J. Cent. South Univ. (2012) 19: 2130-2135
DOI: 10.1007/s11771-012-1255-3
Microwave radiation one-pot synthesis of chloropropyl-functionalized mesoporous MCM-41
XU Yan-qin(������), CAO Yuan(��Ԩ), XIA Zhi-ning(��֮��)
College of Chemistry and Chemical Engineering, Chongqing University, Chongqing 400044, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: Chloropropyl-functionalized mesoporous MCM-41(MCM-41-(CH2)3Cl) was synthesized in alkaline medium by the microwave radiation one-pot method, using cetyltrimethy-lammoniumbromide (CTAB) as novel template, tetraethoxysilane (TEOS) as silica source, and chloropropyltriethoxysilane (ClPTES) as the coupling agent. The microstructure of MCM-41-(CH2)3Cl was characterized by the means of X-ray diffraction (XRD), nitrogen absorption-desorption, Fourier transform infrared spectroscopy (FT-IR), scanning electron microscopy (SEM), and transmission electron microscopy (TEM). The results show that a successful synthesis of MCM-41-(CH2)3Cl with well structure is obtained. The optimal microwave power is 120 W and the best microwave time is 50 min. The dosage of chloropropyltriethoxysilane on the structure of chloropropyl-functionalized MCM-41 was also investigated. It is found that the chloropropyltriethoxysilane volume between 0.8 mL and 1.6 mL is favorable for the formation of highly ordered MCM-41-(CH2)3Cl mesostructure.
Key words: microwave; one-pot synthesis; chloropropyl; modification; MCM-41
1 Introduction
The M41S family of mesoporous molecular sieve (MMS) has been first introduced by MOBIL since 1992 [1]. Due to their unique properties, mesoporous molecular sieves have good application potentials in catalytic [2], luminescent materials [3], adsorption [4], drug-loading [5] and so on. In particular, MCM-41 is the most widely used MMS because of its ordered two-dimensional channel structure. MCM-41 is synthesized through sol-gel process, involving two-step agglomeration process [6] (e.g., using tetraethyl silicate as silicon source) as follows.
Hydrolyzation process:
Si(OC2H5)4+xH2O��
(C2H5O)3-x��Si��(OH)x+xC2H5OH
Crystallization process:
��Si��OC2H5 + Si��OH����Si��O��Si�� +C2H5OH
��Si��OH+ ��Si��OH����Si��O��Si�� +H2O
Nowadays, mesoporous materials have generally synthesized by conventional hydrothermal method, which has the disadvantages of time-consuming and energy-consuming. In addition, high temperature is in favor to form amorphous structure. Nevertheless, using microwave method could greatly shorten reaction time, make heating more homogeneous and promote nucleation of silicon source on the surface of template so that the crystallization rate increases and high-quality samples can be obtained [7-8]. On the other hand, grafting of organic functional group to the surface and channels of MMS can improve its surface properties, which makes it such a widely used material [9-10]. MMS can be organo-functionalized by stepwise synthesis and one-step synthesis technique. The superiority of one-step synthesis could be classified as follows: improved grafting extent [11], uniform surface coverage [12], better surface stability for organic functional groups [13], simple synthesis procedure, little reaction time and so on. In this work, one-pot method was reported to prepare chloropropyl-functionalized mesoporous MCM-41 (MCM-41-(CH2)3Cl) under microwave radiation. The obtained materials were further characterized by several spectroscopic techniques. So, it is a very valuable and can be extended to synthesize other related materials by our method.
2 Experimental
2.1 Preparation of MCM-41
Figure 1 shows synthesizing mechanism. 0.5 g CTAB was dissolved into 500 mL water, then 3.5 mL 2.0 mol/L NaOH was added and stirred for 10 min. 4 mL TEOS was added into the solution with stirring for 3 h, and the mixture was loaded into microwave oven and heated for 50 min under 120 W. The resultant solid was filtered and rinsed with deionized water. Then it was dried at 60 ��. After that, the dried samples were heated to 600 �� and held for 6 h for removing template (CTAB).
