
Pressure leaching of zinc silicate ore in sulfuric acid medium
LI Cun-xiong(�����), XU Hong-sheng(���ʤ), DENG Zhi-gan(��־��),
LI Xing-bin(���˱�), LI Ming-tin(��F͢), WEI Chang(κ ��)
Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology,
Kunming 650093, China
Received 6 July 2009; accepted 25 December 2009
Abstract: Zinc silicate ore was characterized mineralogically and the results showed that zinc exists mainly as hemimorphite and smithsonite in the sample. Sulfuric acid pressure leaching of zinc silicate ore was carried out to assess the effect of particle size, sulfuric acid concentration, pressure, reaction time and temperature on the extraction of zinc and the dissolution of silica. Under the optimum conditions employed, up to 99.25% of zinc extraction and 0.20% silica dissolution are obtained. The main minerals in leaching residue are quartz and small amounts of undissolved oxide minerals of iron, lead and aluminum are associated with quartz.
Key words: zinc silicate ore; sulfuric acid pressure leaching; extraction of zinc; dissolution of silica
1 Introduction
Conventionally, zinc is produced mostly from zinc sulfide ores because sulfides are easy to separate from gangue and to concentrate by conventional flotation techniques. With declining of global reserves of zinc sulfides as well as the restriction on sulfur emissions during their processing, there is an increasing focus on the processing of the huge reserves of zinc oxide ores. Oxidized ores contain zinc in various carbonate and silicates minerals such as smithsonite (ZnCO3), hydrozincite (2ZnCO3��3Zn(OH)2), zincite (ZnO), willemite (Zn2SiO4) and hemimorphite (Zn4Si2O7(OH)2��H2O). Extensive investigations have been performed to upgrade the zinc oxide ores by flotation, but had a limited effect on their qualities. The reason is the fine intergrowth of mineral phases[1]. In the literatures available, many investigations focused on using the hydrometallurgical route for leaching zinc oxide ores in sulfuric acid or alkaline[2]. Mostly, the zinc oxide ores have a large quantity of silica, which dissolves during acid leaching, forming silicic acid and transforming to a gel. This prevents the separation of the zinc sulphate solution from the slurry[3-5]. Currently, measures either by controlling the pH or adding some flocculating agents to leaching solution are used for coagulation of silicic acid[6-10], and investigations on leaching kinetics are also carried out from the economic point of view[11-12]. Because the zinc silicate ore usually contains some acid consuming basic gangue such as CaO and MgO, researches on zinc silicate ore leaching in alkaline medium have also been carried out[13-18]. However, the volatilization of ammonia is a great threat for operators�� health, and zinc in zinc sulphide concentrate cannot be extracted in alkaline medium, which results in a low zinc extraction. In the present work, zinc silicate ore is treated by method of pressure leaching in sulphuric acid medium. The optimum conditions for zinc extraction and silica dissolution from zinc silicate ore in sulphuric acid medium under pressure are systematically investigated.
2 Experimental
2.1 Materials and procedure
The experimental study was carried out using zinc silicate ore from Guangxi province of China. Table 1 shows the chemical composition of the zinc silicate ore used in this study. X-ray diffraction(XRD) analysis of the ore showed that the ore contains mainly about 65.1% hemimorphite (Zn4Si2O7(OH)2H2O), 14.25% smithsonite (ZnCO3), 16.48% quartz (SiO2), 0.39% cerussite (PbCO3), and 2.78% talc ((Fe, Mg)(Fe, Al)(AlSi7O20)-(OH)4). Table 2 shows the mineral composition. Mineralogical analysis of the sample was also performed by scanning electron microscope (SEM), and the result was shown in Fig.1.
The SEM image in Fig.1 suggests that zinc is generally present in gangue materials in three modes: 1) associated with silica, 2) weakly adsorbed to the crystalline quartz surface, and 3) substituted isomorphously for iron in the chlorite structure. Zinc silicate deposits even from the same region can be quite variable in nature. The mineralogy of the zinc silicate ore in Guangxi province of China displays highly variable composition for zinc, iron, calcium, magnesium and silica though the dominant mineral is quartz.
