����ɸ������ˮ�ڸ�ѡҩ���ϳ��е�Ӧ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���11��
�������ߣ����� ��� ���� ������ ����� ���� ����Ȩ �Ž� ��Ʊ�
����ҳ�룺3093 - 3102
�ؼ��ʣ�����ɸ����ˮ��֬�����������ѡ�����ٿ�
Key words��zeolite; wastewater; fatty acid; saponification; flotation; scheelite
ժ Ҫ��ͨ��������Ӧ�����ѡ���飬�Էϼ�Һ��Ϊ�������Ƶ����Ʒ��������������֬����IJ������ܵ�Ӧ�ý����о�����������������������Ӧ���������Ϊ�������ٶ�55 r/min����ʼ�¶�40 ��C�ͽ���ʱ��45 min��ͬʱ��ʵ���ҹ�ģ��ҵ��ģ�ĸ�ѡ�����������ˮ�ϳɵ�֬�����εõ����������ƺϳɵ�֬�������൱��ָ�ꡣ��ˣ������Է���ɸ���������в����ķ�ˮ����������������֬���������ǿ��еġ��ϼ�Һ�Ŀ�����ò������ٻ�����Ⱦ��ͬʱ���������õľ���Ч�档
Abstract: The application of waste alkali liquids as a substitute of sodium hydroxide for the saponification to improve the collection performance of fatty acids was investigated by saponification reaction test and flotation test. The results of the saponification reaction test indicated that the optimal conditions for the saponification were stirring rate of 55 r/min, initial temperature of 40 ��C and stirring time of 45 min. Meanwhile, the laboratory scale and industrial scale flotation experiments showed that the fatty acid salt synthesized by wastewater achieved an index comparable to fatty acid sodium synthesized by sodium hydroxide. As a consequence, it was feasible to replace sodium hydroxide with the wastewater from zeolite production for fatty acid saponification. The cross-border utilization of waste alkali liquids not only reduced environmental pollution, but also produced excellent economic benefits.
Trans. Nonferrous Met. Soc. China 30(2020) 3093-3102
Xu WANG1,2,3, Hao SONG3,4, Fen JIAO1,2, Wen-qing QIN1,2, Cong-ren YANG1,2,Yan-fang CUI1,2, Zheng-quan ZHANG1,2, Jian ZHANG5, Hao-bing LI5
1. School of Mineral Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Hunan Province for Clean and Efficient Utilization of Strategic Calcium-containing Mineral Resources, Central South University, Changsha 410083, China;
3. Luoyang Zhenbei Industry and Trade Co., Ltd., Luoyang 471000, China;
4. College of Engineering, Drexel University, Philadelphia, PA 19102, USA;
5. Luoyang Yulu Mining Co., Ltd., Luoyang 471000, China
Received 13 January 2020; accepted 16 July 2020
Abstract: The application of waste alkali liquids as a substitute of sodium hydroxide for the saponification to improve the collection performance of fatty acids was investigated by saponification reaction test and flotation test. The results of the saponification reaction test indicated that the optimal conditions for the saponification were stirring rate of 55 r/min, initial temperature of 40 ��C and stirring time of 45 min. Meanwhile, the laboratory scale and industrial scale flotation experiments showed that the fatty acid salt synthesized by wastewater achieved an index comparable to fatty acid sodium synthesized by sodium hydroxide. As a consequence, it was feasible to replace sodium hydroxide with the wastewater from zeolite production for fatty acid saponification. The cross-border utilization of waste alkali liquids not only reduced environmental pollution, but also produced excellent economic benefits.
Key words: zeolite; wastewater; fatty acid; saponification; flotation; scheelite
1 Introduction
Water is an indispensable resource for the survival of animals and plants and the most essential component of living organisms. It is closely related to agriculture, industry and other human activities [1-3]. Due to the limited availability of freshwater resources on the earth, the treatment of industrial wastewater and the recycling of water resources have always been the focus of research [4-10].
Zeolite is widely used as a catalyst or adsorbent for industrial processes, such as air purification, removal of heavy metal ions and organic pollutants in water, and catalytic cracking of oil because of its special properties [11-17]. However, due to the low utilization rate of alkali and low ion exchange rate, a large amount of high-concentration alkali-containing wastewater is generated during the hydrothermal synthesis of zeolite [18-21]. Alkali-containing wastewater is a hazard and has strong corrosive properties. When the waste alkali liquid is discharged into the water body, the water body will lose its self-purification ability, resulting in the reduction or destruction of water resources, causing soil salinization and destroying the ecological environment [3]. If humans drink alkaline water with a high concentration, the metabolism will be out of balance, leading to digestive disorders [22]. Therefore, it must be properly treated before it can be discharged into the storage body.