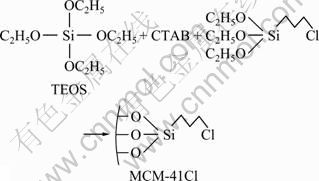
Fig. 1 Synthetic route of MCM-41-(CH2)3Cl by one-pot
2.2 Preparation of MCM-41-(CH2)3Cl
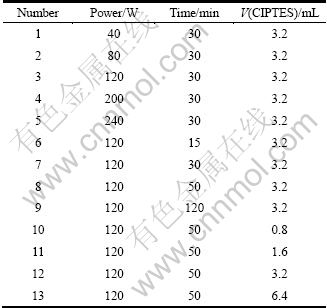
0.5 g CTAB was dissolved into 500 mL water, then 3.5 mL 2.0 mol/L NaOH was added and stirred for 10 min. 4 mL TEOS and 0.8 mL ClPTES were added into the solution with stirring for 3 h, and the mixture was loaded into microwave oven and heated for 50 min under 120 W. The resultant solid was filtered and rinsed with deionized water. After that, it was dried at 60 ��. Single factor analysis was used to determine the optimum synthesis conditions, and the detailed parameters are illustrated in Table 1. 1 g of dried samples was dissolved into 100 mL mixture of hydrochloric acid and alcohol (V/V=1:9) with stewing for 24 h at 80 ��. Then it was filtered and rinsed with alcohol. After that, it was dried at 60 �� and MCM-41-(CH2)3Cl was obtained.
2.3 Characterization
The structure and crystallinity of the materials were determined using X-ray diffraction (XRD, Rigaku D/max-IIIC), with Cu K�� radiation. N2 adsorption- desorption analysis isotherms and pore characterizations were obtained with a 3H-2000 automatic specific surface area analyzer. Fourier transform infrared spectroscopy (FT-IR) was conducted on Nicolet 5DX/550II. Thermo gravimetric analysis-differential thermal analysis (TGA-DTA) was performed on Shimadzu DTG-60H from room temperature to 1 000 ��, with a heating rate of 10 ��/min. The morphologies and microstructures were characterized with a scanning electron microscope (SEM, JSM-6490LV) and transmission electron microscope (TEM, Tecnai20).
Table 1 Detailed parameters of different synthetic methods
3 Results and discussion
3.1 XRD analysis of MCM-41-(CH2)3Cl synthesized at different levels of microwave power
Figure 2 shows the XRD patterns of MCM-41- (CH2)3Cl synthesized at different levels of microwave power. As observed, besides d110 and d200 diffraction peaks, there is a sharp d100 diffraction peak in the range of 2��=2��-4�� when the microwave power is 120 W, indicating a highly ordered mesostructure [14-15]. However, there is only a weak peak when the microwave power is less than 120 W. This is because that the crystallization process is insufficient due to inadequate heating of solution under low microwave power level, resulting in a less ordered structure. With increasing the microwave power, no peak is found. It is probable that high reflux temperature would destroy the ordered structure of MCM-41-(CH2)3Cl, leaving amorphous product. Thus, the best microwave power is 120 W.
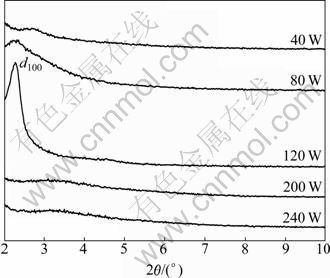
Fig. 2 XRD patterns of MCM-41-(CH2)3Cl at different levels of microwave power for 30 min
3.2 XRD analysis of MCM-41-(CH2)3Cl synthesized at different microwave time
Figure 3 shows the XRD patterns of MCM-41- (CH2)3Cl synthesized at different microwave time. It can be seen that all the samples display a d100 diffraction peak at 2��=2��-3��. Among four microwave time, the samples synthesized at 50 min and 120 min exhibit a relative sharp diffraction peak, indicating the ordered channel structure of the samples. Note that increasing microwave time could increase crystallization time, which is conducive to the formation of regular channel. When the reflux time is longer than 50 min, the time factor has little effect on preparation of MCM-41- (CH2)3Cl. Therefore, from the perspective of energy- saving, 50 min is the best preparation time. In comparison to conventional hydrothermal method, microwave synthesis of mesoporous MCM-41 not only saves much time but also obtains the final products with good structures.
