric properties, which is widely used in solar cells [1-3], biological antibacterial [4-6], photocatalytic degradation [7, 8], gas sensor [9] and other fields. Pure zinc oxide has wide direct band gap (3.37 eV) and high exciton binding energy (60 meV) [10]. And its optical response range is mainly located in the ultraviolet region, greatly limiting its photocatalytic efficiency under sunlight irradiation. A lot of researches to improve its photocatalytic performance have been carried out, and some encouraging breakthroughs have been made recently. Doping or composition modification of ZnO by transition metals such as Co [11], Ag [12], Mn [13], Fe [14], Al [15], Cu [16], rare earth metals such as Ce [17], Eu [18] and non-metallic elements or compounds such as N [19], C3N4 [20] can improve the photocatalytic performance by the introduction of heterojunctions or lattice defects.
Many factors such as crystal morphology, specific surface area and surface defects have important influence on the photocatalytic performance of ZnO. Nanometer ZnO with different morphologies such as sheet [21-23], line [24], rod [25], needle [26], comb [9, 27], flower TAN Xu and ZHOU Shan contributed equally to this work.[28] and microsphere [29] were synthesized successfully. The morphology of sheet possesses the relatively higher specific surface area, greater proportion of exposed active surface, and higher photocatalytic activity. But how to control the preparation conditions to get such morphology of ZnO is still a complicated problem for the fact of the complex factors influencing microstructure and the different growth mechanisms in different methods.
At present, not only the research on the preparation of ZnO nanosheets by hydrothermal method without templates or additives is scarce, but also the control of preparation conditions on the size and thickness of ZnO nanosheets is still immature. Therefore, this work is aimed at developing a simple, safe and efficient hydrothermal method without templates and additives to prepare ZnO and Ag/ZnO nanosheets with good photocatalytic performance through the optimization of preparation conditions.
2 Experimental
2.1 Preparation of Ag/ZnO nanosheet photocatalyst
A certain amount of Zn(NO3)2��6H2O, NaOH, and AgNO3 were dissolved respectively in distilled water to form three kinds of solutions. The NaOH solution was then added slowly into the Zn(NO3)2��6H2O solution under continuous stirring until the mixed solution formed a stable suspension, as the precursor solution. One portion of the precursor solution was put into a hydrothermal reactor with some distilled water and treated at 120 ��C for 6 h to obtain pure ZnO sample. At the same time, different amounts of AgNO3 solutions were added slowly into the precursor solution with continuous stirring to form different uniform and stable solutions for further hydrothermal syntheses, as the same as the preparation of pure ZnO. The hydrothermal products were washed with distilled water and absolute ethyl alcohol, then dried and ground into powders. These samples were labeled as 1%Ag/ZnO, 3%Ag/ZnO, 5%Ag/ZnO, and 7%Ag/ZnO respectively according to the atomic percentages of added Ag.
2.2 Characterization of photocatalyst
The crystal structure was characterized by X-ray powder diffraction (XRD) analysis (Rigaku D/Max 2550) using Cu K�� radiation with ��= 0.15406 nm. The morphology was observed by scanning electron microscope (SEM), FEI Nova NanoSEM 230) and transmission electron microscope (TEM, JEOL JEM-2100F). Surface components and element valence states were detected by X-ray photoelectron spectroscopy (XPS), (Thermo Fisher K-Alpha 1063). The photoluminescence (PL) spectrum was tested using an F-2500 fluorescent spectrophotometer with an excitation wavelength of 315 nm.
2.3 Photocatalytic performance test
The photocatalytic activities of ZnO and Ag/ZnO samples were tested by the photocatalytic degradation experiment of methyl orange (MO) under UV light irradiation by a 250 W mercury lamp. 0.1 g photocatalyst was mixed with 400 mL of MO solution with the concentration of 10 mg/L. The mixture was magnetically stirred in the dark for 10 min to achieve adsorption-desorption equilibrium. Then the photocatalytic performance test was carried out for 30 min, and 5 mL solution was taken out at an interval of 5 min for six times. All solutions were settled for over 24 h before the transmittance of their supernatant liquor was measured at a determined wavelength of 464 nm by a UV-visible spectrophotometer. The photocatalytic reaction rate of MO was calculated by the Lambert-Beer law.