Neutralization is the most common method for the treatment of the waste alkali liquids, but the salts produced by the neutralization process require further processing [23,24]. Nanofiltration and membrane filtration are also used to treat alkali- containing wastewater [5,25]. By using membrane bioreactor, nanofiltration and reverse osmosis to treat woolen textile wastewater, the reuse of wastewater can be achieved [26]. Oxidation and desalination are used to treat the industrial organic alkali-containing wastewater, but their treatment effect on high-concentration alkali-containing wastewater is not good [2,27,28]. In recent years, the use of biotechnology to treat wastewater has become the focus of research due to its environmentally friendly process [29,30].
Fatty acid is a common collector of oxide ore. It is widely used in the flotation of calcium- containing minerals such as scheelite, apatite, fluorite and calcite [31-34]. The ability of fatty acid collection is relatively strong, but its dispersion is poor, especially at the low temperatures. Fatty acids are usually saponified to produce fatty acid salts to improve their solubility and dispersion in slurry [35-37]. The fatty acid is commonly saponified with an alkali solution prepared with sodium hydroxide. Based on the above situation, the research ideas of wastewater produced in zeolite production as a substitute for sodium hydroxide in the synthesis of fatty acid salts were proposed, as shown in Fig. 1.
In this work, the application of waste alkali liquids from the zeolite production process instead of sodium hydroxide in the saponification of fatty acids was provided. The saponification reaction conditions and the effect of the application effect of final product in scheelite beneficiation plant were studied. The potential economic benefits of wastewater used in the production of flotation reagent were investigated. The new ideas for cross-border utilization of waste alkali liquids were explored.
2 Experimental
2.1 Sample of wastewater
The wastewater sample was taken from Luoyang Tianping Molecular Sieve Co., Ltd., (LYTP) which mainly produces zeolite A, X and Y by hydrothermal synthesis. Due to technical limitations, the alkali in the mother liquor cannot be fully utilized, and the waste mother liquor was mixed with filtered water and discharged into the wastewater tank. The chemical composition of wastewater was relatively stable. The collected water was mixed samples taken continuously every two hours for a week from the inlet of wastewater tank. The chemical compositions of the wastewater are shown in Table 1.
The OH- concentration of wastewater was 4.00 mol/L. The concentrations of other ions were 53.16 g/L K, 107.38 g/L Na, 0.14 g/L Ca, 0.38 g/L Mg, 1.04 g/L Al and 0.55 g/L Si.
2.2 Methods
2.2.1 Saponification reaction
The relationship between fatty acids and hydroxide ions in the saponification reaction is described by Eq. (1) [37]:
RCOOH+OH-=RCOO-+H2O (1)
The fatty acid used in the saponification was sampled from raw materials of fatty acid sodium production in Luoyang Zhenbei Industry and Trade Co., Ltd., (LYZB), China. The fatty acid salt (FAS) produced by LYZB was applied to Luanchuan, the largest associated scheelite mining area in China [38]. The indicators of fatty acid are shown in Table 2.
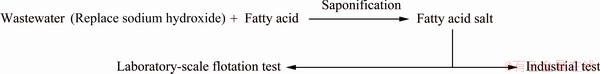
Fig. 1 Research route of wastewater reuse
Table 1 Chemical composition of wastewater (g/L)
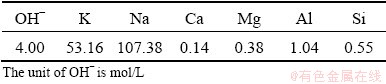
Table 2 Main indicators of fatty acid
The saponification value of fatty acids was 148.7 mg/g KOH, which was equivalent to 2.66 mmol of OH- required for complete saponification of fatty acids per gram.
If the fatty acid saponification was insufficient, the low-temperature collection performance of FAS would still be poor. It was necessary to find suitable saponification conditions so that the collector can achieve a good low-temperature collection performance. According to the relevant studies, the stirring rate, reaction time, initial temperature, and the amount of fatty acid and alkali water added were the main factors affecting the saponification reaction [39-41]. When the amount of alkali water added was increased, the saponification reaction rate was accelerated, but the active ingredient of the final product was lowered. To ensure the active ingredient content of the final product, the amount of alkali water added was fixed as the theoretical value required for complete saponification of the fatty acid. Therefore, the effects of the stirring rate, the initial temperature and the stirring time on the saponification reaction were investigated.
The saponification test procedures were described as follows: 200 g fatty acids were first added to a 500 mL breaker in a water bath, stirred and heated, followed by the addition of wastewater, saponification began. Full saponification of 200 g fatty acids theoretically required about 133 mL of wastewater. The wastewater was divided into three equal parts and added to the beaker in the same time interval. After the stirring time was reached, the reaction was completed to obtain the collector product. Because saponification reaction was exothermic, the water bath pot stopped to be heated after the reaction began [39]. The appropriate saponification reaction conditions were determined by the collection performance of FAS to scheelite at a low temperature.
2.2.2 Flotation test
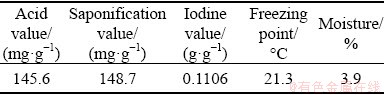
The scheelite sample used in the experiment was taken from molybdenum tailings processed by Luoyang Yulu Mining Co., Ltd., (LYYL). The samples were homogenized and split into representative sub-samples for laboratory tests. The main chemical element analysis results of samples by X-ray fluorescence spectrometry are shown in Table 3.
