
Influence of NaCl on dechromization of Cu-Cr-Al alloy in H3PO4 solution
LIU Yu-wen(������)1, 2, LI Ya-li(������)3, ZHANG Rui-jun(�����)1, WANG Hai-yan(������)2,
PENG Gui-rong(������)1, LIU Jian-hua(������)1
1. State Key Laboratory of Metastable Materials Science and Technology,Yanshan University, Qinhuangdao 066004, China;
2. Department of Basic Medical, Tangshan Vocational Technology College, Tangshan 063000, China;
3. Department of Environmental and Chemical Engineering, Yanshan University, Qinhuangdao 066004, China
Received 13 October 2008; accepted 2 February 2009
Abstract: The dechromization of Cu-Cr-Al alloys was investigated by a static immersing corrosion test. The influence of NaCl on the dechromization was analyzed by electrochemical test, scanning electron microscopy, optical microscopy and X-ray fluorescence spectroscopy. The results show that the dechromization of Cu-Cr-Al alloys will take place in a H3PO4 solution of 11.32 mol/L at 55 ��. And the corrosion process occurs initially at the interface between Cu and Cr phase, and then extends inward Cr-phase inside. The addition of NaCl into H3PO4 solutions can shorten the dechromization incubation time, decrease the dechromization temperature and concentration of H3PO4 solution, and increase the tendency of dechromization. But, the NaCl scarcely has an effect on the form and structure of chromium depleted-layer.
Key words: dechromization; Cu-Cr-Al alloy; H3PO4 solution; NaCl
1 Introduction
Cu-Cr alloy is widely applied in functional materials and parts with high conductivity and high strength due to its high intensity and excellent heat and electrical conductivity[1-4]. With the extending application fields of Cu-Cr alloys, Cu-Cr alloys are potential in use as corrosion-resistant parts applied in environmental and chemical engineering. The research on their corrosion behavior in solutions has been attracting great attention of many researchers[5-7]. Recently, we studied the corrosion behavior of a Cu-Cr alloy in H3PO4 solution and concluded that it is dechromization phenomenon that would occur, not decopperation if the temperature and concentration of H3PO4 solutions are compatible. Some results about dealloying of Cu-Cr alloys have been reported[8-10], which mainly contained dealloying conditions and effect factors. Furthermore, they concentrated on HCl and H2SO4 solutions. H3PO4 solution is a common kind of acid solutions used in industry. Adding NaCl into the solutions can deteriorate corrosion-resistant properties of Cu alloys[11-12]. Many research papers on Cu alloys in H3PO4 solutions have been published. However, study on the dechromization corrosion of Cu-Cr alloy in H3PO4 solutions and the effect of NaCl in the solution on corrosion behavior has scarcely been reported. The addition of Al element into Cu-Cr alloys could increase the strength and anti-oxidation of alloys[13]. Therefore, in this work, the dechromization corrosion behavior of Cu-Cr-Al alloy, and the effects of NaCl in H3PO4 solutions on the dechromization incubation time, and the microstructure of dechromization layer were discussed.
2 Experimental
Cu-Cr-Al alloy containing 50.12%Cu, 48.02%Cr, 1.07%Al, 0.32%Si, 0.45%Ni and 0.03% others (mass fraction) was used to prepare samples with the size of 5 mm��5 mm��10 mm. These samples were polished with silicon carbide paper up to 1200# grit, washed by water, dried and then immersed into H3PO4 solutions with different NaCl contents for dechromization experiments. Some parameters of dechromization of the Cu-Cr-Al alloy were measured, such as the incubation time of dechromization (the duration from the beginning of the sample immersed into the solution to air bubble coming into being), the temperature and the lowest concentration of the H3PO4 solutions. Tests were carried out in a HHS-12 type electric thermostatic water container.
The anodic polarization curves of the alloy in H3PO4 solutions were traced by CH1660A electrochemical instrument with a saturated calomel electrode as the reference electrode and a platinum piece as the counter electrode at a scan rate of 0.5 mV/s. The microstructure and characteristics of the dechromization layer were observed by Neophot21 optical microscope, KYKY-2800 SEM (with EDS), and XP-2TM stylus profilometer. In addition, the compositions of Cu-Cr-Al alloy surface before and after corrosion were analyzed by ADVANTPXP-381 X-ray fluorescence spectrometer.
3 Results and discussion
3.1 Dechromization of Cu-Cr-Al alloys
The original microstructure of Cu-Cr-Al alloy shown in Fig.1 is constituted by a bright white Cr phase and Cu solid state solution (named Cu phase). As shown in Fig.1, the irregular shaped grains of Cr phase scatter in the Cu phase substrate.

