Effects of niobium addition on microstructure and properties of CPM121 powder metallurgy high-speed steel
来源期刊:中南大学学报(英文版)2021年第4期
论文作者:肖逸锋 张乾坤 李苏望 吴靓 钱锦文 陈泽民 沈玮俊 吝楠 贺跃辉
文章页码:1206 - 1218
Key words:CPM121; niobium-alloying; microstructure; mechanical properties; temper resistance; wear resistance; rotary forging; powder metallurgy
Abstract: Massive vanadium additions as hard phases in powder metallurgy high-speed steels (PM HSS) lead to higher cost and bad machinability. In this study, ultrahigh alloy PM HSS with CPM121 (10W-5Mo-4Cr-10V-9Co, wt.%) as the basic composition, was directly compacted and activation sintered with near-full density (>99.0%) using pre-oxidized and ball-mixed element and carbide powders. Niobium-alloyed steels (w(V)+w(Nb)=10 wt.%) show higher hardness and wear resistance, superior secondary-hardening ability and temper resistance. But excess niobium addition (>5 wt.%) leads to coarsened carbides and deteriorated toughness. EPMA results proved that niobium tends to distribute in MC carbides and forces element W to form M6C and WC carbides. Further, the role of rotary forging on properties of niobium-alloyed steels (S3) was researched. After rotary forging with deformation of 40%, the bending strength and fracture toughness of niobium-alloyed steels could be further improved by 20.74% and 43.86% compared with those of sample S3 without rotary forging, respectively.
Cite this article as: ZHANG Qian-kun, LI Su-wang, XIAO Yi-feng, WU Liang, QIAN Jin-wen, CHEN Ze-min, SHEN Wei-jun, LIN Nan, HE Yue-hui. Effects of Niobium addition on microstructure and properties of CPM121 powder metallurgy high-speed steel [J]. Journal of Central South University, 2021, 28(4): 1206-1218. DOI: https://doi.org/ 10.1007/s11771-021-4690-1.

J. Cent. South Univ. (2021) 28: 1206-1218
DOI: https://doi.org/10.1007/s11771-021-4690-1
ZHANG Qian-kun(张乾坤)1, LI Su-wang(李苏望)1, XIAO Yi-feng(肖逸锋)1, WU Liang(吴靓)1,
QIAN Jin-wen(钱锦文)1, CHEN Ze-min(陈泽民)1, SHEN Wei-jun(沈玮俊)2,
LIN Nan(吝楠)3, HE Yue-hui(贺跃辉) 2
1. School of Mechanical Engineering, Xiangtan University, Xiangtan 411105, China;
2. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
3. College of Materials Science and Engineering, Hunan University, Changsha 410082, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Abstract: Massive vanadium additions as hard phases in powder metallurgy high-speed steels (PM HSS) lead to higher cost and bad machinability. In this study, ultrahigh alloy PM HSS with CPM121 (10W-5Mo-4Cr-10V-9Co, wt.%) as the basic composition, was directly compacted and activation sintered with near-full density (>99.0%) using pre-oxidized and ball-mixed element and carbide powders. Niobium-alloyed steels (w(V)+w(Nb)=10 wt.%) show higher hardness and wear resistance, superior secondary-hardening ability and temper resistance. But excess niobium addition (>5 wt.%) leads to coarsened carbides and deteriorated toughness. EPMA results proved that niobium tends to distribute in MC carbides and forces element W to form M6C and WC carbides. Further, the role of rotary forging on properties of niobium-alloyed steels (S3) was researched. After rotary forging with deformation of 40%, the bending strength and fracture toughness of niobium-alloyed steels could be further improved by 20.74% and 43.86% compared with those of sample S3 without rotary forging, respectively.
Key words: CPM121; niobium-alloying; microstructure; mechanical properties; temper resistance; wear resistance; rotary forging; powder metallurgy
Cite this article as: ZHANG Qian-kun, LI Su-wang, XIAO Yi-feng, WU Liang, QIAN Jin-wen, CHEN Ze-min, SHEN Wei-jun, LIN Nan, HE Yue-hui. Effects of Niobium addition on microstructure and properties of CPM121 powder metallurgy high-speed steel [J]. Journal of Central South University, 2021, 28(4): 1206-1218. DOI: https://doi.org/ 10.1007/s11771-021-4690-1.
1 Introduction
In modern industry, traditional tool materials such as high-speed steels (HSSs), cemented carbide and cermet are already incompetent under the condition requiring both high toughness and wear resistance. Common HSSs show insufficient wear resistance, while cermet has poor toughness. High alloyed PM HSSs possess much higher strength and toughness than cermet, and are superior to common HSSs in hardness and wear resistance [1-3]. From W, Mo and W-Mo system [4] to high Co system [5], high V and W-V system [6-9], the development of high alloyed and super-hard HSSs with high abrasive resistance, toughness and red hardness has become a trend of the powder metallurgy (PM) ones [10] applied under certain conditions such as a broach, stamping die, thread forming taps and drills [11, 12].
