J. Cent. South Univ. (2012) 19: 2421-2424
DOI: 10.1007/s11771-012-1291-z
Synthesis and thermoelectric properties of Mg2Si1-xSnx solid solutions by microwave irradiation
ZHOU Shu-cai(�����)1,2, BAI Chen-guang(�׳���)1
1. College of Materials Science and Engineering, Chongqing University, Chongqing 400044, China;
2. School of Metallurgy and Material Engineering,Chongqing University of Science and Technology, Chongqing 401331, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: In order to reduce the oxidizing and volatilizing caused by Mg element in the traditional methods for synthesizing Mg2Sil-xSnx (x=0.2, 0.4, 0.6, 0.8) solid solutions, microwave irradiation techniques were used in preparing them as thermoelectric materials. Structure and phase composition of the obtained materials were investigated by X-ray diffraction (XRD). The electrical conductivity, Seebeck coefficient and thermal conductivity were measured as a function of temperature from 300 to 750 K. It is found that Mg2Sil-xSnx solid solutions are well formed with excessive content of 5% (molar fraction) Mg from the stoichiometric Mg2Sil-xSnx under microwave irradiation. A maximum dimensionless figure of merit, ZT, of about 0.26 is obtained for Mg2Sil-xSnx solid solutions at about 500 K for x=0.6.
Key words: Mg2Sil-xSnx; thermoelectric material; solid solution; microwave synthesis
1 Introduction
Thermoelectric (TE) materials are functional materials that can directly convert thermal energy into electrical energy by the TE effect, and vice versa. However, they are not widely used due to their low efficiency. Dimensionless figure of merit ZT=��2��T/k of TE materials is usually used to evaluate the efficiency of a TE device, where �� is the Seebeck coefficient, �� is the electrical conductivity, k is the thermal conductivity and T is the temperature in Kelvin. The Mg2Si-based alloy has been regarded as a new potential thermoelectric material in the temperature range from 500 to 800 K [1-2] due to its advantages of environmental-friendly materials, such as the abundance of its constituent elements in the earth��s crust and the non-toxicity of processing by-products [3]. However, the TE properties of Mg2Si are moderately low because the thermal conductivity of Mg2Si is relatively high for thermoelectric applications, about 6-8 W��m-1��K-1 [2, 4]. Generally, the thermal conductivity can be significantly reduced through the formation of solid solutions due to the enhanced point defect phonon scattering. Therefore, Mg2Si1-xSnx is chosen for extensive studies to further improve thermoelectric properties of this material system because of the great difference in atomic mass between Si and Sn, which can scatter the low frequency phonons more effectively to decrease the thermal conductivity. However, fabrication of Mg2Sil-xSnx solid solution is difficult due to the large difference in melting points among its constituents, and the high saturated vapor pressure and chemical reactivity of magnesium. Various fabrication methods have been reported, such as direct melting [2-4], solid state reaction [5], mechanical alloying [6-7], hot pressing [8] and spark plasma sintering (SPS) [9-10], which are quite time consuming and complex. Moreover, unintentional inclusions can be introduced very easily by the above preparation methods.
These problems mentioned above motivate the development of a new synthetic method that overcomes these problems and facilitates product formation. The microwave-assisted synthesis method is firstly introduced for this system. This method is a very promising preparation way for many materials because it is fast, clean, energy-efficient and does not suffer from the disadvantages of classical preparation techniques. Many materials have been synthesized by the microwave radiation method at considerably lower temperature and shorter time than the conventional methods [11-12]. However, applicability of microwave sintering to metals is ignored due to the fact that they reflect microwaves. ROY et al [11] reported that particulate metals can be heated rapidly in microwaves. This has led to the use of microwaves to consolidate a range of particulate metals and alloys [13]. This is very interesting from the application point of view.
Based on previous studies, Mg2Sil-xSnx solid solutions were synthesized by microwave irradiation in this work. The effects of Sn doping on the thermoelectric properties of Mg2Sil-xSnx solid solutions were investigated systematically.
2 Experimental
Figure 1 shows a schematic illustration of the experimental equipment, which is used for the production of activated Mg and Si particles. It mainly consists of a microwave oven and a heat preservation system.
