Trans. Nonferrous Met. Soc. China 31(2021) 2493-2500
A novel green depressant for flotation separation of scheelite from calcite
Chen CHEN1,2, Wei SUN1, Hai-ling ZHU1, Run-qin LIU1
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. School of Metallurgy and Environment, Central South University, Changsha 410083, China
Received 7 August 2020; accepted 8 April 2021
Abstract: Polyaspartic acid (PASP) was used as a novel environmental-friendly depressant, and its inhibition effect on flotation performance of scheelite and calcite using sodium oleate (NaOL) as a collector was investigated by ways of flotation experiments, zeta potential measurement, contact angle measurement and infrared spectroscopic analysis (IR). The results show that PASP exhibits stronger inhibition capability and selectivity than acidified water glass, and the flotation separation of scheelite from calcite can be realized in the presence of 6 mg/L PASP and 20 mg/L NaOL at pH>6. In the presence of PASP, the zeta potential of calcite surface almost keeps unchanged after adding NaOL, and the contact angle decreases sharply. Therefore, it is indicated that PASP significantly decreases the adsorption of NaOL on calcite surface. By contrast, it has no distinct effect on the adsorption of NaOL on the scheelite surface, which is further confirmed by IR results.
Key words: polyaspartic acid; scheelite; calcite; flotation separation; inhibition
1 Introduction
Scheelite is one of the major tungsten ores which have industrial exploitation value, and it should be separated from other associated minerals for further use, generally through flotation [1-4]. Calcite is the common calcium-containing gangue mineral, nevertheless, it is difficult to separate scheelite from calcite owing to their similar surface properties and chemical reactivity with reagents, especially when using the most frequently used fatty acids collectors [5-7]. Therefore, the selective inhibition of calcite is of crucial importance in the beneficiation of scheelite.
At present, water glass or modified water glass is the most common depressant in industrial production of scheelite flotation [6-11]. In addition, researches have shown that the inorganic sodium fluorosilicate [12] and the organic reagents such as etidronic acid [13], guar gum [14] and sodium humic acid [15] can also inhibit calcite flotation effectively.
Polyaspartic acid (PASP), a representative green biodegradable polymer, has attracted increasing attention as a scale inhibitor and dispersant during water treatment due to the typical non-toxic and biodegradable nature. It can be seen from the published literatures that PASP has fine chelating ability and dispersibility, and it could easily react with calcium ions [16-18]. In this study, we attempt to use PASP as a novel depressant in scheelite flotation, and the conventional depressant acidified water glass (AWG) is used as comparison. The inhibition effect of PASP on the flotation behaviors of scheelite and calcite was investigated through flotation experiments, and the depression mechanism was analyzed through zeta potential measurement, contact angle measurement and infrared spectroscopic analysis (IR).
2 Experimental
2.1 Samples and reagents
Pure scheelite and calcite minerals were obtained from Hunan province, China. Results of chemical analysis show that the content of WO3 is 77.33% with a scheelite purity of 94.66%, and the content of CaCO3 is 98.01%, which are further confirmed by the X-ray diffraction (XRD) patterns shown in Fig. 1.
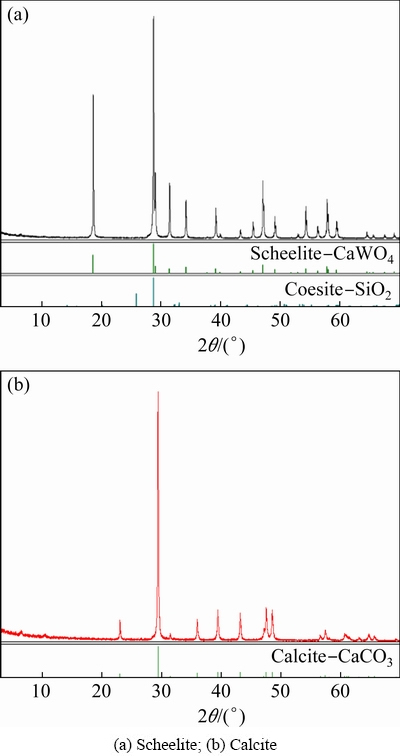
Fig. 1 XRD patterns of pure mineral samples
Analytically pure hydrochloric acid (HCl) and sodium hydroxide (NaOH) were used as the pH regulators. Chemically pure sodium oleate (NaOL), which was used as the collector, was purchased from BSQ Chemistry Technique, Shanghai, China. The commercial PASP was purchased from Taihe Water Treatment Technologies Co., Ltd., China, and its chemical structure is shown in Fig. 2. Distilled water with a resistivity of 18.2 MΩ・cm was used in all experiments.
