
Flotation and adsorption of quaternary ammonium cationic collectors on diaspore and kaolinite
JIANG Hao, XU Long-hua, HU Yue-hua, WANG Dian-zuo, LI Chang-kai, MENG Wei, WANG Xing-jie
School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China
Received 18 November 2010; accepted 15 May 2011
Abstract: The flotation and adsorption behaviors of dodecyltrimethylammonium chloride(DTAC) and cetyltrimethylammonium chloride(CTAC) on diaspore and kaolinite were studied. Solution depletion methods were used to determine adsorption isotherms. Fluorescence probe test along with Zeta potential measurement was also conducted for further investigation into the adsorption of quaternary amines at the mineral-water interface. The results show that the flotation recovery of kaolinite decreases with an increase in pH when DTAC and CTAC are used as collectors, while diaspore is on the contrary. As the carbon chain length of the collectors increases, the flotation recoveries of minerals increase. However, the increment rate of kaolinite is significantly lower than that of diaspore. In the low surfactant concentration range, the cationic surfactants adsorb readily on diaspore surfaces just due to electrostatic interactions. As for kaolinite surfaces, ion exchange process also exists. With a further increase in surfactant concentration, the adsorption was ascribed to the hydrophobic association of chain-chain interactions. Micro-polarity of mineral surfaces study shows that CTAC has a better hydrophobic characteristic than DTAC. Larger aggregates are formed with CTAC on diaspore than on kaolinite in the same solution concentration. The results also indicate that the chain length of cationic surfactants has a greater influence on the adsorption of diaspore than on kaolinite, which is consistent with the flotation result.
Key words: diaspore; kaolinite; DTAC; CTAC; flotation; adsorption; ion exchange; micro-polarity
1 Introduction
Diasporic-bauxite is a major source for aluminumoxide production in China, which is characterized by its low mass ratio of Al2O3 to SiO2 [1]. Diaspore is valuable mineral and aluminosilicate minerals are predominant gangues, such as pyrophyllite, illite, kaolinite and chlorite. This kind of bauxite ores cannot be treated economically by Bayer��s process unless the mass ratio of Al2O3 to SiO2 in the ores is improved higher than 10. Many researches show that the high quality material for Bayer��s process can be provided by flotation to desilica for the diasporic-bauxites [2-3].
In China, reverse flotation has been proved to be an efficient method for bauxite desilication [4]. Numerous researchers [5-6] have focused on the flotation solution chemistry of aluminosilicates minerals and the relationship between crystal structure and flotation. Efforts have been made to develop new and efficient collectors [7]. It has been known that the effect of reverse flotation to desilicon depends on the adsorption of cationic surfactants selectively on aluminosilicaes and diaspore surface. Adsorption of cationic surfactants on charged mineral oxides has been studied extensively [8]. It is generally accepted that cationic surfactant absords readily due to electrostatic interactions on oppositely charged surfaces in a low concentration range. With an increase in surfactant concentration, the adsorption increases sharply. With a further increase in surfactant concentration, the adsorption runs to saturation. But in the case of clay minerals, adsorption of cationic surfactants on clay minerals is attributed to ion exchange [9-10].
In this work, the difference of flotation behaviors of diaspore and kaolinite with the cationic surfactants dodecyltrimethylammonium chloride (DTAC) and cetyltrimethylammonium chloride (CTAC) was discussed. Adsorption mechanisms of quaternary ammonium cationic collectors on diaspore and kaolinite were investigated by Zeta potential measurement and adsorption isotherm study. Micro-polarity was further investigated by pyrene fluorescence probe technique. This study focused on elucidating the influence of surfactant chain lengths on the adsorption of quaternary ammonium on diaspore and kaolinite. The interaction between cationic surfactants and mineral surfaces was determined.