In all the leaching experiments, the analytical grade sulfuric acid with a mass fraction of 98% and a density of 1.84 g/mL was employed. The solid to liquid ratio of reaction mixture was maintained constant at 1?3 g/mL,and was agitated at a speed rate of 550 r/min.
Table 1 Main chemical composition of zinc silicate ore (mass fraction, %)

Table 2 Main mineral composition of zinc silicate ore (mass fraction, %)
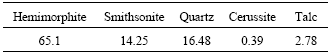
Fig.1 SEM image (a) and EDS patterns (b)-(e) of zinc silicate ore
2.2 Leaching experiments
The leaching experiments of the zinc silicate ore were conducted in a 2 L, acid corrosion resistant titanium autoclaves. This autoclave was equipped with a heating mantle, a PID temperature controller, a variable speed stirrer and an internally mounted cooling coil. The stirring system had two axial impellers with 6 blades with 45? downdraft mounted at 4.5 cm apart. All the experiments were carried out batch-wise using 100 g of zinc silicate ore, various concentrations of H2SO4 and a certain pressure. The procedure consisted of slurring the ore with sulphuric acid and water at the set liquid to solid ratio in autoclave, and heating the solution to the temperature of the experiment. At the set temperature, the gas was admitted and the pressure was adjusted to the desired value, which was maintained constant in the duration of the experiment. At the end of the experiment, the gas flow was shut down; the autoclave was rapidly water-cooled; and the solution was filtered for collecting the solid residues. The solution was analyzed for zinc and silica and some solid residues were observed by SEM. Under this process, the zinc is extracted into the acidic solution, while silica remains as a solid residue.
3 Results and discussion
The main variables studied were the particle size of the sample, concentration of sulfuric acid in the leaching solution, pressure, temperature and time of reaction.
3.1 Effect of particle size
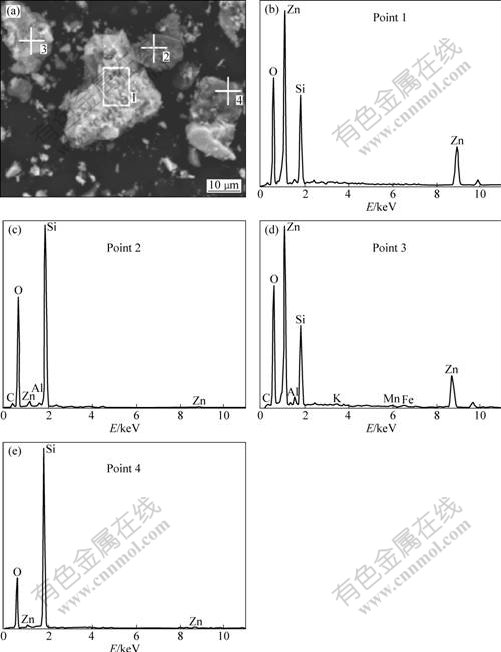
The effect of particle size on the leaching of zinc silicate ore was studied in the range from 50 ��m to 300 ��m. The result is shown in Fig.2.
It is shown in Fig.2 that zinc extraction depends strongly on the particle size of zinc silicate ore in the range of 106-300 ��m, and a significant increase in the
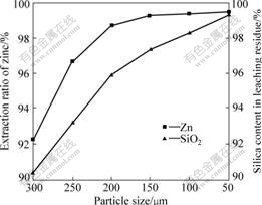
Fig.2 Effect of particle size on leaching of zinc silicate ore (Sulfuric acid concentration of 120 g/L, pressure of 1 000 kPa, reaction time of 90 min and temperature of 150 ��)
zinc extraction is obtained. However, a further decrease in the particle size from 75 ��m to 50 ��m does not produce a substantial increase in the extraction. We can also see from Fig. 2 that the particle size also has a pronounced effect on silica dissolution. Decrease in the particle size can decrease the silica dissolution. So, the ore particle size should be less than 106 ��m in order to obtain a higher zinc extraction and lower silica dissolution. The sample used in the following experiment was sieved and about 85% of it was in size range of ��106 ��m.