The samples were prepared as follows. According to the stoichiometric proportion of Mg2Sil-xSnx (0��x��1.0), Mg (>99.5% purity, 74 ��m), Si (>99.5% purity, 50 ��m) and Sn (>99.5% purity, 50 ��m) were used as raw materials. Mg (5%, molar fraction) was added excessively to compensate for evaporation of Mg in the reaction process. The required amount of these materials was well mixed by an ultrasonic homogenizer in ethanol for 45 min. After being completely dried in the cabinet, they were compressed to form pellet of 10 mm in thickness and 20 mm in diameter under appropriate pressure. The samples were then placed in alumina crucible in the center of the microwave oven and the reaction was carried out under the protection of high purity Ar (99.9%). The microwave oven used in the present work consists of a 2.45 GHz microwave generator with continuous adjustable power output of 0-15 kW. An infrared pyrometer was used (Raytek, Marathon series) for temperature measurement. Microwave heating used in the present work was carried out at the power level of 2.0-3.5 kW and heated to a designed temperature (580-600 ��C) and kept at this temperature (with the power level of 1.0-1.5 kW) for 20-40 min. The samples were cooled to room temperature for further control experiment.
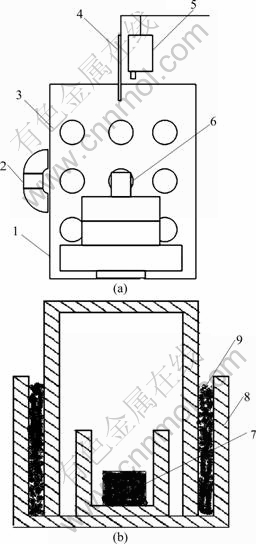
Fig. 1 Schematic view of microwave system setup (a) and crucible (b): 1-Rotating table; 2-Peep hole; 3-Magnetron tube; 4-Infrared thermometer; 5-Thermocouple; 6-Crucible; 7-Mg, Si, Sn samples; 8-Alumina; 9-Mg powder
Phases of the fabricated materials were detected by X-ray diffraction (D/Max-IIIA) with Cu K�� radiation (��=0.154 06 nm). The electrical conductivity (��) and Seebeck coefficient (��) were simultaneously measured by the four-probe dc method in helium atmosphere from room temperature to 800 K using a computer-assisted device. The error of the Seebeck coefficient and the electrical resistivity measurements was estimated to be less than ��5%. The thermal diffusivity (D) and the specific heat capacity (cp) of the samples were measured by a laser flash apparatus (Netzsch LFA 457) and a thermal analyzer (Netzsch DSC 404), respectively. The thermal conductivity was calculated from the relationship k=��Dcp, where �� is the density of the material, measured using Archimedes method at room temperature. Relative densities of the compacts measured by the Archimedes method were about 96% of the theoretical values.
3 Results and discussion
3.1 Synthesis of Mg2Sil-xSnx (0��x��1.0) solid solutions via microwave irradiation
Figure 2 shows the XRD patterns for Mg2Si1-xSnx (x=0.2, 0.4, 0.6 and 0.8) solid solutions synthesized by microwave irradiation. The XRD patterns of pure Mg2Si and Mg2Sn are also given for comparison. The major peaks of the patterns can be indexed to an anti-fluorite type structure (space group, Fm3m), and all the peaks of the solid solutions locate between the characteristic peaks of pure Mg2Si and Mg2Sn, suggesting that the peak shifts occur due to the solid solution. A trace amount of MgO impurity phase is also detected, but there are no discernible peaks for Mg, Si or Sn-related impurity phases. Simultaneously, all the peaks gradually shift left with the increase of x. According to NIKITIN et al [14], Mg2Sil-xSnx is not a continuous solid solution by solid state reaction and melting and there is a transforming melting reaction at high temperature. So solid solution with a Sn content of 0.4-0.6 would not exist. But after microwave irradiation, the split of peaks disappears, and all the peaks are single-pinnacle peaks, as shown in Fig. 2, which shows that it benefits for Mg2Sil-xSnx solid solutions forming.
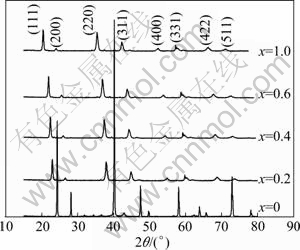
Fig. 2 XRD patterns for Mg2Si1-xSnx (0��x��1.0) compounds prepared by microwave-assisted activation
3.2 Thermoelectric properties of Mg2Sil-xSnx solid solutions
The electrical conductivities vs the temperature of the samples prepared by microwave irradiation methods are presented in Fig. 3. In all samples, the electrical conductivities of Mg2Si1-xSnx increase monotonically with temperature in a range of 300-750 K, indicating that semiconductor behavior occurs, except when x=0.2. The band gaps, Eg, are 0.77 and 0.35 eV for Mg2Si and Mg2Sn, respectively [15]. Eg might decrease with increasing Sn content, which makes it easier for electrons to jump from the valence band to the conduction band. Thus, the electrical conductivity might increase.