2 Experimental
2.1 Materials and chemicals
Kaolinite and diaspore were obtained from Xiaoguan in Henan province, China and Xiaoyi in Shanxi province, China, respectively. The samples were hand-picked, ground by porcelain mill and screened to size smaller than 0.098 mm. Chemical composition analysis and X-ray diffractometry (XRD) were used to study the characteristics of chemical and mineral compositions. The results showed that the purity of diaspore and kaolinite were about 90%. The surface areas for diaspore and kaolinite were 1.26 and 16.54 m2/g, respectively.
DTAC and CTAC of analytical quality from Nanjing Robiot Co., Ltd were used as collectors. Pyrene from Sigma-Aldrich (Shanghai) Trading Co., Ltd., China was recrystallized twice by anhydrous ethanol. Aqueous solution of analytical pure KNO3 was used as background electrolyte solution. Solutions of HCl and NaOH were used to adjust the pH of the system. Double distilled water was used in all tests.
2.2 Flotation
Mineral particles (3 g) were placed in a plexiglass cell (40 mL), and then filled with distilled water. pH regulators were added to adjust the desired pH. After adding desired amount of reagents, the suspension was agitated for 3 min. The flotation was conducted for 6 min. The froth products and tails were weighed respectively after filtration and drying, and the recovery was calculated based on the mass of the products, ruling out the effect of impurities.
2.3 Zeta-potential measurements
A suspension containing 0.1% (mass fraction) mineral particles gournd to <5 ��m in an agate mortar was prepared in 1 mmol/L KCl solution, then conditioned by magnetic stirring apparatus for 5 min. After settling for 10 min, the supernatant of dilute fine particle suspension was taken for Zeta potential measurement.
The Zeta-potentials were measured using a Coulter Delsa 440sx Zeta potential analyzer (USA) equipped with a rectangular electrophoresis cell. The conductivity and pH of the suspension were monitored continuously during the measurement and the environmental temperature was maintained at 22 ��C.
2.4 Adsorption measurement by solution depletion method
Appropriate volumes of dry volumetric flasks were prepared, each containing a certain amount of diaspore. Desired amount of reagent solution was added to each flask. The flasks were then stoppered and shaken at the same stirring rate for 2 h and the system was kept at 22 ��C. When the equilibrium was reached, the solid particles were separated by centrifugation at 12 000 r/min for 20 min. The supernatant solution was analyzed by two-phase titration method [11-12]. The amount of collector adsorbed on the diaspore particles was calculated as [13]:
��=(C0-C)V/(mA) (1)
where C 0 and C are the initial and supernatant concentrations, respectively; V is the solution volume; m is the amount of the particles per sample; A is the particle specific surface area.
2.5 Fluorescence emission spectroscopy
Samples for fluorescence emission spectroscopy were prepared by mixing pyrene stock solution with surfactant and mineral pulp, and allowed to stand for 2 h to equilibrate. The pyrene steady-state emission spectra in the suspension was obtained by using a Hitach F-4500 fluorescence spectrophotometer. The excitation wave length of pyrene was 335 nm. The pyrene stock solution was prepared by dissolving pyrene in hot water until saturation, then cooled to 25 ��C and filtered. The concentration of pyrene in the solution was determined to be 6.53��10-7 mol/L [14].
3 Results and discussion
3.1 Flotation
Flotation tests are conducted to determine the collecting properties of DTAC and CTAC to kaolinite and diaspore. Tests with 0.2 mmol/L collectors are performed to investigate the effect of pH on flotation. Similarly to other oxidized ores, a gradual increase in the recovery for diaspore with increasing pH is observed in the recovery��pH curves (Fig. 1). When the pH reaches about 5.0, the recovery of diaspore with CTAC is up to 90%, while it is just about 70% with DTAC. Apparently, CTAC is more efficient than DTAC in the pH range from 1.52 to 11.19. The case of kaolinite is opposite, namely, the flotation recovery of kaolinite decreases with the increase in pH. It is obviously that the flotation behavior of kaolinite is different from the other oxides and silicates. So, it is believed that the anomalous behavior is related to the kaolinite crystal structure [15]. At the same time, as the carbon chain of collector increases, the recovery of kaolinite increases, but the increase rate is significantly lower than that of diaspore.
