
Thermodynamic calculation of Er-X and Al-Er-X
compounds existing in Al-Mg-Mn-Zr-Er alloy
LIN Shuang-ping(��˫ƽ), NIE Zuo-ren(������), HUANG Hui(�� ��),
ZHAN Chun-yao(ռ��ҫ), XING Ze-bing(�����), WANG Wei(�� Ϊ)
School of Materials Science and Engineering, Beijing University of Technology, Beijing 100124, China
Received 16 December 2008; accepted 28 April 2009
Abstract: Based on the Miedema model and Extended Miedema theory, the formation energies of different solute components in Er-X binary system and Al-Er-X ternary systems were calculated. The results show that the variation of erbium content has little influence on the chance rate of the formation Al-Er compound in the Al-Mg-Mn-Zr-Er system. The calculated formation energies in the Er-X binary system and Al-Er-X ternary systems indicate that Al3Er phase can take priority of depositing, Al-Er-Zr ternary compounds may also found which agrees with the experimental results in references. The consistency of calculation and experiment proves that the intermetallic compounds in the Al-Mg-Mn-Zr-Er system can be predicted directly by calculating the formation energies of the reactions in Er-X binary system and Al-Er-X ternary systems with the Miedema model and Extended Miedema theory.
Key words: Al3Er; Miedema model; Extended Miedema theory; formation energy
1 Introduction
The Al-Mg alloys are widely used in automotive, marine, construction industry due to their high strength to mass ratio, reasonable corrosion resistance and superelasticity[1-2]. However, the strength of the alloys is not high enough to be used as structural materials. Al-Mg alloys derive their strength primarily from the solid solution strengthening by magnesium element, and the strength is enhanced with the increment of magnesium. An alternative effective method for increasing the strength of aluminum alloys involves the addition of some suitable alloying elements such as La, Ce, Y and especially Sc, while the effects of other single elements are scarcely referred[3]. Recent studies show that erbium (Er) is a promising alloying element in aluminium (Al) alloys, and it has been shown that a small Er-addition can improve the material properties in several Al-alloy systems[4-7]. Most of the beneficial effects from the Er-addition are linked to the formation of the phase containing erbium in the alloy.
Traditionally, formation energy for an alloy system plays an important role in the research and application of materials. Miedema model has been widely used to calculate some thermal properties of binary alloys[8-9]. OUYANG et al[10-11] calculated the formation enthalpies of alkaline metal binary alloys and Fe�CAl�CRE ternary alloys. GONCALVES and ALMEIDA[12] proposed an extended Miedema model to predict the formation energy of intermetallic compounds with more than two elements. In that scheme the crystalline structure of an alloy is used to determine the main parameters of the calculations. YAN et al[13] combined the Miedema model and the Toop model to calculate the formation energy for three alloy systems, but the agreement between calculations and experimental data still needs some improvement. ZHANG and JESSER[14] proposed a simple but effective method to extend the Miedema theory to ternary and n-component alloy systems. However, up to now the thermodynamic calculation of the intermetallic compounds in the modified 5083 aluminum alloy containing erbium (noted as Al-Mg-Mn- Zr-Er system) has not been reported. The purpose of this work is to calculate the formation energies of different solute components in Er-X binary system and Al-Er-X ternary systems and to analysis the deposited phases in the Al-Mg-Mn-Zr-Er system based on the Miedema model and Extended Miedema theory.
