
Comparative studies on flotation of illite, pyrophyllite and kaolinite with Gemini and conventional cationic surfactants
XIA Liu-yin(������)1,2, ZHONG Hong(�� ��)2, LIU Guang-yi(������)2,
HUANG Zhi-qiang(��־ǿ)2, CHANG Qing-wei(����ΰ)2, LI Xin-gang(���θ�)1
1. School of Chemical Engineering and Technology, Tianjin University, Tianjin 300072, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China
Received 15 April 2008; accepted 24 September 2008
Abstract: To search a novel class of effective silicate mineral collectors, the Gemini quaternary ammonium salt surfactant (butane-��, ��-bis(dimethyl dodeculammonium bromide), 12-4-12) and its corresponding conventional monomeric surfactant (dedecyl trimethyl ammonium bromide, DTAB) were adopted to comparatively study the flotation behaviors of illite, pyrophyllite and kaolinite. Three silicate minerals with the Gemini surfactant as collector reveal floatability far better than with the corresponding traditional one. At pH 6, the best recoveries of illite, pyrophyllite and kaolinite with 3.5��10-4 mol/L 12-4-12 are 99.2%, 91.7% and 99.6%, respectively. The fluorescence and contact angle measurement were also conducted for the further investigation of surfactants aggregation behavior and silicate mineral surface hydrophobic properties. FTIR spectra analysis and electrokinetic analysis show that the mechanism of adsorption of collector molecules on mineral surfaces is almost identical for the electronic attraction and hydrogen bonds effect. The superior collecting power of dimeric collector may be attributed primarily to its special structure and its essential properties.
Key words: Gemini cationic surfactant; forth flotation; silicate minerals; kaolinite
1 Introduction
For the economical utilization of low-grade aluminum resources, in China, reverse flotation has been proved to be an efficient method[1-5]. The dominant silica-bearing minerals in Chinese diasporic bauxites are illite, pyrophyllite, kaolinite, etc, thus the reverse flotation for silica removal is to separate these silicate minerals from diaspore. To improve the floatability of illite, pyrophyllite and kaolinite, various cationic collectors have been extensively studied in recent year. Alkylamines were used as collectors to float illite, pyrophyllite and kaolinite by JIANG et al[6]. N-dodecyl-1,3-diaminopropane(DN12) and ��-alkoxy- propylamines(ONs) were studied as flotation collector by CAO et al[7-8]. A series of aminoamides, such as N-(3-aminopropyl)-dodecanamide(APDA), N-(2- aminoethyl)-1-naphthaleneacetamide(AENA) and N-(2- aminoethyl)-dodecanamide(AEDA) were employed by ZHAO et al[9-10]. In summary, these cationic surfactants used as collectors in silicate minerals flotation are conventional surfactants with a characteristic of one chain and one head group.
Gemini surfactant is a new family of amphiphilic molecules, and it is a dimeric surfactant consisting of two identical amphiphilic moieties (twin) covalently joined by a spacer group at or close to the ionic head groups, usually represented by m�Cs�Cm, where m and s are the carbon number in the alkyl chains and of the alkanediyl spacer[11]. Such a surfactant has stimulated extensive interest of stronger surface activity, better solubilizing, wetting, foaming, and lime-soap dispersing capability than the conventional surfactants[12]. An increasing number of papers have been published addressing their physicochemical properties of the dimeric surfactants in comparison with those of the corresponding conventional monomeric surfactants (one chain/one head group). Firstly, bis-quaternary ammonium surfactants are much more efficient than the corresponding monomeric surfactants with decreasing the surface tension of water[13]. This is beneficial to floating mineral particles due to the formation of stable bubbles underwater. Secondly, they are characterized by the critical micelle concentration(CMC) that is one to two orders of magnitude lower than that for the corresponding conventional monomeric surfactants [14-16]. The lower the CMC, the better hydrophobicity the surfactant presents. Thirdly, some Gemini surfactants with short spacers have very high viscosity at relatively low surfactant concentration whereas the solution of the corresponding monomer remains low viscosity[17].
