
Effect of carbide formers on microstructure and thermal conductivity of diamond-Cu composites for heat sink materials
XIA Yang(夏 扬), SONG Yue-qing(宋月清), LIN Chen-guang(林晨光),
CUI Shun(崔 舜),FANG Zhen-zheng(方针正)
General Research Institute for Nonferrous Metals, Beijing 100088, China
Received 24 October 2008; accepted 24 March 2009
Abstract: Diamond-copper composites were prepared by powder metallurgy, in which the diamond particles were pre-coated by magnetic sputtering with copper alloy containing a small amount of carbide forming elements (including B, Cr, Ti, and Si). The influence of the carbide forming element additives on the microstructure and thermal conductivity of diamond composites was investigated. It is found that the composites fabricated with Cu-0.5B coated diamond particles has a relatively higher density and its thermal conductivity approaches 300 W/(m・K). Addition of 0.5%B improves the interfacial bonding and decreases thermal boundary resistance between diamond and Cu, while addition of 1%Cr makes the interfacial layer break away from diamond surface. The actual interfacial thermal conductivity of the composites with Cu-0.5B alloy coated on diamond is much higher than that of the Cu-1Cr layer, which suggests that the intrinsic thermal conductivity of the interfacial layer is an important factor for improving the thermal conductivity of the diamond composites.
Key words: diamond-Cu composite; carbide forming elements; thermal conductivity; thermal boundary resistance; interfacial layer
1 Introduction
Thermal considerations in electronic package design have become critically important because the growing power of devices generates more and more heat. Therefore, efficient thermal conductivity and good match of coefficient of thermal expansion(CTE) are required for heat sink materials[1]. Diamond composites are promising for such application. The thermal conductivity of synthetic diamonds of Ib type is estimated in the range of 1 500-2 000 W/(m・K)[2-3]. Cu is one of the most important materials for thermal and electronic applications. It possesses a good thermal conductivity of 400 W/(m・K) and a relative higher CTE of 16.32×10-6 K-1. The use of diamond as reinforcements in copper-based composites is considered attractive to meet the increasing demands for high performance heat sinks[4]. DINWIDDIE et al[5] have reported that thermal conductivity up to 600 W/(m・K) has been achieved by sintering diamond with Cu.
However, composites made by powder metallurgy have been demonstrated to feature weak interfacial bonding because pure liquid copper doesn’t wet diamond [6]. Interfacial thermal resistance becomes a problem as the weak bonding and the mismatch of CTE occur at the interface[7-8]. Experimental data have suggested that the interfacial gaps lowered the effective thermal conductivity of composites in the nickel-sodium borosilicate system[8].
In this work, the copper-based composite is prepared by sparking plasma sintering(SPS) with diamond surface modified by magnetic sputtering previously. Carbide forming elements including Cr, B, Si, Ti are added to study the effect on improving the adhesion and thermo-physical property of diamond-Cu composite. Contribution of interfacial layer to the thermal conductivity of the composites is discussed.
2 Experimental
The diamond particles were pre-coated with Cu alloys by magnetic sputtering. In order to study bonding and thermal boundary resistance in detail, four types of carbide formers were added in Cu. The compositions were Cu-4%Ti, Cu-3%Si, Cu-1%Cr and Cu-0.5%B (mass fraction), respectively.
Commercially available diamonds of 38-44, 124-150, 178-200 μm were used. Volume fractions of diamond in the composites varied from 60% to 80%. In the magnetic sputtering, diamond powders were put in a plate and agitated. By controlling frequency and amplitude of vibration, uniform films were sputtered on each face of the particles, with current of 1 A, voltage of 200 V, vacuum of 1.0×10-2 Pa and sputtering time of 21 min. Finally, the sputtered powders were reduced in H2 atmosphere for 5 h and sealed in vacuum.
The pre-treated powder was mixed with a designated amount of Cu powder, then was heated to 1 000 ℃ and held for 5 min in SPS sintering to provide enough time for the carbide formation.
Based on Archimedes’ law, the bulk density of the composite was measured. The thickness of sputtered layers was tested by X-ray diffraction analysis and gravimetric method as well. Thermal conductivities of the composites were obtained by laser flash method, and thermal diffusion coefficients were tested by LFA427 laser equipment from Germany. Fracture surfaces of the composites were analyzed by scanning electron microscopy using Hitachi S-3500N equipment from Japan. Energy dispersive spectroscopy(EDS) was used to analyze the composition.
3 Results and discussion
3.1 Wetting characteristic
As the surface area of diamond particles is limited, high purity graphite flake is used to get the wetting information of Cu-X alloys on carbon material. The chemical characteristics of diamond and graphite are quite similar, and wetting angles between Cu-X alloys and graphite are derived by micro projection method, as shown in Table 1.