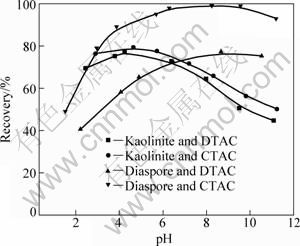
Fig. 1 Flotation recoveries of kaolinite and diaspore as function of pulp pH
As shown in Fig. 2, the DTAC and CTAC cationic collectors display a similar flotation rule. The recovery-collector dosage curves of diaspore obtained at pH=5 (Fig. 2) show that with increasing collector dosage, there is a sharp increase in the recovery of diaspore in the low concentration range between 0.01 and 0.2 mmol/L. When the concentrations of the two solutions are between 0.2 and 0.5 mmol/L, further increase in recovery becomes slow. When the concentration is above 0.6 mmol/L, a flat horizontal is presented and the recovery reaches the maximum value.

Fig. 2 Flotation recoveries of kaolinite and diaspore as function of collector dosage
This discrepancy could be attributed to the length of carbon chain. There have been many studies focused on the flotation mechanism of cationic collectors, finding that the adsorption characteristic is relevant to mineral surface charge potential. It is expected that for pulp pH value above the point of zero charge (PZC) of mineral, there exists an electrostatic attraction between the negative charged surfaces and the cationic collectors.
3.2 Zeta potential
The adsorption of collectors in the flotation of oxides is controlled by the electrical double layer on the mineral/water surface. When the mineral surface is charged oppositely, anionic or cationic collectors are adsorbed to the diaspore physically. Diaspore, a type of oxides, forms hydroxylated surface when it contacts with water. Adsorption of OH- or dissociation of H+ from the surface hydroxyls can account for the surface charge on the diaspore as [16-17]:
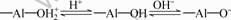 (2)
(2)
Kaolinite has two kinds of different crystal planes, one is the basal planes (001), the other is the edge surfaces (010) and (110). The basal planes are permanently negative charged, which is attributable to isomorphous substitution of lattice elements. Si4+ and Al3+ can be replaced by Al3+ ions and Mg2+/Fe2+, respectively. This permanent negative charge is pH-independent [18]. In the case of the edges, the charge is pH-dependent, which can be described using the traditional model for oxides as:
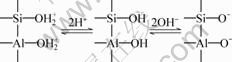 (3)
(3)
Adjusting the pH value of the system can change the adsorption of collectors on the diaspore surfaces. Thus, the PZC of the mineral is the most important property of a mineral in such systems [19].
In order to investigate the effects of pH on Zeta potential with different collectors, the Zeta potential is studied in the absence and presence of DTAC and CTAC as a function of pH, the result is shown in Fig. 3. The PZC of diaspore is obtained in aqueous solution of pH=6.2 above which the negative Zeta potential increases in magnitude. In the presence of DTAC and CTAC, the Zeta potential and PZC of diaspore shift towards the positive direction. It is indicated that the cationic collector molecules have been adsorbed onto the diaspore surfaces with negative charge by electrostatic force. However, collectors have also been adsorbed onto the diaspore surfaces even when the diaspore surface is positively charged at pH below PZC, which might result from the hydrogen bonding interaction [19]. The CTAC has apparently stronger electrostatic interaction with the diaspore than DTAC. It is demonstrated the chain length is of critical importance in determining the adsorption behaviour of a surfactant on diaspore.
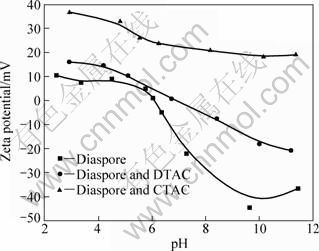
Fig. 3 Zeta potential of diaspore as function of pH
However, in the case of kaolinite, the flotation recovery decreases with the increase in pH. Kaolinite exhibits better floatability in acidic solution than in alkaline solution. This anomalous flotation behavior of kaolinite cannot be explained from electrokinetic measurements.