Figure 4 shows the variation of Seebeck coefficient with the holding temperature for Mg2Sil-xSnx solid solutions. A negative Seebeck coefficient suggests that the obtained materials exhibit n-type conductivity. The highest absolute value of Seebeck coefficient of 460 ��V/K is obtained at about 450 K with x=0.6. The absolute values of all the samples monotonically decrease with the increase of x. According to the formula |��|=��-lnnc, where �� is the scattering factor and nc is the carrier concentration [16], the absolute value of the Seebeck coefficient will be depressed with increasing carrier concentration. In the meantime, energy gaps reduce with the addition of Sn, then carrier concentration nc increases, resulting in the decrease of the absolute value of the Seebeck coefficient with increasing Sn content. The results in Fig. 4 reflect the adjustment of the band structures by the addition of Mg2Sn.

Fig. 3 Variation of electrical conductivity with holding temperature for Mg2Sil-xSnx
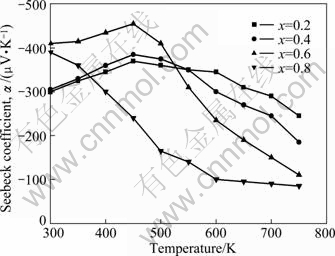
Fig. 4 Variation of Seebeck coefficient with holding temperature for Mg2Sil-xSnx
Variation of thermal conductivity for Mg2Sil-xSnx solid solutions, k, with the holding temperature is depicted in Fig. 5. The overall thermal conductivity k is given by k=ke+kph, where ke is the electronic thermal conductivity and kph is the lattice thermal conductivity. ke is related to electrical conductivity by Wiedemann-Franz law as ke=L0��T, where the degenerate Lorenz number L0 is given a value of 2.45��10-8 V2��K-2. The kph can be obtained by subtracting ke from the total thermal conductivity. For Mg2Sil-xSnx solid solutions, all the electronic thermal conductivities calculated in these samples are far smaller than total k. That is to say, kph plays predominant role in total k. kph is about 78%-96% of k, indicating that phonon-phonon interactions are the primary sources of thermal resistance.
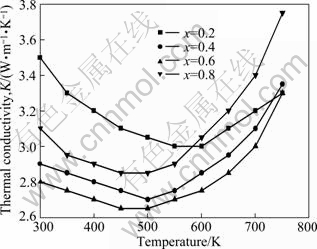
Fig. 5 Variation of thermal conductivity with holding temperature for Mg2Sil-xSnx
The temperature dependences of the dimensionless figure of merit ZT=��2��T/k of Mg2Sil-xSnx solid solutions are presented in Fig. 6. Relatively high figure-of-merit is obtained for x=0.6 in the measuring temperature range. A maximum figure of merit, ZT=0.26, is obtained for Mg2Sil-xSnx at about 500 K.
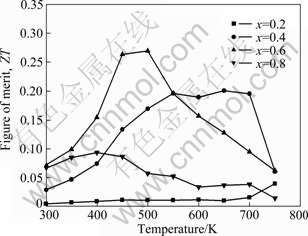
Fig. 6 Temperature dependency of figure of merit for Mg2Sil-xSnx
4 Conclusions
1) Mg2Sil-xSnx solid solutions are successfully prepared by microwave-assisted synthesis techniques. X-ray diffraction (XRD) patterns show that Mg2Sil-xSnx solid solutions are well formed under microwave irradiation.
2) With the electrical conductivity, Seebeck coefficient and thermal conductivity measured from room temperature to 750 K, the dimensionless figure of merit is calculated and discussed. The addition of Sn increases the electrical conductivity monotonically, reduces the thermal conductivity and changes the absolute Seebeck coefficient. The highest ZT value is 0.26 at 500 K for Mg2Sil-xSnx solid solutions. It is also demonstrated that microwave irradiation technique is a suitable method for preparing Mg2Sil-xSnx thermoelectric materials.
References
[1] TANI J I, KIDO H. Thermoelectric properties of Sb-doped Mg2Si semiconductors [J]. Intermetallics, 2007, 15(9): 1202-1207.
[2] ZAITSEV V K, FEDOROV M I, GURIEVA E A, EREMIN I S, KONSTANTINOV P P, SAMUNIN A Y, VEDERNIKOV M V. Highly effective Mg2Si1-xSnx thermoelectric [J]. Physical Review B, 2006, 74(4): 045207.