Table 3 Main element content of molybdenum tailings (wt.%)
The content of WO3 was 0.067 wt.%. The collection performance of the collector was evaluated by the WO3 recovery of scheelite roughing at a low temperature. The flotation slurry and the supplementary water were frozen to ensure that the slurry temperature was lower than 13 ��C during flotation. The flowsheet of scheelite flotation tests is presented in Fig. 2.
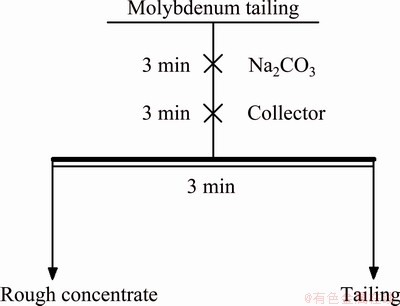
Fig. 2 Flowsheet of scheelite flotation test
The flotation tests were carried out with a 1.5 L-XFD flotation machine (Jilin Exploring Machinery Plant, China). The solid content of flotation pulp was controlled at 40%-42%. The sodium carbonated dosage was 1800 g/t, the collector dosage ranged from 160 to 260 g/t. These reagents were diluted in normal temperature water to a concentration of 5% before being added to the slurry. The mixing time of the reagent was 3 min and the flotation time was also 3 min. The recovery of WO3 was quantified by Eq. (2):
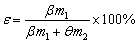 (2)
(2)
where �� is the roughing recovery of WO3; �� and �� are the WO3 grades of rough concentrate and tailing, respectively; m1 and m2 are the mass of rough concentrate and tailing, respectively.
2.2.3 Industrial test
Industrial test was carried out in LYYL, which was one of the three largest scheelite beneficiation plants in Luanchuan area and had three independent and parallel scheelite roughing series. The process flow, reagent regimes, equipment model and ore properties of each roughing series were basically the same. The processing capacity of each series was 6000 t/d. Three series of rough concentrates were combined and thickened for heating cleaning. Industrial tests were performed from January 1, 2018 to January 31, 2018, during the period of Luanchuan area temperature was low and scheelite roughing slurry temperature did not exceed 15 ��C. The collector synthesized with waste alkali liquids was used for No. 1 scheelite roughing series and the collector synthesized with sodium hydroxide was used for No. 2 and No. 3 scheelite roughing series. The roughing indexes of each series were summarized to analyze the application effect of collector synthesized with waste alkali in details. The process of roughing scheelite is shown in Fig. 3.
2.2.4 Analytical methods
The main chemical elements in wastewater were determined by inductively coupled plasma optical emission spectrometry (ICP-OES, SPECTROBLUE, Germany). The contents of different elements of the scheelite were indicated by X-ray fluorescence (XRF, S0902724, Rigaku, Japan). All analyses were performed at the reliable laboratory of analysis and testing of Changsha Institute of Mining and Metallurgy, which was certified by the state.
3 Results and discussion
3.1 Performance of FAS with different saponification conditions
3.1.1 Stirring rate
The effect of stirring rate on the saponification effect was investigated. The stirring rates were set as 40, 45, 50, 55 and 60 r/min, respectively. The initial temperature of the saponification reaction was fixed at 40 ��C, and the stirring time was fixed at 60 min. The dosage of sodium carbonate was fixed at 1800 g/t in the flotation process. The results are presented in Fig. 4.
As shown in Fig. 4, when the stirring rate was lower than 50 r/min, the recovery of scheelite increased with the rise of the stirring rate in the range of the collector dosage from 160 to 260 g/t. After the stirring rate exceeded 50 r/min, the growth rate of the scheelite recovery was slowed down. When the dosage of collector was 220 g/t, the recoveries of scheelite at the stirring rates of 55 and 60 r/min were 83.49% and 83.36%, respectively. There was no significant difference in recovery between the two stirring rates. Therefore, the stirring rate of the saponification reaction was set as 55 r/min.
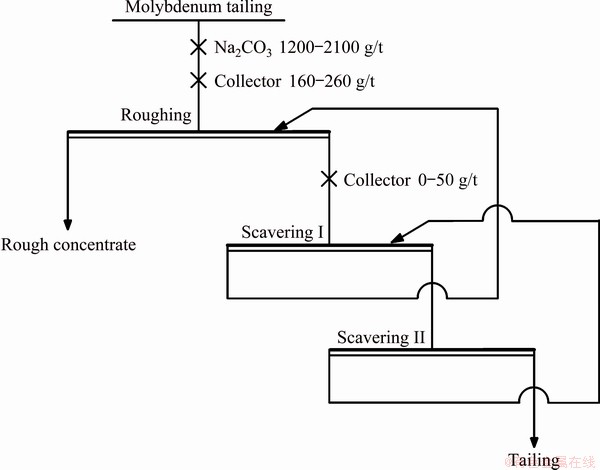
Fig. 3 Roughing flowsheet of LYYL

Fig. 4 Effect of stirring rate on collecting capability of collector
3.1.2 Initial temperature
The saponification reaction started slowly at normal temperature, and a suitable initial temperature allowed the saponification reaction to start rapidly. To explore the effect of initial temperature of the saponification reaction on the collector performance, the initial temperatures were set as 30, 35, 40, 45 and 50 ��C, respectively. The stirring rate of the saponification reaction was fixed at 55 r/min, and the stirring time was tentatively set at 60 min. The dosage of sodium carbonate was fixed at 1800 g/t in the flotation process. The results are presented in Fig. 5.