3 Results and discussion
3.1 XRD patterns
The crystal structures of all samples were the hexagonal wurtzite ones as shown in Figure 1. The diffraction peaks located at about 38.1�� and 44.3�� of Ag/ZnO samples can be assigned to the (111) and (200) planes of Ag. It is found from Figure 1 that the diffraction peak intensity of Ag increased while that of ZnO decreased with the increase of Ag content in Ag/ZnO samples.
3.2 SEM morphologies
The SEM images of pure ZnO and composite Ag/ZnO samples were shown in Figure 2. It is found that all the samples have the morphology of nanosheet and some degree of aggregation, especially for the case of 1%Ag/ZnO sample(Figure 2(b)). The thickness of 3%Ag/ZnO sample (Figure 2(c)) was significantly smaller than that of other samples, implying a largest specific surface area among all the samples.
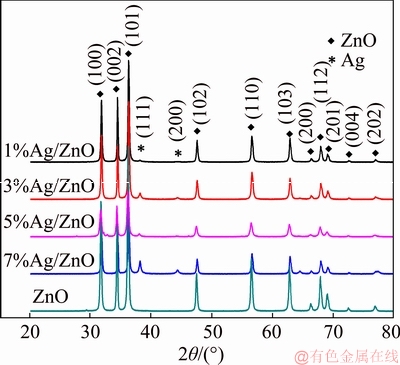
Figure 1 XRD patterns of samples
3.3 XPS spectra
Figure 3 shows the XPS results of samples. Figure 3(a) shows the full spectra of all Ag/ZnO samples with different contents of Ag, where four kinds of elements (C, Zn, Ag and O) are present. C is introduced by the instrument itself, so all the samples only contain Zn, Ag and O elements, agreeing with the XRD results.
Figure 3(b) shows Ag 3d peaks of Ag/ZnO nanosheet composites. The peaks at about 367.2 and 373.2 eV correspond to the 3d5/2 and 3d3/2 electron state of Ag, respectively. The 3d5/2 and 3d3/2 peaks of Ag shift to the lower binding energies compared with the standard values of 368.2 and 374.2 eV of bulk Ag, which confirms the interaction between Ag nanoparticles and ZnO nanocrystals [30, 31]. It is obvious that the peak position of 7% sample has a shift towards high energy, which implies that some monovalent Ag were produced,since the Ag 3d binding energies for Ag2O are about 367.7 eV and 373.7 eV [32 ]. Although there is no obvious evidence of presence of Ag2O in XRD pattern, the XPS results might indicate that the surface of sample contains it [32].

Figure 2 SEM images of samples:
Figure 3(c) presents the O 1s peaks of Ag/ZnO composites with different content of Ag. The peak at about 530.4 eV can be ascribed to O atoms at normal location of ZnO lattice [33], while the other one at about 531.5eV can be attributed to O atoms in the hydroxyls on the surface [34]. The hydroxyls on ZnO surface play a significant role in improving the photocatalytic activity of ZnO sample [12, 31]. With the increase of Ag addition, the binding energies of O 1s both at the normal location and in the hydroxyl increase gradually. The 7% Ag/ZnO sample has the most obvious surface hydroxyl characteristic among all the samples, which has a beneficial effect on the improvement of photocatalytic efficiency.
Figure 3(d) shows the Zn 2p peaks of Ag/ZnO composites. The peaks located at about 1044.3 and 1021.8 eV represent Zn 2p1/2 and Zn 2p3/2 state, respectively [33]. Because of Ag2O, the oxygen vacancy on ZnO surface increased, thus, the Zn 2p peaks of 7% Ag/ZnO sample shift towards high energy as same as Ag 3d [32, 35].
