Structure and electrochemical performance of spinel LiMn2O4 synthesized by mechanochemical process
LI Yun-jiao(�����), HONG Liang-shi(������), LI Hong-gui(����),ZHAO Zhong-wei(����ΰ), HUO Guang-sheng(������)
(School of Metallurgical Science and Engineering,Central South University, Changsha 410083, China)
Abstract: Spinel LiMn2O4 of cathode materials for lithium rechargeable batteries were synthesized by mechanochemical process, using Li2CO3 and different manganese compounds as EMD, Mn2O3 and Mn3O4. The influence of technological conditions on the phase structure and electrochemical performance of samples were systematically investigated by XRD, SEM, BET and constant current cyclic tests, respectively. It is found that the species of manganese compounds have strong effect on the structure and electrochemical performance of the final products. All the final products show spinel structure but their crystallization has feeble difference. And sample from Mn2O3 shows a typical octahedron shape. The initial discharge capacities of the samples are 131.44mA��h/g(made by EMD), 126.17mA��h/g(made by Mn2O3) and 126.34mA��h/g(made by Mn3O4), respectively. The product made by EMD shows well cyclic capability, and it can be used as a promising cathode material for 4V lithium batteries.
Key words: manganese source; lithium ion battery; LiMn2O4; mechanochemistry CLC number: TM912.9
Document code: A
1 INTRODUCTION
Lithium ion battery was used in many areas because of its high voltage, high energy density and long cycle life and so on. Spinel lithium manganese oxide was one of the most promising materials in term of its environmental benign, low cost, easy preparation and temperature safety. Main problem is the poor cycle life. As we all know that both the structure and electrochemical performance of material strongly depended on the preparing method and starting materials. Mechanochemical process is a new solid-state reaction at lower synthesis temperature and shorter reacting time compared with traditional solid-state reaction. It is used in many areas[1-6] because powder prepared by this method has a smaller particle size and well particle size distribution. People also had taken great interest on it in order to improve the properties of spinel lithium manganese oxides by using this synthesis method[7-12].Mn3O4 was one of the most important original sources of Mn-Zn ferrite which has the same spinel structure as LiMn2O4 while few people had taken study on it. Research of Zhang et al[13] showed that there existed Mn3O4 and Mn2O3 phase with lower valences state of Mn than LiMn2O4 in their powder. Ref.[11] also showed that Mn2O3 phase appeared during mechanochemical activatation and heat treatment in LiOH-MnO2 system. In this paper, spinel LiMn2O4 is synthesized by mechanochemical method and subsequent firing. The effects of different manganese compounds on the structure and electrochemical behavior of spinel lithium manganese oxide are studied.
2 EXPERIMENTAL
Lithium salts was Li2CO3(purity large than 99%), and manganese sources were EMD(purity large than 99%, Xiangtan Manganese Co.), Mn3O4(purity large than 99%, Changsha Research Institute of Mining and Metallurgy) and Mn2O3 prepared from MnO2 heated at 973K[10]. The raw materials�� mixture (molar ratio of Li to Mn 1/2) was milled for 1-3h using a planetary ball miller with stainless steel jars and balls(175r/min).The precursors were transferred into a corundum crucible for firing. Then the finial products A (made from Li2CO3+EMD), B (made from Li2CO3+Mn2O3) and C (made from Li2CO3+ Mn3O4) were gotten by thermal treatment of precursor at 973K for 6h. The formation process of LiMn2O4 can be represented by the redox equations as
2Li2CO3+8MnO2=4LiMn2O4+2CO2+O2(1)
2Li2CO3+4Mn2O3+O2=4LiMn2O4+2CO2(2)
6Li2CO3+8Mn3O4+5O2=12LiMn2O4+6CO2(3)
The structure of the final products were measured using a D/max-r A diffractometer with Cu K�� radiation(��=0.15418nm, scan range: 10��-70��) .The samples�� morphology were studied by scanning electron microscopy (SEM). The specific surface area was measured by using a BET apparatus (Novel 2000-Quantum). The cathode consisted of 85% LiMn2O4, 10% PTFE and 5% acetylene black, anode material was Li and separator was Celgard 2400 as well as electrolyte was 1.0mol/L LiPF6 solution in an ethylene carbonate-diethyl Carbonate (EC-DEC, 1/1) mixture. The model cells were assembled in a glove box filled with Ar. The electrochemistry performance was carried out between 3.2 and 4.3V at the rate of 0.1C using a LAND BT1-40 programming battery tester at room temperature.