As shown in Fig. 5, with the same dosage of the collector, the scheelite recovery increased gradually with the rise of the initial temperature. But, the recovery of scheelite under same collector dosages was not significantly different after the initial temperature exceeded 40 ��C. Under the collector dosage of 220 g/t, the recoveries of scheelite at the initial temperature of 40, 45 and 50 ��C were 82.34%, 83.01% and 83.82%, respectively. This indicated that sufficient energy could be provided for the saponification reaction to start quickly at the initial temperature of 40 ��C. Therefore, the initial temperature of the saponification reaction was set as 40 ��C.
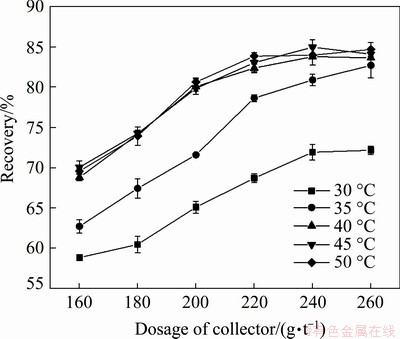
Fig. 5 Effect of initial temperature on collecting capability of collector
3.1.3 Stirring time
The effect of stirring time on collector was investigated under the following conditions: stirring rate of 55 r/min, initial temperatures of 40 ��C, sodium carbonate of 1800 g/t. The stirring time was set as 30, 45, 60 and 75 min, respectively. The results are presented in Fig. 6.
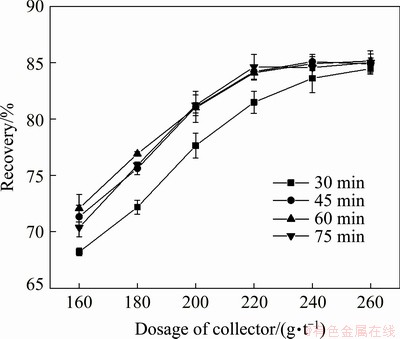
Fig. 6 Effect of stirring time on collecting capability of collector
As shown in Fig. 6, when the dosage of the collector was 220 g/t, the recoveries of scheelite at the stirring time of 30, 45, 60 and 75 min were 81.48%, 84.20%, 84.11% and 84.63%, respectively. The recovery of scheelite was not significantly changed with the increase of stirring time under the same collector dosage after the saponification reaction stirring time exceeded 45 min. This indicated that the saponification reaction might have been relatively complete after the stirring time exceeded 45 min. Therefore, the stirring time of the saponification reaction was set as 45 min. As a result, the stirring rate, initial temperature and stirring time of the saponification reaction were finally determined to be 55 r/min, 40 ��C and 45 min, respectively.
3.2 Comparison of FAS with different alkali sources
The lye having the same concentration of OH- as that of the wastewater was prepared with sodium hydroxide for the synthesis of FAS. The FAS synthesized using two alkali sources was compared through flotation tests. Meanwhile, the effect of other ions in the wastewater on the performance of the collector was also investigated by comparative experiments.
The effects of FAS synthesized by two alkali sources on the flotation of scheelite were studied under different dosages. The dosage of sodium carbonate was fixed at 1800 g/t. The results are presented in Fig. 7.
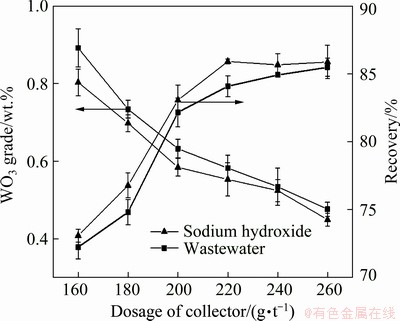
Fig. 7 Comparison of FAS synthesized from two alkali sources at different dosages
Figure 7 shows that the scheelite recovery gradually increased and the WO3 grade in the rough concentrate gradually decreased as the collector dosage increased. After the collector dosage exceeded 220 g/t, the recovery of scheelite hardly increased, and the grade of WO3 in the rough concentrate decreased. Therefore, the optimum dosage of both collectors was 220 g/t. Under the optimal dosage, when the FAS synthesized by sodium hydroxide was used as the collector, the recovery of scheelite was 85.92 wt.%, and the grade of WO3 of rough concentrate was 0.553 wt.%. When using FAS synthesized from wastewater as the collector, the recovery of scheelite was 84.09%, and the grade of WO3 of rough concentrate was 0.582 wt.%.