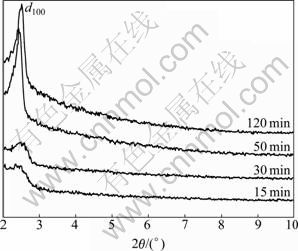
Fig. 3 XRD patterns of MCM-41-(CH2)3Cl at different micro- wave time under power of 120 W
3.3 XRD analysis of MCM-41-(CH2)3Cl synthesized from different dosages of ClPTES
As shown in Fig. 4, there is a noticeable difference in the order degree of MCM-41-(CH2)3Cl synthesized from different dosages of ClPTES. The pure MCM-41 has the best orderliness and remains a better orderliness with addition of 0.8 mL ClPTES. However, it is obviously seen that the orderliness tends to get worse with increasing the dosage of ClPTES. After adding 6.4 mL ClPTES, the material converts to amorphous state. The results show that the introduction of excessive 30% (molar fraction) ClPTES will perturb the crystallization process and it is difficult to form template micelles as well.
3.4 Low temperature N2 adsorption/desorption analysis
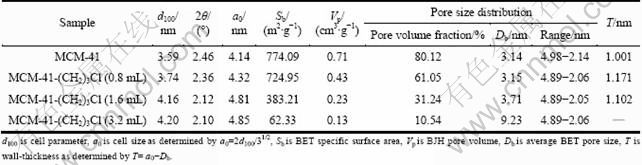
The adsorption/desorption isotherms of MCM-41 and MCM-41-(CH2)3Cl with different amounts of ClPTES are shown in Fig. 5. The MCM-41 samples with pure silicate and MCM-41-(CH2)3Cl prepared with 0.8 mL and 1.6 mL ClPTES show Langmuir IV adsorption isothermal, which is in agreement with the low-temperature N2 adsorption curve of typical mesoporous material. From data presented in Table 2, it is found that the synthesized materials can satisfy the two-dimensional hexagonal model. Further calculations show that the modified MCM-41 has a smaller pore volume but thicker wall, which results from the surface modification by means of ClPTES in combination with silanol groups of MCM-41. Nevertheless, MCM-41- (CH2)3Cl prepared with 3.2 mL ClPTES does not show a typical Langmuir IV adsorption isothermal. Relevant calculations show that the specific surface area is smaller and pore size is larger [16]. Moreover, the wall-thickness cannot be determined by two-dimensional hexagonal model. In other words, the synthesized material is not MCM-41-type mesoporous material. Thus, according to the above analyses, ClPTES with the dosage of 0.8-1.6 mL is more suitable for the preparation of MCM-41- (CH2)3Cl.
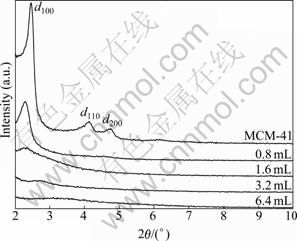
Fig. 4 XRD patterns of MCM-41 and MCM-41-(CH2)3Cl at different dosages of ClPTES
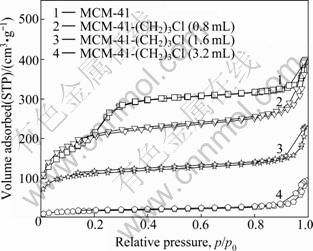
Fig. 5 N2 absorption-desorption curves of MCM-41 and MCM-41-(CH2)3Cl at different dosages of ClPTES
Table 2 Structural characteristics of materials
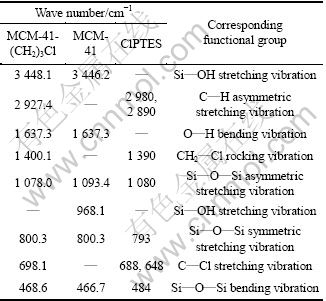
3.5 FT-IR analysis
Figure 6 shows the FTIR spectra of MCM-41- (CH2)3Cl (120 W, 50 min, ClPTES 0.8 mL), MCM-41 and ClPTES. It is found that the modified MCM-41-(CH2)3Cl exhibits the characteristic peaks of ClPTES, besides MCM-41. Moreover, the disappearance of Si��OH on surface of MCM-41-(CH2)3Cl molecular sieve indicates that the hydroxyl has been modified by chloropropyl. The concrete data are presented in Table 3.