3.2 Effect of sulfuric acid concentration
The effect of sulfuric acid concentration on the leaching of zinc silicate ore was studied in the range of 108-156 g/L. The result is shown in Fig.3. As we can see from Fig.3 that an increase in acid concentration from 108 to 132 g/L increases zinc extraction; however, further increase to 156 g/L affects negatively the zinc extraction. The dissolution of silica is increased slowly under this condition and most of silica in zinc silicate ore is converted into solid leaching residues. The sulfuric acid concentration is maintained at 132 g/L in the following experiments which is considered to be appropriate to eliminating the effect of this variable.
Fig.3 Effect of sulfuric acid concentration on leaching of zine silicate ore (Particle size ��106 ��m, pressure of 1 000 kPa, reaction time of 90 min and temperature of 150 ��)
3.3 Effect of pressure
The effects of pressure on extraction of zinc and dissolution of silica were studied. The results can be seen in Fig.4. We observe that the pressure has a pronounced effect on zinc extraction. Increasing pressure increases the zinc extraction. We can also see from Fig.4 that the dissolution of silica is affected little by changes of the pressure from 600 kPa to 1 600 kPa, and the maximum dissolution rate of silica is only 0.8%.
3.4 Effect of reaction time
The effect of reactive time on the leaching of zinc silicate ore was studied in the reaction time range of 30- 150 min. Fig.5 shows the extraction of zinc and dissolution of silica as a function of reaction time in the leaching process. We can observe that an increase in leaching time increases significantly the extraction ratio of zinc. From Fig.5, it can also be seen that the dissolution of silica decreases with increasing leaching time up to 120 min and increases afterwards.
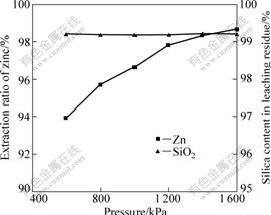
Fig.4 Effect of pressure on leaching of zinc silicate ore (Particle size ��106 ��m, sulfuric acid concentration of 132 g/L, reaction time of 90 min and temperature of 150 ��)
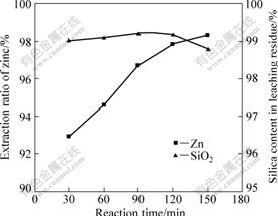
Fig.5 Effect of reaction time on leaching of zinc silicate ore (Particle size ��106 ��m, sulfuric acid concentration of 132 g/L, pressure of 1 200 kPa and temperature of 150 ��)
3.5 Effect of temperature
Zinc silicate ore was leached at temperatures varying from 100 to 160 �� to determine the effect of temperature on the zinc extraction and silica dissolution. The result is shown in Fig.6. From Fig.6, it can be seen that the extraction ratio of zinc is affected significantly by changes in the temperature from 100 �� to 160 ��, and the maximum zinc extraction can reach 98.8% at 160 ��. We can also see from Fig.6 that the silica dissolution depends strongly on the temperature in the range of 100-120 ��, and a significant increase in the silica dissolution is obtained. However, a further increase in
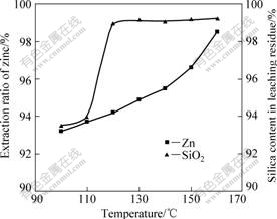
Fig.6 Effect of temperature on leaching of zinc silicate ore (Particle size ��106 ��m, sulfuric acid concentration of 132 g/L, pressure of 1 200 kPa and reaction time of 90 min)
the temperature from 120 ��C to 160 ��C does not produce a substantial increase in the dissolution. Consequently, according to these data for a selective extraction of zinc from zinc silicate ore, the leaching should be carried out at temperature above 120 ��.
