
Thin-film lithium-ion battery derived from Li1.3Al0.3Ti1.7(PO4)3 sintered pellet
XIAO Zhuo-bing(��), MA Ming-you(������), WU Xian-ming(������),
HE Ze-qiang(����ǿ), CHEN Shang(�� ��)
College of Chemistry and Chemical Engineering, Jishou University, Jishou 416000, China
Received 24 November 2005; accepted 20 March 2006
Abstract: Thin-film lithium-ion battery of LiMn2O4/Li1.3Al0.3Ti1.7(PO4)3/LiMn2O4 was fabricated using Li1.3Al0.3Ti1.7(PO4)3 sintered pellet as both substrate and electrolyte. Li1.3Al0.3Ti1.7(PO4)3 sintered pellet was prepared by sol-gel technique, and the thin-film battery was heat-treated by rapid thermal annealing. Phase identification, morphology and electrochemical properties of the components and thin-film battery were investigated by X-ray diffractometry, scanning electron microscopy, electrochemical impedance spectroscopy and galvanostatic charge-discharge experiments. The results show that Li1.3Al0.3Ti1.7(PO4)3 possesses a electrochemical window of 2.4 V and an ionic conductivity of 1.2 ?10��4 S/cm. With Li1.3Al0.3Ti1.7(PO4)3 sintered pellet as both substrate and solid electrolyte, the fabricated thin-film battery with an open circuit voltage of 1.2V can be easily cycled.
Key words: LiMn2O4; Li1.3Al0.3Ti1.7(PO4)3; solid electrolyte; lithium ion battery; thin-film battery; sintered pellet
1 Introduction
All-solid-state thin-film batteries have drawn a lot of attention due to their many possible applications, such as smart cards, CMOS-based integrated circuits and microdevices[1-11]. However, most of thin-film batteries employ substrate, which makes the fabrication of thin-film battery inconvenient.
In the synthesis of Li1.3Al0.3Ti1.7(PO4)3 sintered pellet, although the pressure to press Li1.3Al0.3Ti1.7(PO4)3 powders into pellet is not high and no sintering additive is added, the sintered pellets possess strong intensity and are not easy to break. This makes it important to prepare a kind of thin-film battery using this sintered pellet as both substrate and electrolyte.
The average ionic conductivity of LIPON (lithium phosphorus oxynitride) thin film is about 2.3?10-6 S/cm[12] and that of Li1.3Al0.3Ti1.7(PO4)3 is 7?10-4 S/cm[13]. So the resistance of 300 mm-thick Li1.3Al0.3Ti1.7(PO4)3 sintered pellet (thickness of most substrates is about 300 mm) is almost the same as that of 1 mm-thick LIPON thin film if their surface areas are the same. Therefore, compared with most other thin-film lithium-ion batteries which employ substrate and electrolyte of LIPON film, the thin-film lithium-ion battery using Li1.3Al0.3Ti1.7(PO4)3 sintered pellet as both substrate and solid electrolyte will not increase the internal resistance and the thickness of thin-film battery, but make the fabrication of thin-film battery simpler because this kind of thin-film battery no longer needs extra substrate.
Based on the above considerations, the fabrication of thin-film lithium-ion battery of LiMn2O4/ Li1.3Al0.3Ti1.7(PO4)3/LiMn2O4 was carried out to investigate the feasibility of the thin-film battery using Li1.3Al0.3Ti1.7(PO4)3 sintered pellet as both substrate and electrolyte.
2 Experimental
2.1 Preparation of Li1.3Al0 3Ti1.7(PO4)3 sintered pellet
Dissolved stoichiometric amount of Li(CH3- COO)��2H2O and Al(NO3)3��9H2O in CH3OCH2CH2OH. Dripped PO(OC4H9)4 and (Ti(OC4H9)4) into the mixture under constant stirring. Then added several drops of deionized water into the solution and the solution was finally dried at 140 �� for 4 h to form Li1.3Al0.3Ti1.7(PO4)3 gel. The gel was first calcined at 900 �� for 2 h and then grounded into fine powder. With addition of a small amount of 3% polyvinyl-alcohol solution as binder, the fine powder was then pressed into pellets at pressure of 50 MPa. Finally the pellets were calcined at 900 �� for 2 h.