With the same collector dosage, the recovery of scheelite by using FAS synthesized with sodium hydroxide was slightly higher than that of wastewater. The rough concentrate grade obtained by using FAS synthesized with wastewater was higher than that of sodium hydroxide. In general, the performance of FAS synthesized by the two alkali sources was comparable. Other ions had little effect on the performance of FAS synthesized with wastewater.
3.3 Industrial test
According to the best conditions in the laboratory, the large-scale production of collectors is carried out in a reaction kettle. The No. 1 series of roughing used FAS synthesized by wastewater, and the No. 2 and No. 3 series used FAS synthesized by sodium hydroxide during the industrial experiment conducted in LYYL. The reagents added to the scheelite roughing section were sodium carbonate and FAS. The dosages of flotation reagents were adjusted according to the nature of the ore and the actual situation of on-site production. The dosage of sodium carbonate was in the range of 1200-2100 g/t, and the dosage of FAS was in the range of 160-260 g/t. Flotation column was used for scheelite flotation in LYYL. Compared with the laboratory flotation machine, the WO3 grade of rough concentrate obtained by flotation column was higher, but the recovery was lower. The temperature of the slurry was between 10 and 15 ��C during industrial test. The statistical results of production indicators during the industrial test are shown in Figs. 8 and 9. The average dosage of flotation reagents of each series during the industrial test is shown in Table 4.
Figure 8 reveals that the daily recovery rates of the various series were constantly fluctuating throughout the industrial trial. The overall fluctuation trend of the three series of recovery rates was similar. Figure 9 indicates that the monthly cumulative recoveries of the three roughing series were 75.79%, 75.82% and 75.68%, respectively.
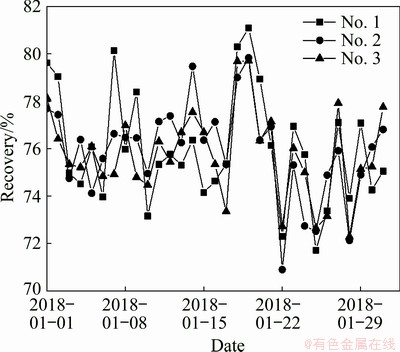
Fig. 8 Statistics of scheelite recovery for each flotation series
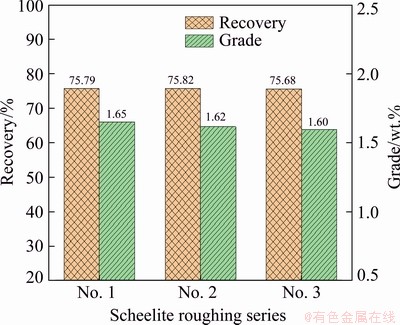
Fig. 9 Monthly cumulative production index for each scheelite roughing series
Table 4 Monthly average dosage of reagents for each scheelite roughing series
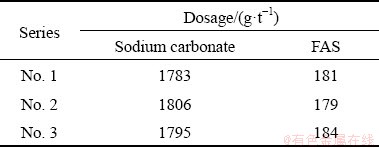
The monthly cumulative grades of WO3 in rough concentrates of three roughing series were 1.65 wt.%, 1.62 wt.% and 1.60 wt.%, respectively. The monthly cumulative production indexes of the three series were comparable. Table 4 shows that the lowest monthly average consumption of sodium carbonate was 1783 g/t of the No. 1 series, followed by 1795 g/t of the No. 3 series and 1806 g/t of the No. 2 series. The monthly average dosages of FAS of three series were similar, with 181, 179 and 184 g/t, respectively.
The effect of two kinds of FAS was equal under the same production conditions. Industrial test results confirmed that the waste alkali liquids from the production process of zeolite could completely replace sodium hydroxide for the synthesis of FAS.
3.4 Technical and economic evaluation
The utilization of waste alkali liquids for the synthesis of flotation reagents had absolute advantages over other treatment technologies. The high-concentration alkali-containing waste liquid treated by the neutralization method had relatively high efficiency, but a large amount of salt substances were generated. These salts were still highly corrosive and caused secondary pollution, which required further desalination before they can be discharged [23]. Most of the ions in the wastewater can be removed by membrane filtration techniques [25,26]. However, the filter membrane was highly susceptible to contamination during operation, resulting in a significant decrease infiltration flux and a deteriorating treatment effect [42,43]. Although biodegradation and oxidation were environmentally friendly and often used to treat organic wastewater, they were not suitable for high-concentration alkali-containing waste liquids, because the growth and reproduction of microorganisms were extremely demanding on water quality [29,30,44]. As an alternative to sodium hydroxide, the wastewater was 100% utilized, and secondary pollution was avoided.