3 RESULTS AND DISCUSSION
3.1 Effects of mechanochemistry on raw materials
Fig.1 shows the X-ray patterns of Li2CO3 and Mn3O4 mixtures without activated or activated for 1h and 3h. After milled for 1h, the diffraction peaks become broader and decrease markedly; some peaks (mainly of Li2CO3) are presented merely at the background level, because Li2CO3 is[CM(22]brickle, during ball-milling it is broken up and[CM)]turns from crystal to amorphous and at the same time, its reaction activity increases. It is connected with the decrease of particle size (see Fig.2) and lattice strain due to mechanochemical activation. During ball-milling, mechanical energy transformed into intrinsic energy of the materials. It is beneficial to form LiMn2O4. In order to investigate the effect of ball-milling time on the mixtures, another sample which was ball-milled for 3h were prepared. The diffraction peaks is slightly broader and lower than the 1h one. According to ball-mill-ing effect, experiment time and energy consump-
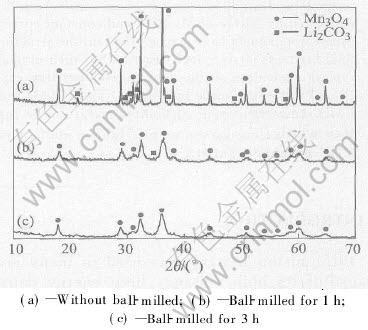
Fig.1 X-ray patterns of Li2CO3-Mn3O4 system

Fig.2 SEM images of Li2CO3 and Mn3O4
tion, all subsequent experiments were followed by milling for 1h.
Under the same condition high-energy ball-milling has different effect on different materials as shown in Fig.3. All X-ray patterns of samples changed obviously after ball-milled for 1h, but the change is quite different. The peaks decrease visibly especially for Mn3O4 because it has a well tetragonal spinel structure, and it is the best one among the three manganese compounds. While the effect of mechanical activation on Mn2O3 is not the same evident as that on the Mn3O4. EMD has a weaker crystallization (nearly amorphous)than Mn2O3 and Mn3O4, as a result, its diffraction curve is quite flat and the mechanochemical effect is invisible. Its diffraction peaks are the broadest but lowest after ball-milled for 1h among the three. At the same time, there are no LiMn2O4 phase appears during mechanochemical process. It is different from Ref.[8] because of too short ball-milling time and relative low rotary speed of planetary ball miller in this situation.
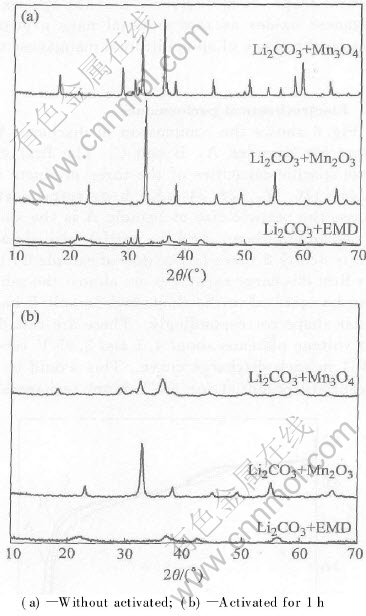
Fig.3 X-ray patterns of mixtures
Fig.4 shows X-ray patterns of Samples A, B and C. After heat treatment at 973K for 6h, the main diffraction peaks of cubic spinel LiMn2O4 phase were well developed and the diffraction peaks are quite narrow and symmetric, and no other peaks were found as shown in Fig.4(a). It indicates that all samples reveal well-defined cubic spinel structure and pure homogeneous. It also implies that the reaction between two phases is conducted completely. Sample B shows a more flat basic line because the precursor Li2CO3 and Mn2O3 have well crystallization than others after ball-milled for 1h. However, the widths at high angle are different and Sample B is comparatively sharper as shown in Fig.4(b). It seems that some peaks were split to some extent. Table 1 gives the comparison of diffraction degrees and crystal lattice parameters of the samples. The peaks of Sample B appear in a lower 2�� position than A and C, which indicated that the lattice parameter increased which was confirmed from Table 1. It was found from the result that the initial oxidation state of manganese in the raw material has an influence on the structure of LiMn2O4 synthesized by mechanochemical method.
3.2 Analysis of final samples
SEM images of samples are presented in Fig.5.