3.4 HRTEM images
The high resolution transmission electron microscope (HRTEM) images of all Ag/ZnO sample are shown in Figure 4. The existence of Ag in all samples is confirmed as shown in the XRD patterns for the measured spacings of lattice fringes of (111) or (200) planes. In addition, the morphologies of Ag nanoparticles on the surface of ZnO matrix are nearly spherical with a diameter of about 15-30 nm. The interface and interaction between Ag nanoparticles and the ZnO matrix have changed the state of oxygen on the surface of ZnO as shown in the XPS spectra.
3.5 PL spectra
The PL spectra are shown in Figure 5. The spectra consist of near ultraviolet band edge emission and visible emission. The former is originated from excitonic transition and the latter is ascribed to different defects such as zinc vacancy (VZn), zinc interstitial (Zni), oxygen vacancy (VO) and oxygen interstitial (Oi) [36, 37]. The spectrum of pure ZnO includes several peak wavelengths. The emission at about 400 nm results from electron transition. The emissions at 450 nm and 468 nm correspond to two different electron transitions associated with the zinc vacancy. The emission at 508 nm may be attributed to the electron transition from Zni to VO level with the transition energy of 2.44 eV. The emission at about 590 nm may be an overlap of several emission bands corresponding to different electro-hole recombination related to the oxygen interstitial [38, 39]. At the same time, the spectra of Ag/ZnO samples have an obvious intensity decrease for the possible non-radiative defect produced by the addition of Ag [40], which indicates that the addition of Ag may have effectively reduced the recombination rate of photo-generated electrons and holes and thus improved the photocatalytic performance of ZnO.
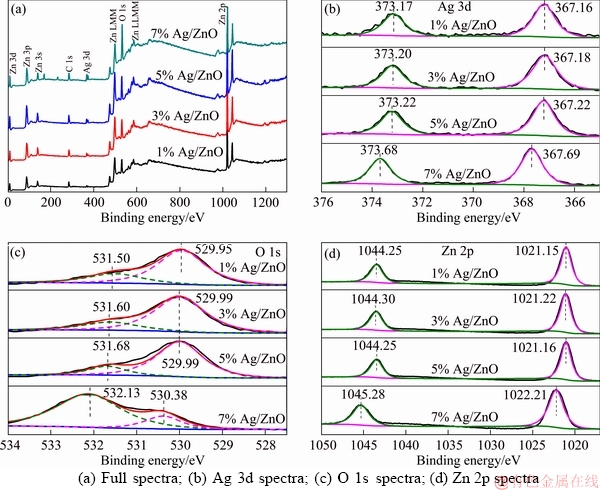
Figure 3 XPS spectra of Ag/ZnO samples with different content of Ag:
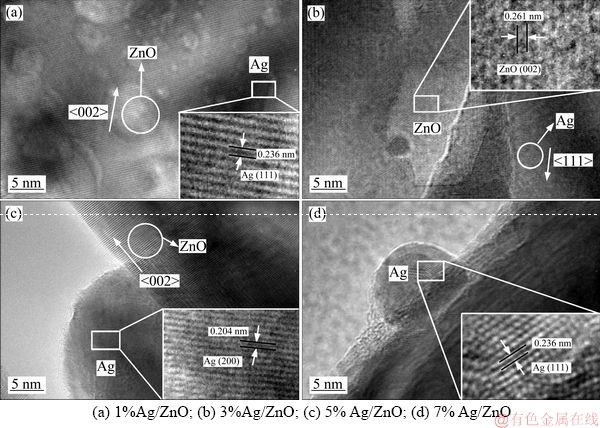
Figure 4 HRTEM images of samples:
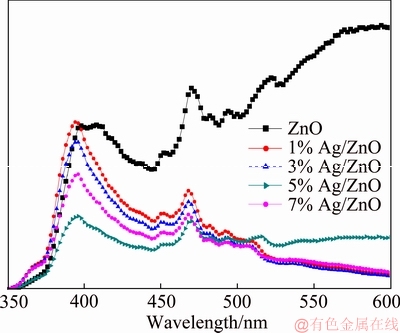
Figure 5 Photoluminescence (PL) spectroscopy of samples
3.6 Photocatalytic performance
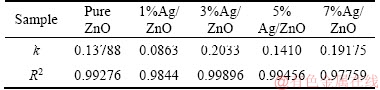
The concentration change of MO under UV-light irradiation is shown in Figure 6, where C0 and C are the initial and real concentration respectively, and the MOs are almost completely degraded within 30 min. All prepared Ag/ZnO samples except 1%Ag/ZnO behave better in photocatalytic activity than pure ZnO, which may be due to worse dispersion of the 1%Ag/ZnO sample as presented in SEM image, leading to smaller specific surface, and less active reaction site for photocatalytic degradation. The