3.3 Adsorption isotherm
Adsorption isotherm is the traditional method for investigating surfactant adsorption phenomena [20]. The measured adsorption isotherms of DTAC and CTAC on diaspore are depicted in Fig. 4, with adsorbed amount as a function of equilibrium concentration (Ce). Figure 4 shows that the amount of adsorbed surfactants is in the order of CTAC>DTAC, their critical micelle concentrations (CMC) are 1.1?10-3 and 1.6?10-2 mol/L, respectively. When the equilibrium concentrations of CTAC reach the corresponding CMC value, adsorption equilibrium is achieved. But the CMC of DTAC is too high to have hemimicelle formation occurring at a higher solution concentration.
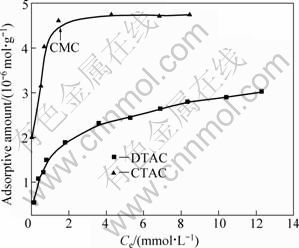
Fig. 4 Adsorption isotherm of quaternary ammonium salts on diaspore surface when pH=5-6
This is consistent with the common observation that the adsorption of ionic surfactants on oxide ore follows the mechanism model, electrostatic adsorption- hydrophobic association-adsorption equilibrium.
It is suggested in Fig. 4 that adsorption mechanism of cationic collectors on oxide minerals can be divided into four stages. In the first stage, cationic surfactants are electrostatically adsorbed to the surface of minerals. The surface adsorption is determined mainly by the surface charge. In the second stage, the mineral surface charge has been neutralised, and a first plateau is presented. But the solution activity of the surfactant is not sufficient to lead to any form of aggregation at the interface, thus, surfactants are still adsorbed as monomers. The abrupt increase in adsorption at the CMC denotes the onset of the third stage. In this stage, the solution surfactant concentration is sufficient to lead to hydrophobic interactions between monomers. The fourth stage occurs above the CMC, the formation of aggregates fully forms and the saturation levels of surface coverage reach [20].
Figure 5 shows the adsorption isotherms of DTAC and CTAC on kaolinite at pH=5. It can be seen that the shapes of both the isotherms are essentially the same. In the low surfactant concentration range, the adsorbed amount increases greatly with the concentration of cationic surfactants. The CTAC adsorbed is more than DTAC. The adsorption isotherms of DTAC and CTAC on kaolinite are different from those of DTAC and CTAC on diaspore.
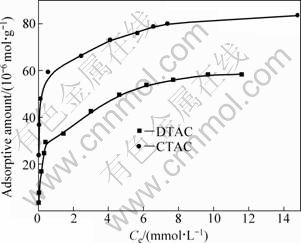
Fig. 5 Isotherm of adsorption of quaternary ammonium salts on kaolinite when pH=5-6
The adsorption curve shape is the same as cationic surfactants on the montmorillonite [21], and the cation exchange presents in the adsorption of cationic surfactants on the clay mineral surfaces [22]. The adsorption of DTAC and CTAC cationic collector on the kaolinite surface can be divided into two stages. In the low surfactant concentration range, the cationic surfactants adsorb readily on kaolinite surfaces due to electrostatic interactions and ion exchange. With further increase in surfactant concentration, the adsorption is ascribed to the hydrophobic association of chain-chain interactions through van der Waals forces.
The study on adsorption isotherm shows the adsorption of cationic surfactants on mineral is electrostatic adsorption and hydrophobic interactions. Ion exchange plays a dominant role in adsorption of the cationic surfactants on kaolinite surfaces. When other factors are in the same situation, the adsorption amounts of DTAC and CTAC on kaolinite are greatly more than those on diaspore. The study also highlights the significance of the chain length of quaternary ammonium salts. As the carbon chain length of a surfactant molecule is increased, the absorbed amount of surfactants on mineral surface increases obviously. To our knowledge, surfactants with longer carbon chains have a much greater driving force for aggregation, which dramatically reduces the solution CMC. All these lead to the different flotation recoveries of diaspore and kaolinite with DTAC and CTAC.
