
Microstructures and properties of Sn-Ag-Cu lead-free solder alloys containing La
ZHOU Ying-chun(周迎春), PAN Qing-lin(潘清林), HE Yun-bin(何运斌), LIANG Wen-jie(梁文杰)
LI Wen-bin(李文斌), LI Yun-chun(李运春), LU Cong-ge(路聪阁)
School of Materials Science and Engineering, Central South University, Changsha 410083, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Trace amounts of La were utilized to improve the melting behaviors, microstructures, tensile properties and microhardness of Sn-3.0Ag-0.5Cu lead-free solder alloy. La has little effect on the melting behavior of Sn-3.0Ag-0.5Cu alloy according to the differential thermal analysis (DTA) tests. The X-ray diffraction (XRD) patterns show that β-Sn, Ag3Sn and Cu6Sn5 coexist in the as-cast solder alloys and LaSn3 phases emerge when adding 0.4% La. The microstructures modified by La are more uniform and much finer than that of baseline alloy, and the coarse LaSn3 particles with complex dendrites are observed by optical microscopy (OM) and scanning electron microscopy (SEM) when the addition of La is up to 0.4%. The composition of the LaSn3 phases is identified by energy-dispersive spectroscopy (EDS). There are considerable improvements in mechanical properties with 0.05% and 0.1% addition, but degenerations by adding 0.4%La. The Vickers microhardness of β-Sn and eutectic area are both enhanced with the addition of La and the microhardness of LaSn3 is much larger than those of β-Sn and eutectic area.
Key words: lead-free solder; Sn-Ag-Cu alloy; rare earth element; La; tensile properties
1 Introduction
Increasing environment and health concerns over the toxicity of lead have prompted widely researches to find alternatives for the traditional Sn-Pb solders, which possess advantages such as low melting temperatures, good workability, and excellent wetting property. The Sn-Ag-Cu ternary alloys were considered as one of the most promising candidates for substituting the Sn-Pb solder alloy in microelectronic packaging and interconnects, owing to their generally superior mechanical properties and relatively low temperature compared with Sn-Ag and Sn-Cu binary solder alloy[1-3].
The effects of pure rare earth (RE) elements on microstructure and properties have not been fully studied, compared with the fully researches of mixture rare earth elements[4-6]. Moreover, though RE elements have been employed to modify the microstructures and properties, there are some problems need to be resolved. The previous literatures did not show XRD patterns of RE phases though some RE-containing phases have been observed [4-6]. And the standpoints on the effects of RE on mechanical properties were not in good agreement [5-7]. In this sense, the study on the effects of pure rare earth elements on the microstructures and properties of lead-free solder alloys was strongly required.
In this study, the effects of trace amounts of La on melting behaviors, microstructures, tensile properties, and microhardness of Sn-3.0Ag-0.5Cu alloys were investigated.
2 Experimental
Pure Sn, Ag, Cu and La were used as raw material. They were melted in furnace at 750 ℃ for 2 h, and then chill cast as an ingot in a stainless steel mould. The chemical compositions of the studied alloys are listed in Table 1. The melting behaviors were defined using TAS 100. The possible phases were revealed by a D/max 2550 X-ray diffractometer. A POLYVER-MET optical microscopy (OM) and an FEI Sirion 200 scanning electron microscopy (SEM) equipped with energy-dispersive spectroscope (EDS) of EDAX Gensis 60 were both used to observe the microstructure. Dog-bone-type specimens with 30 mm in gauge length and 6 mm in diameter were prepared according to GB228―2002. The tensile tests were carried out at room temperature and a strain rate of 2 mm/min on CSS-44100 tensile testing machine.
Vickers hardness was determined by WOLPERT 401MVD microhardness tester at a load of 0.1 N for 10 s. The corresponding Vickers hardness values, HV were identified as[8]:

where F is the applied load and d is the average length of diagonals. Each reported microhardness value is the average of ten individual measurements.
Table 1 Chemical compositions of alloys (mass fraction, %)

3 Results and discussion
3.1 Melting characteristics
The melting behavior is one of the most important properties of solder alloys because it has a direct impact on the soldering process. The typical DTA curves of the solder alloys are shown in Fig.1. The DTA results of Sn-Ag-Cu solder alloys affected by the La content are listed in Table 2.