function on time t, ln(C0/C)=kt, is used to obtain the reaction rate constant k by the linear fitting of ln(C0/C) versus t, and the result is listed in Table 1. Because the degradation rate was over 90% in 15 min for most samples, the data before 15 min are considered for such fitting. It is found in Table 1 that the 3%Ag/ZnO sample exhibits the highest reaction rate constant of 0.2033 min-1, which indicates that an appropriate addition of Ag has a positive effect on the improvement of photocatalytic performance and the best effect can be obtained on the 3%Ag/ZnO sample.
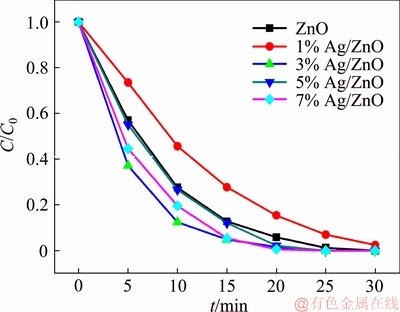
Figure 6 Result of samples in photocatalytic reaction
Table 1 Photocatalytic reaction rate constant of samples
3.7 Photocatalytic mechanism
The photocatalytic mechanism of Ag/ZnO composite could be illustrated by Figure 7. When the catalyst is illuminated by UV light, the electrons in the valence band of ZnO would be excited to its conduction band, because the energy of UV light is higher than the intrinsic band gap energy of ZnO. Accordingly, equal amount of holes are left in the valence band [41]. With the modification by Ag, a new Femi energy lever is established due to the Schottky barrier at the Ag/ZnO interface [42]. Therefore, the photo-generated electrons could be easily transferred to Ag sink, reducing the recombination rate of free electron-hole pairs and enhancing the photocatalytic performance. The adsorbed O2 molecules on the surface of Ag can trap the free electrons, and transform into superoxide anion radicals (��O2-), while the holes can be trapped by H2O molecules to produce hydroxyl radicals (��OH) [43]. Thanks to this two strong oxidizing agents, the organic dye (such as methyl Orange) could be thoroughly decomposed.
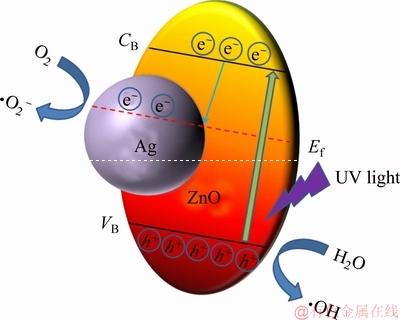
Figure 7 Schematic illustration of photocatalytic mechanism of Ag/ZnO composite under UV light
4 Conclusions
The Ag/ZnO nanosheet composites are successfully prepared by a simple hydrothermal method. It is found that the spherical Ag nanoparticles with the diameter of 15-30 nm are distributed on the surface of ZnO matrix. The interaction between Ag and ZnO can change the surface element distribution of ZnO matrix and improve the photoelectric performance of Ag/ZnO by reducing the recombination rate of photo- generated electrons and holes. The photocatalytic degradation study further proves that appropriate Ag addition is beneficial to the improvement of photocatalytic performance of ZnO and the 3%Ag/ZnO sample has the best photocatalytic effect for both the smaller size and recombination rate of photo-generated electrons and holes.
