
Influence of Li3PO4 addition on properties of lithium ion-conductive electrolyte Li1.3Al0.3Ti1.7(PO4)3
XIAO Zhuo-bing 1, CHEN Shang2, GUO Man-man1
1. Key Laboratory of Hunan Forest Products and Chemical Industry Engineering, Zhangjiajie 427000, China;
2. College of Chemistry and Chemical Engineering, Jishou University, Jishou 416000, China
Received 18 April 2010; accepted 19 September 2011
Abstract: The sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 were prepared by sol-gel method. The structure, surface morphology, electrochemical window, ionic conductivity and activation energy of the sintered pellets were studied by X-ray diffraction, scan electron microscopy, cyclic voltammetry, and electrochemical impedance spectroscopy. The results show that all the sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 have similar X-ray diffraction patterns. The sintered Li1.3Al0.3Ti1.7(PO4)3 pellet with Li3PO4 shows a lower porosity and is denser than the one without Li3PO4. The addition of Li3PO4 has a large effect on the porosity and density of the sintered Li1.3Al0.3Ti1.7(PO3)4 pellet, and little effect on its electrochemical window. Among the sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with different mole fractions of Li3PO4, the one with 1% Li3PO4 shows the activation energy of 0.314 2 eV and the highest ionic conductivity of 6.15?10-4 S/cm.
Key words: sol-gel; electrolyte; ionic conduction
1 Introduction
Lithium titanium phosphate, LiTi2(PO4)3, which is based upon the NASICON (Na+super-ionic+conductor) framework with a space group of R C, has been studied as a lithium-ion-conductive solid electrolyte [1-2]. It is one of the promising lithium ion-conductive solid electrolytes. However, its conductivity is not high enough for practical applications [3-4
C, has been studied as a lithium-ion-conductive solid electrolyte [1-2]. It is one of the promising lithium ion-conductive solid electrolytes. However, its conductivity is not high enough for practical applications [3-4
References and further reading may be available for this article. To view references and further reading you must purchase this article.
]. In order to obtain LiTi2(PO4)3 with a higher ionic conductivity, some researchers replaced Ti with Zr [2], Nb [5], La [6], Y [7], and Ge [8]. However, these studies did not result in satisfied ionic conductivity. AONO et al [9] reported that Al-doping was effective for the enhancement of lithium ion conduction. Since then, a lot of attention has been paid to Li1.3Al0.3Ti1.7(PO4)3 for possible use in solid-state lithium-ion batteries [10-15].
Sol-gel method is one of the most frequently employed techniques to synthesize various materials. The technique has several advantages including good mixture of the reagents, relatively low crystallization temperature, homogenous and small grain size of product. The small grained powders can result in sintered pellet with high density, small voids and grains, which leads to a high ionic conductivity [16].
Flux can influence the sinterability and therefore affect the ionic conductivity and strength of the sintered electrolyte pellet [17]. Considering Li3PO4 a good flux with a relatively low melting temperature of 837 ��C, the characterization of sol-gel derived Li1.3Al0.3Ti1.7(PO4)3 with different mole fractions of Li3PO4 flux is studied in the present work to obtain Li1.3Al0.3Ti1.7(PO4)3 pellet with high ionic conductivity and strength, which can be used in thin-film battery as both electrolyte and substrate. To the author��s knowledge, there are few reports about the influence of Li3PO4 addition on the properties of lithium ion-conductive electrolyte Li1.3Al0.3Ti1.7(PO4)3.
2 Experimental
Li(CH3COO)��2H2O and Al(NO3)3��9H2O at stoichiometric amount were dissolved in CH3OCH2CH2OH. Then PO(OC4H9)4 and Ti(OC4H9)4 were consecutively dripped into the mixture under constant stirring to form Li1.3Al0.3Ti1.7(PO4)3 precursor solution. Then several drops of deionized water were added into the solution followed by drying in an oven at 140 ��C for 4 h to get Li1.3Al0.3Ti1.7(PO4)3 gel. The gel was calcined at 900 ��C for 2 h to obtain Li1.3Al0.3Ti1.7(PO4)3 powders. The powders were mixed with different mole fractions of Li3PO4 and pressed into pellets at the pressure of 100 MPa using 3% polyvinyl-alcohol solution as the binder. The pressed pellets were calcined at 900 ��C for 2 h to obtain Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4.