There is interest in determining whether the Gemini surfactants would be more efficient and/or effective than the convention surfactants containing single similar hydrophilic and hydrophobic group in the molecules in forth flotation. The essential properties of Gemini surfactants illustrated previously may be shown to be useful for beneficiation.
In this work, the collecting ability of a Gemini quaternary ammonium salt (butane-��,��-bis(dimethyl dodeculammonium bromide), 12-4-12, the chemical structure of the gemini surfactant is shown in Fig.1) and a corresponding conventional monomeric surfactant (dedecyl trimethyl ammonium bromide, DTAB) was comparatively studied by microflotation tests of illite, pyrophyllite and kaolinite. The interactions of surfactants and clay minerals were investigated by infrared surface analysis and Zeta potential measurement. The difference of flotation behavior of the two surfactants was discussed by fluorescence measurements and contact angle studies.

Fig.1 Schematic molecular structure of Gemini surfactant
2 Experimental
2.1 Minerals and chemicals
Handpicked illite and pyrophyllite were obtained from Ohai Mine, Qingtian Mine in China. Kaolinite was obtained from the geological museum of China. They were 90% in purity by mineralogical analysis, X-ray diffractometry, and chemical analysis.
All minerals were ground in porcelain to less than 0.076 mm. The specific surface area was measured to be 7.698 8 m2/g for illite, 9.970 5 m2/g for pyrophyllite and 14.185 2 m2/g for kaolinite.
Butane-��,��-bis (dimethyl dodecyl ammonium bromide) and dedecyl trimethyl ammonium bromide were provided by Daochun Chemical Engineering and Technology Corporation of Henan Province, China. They were 99% in purity. Pyrene was bought from Sigma-Aldrich (Shanghai) Trading Co., Ltd., China. KNO3 in analytical purity was used as background electrolyte solution. Solutions of HCl and NaOH were used to adjust the pH of the system.
2.2 Microflotation
Pure mineral particles (3 g) were placed in a plexiglass cell (40 mL), then filled with distilled water. After adding the desired amount of reagents, the suspension was agitated for 3 min, and the pH was adjusted before flotation. The flotation was conducted for 6 min. The products and tails were weighed separately after filtration and drying, and the recovery was calculated. The Al2O3-to-SiO2 mass ratio of artificially mixed minerals was determined by silicon-molybdenum blue colorimetry.
2.3 FTIR spectrum
Diffuse reflectance infrared spectroscopy (DIR) was used to characterize the surface species on the mineral particles treated. Samples were ground to be less than 5 ?m and prepared with the same method as used for the micro-flotation tests. The spectrum was obtained using a DIR Nicolet accessory (Nicolet spectrometer, AVATAR360, USA), and presented without any baseline correction.
2.4 Electrokinetic measurements
Zeta potentials were measured using a Brookhaven ZetaPlus Zeta-potential analyzer (USA). All measurements were conducted in a 0.1 mol/L KNO3 background electrolyte solution. Samples were ground to be less than 5 ?m. 0.05 g sample was placed in a 100 mL breaker, stayed for 5 min, then added with 80 mL distilled water, and the pH was adjusted and measured. The results presented in this work were the average of three independent measurements with a typical variation of ��2 mV. Repeat tests showed that the conditioning procedure was capable of producing mineral surfaces suitable for studying the effect of various treatments.
2.5 Fluorescence emission spectroscopy
Samples for fluorescence emission spectroscopy were prepared by mixing pyrene stock solution with surfactant/mineral pulp, and allowed to stand for 3 d to equilibrate. Samples with precipitates appeared were centrifuged at 12 000 r/min for 20 min to remove the precipitates. The pyrene stock solution was prepared by dissolving pyrene in hot water up to saturation, cooled to 25 ��, and filtered. The concentration of pyrene in the solution was determined to be 6.53��10-7 mol/L[18]. Emission spectrum (��=335 nm) of the mixed solution was recorded with F4500 (HITACHI) at 25 ��. A typical emission spectrum has five peaks at 373, 379, 384, 390, and 397 nm, respectively. The intensity ratio of the first to the third vibronic peaks I1/I3 is sensitive to the local environment of pyrene[19�C21].