References
[1] BRAMMERTZ G, BUFFI RE M, OUESLATIS, EIANZEERYH, MESSAOUDK B,SAHAYARAJ S,K
RE M, OUESLATIS, EIANZEERYH, MESSAOUDK B,SAHAYARAJ S,K  BLE C,MEURIS M, POORTMANSJ. Characterization of defects in 9.7% efficient Cu2ZnSnSe4-CdS-ZnO solarcells [J]. Applied Physics Letters, 2013, 103(16): 163904-163911.
BLE C,MEURIS M, POORTMANSJ. Characterization of defects in 9.7% efficient Cu2ZnSnSe4-CdS-ZnO solarcells [J]. Applied Physics Letters, 2013, 103(16): 163904-163911.
[2] PEI Juan, FENG Kang-ning, ZHAO Xue, HAO Yan-zhong, WEI Ya-nan, SUN Bao, LI Ying-pin, CHEN Shao-rui, LV Hai-jun. ZnO-based inverted hybrid solar cells: Technical adjustment for performance optimization step by step [J]. Optics Communications, 2018, 427(15): 294-300.
[3] DAGAR J, SCAVIA G, SCARSELLI M, DESTRI S, CRESCENZI M D, BROWN T M. Coating ZnO nanoparticle films with DNA nanolayers for enhancing the electron extracting properties and performance of polymer solar cells [J]. Nanoscale, 2017, 9(48): 19031-19038.
[4] SAHA R, KARTHIK S, KUMAR P M R S A, SURIYAPRABHA R, RAJENDRAN V. Psidium guajava leaf extract-mediated synthesis of ZnO nanoparticles under different processing parameters for hydrophobic and antibacterial finishing over cotton fabrics [J]. Progress in Organic Coatings, 2018,124: 80-91.
[5] MAHENDIRAN D, SUBASH G, SELVAN D A, REHANA D, KUMAR R S, RAHIMAN A K. Biosynthesis of zinc oxide nanoparticles using plant extracts of aloe vera and hibiscus sabdariffa: Phytochemical, antibacterial, antioxidant and anti-proliferative studies [J]. Bionanoscience, 2017, 7(3): 530-545.
[6] HE Wei-wei, KIM H K, WAMER W G, MELKA D, CALLAHAN J H, YIN Jun-jie. Photogenerated charge carriers and reactive oxygen species in ZnO/Au hybrid nanostructures with enhanced photocatalytic and antibacterial activity [J]. Journal of the American Chemical Society, 2014, 136(2):750-757.
[7] XU Tong-guang, ZHANG Li-wu, CHENG Han-yun, ZHU Yong-fa. Significantly enhanced photocatalytic performance of ZnO via graphene hybridization and the mechanism study [J]. Applied Catalysis B: Environmental, 2011, 101(3, 4): 382-387.
[8] SUN Jia-xin, YUAN Yu-peng, QIU Ling-guang, JIANG Xia, XIE An-jian, SHEN Yu-hua, ZHU Jun-fa. Fabrication of composite photocatalyst g-C3N4-ZnO and enhancement of photocatalytic activity under visible light [J]. Dalton Transactions, 2012, 41(22): 6756-6763.
[9] PAN Xiao-fang, LIU Xi, BERMAK A, FAN Zhi-yong. Self-gating effect induced large performance improvement of ZnO nanocomb gas sensors [J]. American Chemical Society: Nano, 2013, 7(10): 9318-9324.
[10] XU Lin-hua, LI Xiang-yin, YUAN Jun. Effect of K-doping on structural and optical properties of ZnO thin films [J]. Superlattices and Microstructures, 2008, 44(3): 276-281.
[11] PRASAD N, KARTHIKEYAN B. Cu-doping and annealing effect on the optical properties and enhanced photocatalytic activity of ZnO nanoparticles [J]. Vacuum, 2017, 146: 501-508.