The structural identification of the sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 was conducted by X-ray diffraction. The surface morphology was studied by scan electron microscopy. The compositions of the synthesized powders were determined by the inductively coupled plasma (ICP) method. Porosity of the sintered pellets was determined by the Archimedes��s method. For the measurement of electrochemical window, cyclic voltammetry was carried out using micro-electrode fabricated from grounded Li1.3Al0.3Ti1.7(PO4)3 pellet. The micro-electrode was placed in an beaker cell containing 1 mol/L LiPF6 dissolved in ethylene and dimethyl carbonate (1:1, volumetric ratio). Metal lithium was used as both counter and reference electrodes. The ionic conductivity and activation energy were measured by electrochemical impedance spectroscopy. Both sides of the sintered pellet were painted with silver slurry followed by drying at 150 ��C for 30 min before the electrochemical measurements were carried out. Electrochemical impedance measurement was conducted in an Ar-flowed tube over the frequency range of 1 Hz to 100 kHz at different temperatures.
3 Results and discussion
The X-ray diffraction patterns of the sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 are displayed in Fig. 1. As observed in Fig. 1, all the sintered pellets show similar diffraction patterns belonging to rhombohedral symmetry (space group R C) with six formula units in the unit cell. No diffraction peaks of impure phase are identified. All the diffraction peaks of the sintered pellets agree well with the reports for Li1.3Al0.3Ti1.7(PO4)3 [11], suggesting that the sintered pellets are pure Li1.3Al0.3Ti1.7(PO4)3 phase. There are no obviously difference in the lattice parameters between Li1.3Al0.3Ti1.7(PO4)3 pellets with and without Li3PO4, indicating that the addition of Li3PO4 flux has little effect on the crystal structure of Li1.3Al0.3Ti1.7(PO4)3.
C) with six formula units in the unit cell. No diffraction peaks of impure phase are identified. All the diffraction peaks of the sintered pellets agree well with the reports for Li1.3Al0.3Ti1.7(PO4)3 [11], suggesting that the sintered pellets are pure Li1.3Al0.3Ti1.7(PO4)3 phase. There are no obviously difference in the lattice parameters between Li1.3Al0.3Ti1.7(PO4)3 pellets with and without Li3PO4, indicating that the addition of Li3PO4 flux has little effect on the crystal structure of Li1.3Al0.3Ti1.7(PO4)3.
The scan electron micrographs of the sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 are presented in Fig. 2. As seen in Fig. 2, the contact between the particles of the sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with Li3PO4 flux is better than the one without Li3PO4, which can be attributed to the relatively low melting temperature of Li3PO4, namely, Li3PO4 can melt and fill the space between Li1.3Al0.3Ti1.7(PO4)3 particles during the sintering at 900 ��C, leading to better sinterability and densification. However, there are no obvious differences between the sintered pellets with Li3PO4.

Fig. 1 XRD patterns and lattice parameters of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4: (a) 0; (b) 1%; (c) 2%; (d) 3%; (e) 4%
The porosity of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 is displayed in Fig. 3. As shown in Fig. 3, the porosity of sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with 1%, 2%, 3%, and 4% Li3PO4 are 2.45%, 2.38%, 2.17%, and 2.15%, respectively, which are lower than 3.78% for the one without Li3PO4. The reason why the sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with Li3PO4 shows lower porosity than the one without Li3PO4 is that Li3PO4 melts and fills the space between Li1.3Al0.3Ti1.7(PO4)3 particles during the sintering at 900 ��C, resulting in the pellets with better sinterability and densification.
Electrochemical window of electrolyte is an important factor for its practical application, especially in the field of batteries. Figure 4 shows the redox voltages of the sintered pellet with different mole fractions of Li3PO4. As seen in Fig. 4, there are no significant differences between these sintered pellets. All the pellets show the redox voltage beyond 2.4 V. This indicates that the addition of Li3PO4 has little effect on the electrochemical window of Li1.3Al0.3Ti1.7(PO4)3.