2.6 Contact angle measurement
Static contact angle measurements were carried out with a 2 ��L water drop using an optical contact angle meter (Dataphysics Inc., OCA20). In the experiments, at least three mineral wafers were used to repeat the contact angle measurements for each concentration, and for each treated substrate, the contact angles were measured on at least three different areas. The reported values were averages of these measurements.
3 Results
3.1 Microflotation with 12-4-12 and DTAB
Microflotation tests were conducted to determine the collecting power of 12-4-12 and DTAB for illite, pyrophyllite and kaolinite as a function of pulp pH. The recovery��pH curves (Fig.2) tested with 2.0��10-4 mol/L collectors show that with pH increasing there is a little decrease in recovery for illite, pyrophyllite and kaolinite both with the Gemini and the single-chain surfactant. This is consistent with the common observation that the floatability of clay minerals would decrease in alkaline condition when cationic amine collectors are used[6-10]. In this case, however, the flotation recoveries of three silicate minerals with 12-4-12 fall to much less extent than with DTAB, especially in the slight alkaline pH range, e.g. at pH 8.0, the flotation recoveries of illite, pyrophyllite and kaolinite are about 85%, 96% and 98% for the use of Gemini, whereas they are about 63%, 81% and 78% for the monomeric homologues, respectively.

Fig.2 Flotation recoveries of illite (a), pyrophyllite (b) and kaolinite (c) as function of pulp pH with 2.0��10-4 mol/L collectors
Recovery��collector dosage curves of illite, pyrophyllite and kaolinite obtained at pH 6 (Fig.3) indicate that as collector, Gemini is more efficient than DTAB. It is particularly true for the flotation of illite. With 2.0��10-4 mol/L 12-4-12, about 85% of illite can be floated, but that is only 60% when DTAB is used. When the concentration of 12-4-12 is up to 3.5��10-4 mol/L, a plateau is presented for illite flotation, whereas it is reached at 5.0��10-4 mol/L DTAB. The maximum recoveries of illite, pyrophyllite and kaolinite are 99.2%, 91.7% and 99.6% respectively. It can be concluded that 12-4-12 as a collector displays a superior collecting power for the three silicate minerals, and is much more efficient than the corresponding monomeric surfactant.
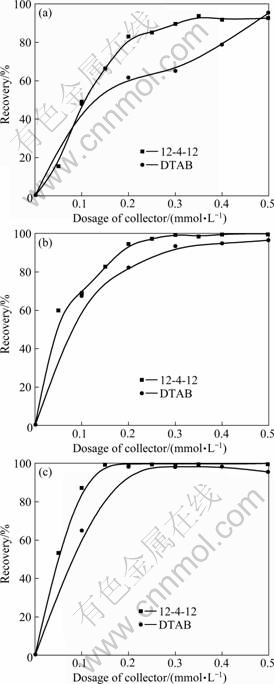
Fig.3 Flotation recoveries of illite (a), pyrophyllite (b) and kaolinite (c) as function of collector dosage at pulp pH 6
3.2 FTIR spectral analysis
FTIR spectra of illite, pyrophyllite, kaolinite and minerals conditioned with 2.0��10-2 mol/L cationic
surfactant solutions are presented in Fig.4. Compared with DTAB, the spectra of clay minerals with 12-4-12 are almost identical, suggesting that the two surfactants have same mechanism of interaction to mineral surfaces.

Fig.4 FTIR spectra of illite (a), pyrophyllite (b) and kaolinite (c) in absence and presence of 12-4-12 and DTAB
In the 3 000-2 500 cm-1 region, new bands at around 2 925 and 2 855 cm-1 for clays with cationic surfactants are previously attributed to the ��CH2 stretching of acyclic compounds. It is evidence that the two surfactants are adsorbed on the mineral surfaces. In the 2 000-1 500 cm-1 region, the pure minerals spectra differ significantly from those previously reported [22-23]. Bands at 1 984-1 821 cm-1 would be Al=O or Si=O stretching frequency due to the relaxation and reconstruction of the planes. These locations are usually assigned to C=O characteristic frequency. The bands at 1 646.3 cm-1 for pyrophyllite and at 1 635.03 cm-1 for kaolinite are attributed to the H��O��H deformation of co-intercalated water molecules. This is based on the study of FROST et al[24], who assigned all the bands present in the 1 575-1 680 cm-1 region to H��O��H bending.