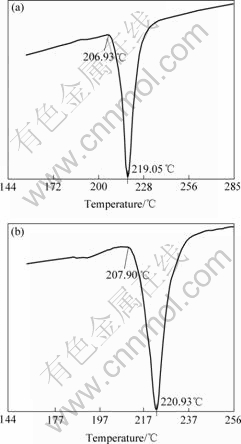
Fig.1 Representative DTA curves of solder alloys: (a) Alloy A; (b) Alloy C
Table 2 Melting behaviors of solder alloys (℃)
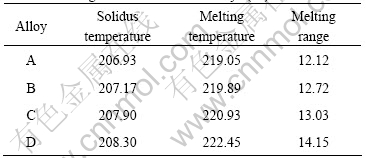
Solidus temperature is defined by the onset point of the DTA curve while melting temperature is the peak point. The analyses indicate that the solidus temperature, melting temperature and melting range are enhanced slightly with increasing the La addition. The previous literatures reported the same increases in melting temperature of Sn-Ag-Cu alloys with the addition of mixture rare earth [5, 9]. The melting temperature of Sn-Ag-Cu solder modified by La does not exceed 223 ℃. Some international electrical-equipment suppliers developed new lead-free reflow oven and lead-free wave oven that can conduct soldering below 250 ℃ using lead-free solder with a relatively higher melting temperature. Therefore, the Sn-3.0Ag-0.5Cu solders with the addition of La can meet the requirement of the present lead-free soldering process.
3.2 X-ray diffraction analysis
The corresponding XRD patterns of different La- containing solder alloys shown in Fig.2 indicate that β-Sn, Cu6Sn5 and Ag3Sn phases coexist in the as-cast solder alloys. The XRD patterns do not show significant differences when La addition does not exceed 0.1%, while the peaks of LaSn3 phases appear when the addition is up to 0.4%, as indicated by the arrows a and b.
3.3 Microstructure evolution
The typical optical microstructures of solders with different La contents are shown in Fig.3. The microstructures of the four alloys are similar to each other, all consisting of bright β-Sn grains surrounded by dark eutectic networks. Coarse primary Ag3Sn and Cu6Sn5 phases are not observed, which is also supported by SEM observation.
Binary and ternary eutectic structures are shown in Fig.4. The region containing tiny disc-shape and needle shape phases is Sn+Cu6Sn5+Ag3Sn ternary eutectic structure in Alloy A, as shown in Fig.4(a). Meanwhile, disc shape Sn+Cu6Sn5 binary eutectic structure and needle shape Sn+Ag3Sn binary eutectic structure in alloy
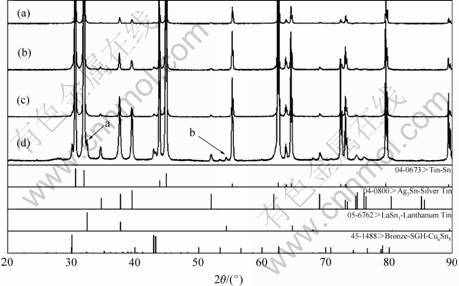
Fig.2 XRD patterns of solder alloys: (a) Alloy A; (b) Alloy B; (c) Alloy C; (d) Alloy D
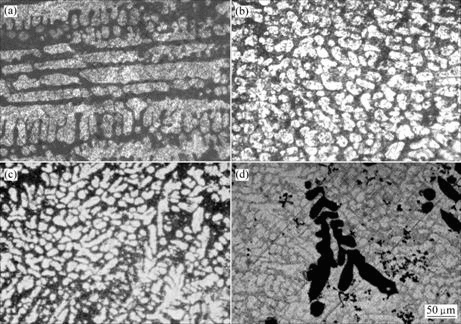
Fig.3 Typical OM microstructure of solder alloys: (a) Alloy A; (b) Alloy B; (c) Alloy C; (d) Alloy D

Fig.4 Typical SEM micrographs of eutectic structures: (a) Alloy A, ternary eutectic structure: (Sn+disk shape: Cu6Sn5+needle shape: Ag3Sn); (b) Alloy C, SAC-0.1La, region with binary and ternary eutectic structures: (Sn+disk shape: Cu6Sn5), (Sn+needle shape: Ag3Sn), and (Sn+disk shape: Cu6Sn5 + needle shape: Ag3Sn)
C are shown in Fig.4(b), as indicated by the arrows. The absence of the large primary Ag3Sn and Cu6Sn5 phases can be attributed to the component and cooling rate reason. KIM et al[10] studied microstructures of three Sn-Ag-Cu alloys under different cooled conditions. Primary Ag3Sn phases were frequently observed for the slowly cooled Sn-3.5Ag-0.7Cu and Sn-3.9Ag-0.6Cu alloys, and for the mildly cooled Sn-3.9Ag-0.6Cu samples, under this primary phase did not appear in Sn-30Ag-0.5Cu under any condition.