2 Calculation model
2.1 Miedema model
According to Miedema model[15], the formation energy, ��Hij, in the binary i-j alloys can be calculated as
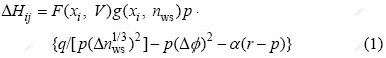



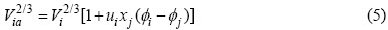
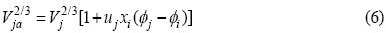
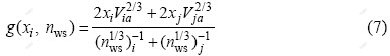
Substituting Eqs.(2)-(7) into Eq.(1), it can be obtained
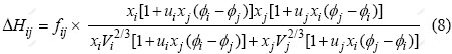
In which
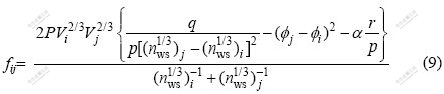
where xi and xj represent the molar fractions of components i and j, respectively; V is the molar volume, 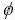 is the electron negativity, nws is the electron density; �� is the empirical constant; u, p, q, a and r are all empirical constants. In this paper, all the empirical constants are selected from Miedema model[16], in which, q/p=9.4, a equals 1.0 for solid alloys and 0.73 for liquid alloys containing a transition metal and a nontransition metal, respectively. As for u=0.14 for the alkali metallic elements, u=0.1 for the divalent metallic elements, u=0.07 for the trivalent metallic elements and Cu, Ag, Au. u=0.04 for the other metallic elements. For constant p, p=12.3 if i and j belong to transition and nontransition elements, p=14.1 if i and j are transition elements, p=10.6 if i and j are nontransition elements; r/p equals 0 when i and j are both transition or nontransition elements. Otherwise, the value of r/p corresponds with the position of i and j in the periodic system of elements[17]. All the parameters above are obtained from Refs.[16-17]. where fij can be got from Eq.(9), and the formation energy of the binary system can be got from Eq.(8).
is the electron negativity, nws is the electron density; �� is the empirical constant; u, p, q, a and r are all empirical constants. In this paper, all the empirical constants are selected from Miedema model[16], in which, q/p=9.4, a equals 1.0 for solid alloys and 0.73 for liquid alloys containing a transition metal and a nontransition metal, respectively. As for u=0.14 for the alkali metallic elements, u=0.1 for the divalent metallic elements, u=0.07 for the trivalent metallic elements and Cu, Ag, Au. u=0.04 for the other metallic elements. For constant p, p=12.3 if i and j belong to transition and nontransition elements, p=14.1 if i and j are transition elements, p=10.6 if i and j are nontransition elements; r/p equals 0 when i and j are both transition or nontransition elements. Otherwise, the value of r/p corresponds with the position of i and j in the periodic system of elements[17]. All the parameters above are obtained from Refs.[16-17]. where fij can be got from Eq.(9), and the formation energy of the binary system can be got from Eq.(8).
2.2 Extended Miedema theory
According to Refs.[18-20], the heat of mixing in a binary system consists of a negative contribution from the electronegativity difference between the two constituents, and a positive contribution from their difference in electron densities. For the alloy combined by i and j constituents, the energy of mixing is given by

For a ternary alloy, only the binary interaction between atoms is considered, and the ternary atomic interactions between the constituents are neglected when the Miedema model extends to the ternary alloys. Then, the chemical interaction contribution is given by
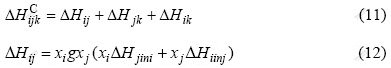
Miedema and Coworkers have calculated the values of ��Hiinj for most binary alloy systems[8-11, 18-20]. The authors calculated the corresponding values that Miedema et al did not published.
Similarly, only the binary contribution is considered in the elastic term, so the elastic contribution in a ternary alloy is given by
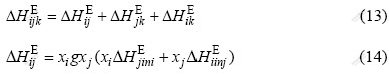
where ?HiinjE is the size-mismatch contribution to the formation enthalpy in a binary system. The FRIEDEL formula[21] is used in the present study, which is given by
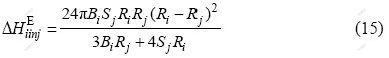
where Bi is the bulk modulus of the solute, and Sj is the shear modulus of the solvent. The values of B and S for elements have been tabulated by GSCHNEIDNER[22]. Ri and Rj are the radii for solute and solvent atoms, respectively.