Upon adsorption, intensities of bands at 1 984- 1 821 cm-1 are reduced, bands at 1 646 and 1 635 cm-1 almost disappear and new bands at 1 465.1 cm-1 for illite, 1 465.4 cm-1 for pyrophyllite and 1 466.23 cm-1 for kaolinite are observed. We assign the new bands to C��H???O twist vibration. Due to the solution chemistry of kaolinite, it is proved that, at acidic pH, the apex oxygen atoms in the silica tetrahedral will be hydrolyzed and dangling OH groups and naked O anions will form on the surface to provide extra hydrogen bonding sites[25]. Thus, the coupled inner-surface hydroxyl sites may leave naked O anions alone, which offers a chance to hydrogen bond to cationic collector molecules.
3.3 Application of electrokinetics in flotation
Electrokinetic measurements can be used to delineate interfacial phenomena, where electrical double layer effects are of relevance to flotation. Of particular importance is the utilization of electrokinetic techniques for distinguishing physical and chemical adsorption of flotation collectors and for delineating conditions under which various kinds of specific adsorption phenomena may occur. The utility of electrokinetic studies in flotation of illite, pyrophyllite and kaolinite with 2.0�� 10-4 mol/L 12-4-12 and DTAB is illustrated in Fig.5.

Fig.5 Zeta potentials of minerals with cationic surfactants of 2.0��10-4 mol/L as function of pH: (a) Illite; (b) Pyrophyllite; (c) Kaolinite
Fig.5 shows the Zeta potential in the absence and presence of cationic collectors. It is shown from the Zeta potential results that the PZC of illite, pyrophyllite and kaolinite are 2.4, 3.0 and 3.4, respectively, above which the negative Zeta potential increases in magnitude. In the presence of 12-2-12 and DTAB, the ��-potential of illite, pyrophyllite and kaolinite shows a pronounced shift towards more positive direction, indicating that collector molecules positively charged have been adsorbed onto clay minerals surfaces through electrostatic force, resulting in the more positive surface. In this regard, the Gemini cationic surfactant has an identical electrostatic adsorption capacity in comparison with the traditional corresponding single-chain surfactant.
4 Discussion
In previous studies, it can be concluded that 12-4-12 as a collector displays a superior collecting power for the three silicate minerals, and reveals much more efficiency than the corresponding monomeric surfactant. Furthermore, FTIR and electrokinetic analyses indicate that Gemini surfactant and the corresponding monomeric surfactant have the same mechanism of adsorption on mineral surfaces because of mainly electrostatic attraction and partially hydrogen bond effect. Performance of Gemini surfactant would be tightly correlated with its special structure and its essential properties. So, fluorescence emission spectroscopy and contact angle measurement are conducted to answer the questions why dimeric surfactant reveals a superior collecting ability over the corresponding conventional one.
4.1 Micro-polarity
I1/I3 in the fluorescence spectrum reflects the intensity of micro-polarity around pyrene, and the change in I1/I3 can be used to detect the formation of micelle and aggregation[26]. Fig.6 shows the dependence of the micro-polarity on surfactant concentration cs in 12-4-12/3 g kaolinite mineral pulp and in DTAB/3 g kaolinite mineral pulp.
At very low cs, I1/I3 is nearly a constant, which is in agreement with pyrene in a surfactant-free solution corresponding to a polar environment, suggesting no hydrophobic microdomain formed in the solution. I1/I3 starts to decease at CAC (critical aggregation concentration), i.e., the onset for the formation of micelle-like structure near the binding site of mineral. Surfactants, both Gemini and conventional, have a nearly identical CAC at the concentration of about 1.0��10-7 mol/L. Furthermore, the charge of mineral surface is so negative that the electrostatic attraction between surfactant molecules and mineral particles is strong, and the CAC in the two circumstances is much lower as shown in Fig.6. Beyond CAC, I1/I3 in the pulp of Gemini remains nearly unchanged until the second decrease, which is defined as CMC (critical micelle concentration).