From these initial experiments, when the solid to liquid ratio of reaction mixture was maintained constant at 1:3 g/mL, and the mixture was agitated at a speed rate of 550 r/min, the optimum leaching conditions were determined to be: ��106 ��m of particle size, 132 g/L of sulfuric acid, 90 min leaching at 120 ��, 1 200 kPa pressure. Experiments were carried out under the optimum conditions. The extraction ratio of zinc was 99.25%, and the dissolution of silica was only 0.20%. Finally, a thin section from the filter cake was prepared and examined by SEM.
3.6 Characterization of residues from leaching
The SEM image and EDS patterns of a leaching residue obtained in leaching at 120 �� for 90 min under the pressure of 1 200 kPa and sulfuric acid concentration of 132 g/L are shown in Fig.7.
The morphology and constituent phases of the residue can be seen clearly in Fig.7. We can observe that the physical aspect of the particles changes a lot as compared with the non-leached zinc silicate particles. In the EDS pattern of Fig.7(b), we can only see the quartz phase as expected since only 0.2% of silica has been dissolved and 99.25% of the associated or adsorbed zinc minerals has been extracted. In the EDS patterns of Figs.7(d), (e) and (f), some undissolved oxide minerals of iron, lead and aluminum are associated with quartz.
The leaching of zinc silicate ores in autoclave in sulfuric acid system is a viable process that effectively extracts most of zinc into solution and rejects silica from its minerals.
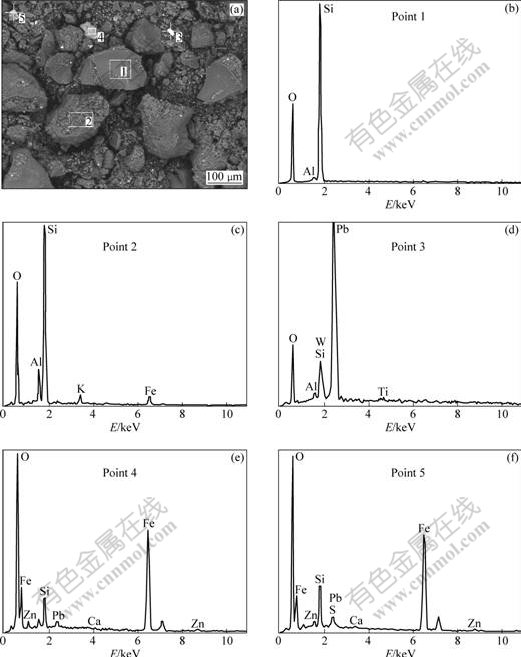
Fig.7 SEM image (a) and EDS patterns (b)-(f) of leaching residue
4 Conclusions
1) The zinc silicate ore from Guangxi province of China contains hemimorphite and smithsonite as economic minerals, and quartz and chlorite as gangue minerals. Zinc is present in three modes: associated with silica, weakly adsorbed to crystalline quartz surface and substituted isomorphously for iron in chlorite structure.
2) When the solid to liquid ratio of reaction mixture is maintained constant at 1:3 g/mL, and the mixture is agitated at a speed rate of 550 r/min, the optimum leaching conditions are ��106 ��m of particle size, 132 g/L of sulfuric acid, 90 min leaching at 120 �� and 1 200 kPa. Under the optimum conditions, the extraction ratio of zinc is 99.25%, and the dissolution of silica is only 0.20%.
3) After leaching at optimum conditions, the morphology and constituent phases of the residue change a lot as compared with the non-leached zinc silicate ore. The main minerals in leach residue are quartz and small amounts of undissolved oxide minerals of iron, lead and aluminum are associated with quartz.
4) Sulfuric acid pressure leaching of zinc silicate ore is a viable process that effectively extracts most of zinc into solution and rejects silica from its minerals.
References
[1] LI Jun-qi. Technology research of hydrometallurgical treatment of zinc silicate ore [J]. Hydrometallurgy, 1996, 4(60): 32-35. (in Chinese)
[2] FRENAY J. Leaching of oxidized zinc ore in various media [J]. Hydrometallurgy, 1985, 15(2): 243-253.