Fig.1 Original microstructure of Cu-Cr-Al alloy
One of the samples was immersed in 50 mL H3PO4 solution of 11.32 mol/L, at 55 ��, and others were immersed in the similar H3PO4 solutions but containing 0.15 g NaCl. It could be observed that the surfaces of the two kinds of samples become pure copper color. When the solution turns into blackish green, plenty of gases appear. The elements on the sample surface were analyzed by X-ray fluorescence spectrometer. Experimental results (Table 1) show that the majority of elements is Cu and the content of Cr decreases obviously and the content of Al is reduced a little. The microstructure of the surface layer of samples is mainly Cu matrix phase (illustrated in Fig.2), which indicates that the corrosion characteristics results from the dechromization. SEM analyses indicate that the dechromization of Cu-Cr-Al alloy occurs initially at the interface between Cu phase and Cr phase, and then extends inward the Cr-phase inside until Cr phase dissolves entirely (illustrated in Fig.3). These results could also be confirmed by EDS analysis. Yet NaCl scarcely has an effect on the microstructure of dechromization layer.
Table1 Compositions of surface of samples immersed in H3PO4 solutions (mass fraction,%)
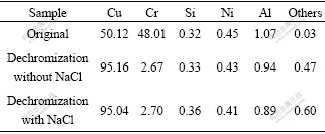
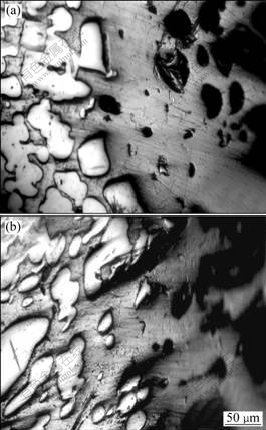
Fig.2 Microstructures of dechromized layer of Cu-Cr-Al alloy under different conditions: (a) Without addition of NaCl; (b) With addition of NaCl
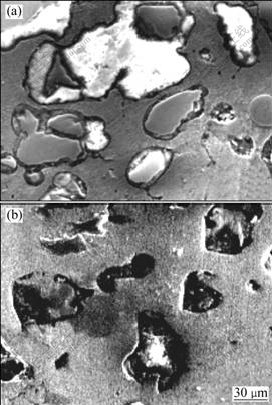
Fig.3 SEM images of dechromized zone of Cu-Cr-Al alloy under different conditions: (a) Without addition of NaCl; (b) With addition of NaCl
3.2 Influence of NaCl on incubation time of dechromization of Cu-Cr-Al alloy
During the experiments, it can be observed that the dechromization occurs after Cu-Cr-Al alloy is immersed in H3PO4 solutions for some time (incubation time of dechromization). Fig.4 shows the relationship between the content of NaCl and the incubation time of dechromization in 20 mL H3PO4 solution of 10.27 mol/L at 60 ��. It can be seen that NaCl can reduce incubation time of dechromization and when the mass of NaCl ranges from 0.10 to 0.25 g, the incubation time decreases with increasing the content of NaCl.
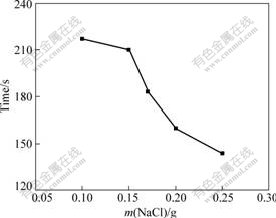
Fig.4 Relationship between content of NaCl and incubation time for dechromization of samples in 10.27 mol/L H3PO4 solution at 60 ��
3.3 Influence of NaCl on concentration and temperature for dechromization of Cu-Cr-Al alloy
Fig.5 shows the relationship between the content of NaCl and the concentration and temperature of H3PO4 solutions when the dechromization happens on the surface of the samples. It can be found that when the temperature (or concentration) is a constant, NaCl can reduce the concentration (or temperature) of H3PO4 solutions for dechromization of the Cu-Cr-Al alloy. The experimental results show that the more the content of NaCl in the solutions is, the lower the concentration or the temperature of H3PO4 solutions is needed for the dechromization. In a word, addition of NaCl in H3PO4 solutions could increase the tendency of dechromization of the Cu-Cr-Al alloy.