Fig.6 Surfactant concentration dependence of micro-polarity in collector/mineral pulp
The CMC value of 12-4-12 in this solution is about 2.5��10-5 mol/L. In this region, micelle-like structure continuously increases, and then pyrene is surrounded by the alkyl tails of the micelles. As a result, the pyrene micro-environment and the micropolarity have no discernable change. The second decrease of the I1/I3 at CMC suggests a much tight arrangement of alkyl tails around pyrene. One reason is that the interactions between micelle-like structures occur due to the stronger hydrophobic interactions between alkyl chains, leading to the formation of cross-linking complex. Another is that the binding sites of mineral slab tend to be less, and excessive Gemini surfactant cannot form new micelle-like structure, but enters into the existing structure to form a larger complex. Either of these factors causes an increase in the density of the surfactant alkyl tails in the micelle-like structures, so that the adsorbed mineral particles are hydrophobic. As for the DTAB, in the region between CAC and CMC, another decrease can be seen, which is similar to CAC. This is attributed to the formation of another micelle-like structure, due to the intra- or intermolecular association. This is also considered that the hydrophobical association in aqueous solutions proceeds in two stages, and such decrease between CAC and CMC is defined as CAC2. CMC of DTAB in kaolinite pulp is 5.0��10-4 mol/L, too much little than that of the Gemini.
Beyond the point of CMC, in both of the circumstances, I1/I3 increases sharply and a peak can be observed. In this process, the micro-polarity increases because of the reduced amount of the surfactant and mineral particles in solution. With further increasing the surfactant concentration, the precipitate re-dissolves and micro-polarity decreases. By hydrophobic forces between the alkyl tails of the added surfactant and those in the precipitate, excessive surfactant intercalates into the precipitate and forms an even larger aggregate. The aggregate is charged again, leading to the redissolution into solution.
The studies of micro-polarity show aggregation behaviors of the two types of surfactants at the solid/ solution interface, in fact, which has been studied on other substrates[27-29]. It is found that the mechanism of adsorption involves two steps[30-31]. The first step occurs at very low concentration and corresponds to a binding of individual Gemini on charged sites on the silica surface by an ion-exchange mechanism. The second step occurs at a concentration slightly below the CMC and corresponds to the formation of surface aggregates. It is also reported that compared with the corresponding conventional surfactants, the Gemini surfactant has much lower c20 value (the value of the surfactant concentration at which the surface tension of water is reduced by 0.02 N/m) and also has a slightly lower value of ��CMC (the value of the surface tension at the CMC). These properties result in a much higher efficiency of Gemini surfactant than the corresponding monomer at the solid/solution interface.
4.2 Contact angle
Normally, surfactant adsorption on solid substrates can modify the surface hydrophobicity, depending on the orientation of adsorbed surfactant molecules. The static contact angles of the mineral surfaces treated with surfactant solutions at different concentrations are shown in Fig.7. All the contact angles given are the initial values, that is to say, the values obtained immediately after 2 ��L water drop was in contact with the mineral surface.

Fig.7 Initial static contact angles of mineral substrates treated with different surfactant concentrations (Error bars indicate degree of reproducibility of measurement)
As seen from Fig.7, the water contact angle on collectors-treated surfaces is concentration-dependent. The plot of contact angle vs surfactants concentration presents a peak of 84? at 0.05 mmol/L for the Gemini and of 70? at 0.75 mmol/L for CTBA. The peaks divide the two plots into two distinct regions. In the upward shift from a very low concentration to the turning point, surfactants monomers are proved to exist on the mineral surfaces, so that the contact angle is much larger than that of bare kaolinite surface (about 30?). However, these molecules could not form micelle on the kaolinite surface at a random state, and their hydrophobic tails may be exposed to air. In this case, the surface free energy is relatively low and it is reasonable to have a relatively hydrophobic surface. At the concentration below the peak, more randomly distributed surfactant molecules are adsorbed with their tails facing air. These molecules cause the further decrease of surface free energy and the upward shift of contact angle. With increasing surfactant concentration, micelles are formed, and the surface area may be occupied by these bilayer micelles. The lower contact angles are the evidences of this increased coverage, but this interpretation needs to be further studied on the difference in the two surfactant micelles orientation and ordering on kaolinite surface. If the explanation about contact angle results is true, the turning point of the curves should correspond to CMC of each surfactants. The vales of 0.05 mmol/L and 0.75 mmol/L basically agree with micro-polarity results.