2.2 Preparation of LiMn2O4 precursor solution
Dissolved stoichiometric amount of lithium acetate and manganese acetate in a small amount of deionized water under heating. Added 2-methoxyethanol to adjust the viscosity and wetting property of the solution. Removed dust and other suspended impurities from the solution by filtering through 0.2 mm syringe filters and thus obtaining the LiMn2O4 precursor solution.
2.3 Preparation of LiMn2O4/Li1.3Al0.3Ti1.7(PO4)3/ LiMn2O4 thin-film lithium-ion battery
After 2 mm-thick Li1.3Al0.3Ti1.7(PO4)3 sintered pellet was polished with 800 grade emery paper(a thick Li1.3Al0.3Ti1.7(PO4)3 sintered pellet was employed to avoid the short circuit of the thin-film battery by the soaking of LiMn2O4 precursor throughout the whole pellet), LiMn2O4 precursor solution was deposited by spin coating on both sides of the sintered pellet and then heated up to 350 �� in air at the heating rate of 10 ��/min and maintained at 350 �� for 20 min. In order to minimize the interfacial reaction between LiMn2O4 and Li1.3Al0.3Ti1.7(PO4)3, the thin-film battery was annealed in a rapid thermal annealing system at 750 �� in air for 2 min.
In order to confirm that LiMn2O4 thin film was formed under the above synthesis conditions, LiMn2O4 precursor solution derived LiMn2O4 thin films deposited on Si substrate was prepared under the same synthesis conditions as that of the preparation of LiMn2O4/ Li1.3Al0.3Ti1.7(PO4)3/LiMn2O4 thin-film lithium-ion battery.
2.4 Measurements
Phase identification of the Li1.3Al0.3Ti1.7(PO4)3 powders and LiMn2O4 thin film were performed with Rigaku D/MAX-gA X-ray diffractometer. The morphology of Li1.3Al0.3Ti1.7(PO4)3 pellet was studied by JSM 5600LV scanning electron microscopy. For the measurements of the electrochemical window of Li1.3Al0.3Ti1.7(PO4)3, Li1.3Al0.3Ti1.7(PO4)3 thin film deposited on Pt-Coated Si substrate (Pt acts as current collector) was placed in an open beaker cell containing 1 mol/L LiPF6 dissolved in ethylene carbonate(EC) and dimethyl carbonate (DMC)(1��1, volume ratio). Lithium metal was used as both counter and reference electrodes. Li1.3Al0.3Ti1.7- (PO4)3 thin film was prepared as follows: a Pt-coated Si substrate was dipped into Li1.3Al0.3Ti1.7(PO4)3 precursor solution and then taken out and followed by heat treatment at 900 �� for 2 h at a heating rate of 8 ��/min.
Gold was deposited on both sides of Li1.3Al0.3Ti1.7- (PO4)3 sintered pellet and LiMn2O4/Li1.3Al0.3Ti1.7(PO4)3/ LiMn2O4 sandwich by direct current sputtering to carry out electrochemical measurements. The ionic conduc- tivity of Li1.3Al0.3Ti1.7(PO4)3 was measured by electro- chemical impedance spectroscopy in the frequency range from 100 kHz to 1 Hz. The galvanostatic charge and discharge measurements of LiMn2O4/Li1.3Al0.3Ti1.7- (PO4)3/LiMn2O4 thin-film battery were conducted in the voltage range 0.5 to 1.6 V.