Turning wastewater from the production of zeolite into a useful resource not only avoided environmental pollution but also saved the cost of wastewater treatment. Taking LYZB and LYTP as examples, the waste alkali liquids discharged by LYTP was about 200 m3 every month, which required about 40 t of sulfuric acid to neutralize. Based on the price of commercially available industrial sulfuric acid at 600 Yuan/t, the annual cost was about 288000 Yuan. If the waste alkali liquids were used in the synthesis of FAS by LYZB, it could replace 32 t of sodium hydroxide every month. Based on the price of commercially available industrial sodium hydroxide at 5000 Yuan/t, the annual savings was 1920000 Yuan. The annual savings of wastewater treatment costs and the cost of purchasing sodium hydroxide totaled 2208000 Yuan. At the same time, it also saved 2400 m3 of industrial water every year.
4 Conclusions
(1) It was completely feasible to replace sodium hydroxide with waste alkali liquids. The optimum stirring rate, the optimum starting temperature and the optimum stirring time for the saponification reaction were 55 r/min, 40 ��C and 45 min, respectively.
(2) The collector synthesized from wastewater has achieved an index comparable to collector synthesized by sodium hydroxide in the laboratory scale and industrial scale flotation tests. Other ions in the wastewater had little effect on the performance of FAS.
(3) The successful utilization of wastewater in the synthesis of FAS not only enabled the full utilization of water resources but also created enormous economic value. It was an environmentally friendly method of treating wastewater, which fundamentally avoided the pollution of wastewater to the environment. At the same time, it also provided new ideas for the treatment of other industrial waste alkali liquids and the sustainable development of the ecological environment.
References
[1] AHMED A. Recycling of polluted wastewater for agriculture purpose using electrodialysis: Perspective for large scale application [J]. Chemical Engineering Journal, 2017, 323: 1-18.
[2] LI Xu-fang, CHEN Wei-yu, MA Lu-ming, WANG Hong-wu, FAN Jin-hong. Industrial wastewater advanced treatment via catalytic ozonation with an Fe-based catalyst [J]. Chemosphere, 2017, 195: 336-343.
[3] MO Jia-hao, YANG Qi, ZHANG Na, ZHANG Wen-xiang, ZHENG Yi, ZHANG Zhi-en. A review on agro-industrial waste (AIW) derived adsorbents for water and wastewater treatment [J]. Journal of Environmental Management, 2018, 227: 395-405.
[4] DELKASH M, BAKHSHAYESH B E, KAZEMIAN H. Using zeolitic adsorbents to cleanup special wastewater streams: A review [J]. Microporous and Mesoporous Materials, 2015, 214: 224-241.
[5] GOH P S, ISMAIL A F. A review on inorganic membranes for desalination and wastewater treatment [J]. Desalination, 2017, 434: 60-80.
[6] LIU He, HAN Peng, LIU Hong-bo, ZHOU Guang-jie, FU Bo, ZHENG Zhi-yong. Full-scale production of VFAs from sewage sludge by anaerobic alkaline fermentation to improve biological nutrients removal in domestic wastewater [J]. Bioresource Technology, 2018, 260: 105-114.
[7] AINIL F M U, HASSIMI A H, MOHD S T, SITI R S A. A review of the potentials, challenges and current status of microalgae biomass applications in industrial wastewater treatment [J]. Journal of Water Process Engineering, 2017, 20: 8-21.
[8] PALMER M, HATLEY H. The role of surfactants in wastewater treatment: impact, removal and future techniques: A critical review [J]. Water Research, 2018, 147: 60-72.
[9] CHAI Li-yuan, PENG Cong, MIN Xiao-bo, TANG Chong-jian, SONG Yu-xia, ZHANG Yang, ZHANG Jing, ALI M. Two-sectional struvite formation process for enhanced treatment of copper�Cammonia complex wastewater [J]. Transactions of Nonferrous Metals Society of China, 2017, 17: 457-466.
[10] HU Bin, YANG Tian-zu, LIU Wei-feng, ZHANG Du-chao, CHEN Lin. Removal of arsenic from acid wastewater via sulfide precipitation and its hydrothermal mineralization stabilization [J]. Transactions of Nonferrous Metals Society of China, 2019, 29: 2411-2421.
[11] ABDULLAHI T, HARUN Z, OTHMAN M H D. A review on sustainable synthesis of zeolite from kaolinite resources via hydrothermal process [J]. Advanced Powder Technology, 2017, 8: 1827-1840.
[12] GASCON J, KAPTEJIN F, ZORNOZA B, SEBASTIAN V, CASADO C, CORONAS J. Practical approach to zeolitic membranes and coatings: State of the art, opportunities, barriers, and future perspectives [J]. Chem Mater, 2012, 24: 2829-2844.
[13] HESAS R H, BAEI M S, ROSTAMI H, GARDY J, HASSANPOUR A. An investigation on the capability of magnetically separable Fe3O4/mordenite zeolite for refinery oily wastewater purification [J]. Journal of Environmental Management, 2019, 241: 525-534.
[14] TAN Yan-Xi, HE Yan-ping, WANG Min, ZHANG Jian. A water-stable zeolite-like metal�Corganic framework for selective separation of organic dyes [J]. RSC Advances, 2013, 3: 1480-1483.