5 Conclusions
1) Gemini collector displays a far stronger collecting power for illite, phrophyllite and kaolinite than the corresponding monomer. At pH 6, the maximum recoveries of illite, pyrophyllite and kaolinite are reached when the dosage of 12-4-12 up to 3.5��10-4 mol/L, and they are 99.2%, 91.7% and 99.6%, respectively.
2) Physical electrostatic effect and hydrogen bonding mainly account for the mechanism of the two cations and minerals. A pronounced shift of Zeta potential in the presence of cationic collectors shows that both of them may have a strong attraction on oppositely charged mineral particles.
3) The merit performance of 12-4-12 on clay minerals flotation is associated with its special structure and its essential properties. Micro-polarity and contact angle studies show that in the mineral pulp, the dimeric surfactant has a much lower CMC than the monomer, resulting in a better hydrophobic characteristic at mineral/solution interface.
References
[1] YOON R H, NAGARAJ D, WANG S, HILDERAND T. Beneficiation of kaolin clay by froth flotation using hydroxamate collectors [J]. Miner Eng, 1992, 5(3/5): 457-467.
[2] HU Y, LIU X, QIU G. Solution chemistry of flotation separation on aluminum and silicate in diasporic-bauxite (I)��Crystal structure and floatability [J]. Mining and Metallurgical Engineering, 2000, 20(2): 11-14. (in Chinese)
[3] HU Y, JIANG H, QIU G. Solution chemistry of flotation separation on aluminum and silicate in diasporic-bauxite [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(1): 125-130. (in Chinese)
[4] HU Y. Progress in flotation de-silica [J]. Trans Nonferrous Met Soc China, 2003, 13(3): 656-662.
[5] LUO Z, HU Y, WANG Y, QIU G. Mechanism of dispersion and aggregation in reverse flotation for bauxite [J]. J Nonferrous Met, 2001, 11(4): 680-683.
[6] JIANG H, HU Y H, QIN W, WANG Y, WANG D Z. Mechanism of flotation for diaspore and aluminum-silicate minerals with alkylamine collectors [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(4): 688-692. (in Chinese)
[7] CAO X F, HU Y H, JIANG Y R. Flotation mechanism of aluminium silicate minerals with N-dodecyl-1,3-diaminopropane [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(4): 696-700. (in Chinese)
[8] CAO X F, HU Y H, XU J. Synthesis of ��-alkoxy-propylamines and their collecting properties on aluminosilicate minerals [J]. J Cent South Univ Technol, 2004, 11(3): 280-285.
[9] ZHAO S M, WANG D Z, HU Y H, LIU B D, XU J. The flotation behaviour of N-(3-aminopropyl)-dodecanamide on three aluminosilicates [J]. Miner Eng, 2003, 16(12): 1391- 1395.
[10] ZHAO S M, WANG D Z, HU Y H, BAO X S, XU J. Flotation of aluminosilicates using N-(2-aminoethyl)-1-naphthaleneacetamide [J]. Miner Eng, 2003, 16(10): 1031-1033.
[11] MENGER F M, LITTAU C A. Gemini-surfactants: Synthesis and properties [J]. J Am Chem Soc, 1991, 113(4): 1451-1452.
[12] ZANA R, XIA J D, ZARA R. Gemini surfactants synthesis, interfacial and solution-phase behavior and applications [M]. New York: Dekker, 2003.
[13] ESPERT A, KLITZING R V, POULIN P, COLIN A. Behavior of soap films stabilized by a cationic dimeric surfactant [J]. Langmuir, 1998, 14(15): 4251-4260.
[14] ZANA R, BBENRRAOU M, RUEFF R. Alkanediyl-��, ��-bis(dimethylalkylammonium bromide) surfactants (I): Effect of the spacer chain length on the critical micelle concentration and micelle ionization degree [J]. Langmuir, 1991, 7(6): 1072-1075.