The formation energy of a ternary alloy system can then be given by

in which the elastic contribution is also considered, otherwise it is represented by Eq.(11).
3 Results
In order to make the calculation representative, four classes of binary systems and three ternary systems have been calculated. They are the alloys with the addition of the transition element erbium, through all this thermodynamic calculation based on the above models, we can analysis the compounds that might be formed in the polynary system containing the promising alloy element erbium.
3.1 Er-X(Al, Mg, Mn, Zr) binary system
In this kind of binary system, Al-Er, Mg-Er, Mn-Er and Zr-Er compounds are considered for calculation of the formation energy. The molar fractions of component Er range from 0 to 1. The results are shown in Fig.1
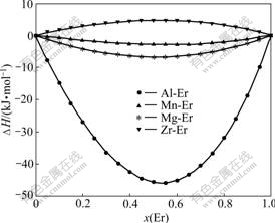
Fig.1 Calculated enthalpy of rare earth element Er reacting with Al, Mg, Mn and Zr, respectively
One can see from Fig.1 that the calculated formation enthalpies mixing heats vs composition do not have intercross, which indicates that the variation of erbium content has little influence on the chance rate of the formation Al-Er compound in the Al-Mg-Mn-Zr-Er system. It can be seen from Fig.1 that Al has good compatible with Er, the minimum formation energy of Al-Er is -47 kJ/mol. Although the formation energy of Mg-Er, Mn-Er shows a negative value, the minimum formation energy of them is only -7 kJ/mol. The formation energy of Zr-Er alloy has a positive value, in which the Zr-Er intermetallic compound does not exist as far as the research concerned. The results from the formation energy of Er-X(Al,Mg,Mn,Zr) binary system show that Al-Er binary compounds have the minimum formation energy, therefore, the transition element erbium will priority bonding with aluminum to form intermetallic compound. According to Al-Er binary phase diagrams, the intermetallic compounds of which the melting point is higher than 928 K include Al3Er, Al2Er, AlEr, Al2Er3 and AlEr2, but the only intermetallic compound exists when the molar fraction of component Er below 0.26 is Al3Er, the possible reactions in Al-Er binary systems at 1 340 K can be given as liquid+ Al2Er=Al3Er. Hence, the intermetallic compound with erbium existing in the multivariant Al-Mg-Mn-Zr-Er system is likely Al3Er phase.
3.2 Al-Er-X(Mg, Mn, Zr) ternary system
Three ternary systems Al-Mg-Er, Al-Mn-Er and Al-Zr-Er compounds are considered for calculation of the formation energy by Extended Miedema model. The results are shown in Fig.2.
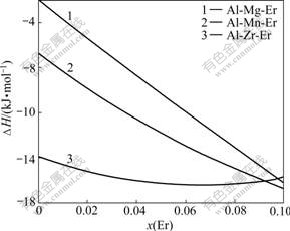
Fig.2 Calculated enthalpy of Al-Er-X(Mg, Mn, Zr) ternary system
For the above calculations, the elastic contribution has not considered, because of the lack of the data of bulk modulus of the solute, and the shear modulus of the solvent. We take the molar fraction of component Al is 0.9, the molar fractions of other components Er and X(Mg, Mn, Zr) range from 0 to 0.1. As shown in Fig.2 that a cross spider is formed when the molar fraction of component Er exceeds 0.09. The formation energy of Al-Er-Zr shows a negative value relative to Al-Er-Mg and Al-Er-Mn ternary compound when the molar fraction of component Er ranges from 0 to 0.09. And the formation energy of Al-Er-Zr is below -14 kJ/mol, which indicates that Al-Er-Zr ternary compound most likely exists. Al-Er-Mn ternary compound may also be found with very large amounts of erbium, which will be only happen when the alloy elements accumulate together; the formation of Al-Er-Mg ternary compounds seems impossible from Fig.2 as the formation energy holds on a high lever. The results from the formation energy of Al-Er-X(Mg,Mn,Zr) ternary system shows that Al-Er-Zr ternary compounds have the minimum formation energy in all the ternary compounds with erbium, therefore, the element erbium will priority bonding with zirconium and aluminum to form ternary intermetallic compound.