[15] YAN Jia-shuai, LI Ye, LI Hao-rui, ZHOU Yu-zhi, XIAO Hua-feng, LI Bo-lin, MA Xiao. Effective removal of ruthenium (III) ions from wastewater by amidoxime modified zeolite X [J]. Microchemical Journal, 2019, 145: 287-294.
[16] LAI Yan-qing, YANG Kai, YANG Chao, TTIAN Zhong-liang, GUO Wei-chang, LI Jie. Thermodynamics and kinetics of fluoride removal from simulated zinc sulfate solution by La(III)-modified zeolite [J]. Transactions of Nonferrous Metals Society of China, 2018, 28: 783-793.
[17] IQBAL N, IQBAL S, IQBAL T, BAKHSHESHI-RAD H R, ALSAKKAF A, KAMIL A, ABDUL KADIR M R, IDRIS M H, RAGHAV H B. Zinc-doped hydroxyapatite-zeolite/ polycaprolactone composites coating on magnesium substrate for enhancing in-vitro corrosion and antibacterial performance [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 123-133.
[18] GARCIA V F, FLORES R E, VERDUGO E C, HUERTAS F. Hydrothermal synthesis of zeolites using sanitary ware waste as a raw material [J]. Applied Clay Science, 2018, 160: 238-248.
[19] LUO Hong-wei, WEI Wee-law, WU Yi-chao, ZHU Wei- ping, YANG En-hua. Hydrothermal synthesis of needle-like nanocrystalline zeolites from metakaolin and their applications for efficient removal of organic pollutants and heavy metals [J]. Microporous and Mesoporous Materials, 2018, 272: 8-15.
[20] SAMA M A, DAVID A H, ABDULLATIF A, NICHOLAS A C, STUART M H. Understanding the seeding mechanism of hierarchically porous zeolite/carbon composites [J]. Microporous and Mesoporous Materials, 2018, 268: 109-116.
[21] NING Wei-wei, YANG Xiao-na, ZHENG Jia-jun, SUN Xiao-bo, PAN Meng, LI Rui-feng. An environmentally friendly route to prepare hierarchical ZSM-12 using waste liquor as partial nutrients [J]. Materials Chemistry and Physics, 2018, 223: 299-305.
[22] SONG Zheng-cheng, LI Ping, DING Li, LI Zhong-gen, ZHU Wei, HE Tian-rong, FENG Xin-bin. Environmental mercury pollution by an abandoned chlor-alkali plant in Southwest China [J]. Journal of Geochemical Exploration, 2018, 194: 81-87.
[23] RAJASULOCHANA P, PREETHY V. Comparison on efficiency of various techniques in treatment of waste and sewage water��A comprehensive review [J]. Resource- Efficient Technologies, 2016, 2: 175-184.
[24] LIU Jun, LI Jun, MEI Rong-wu, WANG Fu-chao, BALASUBRAMANIAN S. Treatment of recalcitrant organic silicone wastewater by fluidized-bed Fenton process [J]. Separation and Purification Technology, 2014, 132: 16-22.
[25] ZHANG Yun, SHAO Shuai, YU Wei-cheng, YANG Feng-lin, XU Xiao-chen. Study on recycling alkali from the wastewater of textile mercerization process by nanofiltration [J]. IERI Procedia, 2014, 9: 71-76.
[26] NAZLI C C, EMRAH O, NEVZAT O Y, MEHMET K. Treatment of woolen textile wastewater using membrane bioreactor, nanofiltration and reverse osmosis for reuse in production processes [J]. Journal of Cleaner Production, 2019, 223: 837-848.
[27] LUCYNA B, KAZIMIERZ B, MARTA G, STANISLAW L. Coupling of electrocoagulation and ozone treatment for textile wastewater reuse [J]. Chemical Engineering Journal, 2019, 358: 992-1001.
[28] SAJJAD A, MOHD Y B M Y, ABDUL A M A, DAVID G H, IHSAN H D, HASSIMI A H. Electrodialysis desalination for water and wastewater: A review [J]. Chemical Engineering Journal, 2019, 380: 122231.
[29] ALICIA P, JULIA C, MARIA J P, JOSE M D. Biological treatment of model dyes and textile wastewaters [J]. Chemosphere, 2017, 181: 168-177.
[30] LU Hai-fei, ZHANG Guang-ming, ZHENG Zi-qiao, MENG Fan, DU Tai-sheng, HE Shi-chao. Bio-conversion of photosynthetic bacteria from non-toxic wastewater to realize wastewater treatment and bioresource recovery: A review [J]. Bioresour Technol, 2019, 278: 383-399.
[31] INNA V F, LEV O F, ZINEB L, ODILE B, DANIEL F. Effect of calcium minerals reactivity on fatty acids adsorption and flotation [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2018, 545: 157-166.
[32] NATHALIE K, MARTUN R. Froth flotation of scheelite��A review [J]. International Journal of Mining Science and Technology, 2018, 28: 373-384.
[33] KEITH Q. Literature review on the use of natural products in the flotation of iron oxide ores [J]. Minerals Engineering, 2017, 108: 12-24.