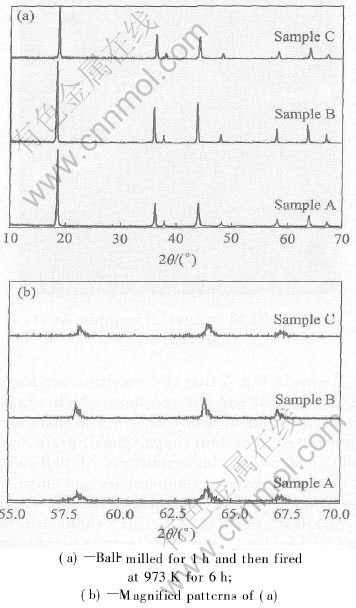
Fig.4 X-ray patterns of Samples A, B and C
Table 1 Comparison of diffraction degrees and crystal lattice parameters of samples
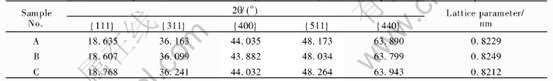
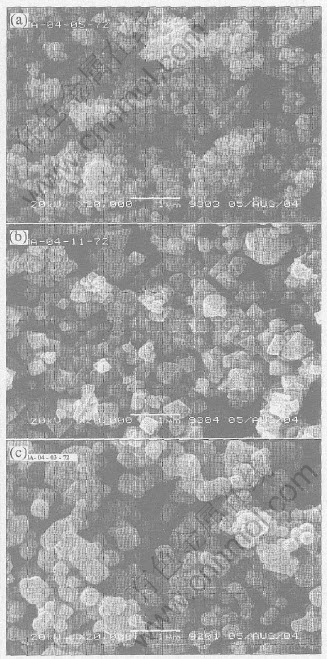
Fig.5 SEM images of Sample A(a), B(b) and C(c)
It is shown in Fig.5 that the resulting products are well crystallized and the powders which composed of small particles of less than 1��m in diameter are agglomerated in certain range. Small grain size can be obtained under the conditions of ball-milling, low thermal treatment temperature and short firing time. Among the three samples, Sample A shows the smallest particle size of submicron-meter range, it provides a new way to prepare nanometer particles.
The average specific areas of the three samples are 1.43m2/g(Sample A), 6.77m2/g(Sample B) and 3.56m2/g(Sample C), respectively. Sample B has a larger grain size than Samples A and C. The particle sizes of all the samples by the present method are smaller than that of synthesized by traditional solid-state reaction apparently[14]. As a matter of factor, particle size affects the electrochemistry performance greatly. The smaller the particle size, the easier the lithium insertion and extraction between the two phases of [Mn]2O4 and LiMn2O4. The research by Lu et al[15] showed that the particle size affected LiMn2O4��s cycle life greatly. It is also found from Fig.5 that Sample A shows a globular shape and a well homogeneous particle size distribution, while Sample B shows a typical octahedron shape and a smooth surface with obvious outline. Sample C has an approximate spherical shape. It is obviously that the species of manganese oxides as raw material have a strong effect on the shape of spinel lithium manganese oxide.
3.3 Electrochemical performance
Fig.6 shows the comparison of discharge behaviors for Samples A, B and C. The first discharge specific capacities of the three products are 131.44, 126.17, 126.34mA��h/g, respectively. Because the particle size of Sample A is the smallest one among them. And the particle size of Sample B is nearly 2 times larger that of sample C, but their first discharge capacities are almost the same. It can be ascribed to the shape and Sample B has a regular shape correspondingly. There are two distinct voltage plateaus about 4.1 and 3.95V versus Li/Li+ in each discharge curve. This would be an appropriate potential for 4V lithium rechargeable
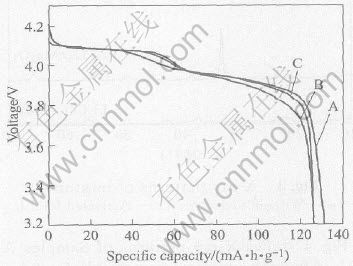
Fig.6 First discharge curves of samples
battery. The first plateau stands for the two-phase equilibrium between MnO2 and Li0.5Mn2O4, while the second flat represents phase equilibrium between Li0.5Mn2O4 and LiMn2O4[15]. But to Sample B, the 4.1V voltage plateau is quite short and the 3.95V voltage plateau isn��t as flat as Samples A and C. Further study would be carried in order to resolve the problem. Samples A and C would be a better cathode materials for 4V lithium ion rechargeable batteries than Sample B only from this aspect. The relationship between discharge capacity and cycle number of samples is illustrated in Fig.7. The average capacity fade of the three Samples A, B and C are about 0.24%, 0.67% and 1.1% per cycle respectively in the first 6 to 10 cycles. It is obvious that the battery performance of LiMn2O4 prepared from EMD is better than those of the other two. The excellent performance of the compound developed in this work is due to the small particle size achieved by mechanochemical method. According to the results, Sample A would be the best cathode material in terms of discharge capacity and cycle life.