[12] SARAVANAN R, KARTHIKEYAN N, GUPTA V K, THIRUMAL E, THANGADURAI P, NARAYANAN V, STEPHEN A. ZnO/Ag nanocomposite: An efficient catalyst for degradation studies of textile effluents under visible light [J]. Materials Science and Engineering C, 2013, 33(4): 2235-2244.
[13] PUTRI N A, FAUZIA V, IWAN S, ROZA L, UMAR A A, BUDI S. Mn-doping-induced photocatalytic activity enhancement of ZnO nanorods prepared on glass substrates [J]. Applied Surface Science, 2018, 439: 285-297.
[14] KIATTISAKSIRI P, KHAMDAHSAG P, KHEMTHONG P, PIMPHA N, GRISDANURAK N. Photocatalytic degradation of 2,4-dichlorophenol over Fe-ZnO catalyst under visible light [J]. Korean Journal of Chemical Engineering, 2015, 32(8): 1578-1585.
[15] ZHANG Jin, QUE Wen-xiu. Preparation and characterization of sol�Cgel Al-doped ZnO thin films and ZnO nanowire arrays grown on Al-doped ZnO seed layer by hydrothermal method [J]. Solar Energy Materials and Solar Cells, 2010, 94(12): 2181-2186.
[16] VAIANO V, IERVOLINO G, RIZZO L. Cu-doped ZnO as efficient photocatalyst for the oxidation of arsenite to arsenate under visible light [J]. Applied Catalysis B: Environmental, 2018, 238: 471-479.
[17] BECHAMBI O, JLAIEL L, NAJJAR W, SAYADI S. Photocatalytic degradation of bisphenol A in the presence of Ce-ZnO: Evolution of kinetics, toxicity and photodegradation mechanism [J]. Materials Chemistry and Physics, 2016, 173: 95-105.
[18] WANG Mei-li, HUANG Chang-gang, HUANG Zhi, GUO Wang, HUANG Ji-quan, HE Hong, WANG Hai, CAO Yong-ge, LIU Quan-lin, LIANG Jing-kui. Synthesis and photoluminescence of Eu-doped ZnO microrods prepared by hydrothermal method [J]. Optical Materials, 2009, 31(10): 1502-1505.
[19] FERRARI-LIMA A M, SOUZA R P, MENDES S S, MARQUES R G, GIMENES M L, FERNANDES- MACHADO N R C. Photodegradation of benzene, toluene and xylenes under visible light applying N-doped mixed TiO2 and ZnO catalysts [J]. Catalysis Today, 2015, 241: 40-46.
[20] JUNG H, PHAM T T, SHIN E W. Interactions between ZnO nanoparticles and amorphous g-C3N4 nanosheets in thermal formation of g-C3N4/ZnO composite materials: The annealing temperature effect [J]. Applied Surface Science, 2018, 458: 369-381.
[21] LIU Chang-zhen, MENG Da-wei, WU Xiu-ling, WANG Yong-qian, YU Xiao-hong, ZHANG Zheng-jie, LIU Xiao-yang. Synthesis, characterization and optical properties of sheet-like ZnO [J]. Materials Research Bulletin, 2011, 46(9): 1414-1416.
[22] ZHU Guo-xing, LIU Yuan-jun, JI Zhen-yuan, BAI Song, SHEN Xiao-ping, XU Zheng. Hierarchical ZnO microspheres built by sheet-like network: Large-scale synthesis and structurally enhanced catalytic performances [J]. Materials Chemistry and Physics, 2012, 132(2, 3): 1065-1070.
[23] HONG Yu, TIAN Chun-gui, JIANG Bao-jiang, WU Ai-ping, ZHANG Qi, TIAN Guo-hui, FU Hong-gang. Facile synthesis of sheet-like ZnO assembly composed of small ZnO particles for highly efficient photocatalysis [J]. Journal of Materials Chemistry A, 2013, 1(18): 5700-5708.