3.4 Micro-polarity
A typical emission spectrum has five peaks at 373, 379, 384, 390 and 397 nm, respectively, as shown in Fig. 6. The intensity ratio of the third to the first vibronic peaks I3/I1 is sensitive to the local environment of pyrene. The value of I3/I1 in hydrophobic environments is higher than that in a hydrophilic environment. The values of I3/I1 are 0.5-0.6 in water, 0.8-0.9 in surfactant micelles and >1 in nonpolar solvents, respectively. Since this ratio can be used to characterize the polarity of environments, it is termed the polarity parameter [23-25].
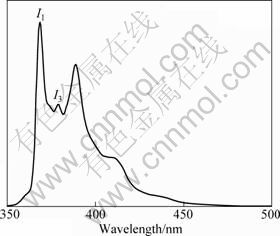
Fig. 6 Fluorescence emission spectrum of pyrene in aqueous solution
Figure 7 shows I3/I1 on diaspore surface as a function of the concentration of DTAC and CTAC. When diaspore interacts with DTAC, I3/I1 increases gradually with increasing DTAC concentration. When the concentration is up to 2?10-2 mol/L, I3/I1 is still less than 0.75. That is to say, the diaspore surface is polar environment and micelles does not form on the diaspore surface in DTAC solution. It can be concluded that DTAC as a collector displays a poor collecting ability to the diaspore. In CTAC solution, as its concentration increases, I3/I1 on diaspore surface increases sharply. Up to a certain concentration, a platform appears and I3/I1 is about 0.95. In this way, CTAC on diaspore surface forms micelle, resulting in a superior collecting ability to the diaspore of CTAC than DTAC. It is consistent with the flotation result.
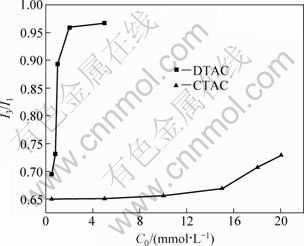
Fig. 7 Relationship between I3/I1 on diaspore surface and concentration of quaternary ammonium salts when pH=5-6 (C0 is initial concentration)
Figure 8 shows the relationship between I3/I1 on kaolinite surface and the concentration of DTAC and CTAC. With increasing concentration of DTAC, I3/I1 changes little in the DTAC solution. DTAC on the kaolinite surface does not form micelles. In CTAC solution, as its concentration is increased, I3/I1 increases to its maximum value of 0.85, which does not meet non-polar environment. The micelle is relatively smaller on kaolinite surface than on diaspore surface in CTAC solution. That is to say, when the CTAC is used as collector, the floatability of diaspore is better than kaolinite. Larger aggregates are formed with CTAC on diaspore than kaolinite in the same solution concentration.

Fig. 8 Relationship between I3/I1 on kaolinite surface and concentration of quaternary ammonium salts when pH=5-6
4 Conclusions
1) When DTAC and CTAC are used as collectors, the flotation recovery of diaspore increases with the increase in pH. But the flotation recovery of kaolinite decreases with the increase in pH.
2) The PZC of diaspore in aqueous solution is obtained at pH=6.2. In the presence of DTAC and CTAC, the Zeta potential and PZC of diaspore shift towards the positive direction. But the anomalous flotation behavior of kaolinite cannot be explained from electrokinetic considerations.
3) In the low surfactant concentration range, the adsorption of cationic collectors is driven by electrostatic interaction on diaspore, while the cationic surfactants adsorb readily on kaolinite surfaces due to electrostatic interactions and ion exchange. With further increase in surfactant concentration, the adsorption is ascribed to the hydrophobic association of chain-chain interactions through van der Waals forces.
4) Micro-polarity study shows that in the mineral pulp, CTAC has a better hydrophobic characteristic than DTAC. With the same CTAC solution concentration, larger aggregates are formed on diaspore than on kaolinite.