4 Discussion
Values of formation enthalpy calculated by the Miedema model and extended Miedema theory with the experiments for the Al-Mg-Mn-Zr-Er system were compared. Overall, the present calculations are in agreement with the experimental data. This intermetallic compound existing in Al-Mg-Mn-Zr-Er system is similar to that for the Er-X binary system and Al-Er-X ternary system calculated by Miedema and extended Miedema theory.
It has been reported that erbium exists three forms in the Al-Mg-Mn-Zr-Er system[23-25]: resolved in ��(Al) based solid solution, forming primary phase A13Er or eutectic compound distributing continuously and dis- continuously on grain boundaries, precipitated in the form of the fine A13Er and Al3(ZrEr) particles during hot-treatment. The solid solution Er would precipitate in the form of fine and dispersely distributed second phase with the size about 20 nm, which would act as effective strengthen phase in Al-Mg-Mn-Zr-Er system. The growth of grains would be hindered by grainy Al3Er distributed continuously and discontinuously on grain boundaries, they are broken during rolling which may be helpful for high-temperature mechanical properties of alloy.
Fig.3 shows the possible compounds existing in Al-0.4Er, Al-4.5Mg-0.4Er, Al-4.5Mg-0.7Mn-0.4Er and Al-4.5Mg-0.7Mn-0.1Zr-0.4Er systems by XRD analysis, it can be seen from the experiment data that they all have the Al3Er phase after erbium addition. Fig.4 indicates that nano-Al3Er precipitation and Al3(ZrEr) phase with a lattice constant of 0.399 3 nm co-exist in the Al-Mg-Mn- Zr-Er system[25]. The results show that Zr dissolves in the Al3Er phase by replacing Er, thus forming Al3(Er1-x, Zrx), the exactly composition of this phase has not been reported.
The consistency of calculation and experiment shows that the Er-intermetallic compounds in the Al alloy with erbium addition are Al3Er and Al3(ZrEr) phases. Of course the experimental data may also be the source of some of the discrepancy. The experimental data not only are derived from different determinations by various authors, but even in the case they are found by the same authors for the determination of the same alloy, there is some variation in the values. Otherwise, we should also consider the content of the alloy elements and the process of the metallurgy, because they can affect the existing form of the alloy elements as it can effect the existing form of the alloy elements.
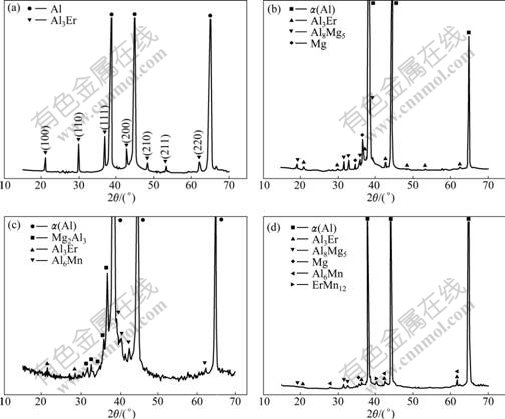
Fig.3 X-ray diffraction patterns of experiment alloys[23-25]: (a) Al-0.4Er; (b) Al-4.5Mg-0.4Er; (c) Al-4.5Mg-0.7Mn-0.1Zr-0.4Er; (d) Al-4.5Mg-0.7Mn-0.4Er
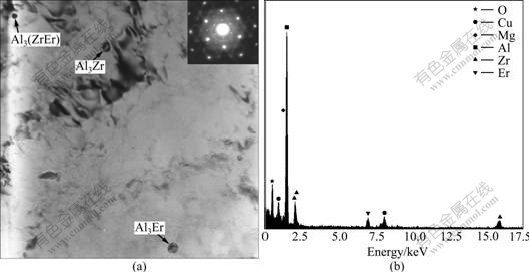
Fig.4 TEM image of Al-4.5Mg-0.7Mn-0.1Zr-0.4Er alloys[25]: (a) TEM image of Al3(ZrEr) corresponding with SAED; (b) EDS of Al3(ZrEr)
5 Conclusions
1) In the Er-X(Al, Mg, Mn, Zr) binary system, the calculation of formation energies of the Er-intermetallic compounds indicates that Er-Al binary compounds have the minimum formation energy. Thus, the possible binary Er-intermetallic compounds in the multivariant Al-Mg-Mn-Zr-Er system are likely Al3Er phase when the molar fraction of component Er is below 0.26. The quantity of erbium has little influence on the chance rate of the formation Al-Er compound.