Fig. 2 Chemical structure of PASP
2.2 Flotation experiments
The pure samples of 38-74 μm in size were used to conduct flotation experiments using a flotation machine with the volume of 40 mL (XFG II, Jilin Exploring Machinery Plant, China). For each test, 2 g minerals and 30 mL distilled water were mixed and conditioned for 1 min to prepare a uniform pulp, followed by the adjustment of pH value with a conditioning time of 2 min. Then, a certain amount of the depressant and collector were successively added and conditioned for 3 min. Finally, the concentrate and tailing were respectively filtered, dried, and weighed to calculate the recoveries of scheelite and calcite.
2.3 Zeta potential measurement
The pure scheelite and calcite samples (<2 μm) were treated with distilled water and reagents according to flotation conditions, and zeta potentials of scheelite and calcite surface as a function of pH value were measured using a zeta-sizer analyzer (Malvern Instrument, England). The measuring error is ±2 mV.
2.4 Contact angle measurement
The crystals samples of scheelite and calcite were used for contact angle measurement. The sample surfaces were ground to flush by diamond grinding disks and then polished with alumina powder solution. According to the experiment requirements, the prepared surfaces were respectively treated with distilled water and reagent solutions for 10 min, then washed using distilled water and vacuum dried at 40 °C. The contact angles were measured using a JY-82C contact angle meter (Dingsheng Tester, China).
2.5 IR analysis
The scheelite and calcite samples were initially treated with PASP, NaOL, PASP+NaOL for 10 min at pH 9-9.5, respectively, then filtered, washed with distilled water and dried in an vacuum oven. The infrared spectra of scheelite and calcite samples with and without reagent treatment were recorded using a Fourier transform infrared spectrometer (Shimadzu, Japan). The adsorption of reagents was analyzed through the changes in the spectra before and after reagent treatment.
3 Results and discussion
3.1 Flotation performance of scheelite and calcite
Figure 3 presents the flotation recoveries of scheelite and calcite using NaOL as the collector without depressant. It can be seen from Fig. 3(a) that both scheelite and calcite show good floatabilities in the pH range of 8-10, and the recovery of calcite is higher than that of scheelite. As shown in Fig. 3(b), the recovery of scheelite gradually increases to 95% when sodium oleate concentration increases to 20 mg/L, while the recovery of calcite is up to 98% at a sodium oleate concentration of only 10 mg/L. It is generally acknowledged that a fatty acid collector adsorbs on a semi-soluble salt-type mineral surface through chemisorption of the carboxylic group binding with lattice cation [19]. The above result clearly shows that due to the same lattice cation (Ca2+), the flotation behaviors of scheelite and calcite are so similar that it is difficult to separate them without depressant.
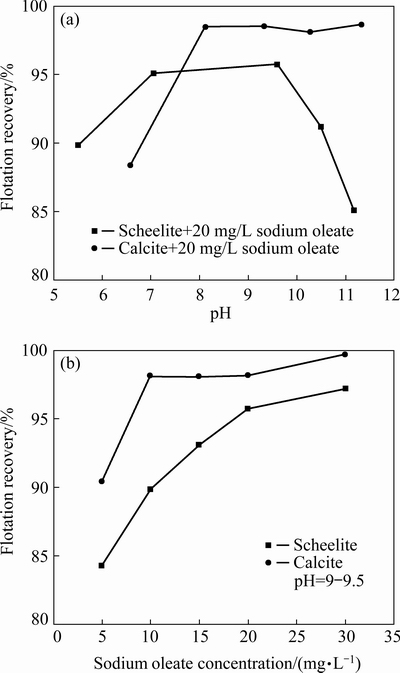
Fig. 3 Flotation recoveries of scheelite and calcite using sodium oleate as collector
AWG has been proven to be an effective depressant in the flotation separation between scheelite and calcite, and the depression mechanism is considered as the selective adsorption of the hydrophilic colloidal silica on calcite surface [9]. In this study, AWG was prepared by mixing WG and H2SO4 with a mass ratio of 3:1. Figure 4 shows the effect of AWG on flotation recoveries of scheelite and calcite using 20 mg/L sodium oleate. The results in Fig. 4(a) show that the recovery of scheelite almost remains at about 77% in the pH range of 6-8, followed by a sharp decrease. While for calcite, the recovery is below 10% when the pH value is higher than 7. Figure 4(b) shows that the recoveries of scheelite and calcite decrease with the increase of AWG concentration, but the reduced degree of calcite is much larger than that of scheelite. When AWG concentration reaches 200 mg/L, the recoveries of scheelite and calcite are approximately 77% and 10%, respectively. Therefore, the flotation separation of scheelite from calcite can be realized using 200 mg/L AWG as the depressant at pH 7-8. However, the recovery of scheelite is not high, and the dose of AWG is large.