Some differences among the microstructures of the current solder alloys owing to different La content are observed. Fig.3 reveals that the dentrites of β-Sn turn to be more uniform and much finer in the La-containing alloys than baseline alloy. The large and coarse La-containing particles with complex dendrites are observed when adding La to 0.4%. However, the LaSn3 phases are observed by OM and SEM in this study, but DUDEK’s work was based on SEM and TEM[4].
Fig.5 shows the EDS analysis result taken from a coarse LaSn3 particle, presenting that the molar ratio of Sn to La is approximately 3?1, and the composition of the particle is LaSn3.
Much more attention should be paid to the mechanism of La on solidification process of solder alloy. When melt is cooled to the temperature lower than the melting temperature of β-Sn, heterogeneous nucleuses arise due to the dissolution of dendrite arm, or oxide formation for the nucleation in alloy A. Because no stirring or convection exists in the liquid phase, which is approaching our condition, the solute rejected from the solid into the liquid only by diffusion at the solid/liquid forms a boundary layer[11].
If R is the rate of advance of the solid interface, the distribution of solute (CL) ahead of the interface as a function of distance from the interface (x) can be described by:
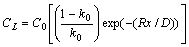
where D is the solute diffusion coefficient in the liquid,k0 is the equilibrium distribution coefficient. The liquids temperature is given by: T = T0-mCL. For the solute boundary layer, the temperature is given by:
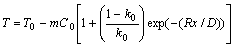
When the actual temperature ahead of the interface is lower than the solidification temperature for the related solute composition, constututional supercooling will occur. The dendrite growth can be viewed not only as branching process in the tip neighborhood, but also as advancing of tips from solid to liquid. The hypoeutectic component determined that hypoeutectic reactions followed dendrite development.
Besides heterogeneous nucleation in alloy A, there is a peritectic reaction for alloys B and C at 235 ℃ that is higher than the melting temperature of β-Sn[12]. The peritectic reaction products can act more effectively for heterogeneous nucleation of β-Sn than that of CeSn3 that is produced by the eutectic reaction at 230 ℃ and eventually refine the dendrite sizes[13]. YU et al[7] pointed out that the adsorption of active elements will prevent dendrite from further growth. Admitting active effects, the La will also be adsorbed at the interface of other heterogeneous nucleation sites, which reduces the interfacial energies and eventually assists the other heterogeneous nucleation. On the other hand, the adsorption of La elements at the interface of solid/liquid will inhibit diffusion of Ag and Cu atoms from solid to liquid to deepen constitutional supercooling. The effect of La on constitutional supercooling plays a positive role in the growth of dendrite structure, which is contrary to the effect of depressing surface energy restraining dendrite development. So the effect of La is the interplay among the heterogeneous nucleates, assisting the other heterogeneous nucleation, decreasing surface energy of β-Sn, and deepening constitutional supercooling.
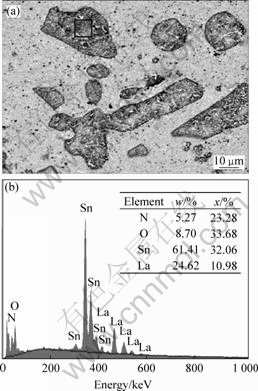
Fig. 5 EDX compositions of LaSn3 particles in alloy D
The formation of dendrite LaSn3 in alloy D can be explained as follows: the Sn atoms are rejected from the solute boundary layer into the liquid to build a boundary layer this time because the k0 has values <1, when regarding the Sn matrix as solute while LaSn3 as matrix according to the La-Sn equilibrium phase diagram. So the constitutional supercooling happened to the dendrite growth of LaSn3.