2) In the Al-Er-X(Mg, Mn, Zr) ternary system, Al-Er-Zr ternary compound has the minimum formation energy in all the ternary Er-compounds, therefore, the transition element erbium, zirconium and aluminum will priority bonding together to form compound; Al-Er-Mn ternary compounds may also found with very large amounts of erbium, this will be only happened when the alloy elements accumulate together.
3) The experimental data are in good agreement with the calculation results, which indicates it is suitable to predict the deposited phases in the Al-Mg-Mn-Zr-Er system by calculating the formation energy of different Er-intermetallic compounds.
References
[1] HECHT R L, KANNAN K, GHOSH A K, BIELER T R. Superplasticity and superplastic forming [M]. Warrendale PA, USA: The Metallurgical Society, 1995: 259-264.
[2] KAIBYSHEV R, MUSIN F, LESUER D R, NIEH T G. Superplastic behavior of an Al-Mg alloy at elevated temperatures [J]. Mater Sci Eng A, 2003, 342: 169-173.
[3] ZHOU Shou-ze, PAN Fu-sheng, DING Pei-dao. Effects of yttrium on as-cast structure of 2024 aluminium alloy [J]. Journal of the Chinese Rare Earth Society, 1993, 11(3): 277-282. (in Chinese)
[4] NIE Zuo-ren, JIN Tou-nan, ZOU Jing-xia. Development on research of advanced rare-earth aluminum alloy [J]. Trans Nonferrous Met Soc China, 2003, 13(3): 509-514.
[5] ZHAO Zhong-kui, ZHOU Tie-tao, LIU Pei-ying, CHEN Chang-qi. Observation of formed Er phase in Al-Zn-Mg-Cu-Li alloys by TEM [J]. Rare Metal Materials and Engineering, 2004, 33(10): 1108-1111. (in Chinese)
[6] LI Yun-tao, LIU Zhi-yi, XIA Qing-kun, YU Ri-cheng, LIU Yan-bin. Effect of trace addition of Er on microstructure and ageing behavior of Al-Cu-Mg-Ag-Zr alloy [J]. Trans Materials and Heat-Treatment, 2007, 28(2): 49-54. (in Chinese)
[7] YANG Fu-bao, LIU En-ke, XU Jun, SHI Li-kai. Effect of Er on the microstructures and mechanical properties of as-cast Al-Mg-Mn-Zn-Sc-Zr-(Ti) filler metals [J]. Acta Metall Sin, 2008, 44(8): 911-914. (in Chinese)
[8] FAN Tong-xiang, YANG Guang, ZHANG Di. Thermodynamic of alloying addition on in-situ reinforced TiB2/Al composites [J]. Metallurgical and Materials Transaction A, 2005, 36A: 225-232.
[9] ZHU Yan, YANG Yan-qing, SUN Jun. Calculation of activity coefficients for components in ternary Ti alloy and intermetallics as matrix of composites [J]. Trans Nonferrous Met Soc China, 2004, 14(5): 875-879.