[3] GAO Hong-li, LIU Xin-xin, ZHU Tie-jun, ZHANG Sheng-nan, ZHAO Xin-bing. Effect of Sb doping on the thermoelectric properties of Mg2Si0.7Sn0.3 solid solutions [J]. Journal of Electronic Materials, 2011, 40(5): 830-834.
[4] HANG Qian, ZHU Tie-jun, YIN Hao, ZHAO Xin-bing. Preparation and thermoelectric properties of Mg2Si1-xSnx [J]. Physica Scripta, 2007, T129: 123-126.
[5] GAO Hong-li, ZHU Tie-jun, ZHAO Xin-bing, LIU Xin-xin, FAN Dong-xiao. Influence of Ca substitution on the thermoelectric properties of Mg2Si [J]. Journal of Materials Science & Engineering, 2010, 28(2): 260-262. ( in Chinese).
[6] ZHANG Qian, HE Jian, ZHU Tie-jun, ZHANG Sheng-nan, ZHAO Xin-bing, TRITT T M. High figures of merit and natural nanostructures in Mg2Si0.4Sn0.6 based thermoelectric materials [J]. Applied Physics Letters, 2008, 93(10): 102109.
[7] CLARK C R, WRIGHT C, SURYANARAYANA C, BABURAJ E G, FROES F H. Synthesis of Mg2X (X=Si, Ge, or Sn) intermetallics by mechanical alloying [J]. Materials Letters, 1997, 33(1-2): 71-75.
[8] SONGA R B, AIZAWA T, SUNA J Q. Synthesis of Mg2Si1-xSnx solid solutions as thermoelectric materials by bulk mechanical alloying and hot pressing [J]. Materials Science and Engineering B, 2007, 136(2/3): 111-117.
[9] LUO Wei-un Luo, YANG Mei-jun, CHEN Fei, SHEN Qiang, JIANG Hong-yi, ZHANG Lian-meng. Fabrication and thermoelectric properties of Mg2Si1-xSnx (0��x��1.0) solid solutions by solid state reaction and spark plasma sintering [J]. Materials Science and Engineering B, 2009, 157(1/2/3): 96-100.
[10] HAN Li-qin, YANG Mei-jun, SHEN Qiang, ZHANG Lian-meng. Reaction sintering of magnesium silicide thermoelectric material by the spark plasma sintering technique [J]. Journal of the Chinese Ceramic Society, 2008, 36(8): 337-340. (in Chinese)
[11] ROY R, AGRAWAL D, CHENG J P, GEDEVANISHVILI S. Full sintering of powdered metal bodies in a microwave field [J]. Nature, 1999, 399(17): 668-670.
[12] LEKSE J W, STAGGER T J, AITKEN J A. Microwave metallurgy: Synthesis of intermetallic compounds via microwave irradiation [J]. Chemistry of Materials, 2007; 19(15): 3601-3603.
[13] PANDA S S, SINGH V, UPADHYAYA A, AGRAWAL D. Sintering response of austenitic (316) and ferritic (434L) stainless steel consolidated in conventional and microwave furnaces [J]. Scripta Materialia, 2006, 54 (12): 2179-2183.
[14] NIKITIN E N, ZAITSEV V K, TKALENKO E N. A study on the phase diagram for the Mg2Si-Mg2Sn system and properties of certain of its solid solutions [J]. J Inorganic Mater, 1970, 4: 1656.
[15] ZAITSEV V K, FEDOROV M I, EREMIN I S, GURIEVA E A. Thermoelectrics handbook: Macro to nano-structured materials [M]. New York: CRC Press, 2005, Chap. 29: 1-11.
[16] HYUN D B, HWANG J S, OH T S, SHIM J D, KOLOMOETS N V. Electrical properties of the 85% Bi2Te3-15% Bi2Se3 thermoelectric material doped with SbI3 and CuBr [J].Journal of Physics and Chemistry of Solids, 1998, 59(6/7): 1039-1044.
(Edited by YANG Bing)
Foundation item: Project(2009BB4228) supported by the Natural Science Foundation of Chongqing City, China; Project(CK2010Z09) supported by the Research Foundation of Chongqing University of Science and Technology, China
Received date: 2011-07-27; Accepted date: 2011-11-18
Corresponding author: ZHOU Shu-cai, Associate Professor, PhD; Tel: +86-13102345792; E-mail: zhoushucai71@126.com