3.4 Tensile properties
The stress-strain curves as a function of La content is shown in Fig.6. It is obvious that the addition of 0.05% and 0.1% La can increase in mechanical properties. The addition up to 0.4% La can decrease the mechanical properties.
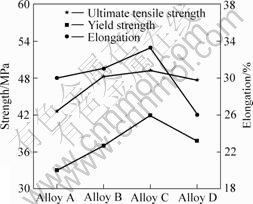
Fig.6 Influence of La content on tensile properties of Sn-3.0Ag-0.5Cu solder alloys
Microstructure refinement and hard RE compounds were regarded as the most important factors for the fluctuations of mechanical properties[5, 7, 14].
However, the microstructure refinement can not improve the ultimate tensile strength and yield strength. Though the tensile tests are performed at room temperature,
T/Tm ≈ (273+20)/(273+220) = 0.59
where T is application temperature in degrees of Kelvin and Tm is melting point. The ratio of T/Tm that is greater than 0.5 indicates that the tensile tests are carried out at elevated temperature[15]. At elevated temperatures, the mechanical properties are governed by grain boundaries sliding because grain boundaries are weaker than the grain interiors. Fine grain size with more grain boundaries will have negative effect on the strength. Moreover, the high affinities of RE elements for oxygen and hydrogen have been widely used in metals, such as aluminum alloy for reducing the content of gases and impurities [16]. The addition of La favors the formation of Sn-3.0Ag-0.5Cu alloy with cleaner grain boundaries because of the fact that greater part of the undesirable impurities is combined with La into stable inclusions within grains thereby the strength and elongation can be improved. The ultimate tensile strength, yield strength and elongation of alloys B and C are higher than that of alloy A owing to cleaner grain boundaries with minimal La addition. The reason for the decline of mechanical properties when adding 0.4% La is that the coarse and brittle LaSn3 has weaker combination with matrix, and can not deform harmoniously with matrix when experiencing stress concentration, which induces the cavity nucleation[15].
3.5 Microhardness
The microhardness tests have been performed in β-Sn and eutectic area of the four alloys as well as the LaSn3 particles. Fig.7 shows the typical optical micrographs of indentation sizes of β-Sn in alloy A and gradual change from β-Sn to LaSn3 particles in alloy D. The gradual change of indentation size from β-Sn to LaSn3 particles indicates evidently that the Vickers microhardness of LaSn3 particles is HV 60.1, which is much larger than that of β-Sn.

Fig.7 Optical micrographs of indentation size: (a) β-Sn, alloy A; (b) gradual change from β-Sn to LaSn3 particles, alloy D
Fig.8 shows the effects of La content on Vickers microhardness of the four as-cast alloys. It is obvious that the microhardnesses of β-Sn and eutectic area with the addition of La are enhanced from HV13.8 to HV16.4 and from HV16.8 to HV18.8, respectively.
The improvement in Vickers microhardness of β-Sn and eutectic area can be associated with the microstructure refinement by the addition of La and reducing the content of gases and impurities by the high affinities for oxygen and hydrogen[17].

Fig.8 Microhardness of β-Sn and eutectic area of Sn-3.0Ag-0.5Cu solder alloys
4 Conclusions
1) La has little effect on the melting behavior of Sn-3.0Ag-0.5Cu alloy.
2) β-Sn, Ag3Sn and Cu6Sn5 phases coexist in the as-cast solder alloys and LaSn3 phases emerge when adding 0.4% La.
3) The dendrites of β-Sn turn to be more uniform and much finer in the La-containing alloys. When the addition of La is up to 0.4%, LaSn3 phases with complex dendrites are observed by OM and SEM, and the composition of the particles is also identified by EDS. The addition of La has a direct effect on the solidification process of Sn-3.0Ag-0.5Cu solder alloy by peritectic reaction, assisting the other heterogeneous nucleation, decreasing in surface energy of β-Sn, and constitutional supercooling.
4) Addition of La up to 0.1% leads to remarkable increases in ultimate tensile strength and yield strength of Sn-3.0Ag-0.5Cu solder alloys, but excessive addition is harmful to them. The addition of La has a similar effect on elongation.