[10] OUYANG Yi-fang, ZHANG Bang-wei, LIAO Shu-zhi, JIN Zhan-peng. Formation enthalpies of alkaline metal binary alloys [J]. Rare Metal Materials and Engineering, 1995, 24(4): 14-18. (in Chinese)
[11] OUYANG Yi-fang, ZHONG Xiao-ping, DU Yong, JIN Zhan-peng, HE Yue-hui, YUAN Zhao-hui. Formation enthalpies of Fe-Al-RE ternary alloys calculated with a geometric model and Miedema��s theory [J]. JAlloyCompd,2006, 416: 148-154.
[12] GONCALVES A P, ALMEIDA M. Extended Miedema model: Predicting the formation enthalpies of intermetallic phases with more than two elements [J]. Physica B, 1996, 228: 289-294.
[13] YAN Xiao-hua, TANG Wei-hua, QIAO Zhi-yu, RAO Guang-hui, LIANG Jing-kui, XIE Si-shen. Calculation of the heat of formation: Ternary alloy system [J]. J Rare Earths, 1993, 4: 252-256.
[14] ZHANG Bang-wei, JESSER W A. Formation energy of ternary alloy systems calculated by an extended Miedema model [J]. Physica B, 2002, 315: 123-132.
[15] DING Xue-yong, FAN Peng, HAN Qi-yong. Models of activity and activity interaction parameter in ternary metallic melt [J]. Acta Metallurgica, 1994, 30(14): 50-60. (in Chinese)
[16] de BOER F R, BOOM R, MATTENS W C M. Cohesion in transition metal alloys [M]. North-Holland: Elsevier Science Publishers BV, 1998: 26-32.
[17] GUO Jing-jie, SU Yan-qing. Thermodynamic analysis of Ti alloy during ISM smelting [M]. Harbin: Harbin Institute of Technology Press, 1998.
[18] MIEDEMA A R. The electronegativity parameter for transition metals: Heat of formation and charge transfer in alloys [J]. J Less-Common Met, 1973, 32: 117-136.
[19] MIEDEMA A R, CHATEL P F, BOER F R. Cohesion in alloys- fundamentals of a semi-empirical model [J]. Physica B, 1980, 100: 1-28.
[20] BOER F R, BOOM R, MATTENS W C M, MIEDEMA A R, NIESSEN A R. Cohesion in metals [M]. North-Holland, Amsterdam, 1988.
[21] FRIEDEL J. Emploi d'une. masse effective dans les semi- conducteurs [J]. Adv Phys, 1954, 20: 998-1001. (in French)
[22] GSCHNEIDNER K A. Concerning the valences of the cerium allotropes [J]. Solid State Phys, 1964, 16: 275-282.
[23] XING Ze-bing, NIE Zuo-ren, ZOU Jing-xia, GAO Xu-dong. Existing form and effect of erbium in Al-Er alloy [J]. Journal of the Chinese Rare Earth Society, 2007, 25(2): 234-238. (in Chinese)
[24] JI Xiao-lan, XING Ze-bing, NIE Zuo-ren, ZOU Jing-xia, ZUO Tie-yong. Effects of Trace erbium on structure and tensile properties of Al-4.5Mg-0.7Mn alloy [J]. Chinese Journal of Rare Earth, 2006, 30(4): 462-465. (in Chinese)
[25] XING Ze-bing. Study on microstructure and properties of Er microalloyed Al-4.5Mg-0.7Mn-0.1Zr alloys [D]. Beijing: Beijing University of Technology Press, 2008: 89-95. (in Chinese)
Foundation item: Projects(2006AA03A207, 2007AA03Z514) supported by the National High-tech Research and Development Program of China
Corresponding author: NIE Zuo-ren; Tel: +86-10-67391536; E-mail: zrnie@bjut.edu.cn
DOI: 10.1016/S1003-6326(09)60198-9
(Edited by LI Xiang-qun)