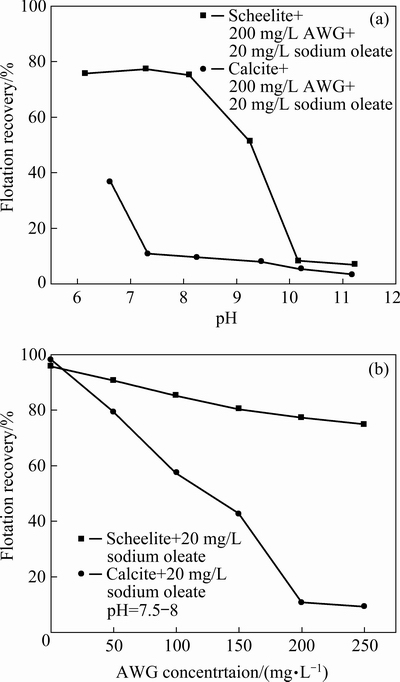
Fig. 4 Effect of AWG on flotation recoveries of scheelite and calcite
Figure 5 presents the inhibition effect of PASP on the flotation performance of scheelite and calcite at a sodium oleate concentration of 20 mg/L. With the increase of pH value, the recovery of scheelite gradually increases, and it is nearly 85% at around pH 9, only a little decrease compared with the recovery (95%) without PASP. However, calcite can hardly float in the whole experimental pH range (Fig. 5(a)). As shown in Fig. 5(b), when PASP concentration is increased to 6 mg/L, the calcite recovery sharply decreases from 98% to 5%, while the recovery of scheelite is still 85%. Then increasing PASP concentration causes no evident change in their recoveries. Therefore, it is illustrated that the flotation separation of scheelite from calcite can be realized in the presence of 6 mg/L PASP at pH>6.
From the above results, it can be seen that compared with the conventional depressant AWG, PASP exhibits the stronger inhibition ability and selectivity due to the lower required concentration, the wider available pH range and the higher scheelite recovery. Therefore, PASP can be used as a new effective depressant in the flotation separation of scheelite from calcite.
3.2 Zeta potentials of scheelite and calcite
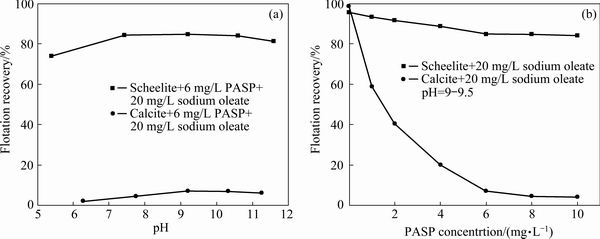
Fig. 5 Effect of PASP on flotation recoveries of scheelite and calcite

Fig. 6 Effect of pH on zeta potentials of scheelite (a) and calcite (b)
Zeta potential of mineral surface has been widely used to interpret the modification of flotation performance caused by the presence of reagents [20]. Figure 6 shows zeta potentials of scheelite and calcite as a function of pH value. As shown in Fig. 6(a), the zeta potential of scheelite in distilled water is always negative in the pH range of 2-12, which agrees with other researches [6-9]. It can be seen from Fig. 6(b) that the surface of calcite without depressant is positively charged at pH<11, and negatively charged at pH>11. After reagent treatment, the zeta potentials of scheelite and calcite also shift to more negative position, and it is indicated that a stronger adsorption of sodium oleate occurs on the calcite surface due to the lager shift value (Table 1). However, in the presence of PASP, the adsorption of sodium oleate on the calcite surface is significantly decreased since no evident decrease is observed between the zeta potentials of “calcite+PASP” and “calcite+PASP+sodium oleate”. But for scheelite, a decrease by 24.4 mV indicates that the adsorption of sodium oleate is little affected.