Table 1 Contact angles between Cu-X alloy and graphite at 1 100 ℃

According to Table 1, contact angles between graphite and Cu are reduced by adding Cr and B elements into Cu, and 1%Cr addition obviously reduces the value to about 54? at 1 100 ℃ in 2.0×10-2 Pa vacuum. By observing the interface, it is found that the bonding strength of Cu-1Cr alloy is stronger than that of Cu-0.5B alloys,which is caused by the strong tendency of the carbide reaction at interfaces[9].
3.2 Thickness of sputtering layer
According to X-ray diffraction analysis of thin films, the thicknesses of Cu-1Cr, Cu-0.5B and Cu-3Si layer on diamond particles are about 1.85, 1.90 and 1.79 μm, respectively. Small angle diffraction is beneficial to diminishing testing errors. In addition, gravimetric method is used to estimate the thickness by accurately compare the mass of diamond powders before and after sputtering. The thickness of layers is calculated on the assumption that each diamond particle is a sphere of the same size and values of 2.42, 2.12 and 2.31 μm are obtained, respectively. As the layer is too thin and some particles lose during sputtering, the calculated results are higher than the X-ray results.
3.3 Microstructures of composites
The relative densities of the CuCr-diamond, CuB- diamond, CuTi-diamond and CuSi-diamond composites are 98.9%, 99.3%, 96.7% and 97.1%, respectively.
Morphologies of the diamond are shown in Fig.1. Surfaces of original (Fig.1(a)) and sputtered (Fig.1(b)) diamond are smooth. EDS area mapping on sputtered surface indicates that the layer mainly consists of Cu. Due to the minor quantity of the additives and sensitivity of the instrument, B and Cr elements are not detected. After sintering, pits with inverted pyramid shapes appear on the {100} surface of a few integrated and highly symmetrical diamond particles released from Cu-0.5B- coated diamond composites, as seen in Figs.1(c) and (d). It can be explained by the different bonding arrangements on {100} and {111} planes of diamond. In terms of the unit cell model of diamond, the {100} surface atoms are two-fold bonded to the bulk atoms while the {111} planes are three-fold bonded to the atom. It is obviously easier to rupture the two bonds than three bonds when the molten Cu alloys cover on diamond surface during sintering. The {100} surface atoms protect the {111} ones from being dissolved by consecutively destroying the two bonds and removing. Then, the atoms on {100} surfaces dissolve layer by layer and pits come out. This also indicates the different reactivity of diamond surfaces. The {100} surfaces are easier to dissolve and participate in the formation of interfacial carbides[10-12].

Fig.1 Microstructures of diamond particles: (a) Original diamond particles; (b) Particles sputtered with Cu-1Cr alloy; (c) Particles released from CuB-diamond composite; (d) Converted pyramids appearing on {100} surface
More interfacial information in Fig.2 is helpful to illustrating the interfacial adhesion of Cu-X-coated diamond composites. Figs.2(a) and (b) show the morphologies of the composite fabricated by pure diamond and copper. Obvious cracks are observed and only mechanical adhesions are formed in diamond-Cu composite. If adding minor B element, interfacial bonding is distinctly improved and diamond is tightly surrounded by matrix, seen in Figs.2(c) and (d). There is not any obvious macro-crack and defect appearing at the interfaces. In Fig.2(f), the cracks appearing in Cu-Si- coated diamond composite suggest that addition of Si cannot enhance interfacial bonding. In Fig.2(g), copper is observed to penetrate into the spaces between diamond particles and cohesions seem good. But a great deal of blisters turn out on diamond surface after SPS, as shown in Fig.2(h). This is possibly due to the reaction of the C atoms with the released gas that is absorbed on diamond surface before sputtering or in the fast sintering process. In addition, CTE mismatch between the Cu-1%Cr metallic coatings and diamond makes it possible to release stress in the regions of relatively weaker adhesion and thus bubbles form as well.

Fig.2 Morphologies of diamond composites: (a) and (b) Diamond-Cu; (c) and (d) Diamond-CuB; (e) and (f) Diamond-CuSi; (g) and (h) Diamond-CuCr
3.4 Thermal conductivity of composites
Thermal conductivity of diamond composites in this work consists of resistance of the diamond/carbide interface, the resistance of the carbide and the resistance of carbide/metal interface[13-14]. Heat conduction of the diamond/carbide interface is discussed in detail.