[34] YIN Wan-zhong, WANG Ji-zhen. Effects of particle size and particle interactions on scheelite flotation [J]. Transactions of Nonferrous Metals Society of China, 2014, 24: 3682-3687.
[35] GUIMARAES R C, ARAUJO A C, PERES A E C. Reagents in igneous phosphate ores flotation [J]. Minerals Engineering, 2005, 18: 199-204.
[36] NAGARAJ D R, FARINATO R S. Evolution of flotation chemistry and chemicals: A century of innovations and the lingering challenges [J]. Minerals Engineering, 2016, 96-97: 2-14.
[37] PEARSE M J. An overview of the use of chemical reagents in mineral processing [J]. Minerals Engineering, 2005, 18: 139-149.
[38] KANG Jian-hua, HU Yue-hua, SUN WeI, LIU Run-qing, YIN Zhi-gang, TANG Hong-hu, MENG Xiang-song, ZHANG Qing-peng, LIU Hang. A significant improvement of scheelite flotation efficiency with etidronic acid [J]. Journal of Cleaner Production, 2018, 180: 858-865.
[39] AUDREY B, HELENE C, JEAN P D. Saponification of fatty slaughterhouse wastes for enhancing anaerobic biodegradability [J]. Bioresource Technology, 2009, 100: 3695-3700.
[40] JIA Rui, ZHU Jian-hua, LIU Hong-yan. Study on saponification kinetics of soybean oil [J]. Cereals and Oils, 2007, 9: 22-25.
[41] ANJA P, MAJDA K, ALJANA P. Saponification of ethyl acetate in the presence of ��-cyclodextrin [J]. Journal of Molecular Liquids, 2018, 272: 313-318.
[42] ZHU You-bing, YU Shui-li, ZHANG Bing, LI Jian-feng, ZHAO Dong-sheng, GU Zheng-yang, GONG Chao, LIU Gui-cai. Antifouling performance of polytetrafluoroethylene and polyvinylidene fluoride ultrafiltration membranes during alkali/surfactant/polymer flooding wastewater treatment: Distinctions and mechanisms [J]. Sci Total Environ, 2018, 642: 988-998.
[43] KASHIF N, GOKCE T G, BULENT K, NADIR D. Investigation of segregated wastewater streams reusability with membrane process for textile industry [J]. Journal of Cleaner Production, 2019, 228: 1437-1445.
[44] YANG Bo, WANG Qing, YE Jin-shao, XU Hui, LIU Yan-biao, LI Fang, SONG Xin-shan, LIU Jian-she, WANG Zhi-wei, WOLFGANG S. Performance and microbial protein expression during anaerobic treatment of alkali-decrement wastewater using a strengthened circulation anaerobic reactor [J]. Bioresour Technol, 2019, 273: 40-48.
�� ��1,2,3���� �3,4���� ��1,2��������1,2�������1,2������1,2������Ȩ1,2���� ��5����Ʊ�5
1. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083��
2. ���ϴ�ѧ ս�Ժ��ƿ�����Դ����Ч���ú���ʡ�ص�ʵ���ң���ɳ 410083��
3. ������ó����˾������ 471000��
4. College of Engineering, Drexel University, Philadelphia, PA 19102, USA;
5. ����ԥ�ؿ�ҵ�������ι�˾������ 471000��
ժ Ҫ��ͨ��������Ӧ�����ѡ���飬�Էϼ�Һ��Ϊ�������Ƶ����Ʒ��������������֬����IJ������ܵ�Ӧ�ý����о�����������������������Ӧ���������Ϊ�������ٶ�55 r/min����ʼ�¶�40 ��C�ͽ���ʱ��45 min��ͬʱ��ʵ���ҹ�ģ��ҵ��ģ�ĸ�ѡ�����������ˮ�ϳɵ�֬�����εõ����������ƺϳɵ�֬�������൱��ָ�ꡣ��ˣ������Է���ɸ���������в����ķ�ˮ����������������֬���������ǿ��еġ��ϼ�Һ�Ŀ�����ò������ٻ�����Ⱦ��ͬʱ���������õľ���Ч�档
�ؼ��ʣ�����ɸ����ˮ��֬�����������ѡ�����ٿ�
(Edited by Xiang-qun LI)
Foundation item: Projects (51604302, 51574282) supported by the National Natural Science Foundation of China; Project (2016RS2016) supported by the Provincial Science and Technology Leader (Innovation Team of Interface Chemistry of Efficient and Clean Utilization of Complex Mineral Resources), China; Project (2018zzts224) supported by the Postgraduate Independent Exploration and Innovation Project of Central South University, China; Project (2018TP1002) supported by the Key Laboratory of Hunan Province for Clean and Efficiency Utilization of Strategic Calcium-containing Mineral Resources, China
Corresponding author: Fen JIAO; Tel: +86-13549683403; E-mail: jfen0601@126.com
DOI: 10.1016/S1003-6326(20)65445-0