Table 1 Zeta potentials of scheelite and calcite at pH 9 (mV)

3.3 Contact angle
The wettability of the mineral surface can be visually expressed by the contact angle. The larger the contact angle, the stronger the hydrophobicity and the better the floatability of mineral [21]. Figure 7 shows the effect of PASP concentration on the contact angles of scheelite and calcite. The contact angles of scheelite and calcite used in this study are 40.4° and 51.2°, respectively in distilled water at pH 9-9.5, and they increase to approximately 95° and 98° owing to the adsorption of sodium oleate. With the increase of PASP concentration, the contact angle of calcite exhibits a large decrease, and it is only 56° when PASP concentration is 6 mg/L, but 86.5° for scheelite. The result further confirms that PASP can greatly decrease the adsorption of sodium oleate on calcite surface, while has little influence on the adsorption of sodium oleate on scheelite surface.
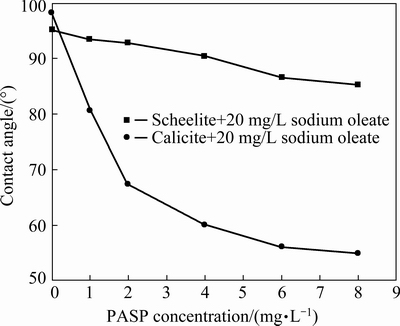
Fig. 7 Effect of PASP concentration on contact angles of scheelite and calcite
3.4 IR spectra
Figure 8 shows the spectrum of PASP. It can be seen that the adsorption peak at 1398.16 cm-1 is due to N―H bond bending and C―N stretching of acylamino, the absorption peak which is assigned to stretching vibration of C=O bond is observed at 1585.23 cm-1, and the characteristic absorption peak of N―H bond stretching vibration appears at 3324.73 cm-1, which well agrees with other researches [17,18].
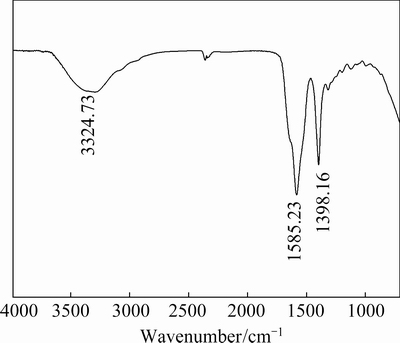
Fig. 8 IR spectrum of PASP
Figure 9 shows the IR spectra of scheelite in the absence and presence of reagent at pH 9-9.5. The peaks at 807.58 and 440.07 cm-1 are attributed to the asymmetric stretching vibration and the out-of-plane bending vibration of W―O bond, respectively. After sodium oleate treatment, three new bands at 1546 cm-1 assigned to C=O stretching, 2853.71 and 2924.91 cm-1 assigned to ―CH3 and ―CH2 stretching are observed whether PASP is added or not. Therefore, it is illustrated that sodium oleate can always adsorb on scheelite surface.

Fig. 9 IR spectra of scheelite
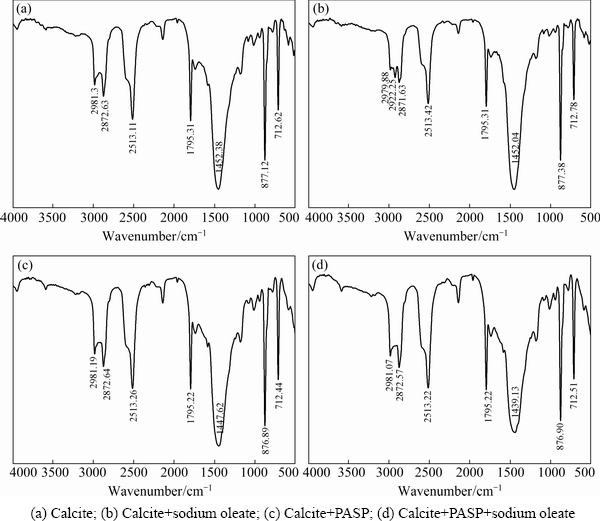
Fig. 10 IR spectra of calcite
Figure 10 shows the IR spectra of calcite in the absence and presence of reagent at pH 9-9.5. The characteristic peaks at 1452.38 cm-1 due to the asymmetric stretching vibration of 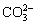 , 877.12 and 712.62 cm-1 due to the υ2 out-of-plane bending vibration and υ4 in-plane bending vibration, are observed in the spectrum of calcite. After sodium oleate treatment, only a new band at 2922.25 cm-1 occurs in Fig. 10(b), the other characteristic bands of sodium oleate, such as 2854 and 1456 cm-1, are likely to be covered by the adsorption bands of
, 877.12 and 712.62 cm-1 due to the υ2 out-of-plane bending vibration and υ4 in-plane bending vibration, are observed in the spectrum of calcite. After sodium oleate treatment, only a new band at 2922.25 cm-1 occurs in Fig. 10(b), the other characteristic bands of sodium oleate, such as 2854 and 1456 cm-1, are likely to be covered by the adsorption bands of 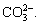 Whereas the spectrum of calcite in Fig. 10(d) almost has no change compared with Fig. 10(a), so it is indicated that the adsorption of PASP on the calcite surface can hinder that of sodium oleate.