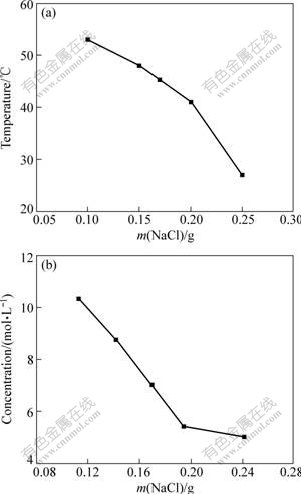
Fig.5 Relationship between content of NaCl and concentration and temperature of H3PO4 solutions in dechromization: (a) NaCl content vs temperature(10.27 mol/L, 20 mL); (b) NaCl content vs concentration of H3PO4 (50 ��, 20 mL)
3.4 Influence of NaCl on corrosion potential and corrosion current of Cu-Cr-Al alloy
In order to further discuss the effect of NaCl on the corrosion behavior, the surface electrochemical polarization and roughness of Cu-Cr-Al alloy in H3PO4 solutions were tested. Fig.6 shows the polarization curves. It can be seen that when NaCl is added in the H3PO4 solution, the corrosion potential becomes more negative and the corrosion current density increases. The results from X-ray fluorescence spectroscopy (Fig.7) show that the surface of sample after being immersed in H3PO4 solutions with NaCl changes much coarser compared with that of sample after being immersed in H3PO4 solutions without NaCl. It can be concluded that NaCl could increase the corrosion sensibility of the alloy in H3PO4 solutions.
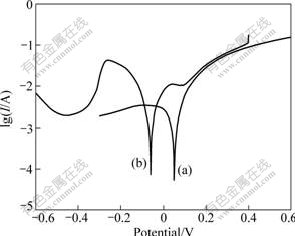
Fig.6 Polarization curves of Cu-Cr-Al alloy in 50 mL H2SO4 solution of 1.60 mol/L at 20 ��: (a) Without NaCl; (b) With addition of 20 g NaCl
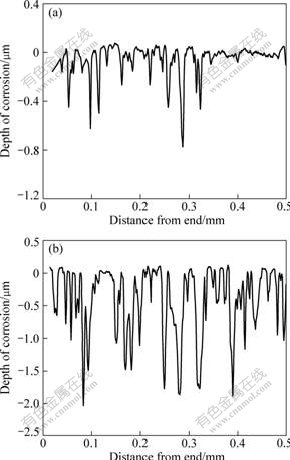
Fig.7 Roughness of sample surface after being immersed in 50 mL H3PO4 solution of 1.60 mol/L at 20 �� for 5 h: (a) Without NaCl; (b) With addition of 20 g NaCl
3.5 Discussion
Cu-Cr-Al alloy is composed of Cu phase and Cr phase, in which the two phases are almost undissolved in each other[14]. It can be seen from the plots of the potential pH that in the acid region, the electrode potential of Cu metal is higher than that of Cr metal, and thus the ionization of Cu metal is more difficult than Cr metal. It can be confirmed that Cu and Cr in the H3PO4 solutions compose a corrosion battery with Cr being the anode and Cu being the cathode, and the electrochemical reactions of the Cu-Cr-Al alloy in H3PO4 solutions can be described simply as follows:
Anode: Cr��Cr3++3e-
Cathode: H++2e-��H2
It would be expected that when Cu-Cr-Al alloy is immersed in H3PO4 solutions under a certain conditions, the corrosion of Cr phase is much easier than Cu phase and the dechromization occurs easily. However, it is well known that the Cr exposed in the air is very easy to be oxidized, and a steady passivation film formed on the surface could protect Cr phase from oxidization. So, in fact, the dechromization of Cu-Cr-Al alloys is more difficult. Therefore, the dechromization occurs after the Cu-Cr-Al alloy is immersed in H3PO4 solutions for some time (incubation time) until the passivation film on the Cr surface is dissolved. As soon as the dechromization occurs, Cr3+ created from the Cr phase surface goes into the solution and thus the surface is almost covered by copper element.
Generally, an acid solution could dissolve the passivation film on metal surface. The higher the concentration and the temperature of acid solutions are, the faster the passivation film dissolves. For Cl- has a smaller radius and strong penetrability[15-16], the addition of NaCl in H3PO4 solutions can provide Cl-, which could accelerate deterioration of the passivation film on the Cr surface. In addition, Cl- could react with Cr3+ to form complex compounds, such as ([Cr(H2O)5Cl]Cl2?H2O, [Cr(H2O)4Cl2]Cl?2H2O and [Cr(H2O)6]Cl2?H2O). So, the addition of NaCl in H3PO4 solutions could promote anodic dissolving process, and thus accelerate dechromization of the Cu-Cr-Al alloy. As a result, the incubation time of dechromization is shortened, the temperature and concentration of H3PO4 solutions for dechromization decrease, and the tendency of dechromization increases. Besides, Al dissolved completely in the Cu substrate has a little effect on dechromization corrosion.