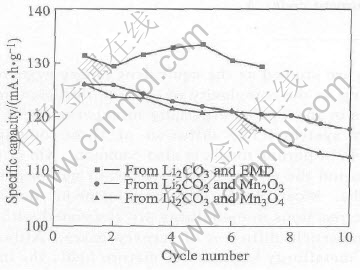
Fig.7 Cycle behavior of samples
4 CONCLUSIONS
1) All samples are phase pure homogeneous after thermal treatment of precursor at 993K for 6h.
2) The prepared powder by mechanochemical process consisted of small particle size of less than 1��m and the sample made from EMD has a submicron-meter grain. Samples started from EMD and Mn3O4 shows a spherical shape, while sample made from Mn2O3 is octahedron shape.
3) The final products made from EMD, Mn2O3 and Mn3O4 have a good specific capability. The first discharge capacities are 131.44, 126.17, 126.34mA��h/g, respectively. And the first sample exhibits a better cycle behavior.
4) Mechanochemical method is a promising way to synthesize cathode material for 4V lithium rechargeable batteries.
REFERENCES
[1]Wang J, Junmin X, Dongmei W, et al. Mechanochemical fabrication of single phase PMN of perovskite structure[J]. J Solid State Ionics, 1999, 124: 271-279.
[2]Pavlova S N, Sadykov V A, Zabolotngya G V, et al. The novel acid catalysts-framework zirconium phosphates: the bulk and surface structure[J]. J Molecular Catalysis A: Chemical, 2000, 158: 319-323.
[3]Balema V P, Pecharsky V K, Dennis K W. Solid state phase transformations in LiAlH4 during high-energy ball-milling[J]. J Alloys and Compounds, 2000, 313: 69-74.
[4]Tsuzuki T, McCormick P G. Synthesis of Cr2O3 nanoparticles by mechanochemical processing. Acta Mater, 2000, 48: 2795-2801.
[5]Menzel M, Sepelak V, Becker K D, et al. Mechanochemical reduction of nickel ferrite[J]. J Solid State Ionics, 2001, 141-142: 663-669.
[6]Dalvi A, Hahi K. Mechanochemically synthesized amorphous superionic systems for solid-state batteries[J]. J Solid State Ionics, 2002, 148: 431-436.
[7]Song G M, Wang Y J, Zhou Y, et al. Synthesis and electrochemical performance of LiCrxMn2-xO4 powders by mechanical activation and rotary heating[J]. J Power Sources, 2004, 128: 270-277.
[8]Jeong W T, Joo J H, Lee K S, et al. Improvement of electrode performances of spinel LiMn2O4 prepared by mechanical alloying and subsequent firing[J]. J Power Sources, 203, 119-121: 690-694.
[9]Soiron S, Roygier A, Aymard L, et al. Mechanochemical synthesis of Li-Mn-O spinels: positive electrode for lithium batteries[J]. J Power Sources, 2001, 97-98: 402-405.
[10]Kosova N V, Uvarov N F, Devyatkina E T, et al. Mechanochemical synthesis of LiMn2O4 cathode material for lithium battery[J]. Solid State Ionics, 2000, 135: 107-114.
[11]Kosava N V, Devyatkina E T, Kozlova S G, et al. Mechanochemical way for preparation of disordered lithium-manganese spinel compounds[J]. J Power Sources, 2001, 97-98: 406-411.
[12]HU Guo-rong, PENG Zhong-dong, YANG Jian-hong, et al. Synthesis of cathode material LiMn2O4 for lithium ion batteries by high-energy ball milling[J]. Trans Nonferrous Met Soc China, 2000, 10(6): 817-819.
[13]Zhang Y L, Shin H C, Dong J, et al. Nanostructures LiMn2O4 prepared by a glycine-nitrate process for lithium-ion batteries[J]. J Solid State Ionics, 2004, 17: 25-31.
[14]Cho J. Correlation of capacity fading of LiMn2O4 cathode material on 55�� cycling with their surface area measured by a methylene blue adsorption[J]. Solid State Ionics, 2001, 138: 267-271.
[15]Lu C H, Lin S W. Influence of the particle size on the electrochemical properties of lithium manganese oxides[J]. J Power Sources, 2001, 97-98: 458-460.
(Edited by LONG Huai-zhong)
Foundation item: Project (50174058) supported by the National Natural Science Foundation of China
Received date: 2004-09-29; Accepted date: 2004-11-17
Correspondence: LI Yun-jiao, Professor, PhD; Tel: +86-731-8830476; E-mail: yunjiaoli@263.net