3 Results and discussion
The X-ray diffraction patterns of the LiMn2O4 thin film and Li1.3Al0.3Ti1.7(PO4)3 powders are shown in Fig.1. Three peaks at diffraction angles of 18.61?, 36.09?, and 43.87? can be observed in Fig.1(a), which correspond to the (111), (311) and (400) reflections of spinel LiMn2O4, indicating that the prepared thin film is LiMn2O4. In Fig.1(b) all the diffraction peaks agree well with the reported results of the bulk material of the Li1.3Al0.3Ti1.7(PO4)3[14, 15], suggesting that the powders are Li1.3Al0.3Ti1.7(PO4)3.
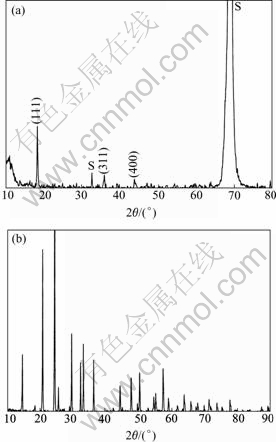
Fig.1 X-ray diffraction patterns of LiMn2O4 thin film and Li1.3Al0.3Ti1.7(PO4)3 powders: (a) LiMn2O4 thin film on Si substrate(peaks marked with S belongs to the substrate); (b) Li1.3Al0.3Ti1.7(PO4)3 powders
Fig.2 shows the SEM image of Li1.3Al0.3Ti1.7(PO4)3 sintered pellet. It can be seen from Fig.2 that the sintered pellet is relatively dense although the pressure to press the Li1.3Al0.3Ti1.7(PO4)3 powders into the pellet is not high and no sintering addictive is added.

Fig.2 SEM image of Li1.3Al0.3Ti1.7(PO4)3 sintered pellet
Electrochemical window of the electrolyte is an important factor for its practical application, especially in the field of batteries. Some difficulty exists in measuring the electrochemical window of Li1.3Al0.3Ti1.7(PO4)3 by using working electrode made up of Li1.3Al0.3Ti1.7(PO4)3 powders, i.e., no redox peaks can be observed in the cyclic voltammogram even at slow rate. This may be caused by the poor electronic conductivity of Li1.3Al0.3Ti1.7(PO4)3 (The electronic conductivity is about 10-11 S/cm in our preliminary studies) and the thick electrode made up of Li1.3Al0.3Ti1.7(PO4)3 powders, i.e., the poor electronic conductivity and relatively thick working electrode make it hard to observe the current response (redox peaks) in the cyclic voltammogram. So Li1.3Al0.3Ti1.7(PO4)3 thin film was used to carry out the measurement. Fig.3 shows the cyclic voltammogram of the sol-gel derived Li1.3Al0.3Ti1.7(PO4)3 thin film recorded at a scan rate of 1mV/s. The figure shows the voltages (vs.Li/Li+) corresponding to the redox peaks are about 2.4 V, which agrees well with the value of the Li1.3Al0.3- Ti1.7(PO4)3 powder tested by coulometric titration[14], indicating that Li1.3Al0.3Ti1.7(PO4)3 possesses a wide electrochemical window.
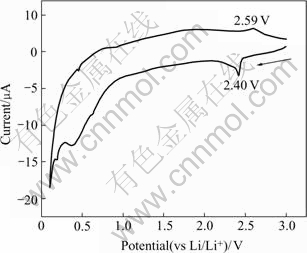
Fig.3 Cyclic voltammogram of Li1.3Al0.3Ti1.7(PO4)3 thin film recorded at scan rate of 1 mV/s
The electrochemical impedance spectroscopy of the Li1.3Al0.3Ti1.7(PO4)3 sintered pellet is shown in Fig.4, as observed from this figure, the spectrum consists of an incomplete semicircle and an inclined line. The incomplete semicircle is caused by the insufficient upper limit of 100 kHz for Electrochemical Workstation CHI660A. From the intercept of the inclined line in the diagram the ionic conductivity can be estimated to be 1.2?10-4 S/cm.