4 Conclusions
1) The addition of NaCl in H3PO4 solutions could shorten the dechromization incubation time of Cu-Cr-Al alloy, decrease the temperature and concentration of H3PO4 solutions for the Cu-Cr-Al alloy to dechromize, and increase the tendency of dechromization, but it has a little effect on the microstructure of chromium depleted-layer.
2) Cl- in H3PO4 solutions could facilitate the dechromization of the Cu-Cr-Al alloy. So, avoiding Cl- into the H3PO4 solutions is benefit for protecting the Cu-Cr-Al alloy from the dechromization corrosion.
References
[1] WANG You-hong, SONG Xiao-ping, SUN Zhan-bo, ZHOU Xuan, GUO Juan. Effects of Ti addition on microstructures of melt-spun CuCr ribbons [J]. Transactions of Nonferrous Metals Society of China, 2007,17(1): 72-76.
[2] SPAIC S. Microstructure and properties of sintered Cu-25Cr alloy [J]. Material Science and Technology, 1989, 5(11): 1069-1073.
[3] MORRIS D G, MORRIS M A. Rapid solidification and mechanical alloying techniques applied to CuCr alloys [J]. Mater Sci Eng A, 1988, 104: 201-213.
[4] HE Wen-xiong, WANG Er-de, CHEN Hui, YU Yang, LIU Jing-lei. Effects of deformation on microstructures and properties of submicron crystalline Cu-5%Cr alloy [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(2): 348-388.
[5] ZHANG Y N, ZI J L, ZHENG M S. Corrosion behavior of copper with minor alloying addition in chloride solution [J]. Journal of Alloys and Compounds,2008, 462(1/2): 240-243.
[6] CAO Zhong-qiu, BIAN Jing, XUE Rong, LIU Wei-hua. Electrochemical corrosion behavior of Cu-40Ni-20Cr alloys with different grain sizes in solutions containing chloride ions [J]. Transactions of Nonferrous Metals Society of China, 2007, 17(6): 1236-1241.
[7] YU Cui-yan, CHANG Li-min, ZHOU Li-min, LIN Hong-yan. Corrosion behavior of a CuCrNiAl alloy in HCl solutions [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(1): 229-233.
[8] LIU Jian-hua, ZHANG Run-jun, YANG Jing-ru, SUN Qi-na. Corrosion behavior of CuNiCrAl alloy in H2SO4 solutions [J]. Rare Metal Materials and Engineering, 2007, 36(S): 295-298. (in Chinese)
[9] XU Tao, GAO Hua-wei, LIU Jian-hua. Effect of ultrasonic vibration on the dechromization corrosion of a CuCr alloy in HCl solution [J]. Rare Metals, 2007, 26(4): 398-402.
[10] YU Cui-yan, LIU Jian-hua, ZHOU Li-min. The influence of surface roughness and deformation on dechromization of Cu-Cr alloy [J]. Rare Metals, 2006, 25(2): 166-167.
[11] BARBUCCI A, FARNE G, MATTEAZZI R, RICCIERI R, CERISOLA G. Corrosion behaviour of nanocrystalline Cu90Ni10 alloy in neutral solution containing chlorides [J]. Corrosion Science, 1998, 41(3): 463-475.
[12] MILOSEV I, METIKOS-HUKOVIC M. The behaviour of Cu-xNi (x=10 to 40wt%) alloys in alkaline solutions containing chloride ions [J]. Electrochemical Acta, 1997, 42(10): 1537-1548.
[13] WANG Zhu-tang. Cu Alloys and it��s working handbook [M]. Changsha: Central South University Press, 2002: 127. (in Chinese)
[14] MA J, HE Y D, WANG D R. Oxidation of two-phase Cu�C50 Cr alloy at low oxygen pressure [J]. Mater Sci Eng A,2006, 434(1/2): 141-146.
[15] SONG Ming-zhao, ZHU Da-chuan, TU Ming-jing. Study on the corrosion behavior of Cu-Te-Li multiple alloy in the 3.5% NaCl solution [J]. Journal of Functional Materials, 2005, 36(2): 209-211. (in Chinese)
[16] ZAKY A M. Role of Cl- in breakdown of Cu�CAg alloys passivity in aqueous carbonate solutions [J]. Electrochimica Acta, 2006, 51: 2057-2062.
Foundation item: Project(Z2007208) supported by the Scientific Research Fund of Hebei Provincial Education Department, China
Corresponding author: ZHANG Rui-jun; Tel: +86-13833596156; E-mail: Zhangrj@ysu.edu.cn
DOI: 10.1016/S1003-6326(09)60108-4
(Edited by YANG Hua)