[15] ZANA R. Alkanediyl-��,��-bis(dimethylalkylammonium bromide) surfactants (II): Krafft temperature and melting temperature [J]. J Colloid Interface Sci, 2002, 252(1): 259-261.
[16] ROSEN M J. Surfactant: Designing structure for performance [J]. Chem Technol, 1985, 15(3): 292-298.
[17] KERN F, ZANA R, CANDAU S J. Rheological properties of semidilute and concentrated aqueous solutions of cetyltrimethylammonium chloride in the presence of sodium salicylate and sodium chloride [J]. Langmuir, 1991, 7(7): 1344-1351.
[18] PISARCIK M, IMAE T, DEVINSKY F, LACKO I. Aggregates of sodium hyaluronate with cationic and aminoxide surfactants in aqueous solution-light scattering study [J]. Colloids Surf A, 2001, 183/185(15): 555-562.
[19] HANSSON P. A fluorescence study of divalent and monovalent cationic surfactants interacting with anionic polyelectrolytes [J]. Langmuir, 2001, 17(14): 4161-4166.
[20] THALERG K, VANSTAM J, LINBLAD C, ALMGREN M. Time-resolved fluorescence and self-diffusion studies in systems of a cationic surfactant and an anionic polylelectrolyte [J]. J Phys Chem, 1999, 95(22): 8975-8982.
[21] HASHIDZNME A, MIZUSAK M, YODA K, MORISHIMA Y. Interaction of unimolecular micelles of hydrophobically-modified polyelectrolytes with nonionic/ionic mixed surfactant micelles [J]. Langmuir, 1999, 15(12): 4276-4282.
[22] PENG W S, LIU G K. Graph of minerals infrared spectra [M]. Beijing: Science and Technology Press, 1982. (in Chinese)
[23] WEN L. Mineral infrared spectroscopy [M]. Chongqing: Chongqing University Press, 1989. (in Chinese)
[24] FROST R T, KRISTOF J, PAROZE G N, KLOPROGGE J T. Role of water in the intercalation of kaolinite with hydrazine [J]. J Colloid Interface Sci, 1998, 208(1): 216-225.
[25] LIU G Y, ZHONG H, HU Y H, XIA L Y. The role of cationic polyacrylamide in the reverse flotation of diasporic bauxite [J]. Miner Eng, 2007, 20(13): 1191-1199.
[26] JIANG H, HU Y H, WANG D Z, QIN W Q, GU G H. Structure of the adsorbed layer of cationic surfactant at diaspore-water interface [J]. J Chin Univ Mining Technol, 2005, 34(4): 500-503.
[27] MENGER F M, KEIPER J S, AZOV V. Gemini surfactants with acetylenic spacers [J]. Langmuir, 2000, 16(5): 2062-2067.
[28] ROSEN M J, MATHIAS J H, DAVENPORT L. Aberrant aggregation behavior in cationic gemini surfactants investigated by surface tension, interfacial tension, and fluorescence methods [J]. Langmuir, 1999, 15(21): 7340-7346.
[29] IN M, BEC V, AGUERRE-CHARIOL O, ZANA R. Quaternary ammonium bromide surfactant oligomers in aqueous solution: Self-association and microstructure [J]. Langmuir, 2000, 16(1):141-148.
[30] CHORRO C, CHORRO M, DOLLADILLE O, PARTYKA S, ZANA R. Adsorption of dimeric (Gemini) surfactants at the aqueous solution/silica interface[J]. J Colloid Interface Sci, 1998, 199(2): 169-176.
[31] CHORRO C, CHORRO M, DOLLADILLE O, PARTYKA S, ZANA R. Adsorption mechanism of conventional and dimeric cationic surfactants on silica surface: Effect of the state of the surface[J]. J Colloid Interface Sci, 1999, 210(1): 134-143.
Foundation item: Project(2005CB623701) supported by the National Basic Research Program of China; Project(50874118) supported by the National Natural Science Foundation of China; Projet(2007B52) supported by the Foundation for the Author of National Excellent Doctoral Dissertation of China
Corresponding author: ZHONG Hong; Tel: +86-731-8830603; E-mail: zhongh@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(08)60293-9
(Edited by YANG Bing)