[3] LIN Zuo-yan, HUA Yi-xin. Technology and mechanism study on leaching high silicon zinc oxide ore with sulfuric acid [J]. Nonferrous Metal (Smelting Section), 2003(5): 9-23. (in Chinese)
[4] DUFRESNE R E. Quick leach of siliceous zinc ore [J]. Journal of Metals, 1976, 28: 8-12.
[5] LI Guo-min. Research on leaching de-silication process of zinc oxide ore with high silicon [J]. China Nonferrous Metal, 2005(4): 32-35. (in Chinese)
[6] PERRY W. Refining zinc silicate ore by special leaching technique [J]. Chemical Engineering, 1966, 73(21): 182-184.
[7] KUMAR R, BISWAS A K. Zinc recovery from Zawar ancient siliceous slag [J]. Hydrometallurgy, 1986, 15(3): 267-280.
[8] MATTHEW I G, ELSNER D. The hydrometallurgical treatment of zinc silicate ores [J]. Metallurgical and Materials Transactions B, 1977, 8(1): 73-83.
[9] BODAS M G. Hydrometallurgical treatment of zinc silicate ore from Thailand [J]. Hydrometallurgy, 1996, 40(1): 37-49.
[10] ABDEL-AAL E A, SHUKRY Z E. Application of quick leaching method to Egyptian zinc silicate ore [J]. Transactions of the Institution of Mining and Metallurgy (Section C: Mineral Processing and Extractive Metallurgy), 1997, 106: 89-90.
[11] ABDEL-AAL E A. Kinetics of sulfuric acid leaching of low-grade zinc silicate ore [J]. Hydrometallurgy, 2000, 39(2): 247-254.
[12] TERRY B, MONHEMIUS A J. Acid dissolution of willemite and hemimorphite [J]. Metallurgical and Materials Transactions B, 1983, 14(3): 335-346.
[13] WANG Rui-xiang, TANG Mo-tang, YANG Sheng-hai, ZHAGN Wen-hai, TANG Chao-bo, HE Jing, YANG Jian-guang. Leaching kinetics of low grade zinc oxide ore in NH3-NH4Cl-H2O system [J]. J Cent South Univ Technol, 2008, 15: 679-683.
[14] YANG Sheng-hai, LI Ying-nian, JU Shao-hua, TANG Mo-tang. Leaching of zinc oxide ore with ammonium chloride solution [J]. Hydrometallurgy, 2006, 25(4): 180-182. (in Chinese)
[15] YANG Sheng-hai, TANG Mo-tang, CHEN Yi-feng, TANG Chao-bo, HE Jing. Anodic reaction kinetics of electro winning zinc in system of Zn(II)-NH3-NH4Cl-H2O [J]. Trans Nonferrous Met Soc China, 2004, 14(3): 626-630.
[16] TANG Mo-tang, OUYANG Min. Preparation of grade zinc oxide using ammonium sulfate [J]. The Chinese Journal of Nonferrous Metals, 1998, 8(1): 118-121. (in Chinese)
[17] ZHANG Bao-ping, TANG Mo-tang, YANG Sheng-hai. Treating zinc oxide ores using ammonia-ammonium chloride to produce electrolysis zinc [J]. Journal of Central South University of Technology: Natural Science, 2003, 34(6): 619-623. (in Chinese)
[18] KUNKUL A, MUHTAR K M, YAPICI M, DEMIRBA A. Leaching kinetics of malachite in ammonia solutions [J]. Mineral Processing, 1994, 41(3): 167-182.
(Edited by YANG Bing)
Foundation item: Project(2007CB613605) supported by the National Basic Research Program of China
Corresponding author: LI Cun-xiong; Tel/Fax: +86-871-5188819; E-mail: licunxiong@126.com
DOI: 10.1016/S1003-6326(09)60236-3