By taking thermal resistance and particle size into consideration, effective thermal conductivity of the composite can be expressed by Eq.(1)[16]:
 (1)
(1)
where Keff, Km, and Kp are the effective thermal conductivities of the composite, metal matrix, and particles respectively; φp is the volume fraction of reinforcements, a is the diameter of the reinforcement and hc is the boundary conductivity.
Thermo-physical properties of carbides are listed in Table 2[15]. According to Eq.(1), theoretical predictions of composites with different carbide interfaces are calculated, as shown in Fig.3, in which particle size is 200 μm and volume fraction of diamond is 80%. It is found that thermal conductivity of diamond composites increases with the intrinsic thermal conductivity of the transition layers if perfect interfacial bondings are achieved. Since thermal conductivity of the transition layers is quite low, it is impossible to advance the heat conduction of the composites only by increasing the thermal conductivity of the reinforcements. Only if the thermal conductivity of the transition layer excesses a certain value, can thermal conductivity of composites be enhanced. Thus, it is instructive to discuss the influence of carbide layers on interfacial thermal conductivity.
Table 2 Thermo-physical properties of carbides
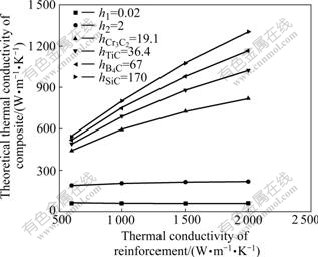
Fig.3 Theoretical thermal conductivity of composites consisting of different interfacial layer and reinforcement
Fig.4 shows the measured thermal conductivity of diamond composites, which is much lower than theoretical value. It is assumed that the thermal conductivities of diamond and copper are 1 500 and 400 W/(m・K), respectively, and the thermal conductivity of diamond is independent of the particle size. The interfacial conductivity calculated using Eq.(1) is also summarized in Fig.4. In the diamond-pure Cu composites, the interfacial conductivity is less than 1.07×106 W/(m2・K). But Cr and B appear to be the good candidates for diamond-Cu composites[17], with which the interfacial conductivities increase to 3.92×106 and 7.27×106 W/(m2・K), respectively. This is due to the low substitutional or interstitial solubility of the two elements in Cu and the medium formation heat at sintering temperature[4]. Carbide forming reaction may not take place in Cu-3Si-diamond system at the sintering temperature[18-19]. Micro-cracks still exist and thus result in the great decline in thermal conductivity of diamond composites. In terms of effective medium theory(EMT), we can deduce that in the prepared diamond-Cu composites, Cu plays key role in effective heat conduction. The large interfacial effect makes the highly conductive particles act as voids or defects. Although addition of 0.5%B and 1%Cr can deduce interfacial defects to a certain extent, great interfacial boundary resistance still exists and destroys the expected thermal property.
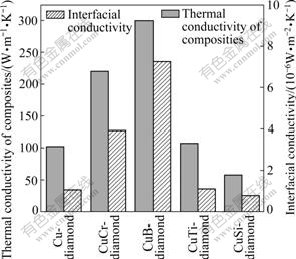
Fig.4 Measured thermal conductivity of composites
Thickness of the intermediate reaction layer should be properly controlled, for CTE mismatch and inner-stress release in such thick carbide layers tend to impair both the bonding characteristics and heat conductive efficiency if the diamond is completely covered by carbide layers. The optimal parameters should offer the reaction layer a growing rate to maintain a minimum thermal boundary resistance in diamond/ matrix interface. GUI et al[19] have demonstrated that a 100 nm-thick Cr3C2 helps to obtain thermal conductivity of 640 W/(m・K). Therefore, the critical issue is to optimize composition and reaction rate of carbide layer to achieve an ideal structure, in which diamond skeleton is the main conductive path and Cu alloy binders are closely adhered to the particles.
4 Conclusions
1) Diamond-Cu composites are prepared by SPS, with four Cu alloys sputtered on diamond particles previously.
2) Cu-0.5B-coated diamond composite has a highest relative density of 99.3% and its thermal conductivity reaches 300 W/(m・K).
3) Carbide forming elements including B and Cr have effect on the microstructure and thermal conductivity of diamond-Cu composites. The addition of 0.5%B renders the atoms in {100} planes of the diamond easier to dissolve and participate in interfacial carbide reaction, and thus greatly improves the interfacial bonding and conductivity between diamond and matrix. While addition of 1%Cr produces a great deal of blisters on sintered diamond surface. Gaps at the interface limit the improvement of interfacial bonding and conductivity.
4) Intrinsic thermal conductivity of interfacial layer accounts for the differences between thermal conductivity of the composites with nearly the same compositions. The greater the intrinsic thermal conductivity is, the higher the theoretically predicted values are.