Electrochemical impedance spectra of the sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with different mole fractions of Li3PO4 conducted at 25 ��C are shown in Fig. 5. All the sintered pellets show an incomplete semicircle and an inclined line. The incomplete semicircle in the spectrum is caused by the insufficient frequency upper limit of electrochemical workstation CHI660B. The high- frequency limit should be more than 1 MHz because the time constant of the bulk process is 1 ns assuming a geometric capacity (cgeo) of 1-10 pF. Obviously, the high-frequency limit in this study of 100 kHz is insufficient and causes the incomplete semicircle, which makes it difficult to determine the bulk ionic conductivity. However, from the intercept of the inclined line in the diagram the total ionic conductivity of the sintered pellets with different mole fractions of Li3PO4 can be calculated.
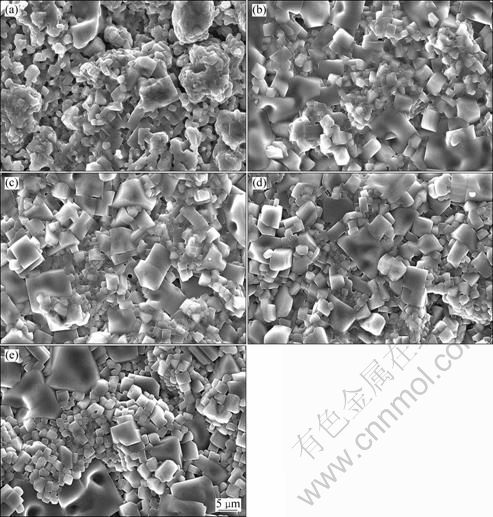
Fig. 2 SEM images of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4: (a) 0; (b) 1%; (c) 2%; (d) 3%; (e) 4%

Fig. 3 Effect of mole fraction of Li3PO4 on porosity of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets

Fig. 4 Redox voltages of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4
Temperature dependence of ionic conductivity for the sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with different mole fractions of Li3PO4 is presented in Fig. 6. All the sintered pellets result in a linear temperature-dependence of conductivity followed on Arrhnius��s law. According to the results in the figure, the activation energy Ea can be obtained from the following equation:

where ��0 is the pre-exponential factor; Ea is the activation energy for conductor; R is the mole gas constant; and T is the thermodynamic temperature.

Fig. 5 Electrochemical impedance spectra of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 conducted at 25 ��C
The ionic conductivity and activation energies of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 are displayed in Fig. 7. As shown in Fig. 7, the ionic conductivities of the sintered Li1.3Al0.3Ti1.7(PO4)3 with 1% and 2% Li3PO4 are higher than the one without Li3PO4. When the mole fraction of Li3PO4 increases from 0 to 1%, the ionic conductivity increases from 3.46?10-4 S/cm to 6.15?10-3 S/cm. However, the ionic conductivity decreases with further increase of Li3PO4. This effect of Li3PO4 addition on the ionic conductivity of Li1.3Al0.3Ti1.7(PO4)3 may be caused by the following reasons: The increase of Li3PO4 leads to the pellet with better sinterability and densification because Li3PO4 is a good flux, which results in closer contact of Li1.3Al0.3Ti1.7(PO4)3 particles and decreases the resistance at grain boundaries, leading to higher ionic conductivity. However, too much Li3PO4 addition also decreases the ionic conductivity of Li1.3Al0.3Ti1.7(PO4)3 because the ionic conductivity of Li3PO4 is considerably lower than Li1.3Al0.3Ti1.7(PO4)3. Figure 7 also shows the activation energies of sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with 0, 1%, 2%, 3%, and 4% Li3PO4 are 0.282 1, 0.314 2, 0.296 3, 0.331 5, and 0.323 7 eV, respectively. The activation energy of sintered Li1.3Al0.3Ti1.7(PO3)4 with Li3PO4 is slightly higher than the one without Li3PO4. This means that the temperature has greater influence on the ionic conductivity of the sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with Li3PO4.