[24] CONSONNI V, SARIGIANNIDOU E, APPERT E, BOCHEUX A, GUILLEMIN S, DONATINI F, ROBIN I C, KIOSEOGLOU J, ROBAUT F. Selective area growth of well-ordered ZnO nanowire arrays with controllable polarity [J]. American Chemical Society: NANO, 2014, 8(5): 4761-4770.
[25] ABBASI M A, IBUPOTO Z H, HUSSAIN M, POZINA G, LU Jun, HULTMAN L, NUR O, WILLANDER M. Decoration of ZnO nanorods with coral reefs like NiO nanostructures by the hydrothermal growth method and their luminescence study [J]. Materials, 2014, 7(1): 430-440.
[26] LIU Jia, CAO Wen-qiang, JIN Hai-bo, YUAN Jie, ZHANG De-qing, CAO Mao-sheng. Enhanced permittivity and multi-region microwave absorption of nanoneedle-like ZnO in X-band at elevated temperature [J]. Journal of Materials Chemistry C, 2015, 3(18): 4670-4677.
[27] PHAN T L, SUN Yue-kui, VINCENT R. Structural characterization of CVD-grownZnOnanocombs [J]. Journal of The Korean Physical Society, 2011, 59(1): 60-64.
[28] AMEEN S, AKHTAR M S, SHIN H S. Speedy photocatalytic degradation of bromophenol dye over ZnO nanoflowers [J]. Materials letters, 2017, 209: 150-154.
[29] ZHANG Li, DAI Chao-hua, ZHANG Xiu-xiu, LIU You-nian, YAN Jian-hui. Preparation and photocatalytic performance of ZnO/ZnGa2O4 composite microspheres [J]. Journal of Central South University, 2016, 23(12): 3092-3099.
[30] GU Chang-dong, CHENG Chun, HUANG Hai-you, WONG Tai-lun, WANG Ning, ZHANG Tong-yi. Growth and photocatalytic activity of dendrite-like ZnO@Agheterostructure nanocrystals [J]. Crystal Growth and Design, 2009, 9(7): 3278-3285.
[31] GAO Shu-yan, JIA Xiao-xia, YANG Shu-xiang, LI Zheng- dao, JIANG Kai. Hierarchical Ag/ZnO micro/nanostructure: green synthesis and enhanced photocatalytic performance [J]. Journal of Solid State Chemistry, 2011, 184(4): 764-769.
[32] SAHU R K, GANGULY K, MISHRA T, MISHRA M, NINGTHOUJAM R S, ROY S K, PATHAK L C. Stabilization of intrinsic defects at high temperatures in ZnO nanoparticles by Ag modification [J]. Journal of Colloid and Interface Science, 2012,366(1): 8-15.
[33] MOULDER J F, STIKLE W F, SOBOL P E, BOMBEN K D. Handbook of X-ray photoelectron spectroscopy:a reference book of standard spectra for identification andinterpretation of XPS data [M]. Eden Prairie: Physical Electronics, Inc. 1995: 213-242.
[34] RAMGIR N S, MULLA I S, PILLAI V K. Micropencils and microhexagonal cones of ZnO [J]. Journal of Physical Chemistry B, 2006, 110: 3995-4001.
[35] TAY Y Y, LI S, SUN C Q, CHEN P. Size dependence of Zn 2p3/2 binding energy in nanocrystalline ZnO [J]. Applied Physics Letters,2006,88(17): 173118.
[36] DJURISIC A B, LEUNG Y H, TAM K H, HSU Y F, DING L,GE W K, ZHONG Y C, WONG K S, CHAN W K, TAM H L, CHEAH K W, KWOK W M, PHILLIPS D L D. Defect emissions in ZnO nanostructures [J]. Nanotechnology, 2005, 18(9): 095702.
[37] TAM K H, CHEUNG C K, LEUNG Y H, DJURI I
I A B, LING C C, BELING C D, FUNG S, KWOK W M, CHAN W K, PHILLIPS D L, DING L, GE W K. Defects in ZnO nanorods prepared by a hydrothermal method. [J]. Journal of Physical Chemistry B, 2006, 110(42): 20865-20871.