References
[1] WILDNER H, KNIPPSCHEER S, LANDGRAF J,BOLLINA R. Manufacturing and applications of diamond composites for thermal management [R]. Dübendorf: High performance diamond composites for TM, 2006.
[2] SCHUBERT T, TRINDADE B, WEIβG?RBER T. Interfacial design of Cu-based composites prepared by powder metallurgy for heat sink applications [J]. Mater Sci Eng A, 2008, 475: 39-44
[3] DAVIS L C, ARTZ B E. Thermal conductivity of metal-matrix composites [J]. J Appl Phys, 1995, 77(10): 4954-4960.
[4] YOSHIDA K, MORIGAMi H. Thermal properties of diamond/ copper composites materials [J]. Microelectronics Reliability, 2004, 44: 303-308.
[5] DINWIDDIE R B, BEECHER S C, ARYA P V. Thermal conductivity of diamond composites [J]. Thermal Conductivity, 1994, 22: 468-476.
[6] SCOTT O M, NICHOLAS M. The wetting and bonding of diamonds by copper-base binary alloys [J]. J Mater Sci, 1975, 10: 1833-1840.
[7] POWELL B R, YOUNGBLOOD G E, HASSELMANN D P H, BENTSEN L D. Effect of thermal expansion mismatch on the thermal diffusivity of glass-Ni composites [J]. J Am Cerm Soc, 1990, 63(9/10): 581-586.
[8] HASSELMANN D P H, JOHNSON L F, SYED R, TAYLOR M P, CHYUNGI K. Heat conduction characteristics of a carbon fiber-reinforced lithium-alumino-silicate glass-ceramic [J]. J Mater Sci, 1987, 22: 701-709.
[9] SCHUBERT T, CIUPI?SKI ?, ZIELI?SKI W, MICHALSKI A, WEIβG?RBER T, KIEBACK B. Interfacial characterization of Cu/diamond composites prepared by powder metallurgy for heat sink application [J]. Scripta Materialia, 2008, 58: 263-266.
[10] FLAQUER J, RíOS A, MARTIN-MEIZISO A, NOGALES S. Effect of diamond shapes and associated thermal boundary resistance on the thermal conductivity of diamond-based composites [J]. Computational Materials Science, 2007, 41(2): 156-163.
[11] KLEINER S, KHALID F A, RUCH P W, BEFFORT O. Effect of diamond crystallographic orientation on dissolution and carbide formation in contact with liquid aluminum [J]. Scripta Materialia, 2006, 55: 291-294.
[12] HU Shao-chung, CHANG Kuen-liang, SUNG James. Ultrahigh pressure sintering of polycrystalline diamond cubes [C]// LI Zhi-hong, ZHAO Bo, YI Yun-lei, LIU Yue-lan. Proceedings of the 5th Zhengzhou International Super-Hard Materials & Related Products Conference. Zhengzhou: China Machine Press, 2008: 70-76.
[13] WEBER L, TAVANGAR R. On the influence of active element content on the thermal conductivity and thermal expansion of Cu-X(X=Cr, B) diamond composites [J]. Scripta Materialia, 2007, 57: 988-991.
[14] DUNN M, TAYA M. The effective thermal conductivity of composites with coated reinforcement and the application to imperfect interfaces [J]. J Apply Phys, 1993, 73: 1711-1722.
[15] САМСОНОВ Г В. Analysis of refractory compound [M]. HUANG Jing-hua. Shanghai: Shanghai Science and Technology Press, 1965: 126-128. (in Chinese)
[16] HASSELMANN D P H, JOHNSON L F. Effective thermal conductivity of composites with interfacial thermal barrier resistance [J]. Journal of Composite Materials, 1987, 21(6): 508-515.
[17] TORQUATO S, RINTOUL D M. Effect of the interface on the properties of composite media [J]. Phys Rev Lett, 1995, 75: 4067-4070.
[18] ZHOU Y C, LONG S G, LIU Y W. Thermal failure mechanism and failure threshold of SiC particle reinforced metal matrix composites induced by laser beam [J]. Mechanics of Materials, 2003, 35(10): 1003-1020. (in Anglais)
[19] GUI Man-chang, KANG Suk-bong, EUH Kwang-jun. Thermal conductivity of Al-SiCp composites by plasma spraying [J]. Scripta Materialia, 2005, 52: 51-56.
Foundation item: Project(82129) supported by the Innovative Foundation of Science and Technology of General Research Institute of Nonferrous Metals, China
Corresponding author: XIA Yang; Tel: +86-10-82241207; E-mail: tjuxy82@yahoo.com.cn
DOI: 10.1016/S1003-6326(08)60422-7
(Edited by YANG Bing)