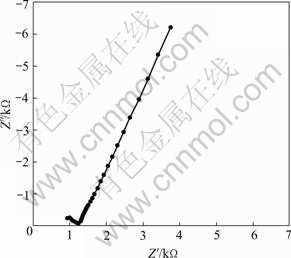
Fig.4 Electrochemical impedance spectroscopy of Li1.3Al0.3Ti1.7(PO4)3 sintered pellet
The battery with surface area of 0.8 cm2 exhibits an open circuit voltage of approximately 1.2 V at ambient temperature. Fig.5 shows the galvanostatic charge and discharge curves of LiMn2O4/Li1.3Al0.3Ti1.7(PO4)3/ LiMn2O4 thin-film lithium-ion battery conducted at a current of 5 mA. Two voltage plateaus can be observed at about 1.1 and 1.2 V in the charge curve, which is the characteristics of LiMn2O4. But no obvious voltage plateau can be seen in the discharge curve. It apparently shows that the thin-film battery has a large irreversible capacity, but in fact it is not the case because the battery only delivers very little capacity from 1.5 to 1.3 V in the discharge curve, i.e. the charge from 1.3 to1.6 V contributes very little capacity to the battery.
The capacity as a function of discharge current is shown in Fig.6 (it is assumed that the capacity of the battery discharging at 5 mA is 100%). As shown in Fig.6, the capacity decreases as the discharge current increases. The extent of the capacity reduction is higher than that of the battery using powder materials as electrodes, which is caused by the higher internal resistance of thin-film battery. Contrary to the battery using liquid electrolyte and electrodes containing electronic materials, thin film battery employs the solid electrolyte and electrode material which does not contain electronic materials. Therefore, thin-film battery possesses higher internal resistance, leading to faster capacity decrease with the increase of the discharge current.
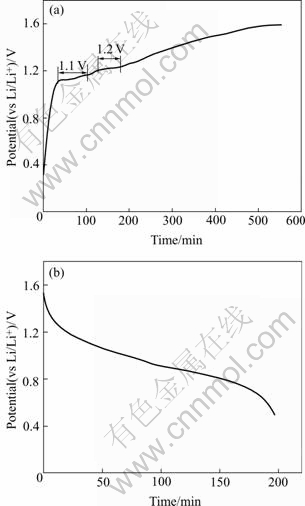
Fig.5 Charge and discharge curves of LiMn2O4/Li1.3Al0.3Ti1.7- (PO4)3/LiMn2O4 thin-film battery: (a) Charge curve; (b) Discharge curve

Fig.6 Capacity variation of thin-film battery with discharge current
Fig.7 shows the cycling behavior of LiMn2O4/Li1.3Al0.3Ti1.7(PO4)3/LiMn2O4 thin-film battery carried out at current of 10 mA. As observed in Fig.7, the capacity decreases with the increase of the cycle number, and the capacity loss per cycle is about 3.2%. The capacity loss is a bit higher than that in Ref.[16]. The reason is that both the cathode and anode are LiMn2O4, which can cause the formation of Li2Mn2O4 and the occurrence of Jahn-Teller effect, thus leading to rapid capacity loss.
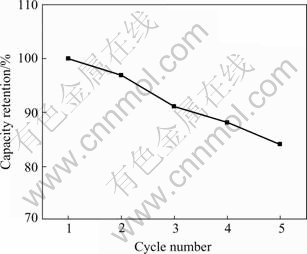
Fig.7 Cycling behavior of LiMn2O4/Li1.3Al0.3Ti1.7(PO4)3/ LiMn2O4 thin-film battery
4 Conclusions
A novel thin-film lithium-ion battery was fabricated by using Li1.3Al0.3Ti1.7(PO4)3 sintered pellet as both substrate and electrolyte. Although the thickness of Li1.3Al0.3Ti1.7(PO4)3 sintered pellet is more than 2 mm and the pressure to prepare the powders into pellets is only 50 MPa, the thin-film battery with an open circuit voltage of 1.2 V can be easily cycled. The thin-film battery using Li1.3Al0.3Ti1.7(PO4)3 sintered pellet as both substrate and electrolyte not only makes the fabrication of thin-film lithium-ion battery much simpler but also makes the operating voltage of the thin-film battery up to 2.4 V. This kind of thin-film lithium-ion battery can be commercialized if improved conditions, proper methods and techniques were available and used.