A B, LING C C, BELING C D, FUNG S, KWOK W M, CHAN W K, PHILLIPS D L, DING L, GE W K. Defects in ZnO nanorods prepared by a hydrothermal method. [J]. Journal of Physical Chemistry B, 2006, 110(42): 20865-20871.
[38] AHN C H,KIM Y Y,KIM D C,MOHANTA S K, CHO H K. Erratum: A comparative analysis of deep level emission in ZnO layers deposited by various methods [J]. Journal of Applied Physics, 2009, 105(1): 013502.
[39] CHEN Z, LI X X, CHEN N, WANG H, DU GP, SUEN AYM. Effect of annealing on photoluminescence of blue-emitting ZnO nanoparticles by sol�Cgel method [J]. Journal of Sol-Gel Science and Technology, 2012, 62(2): 252-258.
[40] SAKAGUCHI I, PARK D, TAKATA Y, HISHITA S, OHASHI N, HANEDA H, MITSUHASHI T. An effect of annealing on In implanted ZnO [J]. Nuclear Instruments and Methods in Physics Research B, 2003, 206(3): 153-156.
[41] WANG Chao, WU Di, WANG Pei-fang, AO Yan-hui, HOU Jun, QIAN Jin. Effect of oxygen vacancy on enhanced photocatalytic activity of reduced ZnO nanorod arrays [J]. Applied Surface Science, 2015,325: 112-116.
[42] LIU Hai-rui, HU Yan-chun, ZHANG Zhu-xia, LIU Xu-guang, JIA Hu-sheng, XU Bing-she. Synthesis of spherical Ag/ZnO heterostructural composites with excellent photocatalytic activity under visible light and UV irradiation [J]. Applied Surface Science, 2015,355: 644-652.
[43] SUN Yong-jiao, ZHAO Zhen-ting, LI Gang, LI Peng-wei, ZHANG Wen-dong, HAN Zhi-tao, LIAN Kun, HU Jie. Synthesis and characterization of Ag@ZnO nanostructures for photocatalytic degradation of rhodamine B: influence of calcination temperature and Ag content [J]. Applied Physics A, 2017,123(2): 116.
(Edited by HE Yun-bin)
���ĵ���
Ag�Զ�ά����Ag/ZnO���ϲ��Ϲ�����ܵ�Ӱ��
ժҪ��ʹ������п���������ƺ�������Ϊԭ�ϣ�ͨ��ˮ�ȷ��Ʊ�����Ag/ZnO�����ϲ��ϣ�����XRD��SEM��HRTEM��XPS��PL �ȱ����ֶζԸ��ϲ��ϵľ���ṹ����ò����ѧ���ܺ�����ܽ������о������Խ������, ��ZnO��Ag/ZnO���ϲ��Ͼ�����ͬ������Ƭ��ò����Ag/ZnO ��Ʒ��, ��״Ag������ZnO����������������˸��ϲ��ϵı����ǻ��������˹⼤�������ӵĸ����ʣ��������˹��Ч�ʡ�����������Ʒ�У�������3 at%Ag����Ʒ���ֳ������ŵĹ�����ܣ�����15 min�ڹ�������ʴﵽ95%��
�ؼ��ʣ���ά����Ag/ZnO�������ˮ�ȷ����������
Foundation item: Project(21373273) supported by the National Natural Science Foundation of China; Project(2019CG033) supported by the Deepening Reform Project of Innovation and Entrepreneurship Education of Central South University, China; Project(2019JG067) supported by the Graduate Education and Teaching Reform Research Project of Central South University, China; Project(201810533278) supported by the National College Students�� Innovative Experiment Project of Central South University, China
Received date: 2018-07-01; Accepted date: 2018-12-28
Corresponding author: TAO Hui-jin, PhD, Associate Professor; E-mail: thj@csu.edu.cn; ORCID: 0000-0002-9486-6045