References
[1] HU Yue-hua. Progress in flotation de-silica [J]. Transactions of Nonferrous Metals Society of China, 2003, 13(3): 656-662.
[2] XIAO Ya-qing. Technology development of China��s aluminum industry [M]. Beijing: Metallurgical Industry Press, 2007: 37-38. (in Chinese)
[3] HUANG Guo-zhi, FANG Qi-xue, CUI Ji-rang. Beneficiation ways and research progress of bauxite ore [J]. Light Metals, 1995(5): 16. (in Chinese)
[4] LUO Zhao-jun, HU Yue-hua, WANG Yu-hua, QIU Guan-zhou. Mechanism of dispersion and aggregation in reverse flotation for bauxite [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(4): 680-683. (in Chinese)
[5] HU Yue-hua, LIU Xiao-wen, XU Zheng-he. Role of crystal structure in flotation separation of diaspore from kaolinite, pyrophyllite and illite [J]. Minerals Engineering, 2003, 16: 219-227.
[6] HU Yue-hua, JIANG Hao, QIU Guan-zhou. Solution chemistry of flotation separation on aluminum and silicate in diasporic-bauxite [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(1): 125-130. (in Chinese)
[7] CAO Xue-feng, HU Yue-hua, JIANG Yu-ren. Flotation mechanism of aluminium silicate minerals with N-dodecyl-1, 3-diaminopropane [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(4): 696-700. (in Chinese)
[8] BRUCE L R, SCAMEHORN J F, HARWEL J H. Adsorption of a mixture of anionic surfactants on alumina [J]. Phenomena in Mixed Surfactant Systems, 1986, 311: 200-215.
[9] WANG Jin-ben, HAN Bu-xing, DAI Ming, YAN Hai-ke, LI Zhi-xin, THOMAS R K. Effects of chain length and structure of cationic surfactants on the adsorption onto Na-kaolinite[J]. Journal of Colloid and Interface Science, 1999, 213: 596-601.
[10] LI Zhao-hui, GALLUS L. Adsorption of dodecyl trimethylammonium and hexadecyl trimethylammonium onto kaolinite-competitive adsorption and chain length effect [J]. Applied Clay Science, 2007, 35: 250-257.
[11] LI Z, ROSEN M J. Two-Phase mixed indieator titration method for the determination of anionic surfactants [J]. Anal Chem, 1981, 53: 1516-1519.
[12] TSBOUCHI M, MITSUSHIO H, YAMKI N. Determination of cationic surfactants by two-phase titration [J]. Anal Chem, 1981, 53: 1957-1959.
[13] BEAUSSART A, VASILEV A M, BEATTIE D A. Adsorption of dextrin on hydrophobic minerals [J]. Langmuir, 2009, 25(17): 9913-9921.
[14] PISARCIK M, IMAE T, DEVINSKY F, LACKO I. Aggregates of sodium hyaluronate with cationic and aminoxide surfactants in aqueous solution-light scattering study [J]. Colloids and Surfaces A, 2001, 183-185(15): 555-562.
[15] HU Yue-hua, SUN Wei, JIANG Hao, MILLER J D, FA Ke-qing. The anomalous behavior of kaolinite flotation with dodecyl amine collector as explained from crystal structure considerations [J]. International Journal of Mineral Processing, 2005, 76: 163-172.
[16] PARIA S, KHILAR K C. A review on experimental studies of surfactant adsorption at the hydrophilic solid�Cwater interface [J]. Advances in Colloid and Interface Science, 2004, 110(3): 75-95.
[17] HU Yue-hua, LIU Xiao-wen, QIU Guan-zhou, HUANG Sheng-sheng. Solution chemistry of flotation separation on aluminum and silicate in diasporic-bauxite (I)��Crystal structure and floatability [J]. Mining and Metallurgical Engineering, 2000, 20(2): 11-14. (in Chinese)
[18] ZHAO Shi-min, WANG Dian-zuo, HU Yue-hua, XU Jing. The flotation behaviour of N-(3-aminopropyl)-dodecanamide on three aluminosilicates [J]. Minerals Engineering, 2003, 16: 1391-1395.