Whereas the spectrum of calcite in Fig. 10(d) almost has no change compared with Fig. 10(a), so it is indicated that the adsorption of PASP on the calcite surface can hinder that of sodium oleate.
4 Conclusions
(1) Flotation separation of scheelite from calcite is likely to be realized by using PASP as the depressant. Compared with the conventional depressant AWG, PASP exhibits the stronger inhibition ability and selectivity due to the lower concentration, the wider pH range and the higher scheelite recovery.
(2) After being reacted with PASP, a large negative shift is observed in zeta potential of calcite surface, indicating a stronger adsorption of PASP on calcite surface.
(3) The presence of PASP can hardly affect the adsorption of sodium oleate on scheelite surface, whereas prevents the adsorption of sodium oleate on calcite surface, thus realizing their separation.
Acknowledgments
The authors are grateful for the financial support from the Youth Science Foundation of China (No. 52004333).
References
[1] KUPKA N, PUDOLPH M. Role of sodium carbonate in scheelite flotation―A multi-faceted reagent [J]. Miner Eng, 2018, 129: 120-128.
[2] ZHAO Chen, SUN Chuan-yao, YIN Wan-zhong, LUO Bin-bin. An investigation of the mechanism of using iron chelate as a collector during scheelite flotation [J]. Miner Eng, 2019, 131: 146-153.
[3] ZHAO Gang, ZHONG Hong, QIU Xian-yang, WANG Shuai, GAO Yu-de, DAI Zi-lin, HUANG Jian-ping, LIU Guang-yi. The DFT study of cyclohexyl hydroxamic acid as a collector in scheelite flotation [J]. Miner Eng, 2013, 49: 54-60.
[4] ZHU Hai-ling, QIN Wen-qing, CHEN Chen, LIU Run-qing. Interactions between sodium oleate and polyoxyethylene ether and the application in the low-temperature flotation of scheelite at 283 K [J]. J Surfactants Deterg, 2016, 19: 1-7.
[5] RAI B, SATHISH J, TANWAR J, MOON K S, FUERSTENAU D W. A molecular dynamics study of the interaction of oleate and dodecylammonium chloride surfactants with complex aluminosilicate minerals [J]. J Colloid Interf Sci, 2011, 362: 510-516.
[6] GAO Zhi-yong, BAI Ding, SUN Wei, CAO Xue-feng, HU Yue-hua. Selective flotation of scheelite from calcite and fluorite using a collector mixture [J]. Miner Eng, 2015, 72: 23-26.
[7] GAO Yue-sheng, GAO Zhi-yong, SUN Wei, YIN Zhi-gang, WANG Jian-jun, HU Yue-hua. Adsorption of a novel reagent scheme on scheelite and calcite causing an effective flotation separation [J]. J Colloid Interface Sci, 2018, 512: 39-46.
[8] LIU Cheng, FENG Qi-ming, ZHANG Guo-fan, CHEN Wei, CHEN Yan-fei. Effect of depressants in the selective flotation of scheelite and calcite using oxidized paraffin soap as collector [J]. Inter J Miner Process, 2016, 157(10): 210-215.
[9] FENG Bo, LUO Xian-ping, WANG Jin-qing, WANG Peng-cheng. The flotation separation of scheelite from calcite using acidified sodium silicate as depressant [J]. Miner Eng, 2015, 80: 45-49.
[10] FENG Bo, GUO Wei, XU Hong-guo, PENG Jin-xiu, LUO Xian-ping, ZHU Xian-wen. The combined effect of lead ion and sodium silicate in the flotation separation of scheelite from calcite [J]. Sep Sci Technol, 2017, 52: 567-573.