Fig. 6 Temperature dependence of conductivity for sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with different mole fractions of Li3PO4

Fig. 7 Ionic conductivity and activation energies of sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 conducted at 25 ��C
4 Conclusions
1) The sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with different mole fractions of Li3PO4 show similar X-ray diffraction patterns. The sintered Li1.3Al0.3Ti1.7(PO4)3 pellets with Li3PO4 show lower porosity and are denser than the one without Li3PO4.
2) The addition of Li3PO4 has little effect on the redox voltages of Li1.3Al0.3Ti1.7(PO4)3. When the Li3PO4 addition increases from 0 to 1%, the ionic conductivity increases from 3.46?10-4 S/cm to 3.95?10-4 S/cm. However, the ionic conductivity decreases with further increase of Li3PO4.
3) The activation energies of sintered Li1.3Al0.3Ti1.7(PO3)4 pellets with 0, 1%, 2%, 3%, and 4% are 0.282 1, 0.246 9, 0.297 1, 0.327 3, and 0.341 9 eV, respectively. The activation of the sintered Li1.3Al0.3Ti1.7(PO3)4 pellet with Li3PO4 is slightly higher than the one without Li3PO4.
References
[1] CHEN L J, ZHAO Y J, LUO J Y, XIA Y Y. Oxygen vacancy in LiTiPO5 and LiTi2(PO4)3: A first-principles study [J]. Phys Lett A, 2011, 375: 934-938.
[2] ARBI K, ROJO J M, SANZ J. Lithium mobility in titanium based NASICON Li1+xTi2-xAlx(PO4)3 and LiTi2-xZrx(PO4)3 materials followed by NMR and impedance spectroscopy [J]. J Eur Ceram Soc, 2007, 27: 4215-4218.
[3] LUO J Y, CHEN L J, ZHAO Y J, HE P, XIA Y Y. The effect of oxygen vacancies on the structure and electrochemistry of LiTi2(PO4)3 for lithium-ion batteries: A combined experimental and theoretical study [J]. J Power Sources, 2009, 194: 1075-1080.
[4] WOLFENSTINE J, ALLEN J L, SUMNER J, SAKAMOTO J. Electrical and mechanical properties of hot-pressed versus sintered LiTi2(PO4)3 [J]. Solid State Ionics, 2009, 180: 961-967.
[5] VINOTH RATHAN S, GOVINDARAJ G. Thermal and electrical relaxation studies in Li(4+x)TixNb1-xP3O12 (0.0��x��1.0) phosphate glasses [J]. Solid State Sciences, 2010, 12: 730-735.
[6] KAZAKEVI?IUS E, ?ALKUS T, DINDUNE A, KANEPE Z, RONIS J, KE?IONIS A, KAZLAUSKIEN? V, MI?KINIS J, SELSKIEN? A, SELSKIS A. La-doped LiTi2(PO4)3 ceramics [J]. Solid State Ionics, 2008, 179: 51-56.
[7] ?ALKUS T, KAZAKEVI?IUS E, KE?IONIS A, KAZLAUSKIEN? V, MI?KINIS J, DINDUNE A, KANEPE Z, RONIS J, DUDEK M, BU?KO M. XPS and ionic conductivity studies on Li1.3Al0.15Y0.15Ti1.7(PO4)3 ceramics [J]. Ionics, 2010, 16: 631-637.
[8] XU X, WEN Z, GU Z, XU X, LIN Z, Lithium ion conductive glass ceramics in the system Li1.4Al0.4(Ge1-xTix)1.6(PO4)3 (x=0�C1.0) [J]. Solid State Ionics, 2004, 171: 207-213.
[9] AONO H, SUGIMOTO E, SADAOKA Y, IMANAKA N, ADACH G. Conductivity of the lithium titanium phosphate (Li1+xMxTi2-x(PO4)3, M=Al, Sc, Y, and La) system [J]. J Electrochem Soc, 1989, 136: 590-591.
[10] XU X X, WEN Z Y, YANG X L, CHEN L D. Dense nanostructured solid electrolyte with high Li-ion conductivity by spark plasma sintering technique [J]. Materials Research Bulletin, 2008, 43: 2334-2341.