Fig. 6 FT-IR spectra of MCM-41, ClPTES and MCM-41- (CH2)3Cl
3.6 TGA-DTA analysis
Figure 7 shows the TGA-DTA curves of MCM-41 and MCM-41-(CH2)3Cl prepared under optimum conditions. The relatively intensive mass loss of MCM-41 occurs when the temperature is lower than 100 ��, which is due to the loss of adsorbed water on the surface of molecular sieve. On the other hand, when the temperature increases above 100 ��, the mass loss is caused by the loss of adsorbed water held within channels. As dehydration process is an endothermic process, there is an obvious endothermic peak at 72.15 �� on the DTG curve. In the case of MCM-41- (CH2)3Cl, the mass loss at around 100 �� corresponds to the loss of adsorbed water, whereas an intensive mass loss at above 300 �� corresponds to the decomposition of chloropropyl from the surface-modified MCM-41. As shown on the DTG curve, there is an exothermic peak at 296.8 ��, suggesting that the decomposition process is an exothermic process, and MCM-41-(CH2)3Cl has been successfully modified by chloropropyl as well. Moreover, both materials show a certain amount of mass loss above 800 ��, due to the dehydration of Si��OH.
Table 3 Results of infrared spectroscopy
3.7 SEM and TEM analysis
Figure 8 shows the typical SEM images of MCM-41 and MCM-41-(CH2)3Cl, and Fig. 9 shows the typical TEM images of MCM-41 and MCM-41-(CH2)3Cl synthesized under the optimum condition. The morphology and microstructure of the products are investigated in detail through Fig. 8 and Fig. 9. The observations reveal that the dispersed MCM-41 particulates are regularly spherical in shape, as shown in Figs. 8(a) and (b). Further morphological characterization of an ordered two-dimensional channel hexagonal structure is shown in Fig. 9. Obviously, it is a typical mesoporous structure. And Fig. 9(b) has the similar structure with Fig. 9(a). This shows that the channel structure of MCM-41 could be maintained after being modified by ��(CH2)3Cl. Therefore, microwave radiation one-pot synthesis can be used as an effective method to synthesize MCM-41-(CH2)3Cl.
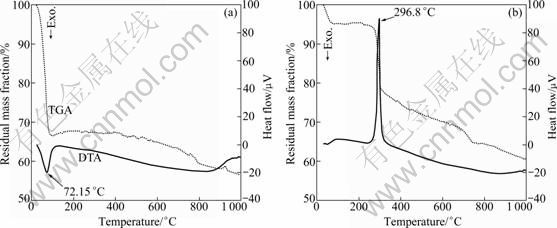
Fig. 7 TGA and DTA curves of MCM-41(a) and MCM-41-(CH2)3Cl (b)

Fig. 8 SEM images of MCM-41 (a) and MCM-41-(CH2)3Cl (b)

Fig. 9 TEM images of MCM-41 (a) and MCM-41-(CH2)3Cl (b)
4 Conclusions
1) MCM-41-(CH2)3Cl is successfully synthesized in alkaline medium by the microwave radiation one-pot method, using cetyltrimethy-lammoniumbromide (CTAB) as novel template, tetraethoxysilane (TEOS) as silica source and chloropropyltriethoxysilane (ClPTES) as the coupling agent.
2) Single factor analysis shows that the optimal microwave power is 120 W and the best microwave time is 50 min. The dosage of ClPTES ranging from 0.8 mL to 1.6 mL is favorable for the formation of highly ordered MCM-41-(CH2)3Cl mesostructure.
References
[1] KRESGE C T, LEONOWICZ M E, ROTH W J, VARTULI J C, BECK J S. Ordered mesoporous molecular sieves synthesized by a liquid�Ccrystal template mechanism [J]. Nature, 1992, 359(6397): 710-712.