5) Microhardnesses of β-Sn and eutectic area are both enhanced with addition of La and the microhardness of LaSn3 is much larger than those of them.
References
[1] WU C M L, YU D Q, LAW C M T, WANG L. Properties of lead-free solder alloys with rare earth element additions[J]. Mater Sci Eng R, 2004, 44: 1-44.
[2] LI G Y, SHI X Q. Effects of bismuth on growth of intermetallic compounds in Sn-Ag-Cu Pb-free solder joints[J]. Trans Nonferrous Met Soc China, 2006, 16(s1): s739-s743.
[3] SUNDELIN J J, NURMI S T, LEPIST? T K, RISTOLAINEN E O. Mechanical and microstructural properties of SnAgCu solder joints[J]. Mater Sci Eng A, 2006, 420: 55-62.
[4] DUDEK M A, SIDHU R S, CHAWLA N, RENAVIKAR M. Microstructure and mechanical behavior of novel rare earth-containing Pb-free solders[J]. Journal of Electronic Materials, 2006, 35: 2088-2097.
[5] CHEN Zhi-gang, SHI Yao-wu, XIA Zhi-dong, YAN Yan-fu. Properties of lead-free solder SnAgCu containing minute amounts of rare earth[J]. Journal of Electronic Materials, 2003, 32: 235-243.
[6] WANG L, YU D Q, ZHAO J, HUANG M L. Improvement of wettability and tensile property in Sn-Ag-RE lead-free solder alloy[J]. Materials Letters, 2002, 56, 1039-1042.
[7] YU D Q, ZHAO J, WANG L. Improvement on the microstructure stability, mechanical and wetting properties of Sn-Ag-Cu lead-free solder with the addition of rare earth elements[J]. Journal of Alloys and Compounds, 2004, 376: 170-175.
[8] SHEN Jun, LIU Yong-Chang, HAN Ya-jing, GAO Hou-xiu, WEI Chen, YANG Yu-qin. Effects of cooling rate on microstructure and microhardness of lead-free Sn-3.5Ag solders[J]. Trans Nonferrous Met Soc China, 2006, 16(1): 59-64.
[9] CHENG Guang-hui, ZHANG Ke-ke, MAN Hua, YANG Jie, LIU Ya-min, YU Yang-chun. Effect of rare earths on microstructure and properties of Sn2.0Ag0.7CuRE solder alloy for surface mount technology[J]. Journal of the Chinese Rare Earth Society, 2005, 23: 486-489. (in Chinese)
[10] KIM K S, HUH S H, SUGANUMA K. Effects of cooling speed on microstructure and tensile properties of Sn-Ag-Cu alloys[J]. Mater Sci Eng A, 2002, 333: 106-114.
[11] MINKOFF I. Solidification and cast structure[M]. Chichester: JHON WILEY and SONS, 1986: 16-17.
[12] ASM. ASM Handbook: Alloy phase diagrams[M]. 1992: 275.
[13] ZHANG Si-qi, HUANG Jin-song. Smelting and casting of nonferrous metal[M]. Beijing: Chemical Industry Press, 2006: 144-145. (in Chinese)
[14] WU C M L, D YU Q, LAW C M T, WANG L. The properties of Sn-9Zn lead-free solder alloys doped with trace rare earth elements[J]. Journal of Electronic Materials, 2002, 31: 921-927.
[15] DING Ying, WANG Chun-qing, TIAN Yan-hong, et al. Influence of aging treatment on deformation behavior of 96.5Sn3.5Ag lead-free solder alloy during in situ tensile tests[J]. Journal of Alloys and Compounds, 2007, 428: 274-285.
[16] XU Guo-fu, MOU Shen-zhou, YANG Jun-jun, JIN Tou-nan, NIE Zuo-ren, YIN Zhi-min. Effect of trace rare earth element Er on Al-Zn-Mg alloy[J]. Trans Nonferrous Met Soc China, 2006, 16(3): 598-603.
[17] MENG De-quan, ZHANG Wei-qiang, ZHANG Guang-feng. Influences of RE element La and Ce on structure and properties of as-cast pure copper[J]. Foundary, 2007, 56: 651-653. (in Chinese)
(Edited by HE Xue-feng)
Corresponding author: ZHOU Ying-chun; Tel: +86-731-8830933; E-mail: valuable1008@163.com