[11] BEST A S, FORSYTH M, MACFARLANE D R. Stoichiometric changes in lithium conducting materials based on Li1+xAlxTi2-x(PO4)3: Impedance, X-ray and NMR studies [J].Solid State Ionics, 2000, 136-137: 339-344.
[12] WANG Y J, PAN Y, KIM D. Conductivity studies on ceramic Li1.3Al0.3Ti1.7(PO4)3-filled PEO-based solid composite polymer electrolytes [J]. J Power Sources, 2006, 159: 690-701.
[13] WU X M, LI X H, ZHANG Y H, XU M F, HE Z Q. Synthesis of Li1.3Al0.3Ti1.7(PO4)3 by sol-gel technique [J]. Mater Lett, 2004, 58: 1227-1230.
[14] PANG M, SUZUKI R, SAITO M, MACHIDA K, HANAWA H, NOJIRI Y, TANASE S. Fabrication and crystal line patterning of Li1.3Al0.3Ti1.7(PO4)3 ion conductive glass by Ni atom heat processing method [J]. Appl Phys Lett, 2008, 92: 041112-1-3.
[15] KOSOVA N V, DEVYATKINA E T, STEPANOV A P, BUZLUKOV A L. Lithium conductivity and lithium diffusion in NASICON-type Li1+xTi2�CxAlx(PO4)3 (x=0; 0.3) prepared by mechanical activation[J]. Ionics, 2008, 14: 303-311.
[16] BROUSSE T, FRAGNAUD P, MARCHAND R, SCHLEICH D M, BOHNKE O, WEST K. All oxide solid-state lithium-ion
cells [J]. J Power Sources, 1997, 68: 412-415.
[17] AONO H, SUGIMOTO E, SADAOKA Y, IMANAKA N, ADACHI G. Ionic
conductivity of LiTi2(PO4)3 mixed with lithium salts [J]. Chem Lett, 1990, 190:
331-334.
���Ӽ�Li3PO4������ӹ�������
Li1.3Al0.3Ti1.7(PO4)3���ܵ�Ӱ��
��1���� ��2��������1
1. �ֲ��������̺���ʡ�ص�ʵ���ң��żҽ� 427000��
2. ���״�ѧ ��ѧ����ѧԺ������ 416000
ժ Ҫ�������ܽ�-�����������Ӳ�ͬ������Li3PO4���ۼ����ϳ�Li1.3Al0.3Ti1.7(PO4)3����ӹ��������ս�Ƭ������X�������䡢ɨ����������о��ϳɲ���Ľṹ����ò������ѭ�������������迹�����о����Ӳ�ͬĦ��������Li1.3Al0.3Ti1.7(PO4)3���������ս�Ƭ�Ľṹ������-��ԭ��λ�����ӵ絼�ʺͻ�ܡ����������������δ����Li3PO4���ۼ���Li1.3Al0.3Ti1.7(PO4)3�ս�Ƭ�������Ƶ�X����������������Li3PO4��Li1.3Al0.3Ti1.7(PO4)3�ս�Ƭ�Ŀ�϶�ʽ�С����Ϊ���ܡ�����Li3PO4��Li1.3Al0.3Ti1.7(PO4)3������-��ԭ��λӰ�첻������������Li3PO4���ۼ���Li1.3Al0.3Ti1.7(PO4)3�ս�Ƭ�У�����1%(Ħ������)Li3PO4 ���ս�Ƭ������ߵ����ӵ絼��6.15?10-4 S/cm����͵Ļ��0.314 2 eV��
�ؼ��ʣ��ܽ�-����������ʣ����ӵ絼
(Edited by YANG Hua)
Foundation item: Project supported by Aid Program for Science and Technology Innovative Research Team (Chemicals of Forestry Resources and Development of Forest Products) in Higher Educational Institutions of Hunan Province; Project (2009FJ3045) supported by the Fund of Science and Technology Department of Hunan Province, China
Corresponding author: XIAO Zhuo-bing; Tel: +86-744-8231386; E-mail: xiaoyddd@163.com
DOI: 10.1016/S1003-6326(11)61036-4