[11] MARTINS J I, AMARANTE M M. Scheelite flotation from Tarouca mine ores [J]. Miner Process Extr Metall Rev, 2013, 34: 367-386.
[12] DONG Liu-yang, JIAO Fen, QIN Wen-qing, ZHU Hai-ling, JIA Wen-hao. Selective depressive effect of sodium fluorosilicate on calcite during scheelite flotation [J]. Miner Eng, 2019, 131: 262-271.
[13] KANG Jian-hua, HU Yue-hua, SUN Wei, LIU Run-qing, YIN Zhi-gang, TANG Hong-hu, MENG Xiang-song, ZHANG Qing-peng, LIU Hang. A significant improvement of scheelite flotation efficiency with etidronic acid [J]. J Cleaner Production, 2018, 180: 858-865.
[14] ZHANG Yong-zhong, GU Guo-hua, WU Xiang-bin, ZHAO Kai-le. Selective depression behavior of guar gum on talc-type scheelite flotation [J]. Inter J Minerals Metall Materials, 2017, 24(8): 857-862.
[15] QIU Ting-sheng, SONG Yi-fu, QIU Xian-hui, LI Xiao-bo. Performance of organic depressants in scheelite flotation system [J]. The Chinese Journal of Nonferrous Metals, 2017, 27(7): 1527-1534. (in Chinese)
[16] LIU Yu-jing, CHEN Hong, BAI Feng-ling, GU Jia-li, LIU Lian-li. Application of sodium poly (aspartic acid) as a binding phase in the technique of diffusive gradients in thin films [J]. Chemi Lett, 2012, 41: 1471-1472.
[17] WANG Yong-li, HUANG Shao-bin, HU He-ping. Synthesis of polyaspartic acid and its scale inhibiting performance [J]. Ind Water Wastewater, 2004, 35: 74-76.
[18] LIU Zheng-yan, SUN Yong-hong, ZHOU Xiao-hui, WU Tao, TIAN Ying, WANG Ying-zhan. Synthesis and scale inhibitor performance of polyaspartic acid [J]. J Environ Sci, 2011, 23(S1): s53-s55.
[19] CHEN Chen, HU Yue-hua, ZHU Hai-ling, SUN Wei, QIN Wen-qing, LIU Run-qing, GAO Zhi-yong. Inhibition performance and adsorption of polycarboxylic acids in calcite flotation [J]. Miner Eng, 2019, 133: 60-68.
[20] LI Fang-xu, ZHOU Xiao-tong, LIN Ri-xiao. Flotation performance and adsorption mechanism of novel 1-(2- hydroxyphenyl)hex-2-en-1-one oxime flotation collector to malachite [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 2792-2801.
[21] CUI Yan-fang, JIAO Fen, QIN Wen-qing, DONG Liu-yang, WANG Xu. Synergistic depression mechanism of zinc sulfate and sodium dimethyl dithiocarbamate on sphalerite in Pb-Zn flotation system [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 2547-2555.
一种用于白钨矿与方解石浮选分离的新型抑制剂
陈 臣1,2,孙 伟1,朱海玲1,刘润清1
1. 中南大学 资源加工与生物工程学院,长沙 410083;
2. 中南大学 冶金与环境学院,长沙 410083
摘 要:通过浮选试验、动电位测试、接触角测量和红外光谱分析研究一种新型环保抑制剂聚天冬氨酸(PASP)对白钨矿和方解石的抑制作用。结果表明,与传统抑制剂酸化水玻璃相比较,PASP具有更强的抑制能力和选择性。当pH>6时,采用6 mg/L PASP和20 mg/L NaOL可以实现白钨矿和方解石的浮选分离。当PASP存在时,加入NaOL后方解石表面的动电位几乎无变化,且接触角大幅降低,表明PASP降低NaOL在方解石表面的吸附。相反,PASP对NaOL在白钨矿表面的吸附无明显影响,这一结果也通过红外光谱分析进一步证实。
关键词:聚天冬氨酸;白钨矿;方解石;浮选分离;抑制
(Edited by Xiang-qun LI)
Corresponding author: Hai-ling ZHU, Tel: +86-731-88837455, E-mail: lingerfeixiang@126.com
DOI: 10.1016/S1003-6326(21)65669-8
1003-6326/ 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press