[2] LIU Wei-qiao, SANG Tong-ming, LI Gong, WU Fei-ke, TONG Hui-juan, SUN Yu-han. Synthesis and catalytic performance of mesoporous material by two-step crystallization [J]. Journal of Inorganic Materials, 2010, 25(3): 272-278. (in Chinese)
[3] YANG H M, DENG Y H, DU C F. Synthesis and optical properties of mesoporous MCM-41 containing doped TiO2 nanoparticles [J]. Colloids and Surfaces A: Physicochem Eng Aspects, 2009, 339(1/2/3): 111-117.
[4] BURKEA A M, HANRAHAN J P, HEALY D A, SODEAU D A, HOLMES J R, MORRIS J D. Large pore bi-functionalised mesoporous silica for metal ion pollution treatment [J]. Journal of Hazardous Materials, 2009, 164(1): 229-234.
[5] CAO Yuan, BAI Ying-hao, XIA Zhi-ning. Synthesis and drug loading/release performance of mesoporous molecular sieve MCM-41 with tunable pore size [J]. Journal of the Chinese Ceramic Society, 2009, 37(9): 1570-1574. (in Chinese)
[6] CHRISTOPHE B, BARTLETT J, KONG L G, KIM F, LIN Hui-qiang, MICHAEL L, SANDRINE C, ALEXANDRA B, GERARARD C. Silia particles: A novel drug-delivery system[J]. Advanced Materials, 2004, 16(20): 1-8.
[7] WANG X G, KYLE S K, JERRY C C, CHENG S. Preparation of ordered large pore SBA-15 silica functionalized with aminopropyl groups through one-pot synthesis [J]. Chem Commun, 2004, 23: 2762-2763.
[8] CAO Yuan, LIU Bo-lin, HUANG Rui, XIA Zhi-ning, GE Song. Flash synthesis of flower-like ZnO nanostructures by microwave- induced combustion process [J]. Materials Letters, 2011, 65(2): 160- 163.
[9] ZENG W, QIAN X F, ZHANG Y B. Organic modified mesoporous MCM-41 through solvothermal process as drug delivery system [J]. Mater Res Bull, 2005, 40(5): 766-772.
[10] DOADRIO J C, SOUSA E M B, IZQUIERDO B I, DOADRIO A L, PEREZ P J, VALLET R M. Functionalization of mesoporous materials with long alkyl chains as a strategy for controlling drug delivery pattern [J]. Mater Chem, 2006, 16: 462-466.
[11] MORI Y, PINNAVAIA T J. Optimizing organic functionality in mesostructured silica: Direct assembly of mercaptopropyl groups in wormhole framework structures [J]. Chem Mater, 2001, 13(6): 2173- 2178.
[12] LIM M H, STEIN A. Comparative studies of grafting and direct syntheses of inorganic-organic hybrid mesoporous materials [J]. Chem Mater, 1999, 11(11): 3285-3295.
[13] KARIN M, THOMAS B, REINHARD X F. Synthesis of ordered mesoporous methacrylate hybrid systems: Hosts for molecular polymer composites [J]. Chem Mater, 1999, 11(3): 665-673.
[14] LIU Hui-ping, LU Guan-zhong, GUO Yun, WANG Yan-qin, GUO Yang-long. Synthesis of spherical-like Pt�CMCM-41 meso-materials with high catalytic performance for hydrogenation of nitrobenzene [J]. Journal of Colloid and Interface Science, 2010, 346(2): 486-493.
[15] LIU Xian-bin, SUN Hui, YANG Yan-hui. Rapid synthesis of highly ordered Si-MCM-41 [J]. Journal of Colloid and Interface Science, 2008, 319(1): 377-380.
[16] SHAHSAVAND A, NIKNAM S M, Reliable prediction of pore size distribution for nano-sized adsorbents with minimum information requirements [J]. Chemical Engineering Journal, 2011, 171(1): 69- 80.
(Edited by HE Yun-bin)
Foundation item: Project(207759096) supported by the National Natural Science Foundation of China; Project(20080440696) supported by the China Postdoctoral Science Foundation
Received date: 2011-06-17; Accepted date: 2011-09-29
Corresponding author: CAO Yuan, PhD, Associate professor; Tel: +86-23-65111748; E-mail: caoyuan108@vip.sina.com