References
[1] Yoon Y S, Kim J S, Choi S H. Structural and electrochemical properties of vanadium oxide thin films grown by d.c. and r.f. reactive sputtering at room temperature[J]. Thin Solid Films, 2004, 460: 41-47.
[2] Tao Y, Chen Z, Zhu B, Huang W. Preparation of preferred oriented LiCoO2 thin films by soft solution processing[J]. Solid State Ionics, 2003, 161: 187-192.
[3] West W C, Whitacre J F, Lim J R. Chemical stability enhancement of lithium conducting solid electrolyte plates using sputtered LiPON thin films[J]. J Power Sources, 2004, 126: 134-138.
[4] Eftekhari A. Fabrication of 5 V lithium rechargeable micro-battery[J]. J Power Sources, 2004, 132: 240-243.
[5] Wu X M, Li X H, Xu M F, Zhang Y H, He Z Q, Wang Z. Preparation of LiMn2O4 thin films by aqueous solution deposition[J]. Materials Research Bulletin, 2002, 37: 2345-2353.
[6] Kim M K, Park K S, Son J T, Kim J G, Chung H T, Kim H G. The electrochemical properties of thin-film LiCoO2 cathode prepared by sol�Cgel process[J]. Solid State Ionics, 2002, 152/153: 267-272.
[7] Nagasubramanian G, Doughty D H. Electrical characterization of all-solid-state thin film batteries[J]. J Power Sources, 2004, 136: 395-400.
[8] Wu X M, Li X H, Wang Z, Xiao Z B, Liu J B, Yan W B. Characterization of solution-derived LiMn2O4 thin films heat-treated by rapid thermal annealing[J]. Materials Chemistry and Physics, 2004, 83: 78-81.
[9] Lee J H, Han K S, Lee B J, Seo S I, Yoshimura M. Fabrication of LiCoO2 films for lithium rechargeable microbattery in an aqueous solution by electrochemical reflux method[J]. Electrochimica Acta, 2004, 50: 467-471.
[10] Bates J B, Dudney N J, Neudecker B, Ueda A, Evans C D. Thin-film lithium and lithium-ion batteries[J]. Solid State Ionics, 2000, 135: 33-35.
[11] Shui J L, Jiang G S, Xie S, Chen C H. Thin films of lithium manganese oxide spinel as cathode materials for secondary lithium batteries[J]. Electrochimica Acta, 2004, 49: 2209-2213.
[12] Zhao S, Fu Z, Qin Q. A solid-state electrolyte lithium phosphorus oxynitride film prepared by pulsed laser deposition[J]. Thin Solid Films, 2002, 415: 108-113.
[13] Takada K, Tansho M, Yanase I, Inada T, Kajiyama A, Kouguchi M, Kondo S, Watanabe M. Lithium ion conduction in LiTi2(PO4)3[J]. Solid State Ionics, 2001, 139: 241-247.
[14] Birke P, Salam F, D?ring S, Weppner W. A first approach to a monolithic all solid state inorganic lithium battery[J]. Solid State Ionics, 1999, 118: 149-157.
[15] Best A S, Forsyth M, Macfarlane D R. Stoichiometric changes in lithium conducting materials based on Li1+xAlxTi2-x(PO4)3: impedance, X-ray and NMR studies[J]. Solid State Ionics, 2000, 136/137: 339-344.
[16] Hideaki O, Yoji S. Characteristics of Li/MoO3-x thin film batteries[J]. Solid State Ionics, 2001, 144: 59-64.
Foundation item: Projects(03JJY6005; 04JJ4038) supported by the Natural Science Foundation of Hunan Province, China; Project(05C140) supported by the Education Office of Hunan Province, China
Corresponding author: XIAO Zhou-bing; Tel: +86-743-8563786; Fax: +86-743-8563911; E-mail: zhuobing@netease.com
(Edited by LONG Huai-zhong)