[19] FUERSTENAU D W, PRADIP. Zeta potentials in the flotation of oxide and silicate minerals [J]. Advances in Colloid and Interface Science, 2005, 114-115: 9-26.
[20] ATKIN R, CRAIG V S J, WANLESS E J, BIGGS S. Mechanism of cationic surfactant adsorption at the solid-aqueous interface [J]. Advances in Colloid and Interface Science, 2003, 103: 219-304.
[21] PEKER S, YAPAR S, BESUN N. Adsorption behavior of a cationic surfactant on montmorillonite [J]. Colloids and Surfaces A, 1995, 104: 249-257.
[22] TAHANI A, KARROUA M, DAMME H V, LEVITZ P, BERGAYA F. Adsorption of a cationic surfactant on Na-montmorillonite: inspection of adsorption layer by x-ray and fluorescence spectroscopies [J]. Journal of Colloid and Interface Science, 1999, 216: 242-249.
[23] JIANG Hao, HU Yue-hua, WANG Dian-zuo, QIN Wen-qing, GU Guo-hua. Structure of the adsorbed layer of cationic surfactant at diaspore-water interface [J]. Journal of China University of Mining & Technology, 2005, 34(4): 500-503. (in Chinese)
[24] MATHIAS J H, ROSEN M J, DAVENPORT L. Fluorescence study of premicellar aggregation in cationic Gemini surfactants [J]. Langmuir, 2001, 17: 6148-6154.
[25] HUANG L, SOMASUNDARAN P. The change in structure of surfactant aggregates during adsorption/desorption processes and its effect on the stability of Alumina suspension [J]. Colloid Surface A, 1996, 117: 235-244.
����������Ӳ��ռ���һˮӲ��ʯ����ʯ
�������������
�� 껣�������������������������������� �⣬���˽�
���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083
ժ Ҫ���о�ʮ����������Ȼ��(DTAC)��ʮ����������Ȼ��(CTAC)��һˮӲ��ʯ����ʯ�ĸ�ѡ��Ϊ��ͨ������Ũ�ȷ��ⶨ���������ߣ�ӫ��̽�뷨��Zeta��λ���Է����о�����������Ӳ��ռ��ڿ�������������������ѡ�����������DTAC��CTAC��Ϊ���ռ���һˮӲ��ʯ�ĸ�ѡ����������pH����������ӣ�������ʯ�ĸ�ѡ����������pH�������½��������ռ���̼������ʱ�����︡ѡ��������ߣ�������ʯ�����ӷ���С��һˮӲ��ʯ�ġ��ڵ�Ũ�ȷ�Χ�ڣ������ӱ�����Լ�ͨ����������������һˮӲ��ʯ���棬�����ڸ���ʯ�����������ӽ������á���Ũ������ʱ�������ӱ�����Լ�ͨ��̼������ˮ�����������ֿ�������һ��������������������о�������CTAC����ˮ�Ա�DTACǿ����ͬ��ҺŨ����CTAC��һˮӲ��ʯ�������γɱ��ڸ���ʯ�������Ľ��ţ���Ҳ˵�������ӱ�����Լ�̼����������һˮӲ��ʯ������Ӱ��Ҫ���븡ѡ������Ǻϡ�
�ؼ��ʣ�һˮӲ��ʯ������ʯ��DTAC��CTAC����ѡ�����������ӽ���������
(Edited by FANG Jing-hua)
Foundation item: Projects (50974134, 50804055) supported by the National Natural Science Foundation of China; Project (2005CB623701) supported by the National Basic Research Program of China
Corresponding author: JIANG Hao; Tel: +86-731-88830204; E-mail: jianghao-1@126.com
DOI: 10.1016/S1003-6326(11)61046-7