Commercial high-alloyed PM HSSs mainly
Foundation item: Projects(51771237, 51704257) supported by the National Natural Science Foundation of China; Project(2019JJ60019) supported by the Joint Fund of Hunan Province, China; Project(17QDZ25) supported by the School Level Fund of Xiangtan University, China
Received date: 2020-11-11; Accepted date: 2021-04-26
Corresponding author: XIAO Yi-feng, PhD, Professor, Tel: +86-731-58292214; E-mail: xiaoyifeng@xtu.edu.cn; ORCID: https://orcid. org/0000-0003-1017-9115
include S290, ASP2060, CPM121 [13], CPM15V [9], etc. The traditional process (such as gas-atomization and hot isostatic pressing (HIP) [14], spray-forming [15]) of PM HSS is often characterized by long process, costly equipment investment, limitation of smelting alloying and atomization [16], and it is difficult to achieve near-net shaping. For high-alloyed PM HSS, higher vanadium content is the key to improve the wear resistance [17]. However, excessive vanadium (>5 wt.%) will deteriorate the grinding performance and toughness of HSS and increase the cost. In addition, excessive vanadium leads to the aggregation and coarsening of carbide [18], which will decrease the strength, toughness, and grinding performance of steel.
Niobium as a strong carbide forming element with lower cost is often added into PM HSS (such as ASP2017 [19]) with trace amounts to improve wear resistance and strength by secondary hardening. TAKAHASHI et al [20] showed that niobium has a higher carbon capture ability than vanadium, and the segregation of trace niobium at defects can effectively pin dislocations. In detail, the formation of the Nb-Cottrell atmosphere and the solid solution of niobium in the matrix can strongly inhibit the recovery of dislocations and enhance the temper resistance of steel [21]. In the cast steels alloyed by vanadium, by adding niobium, the grain size of primary MC carbides can be refined, the solid solution of vanadium in the matrix was increased, thus the secondary hardening ability can be strengthened [15, 22]. However, the results above are based on the low content vanadium and niobium HSS. The effect and strengthening mechanism of high content vanadium/niobium in HSSs are rarely studied, especially for the high alloy HSSs with high vanadium and niobium [8, 23-25].
In this work, element niobium was introduced in quantity into CMP121 steel by partially replacing vanadium, aiming at reducing the content of costly vanadium element [21, 26], optimizing the grinding performance of high vanadium steel without the loss of hardness and wear resistance. In detail, ultrahigh vanadium/niobium PM HSS based on CPM121 (10W-5Mo-4Cr-10V-9Co) was prepared by ball milling, pressing, activated sintering and reactive diffusion, namely the direct fabrication technology (DFT), which shows low pollution, near-net shaping and is easy to prepare ultrahigh-alloyed PM materials as previously studied [27]. The optimized mechanical properties and temper resistance of niobium-alloyed steels and hot deformation by rotary forging, as well as the strengthening mechanisms of alloying and hot deformation, were investigated.
2 Experimental
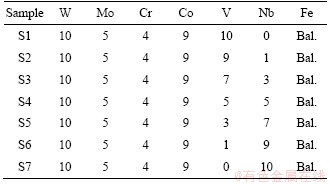
The chemical compositions of CPM121 with different vanadium/niobium contents are listed in Table 1. Using DFT method, the powders were mixed in alcohol by a planetary ball mill using raw carbide powders (WC, Mo2C, Cr3C2, VC, NbC) and element powders (Fe, Co) with the particle size of 2-4 μm and 4-10 μm, respectively. The powders were milled for 48 h with a ball-to-powder weight ratio of 5:1. After milling, the mixture slurry was dried at 78 °C for 8 h. After sieving, the mixture was transferred to a drying chamber with low oxygen partial pressure (at 0.04 MPa) for pre-oxidation to a certain extent. Subsequently, the mixed powders were compacted at 150 MPa and sintered at 1165 °C for 2 h in a vacuum furnace.
Table 1 Chemical composition of CPM121 with different vanadium/niobium content (wt.%)
Then as-sintered samples were austenitized at 1150 °C, oil-quenched and triple tempered at 580 °C for 1.5 h. Besides, as-sintered steels were rotary forged at 1100 °C with the deformation of 10%, 20%, 30% and 40%, controlled by gaskets and dies.
The density of samples was tested by the Archimedes method. The phase transformation points of HSSs were measured by differential thermal analysis (DSC) at a heating rate of 10 °C/min. Rockwell hardness was measured with a load of 150 kg (according to the Chinese National Standard GB/T 230.1―2004) and the transverse rupture strength (TRS) was tested using the three-point bending method with INSTRON 3369. The fracture toughness is tested by the single-edge notched beam (SENB) [28]. The impact toughness was tested on a pendulum-type impact-tester. The friction and wear properties of the sample against with YG-6 hard alloy ball of φ6 mm with a load of 90 N and a rate of 600 r/min was measured by HSR-2M reciprocating friction and wear tester. LECO CS-444 C/S analyzer and LECO TC-436 N/O analyzer were utilized for testing the content of C, S, O, and N of as-sintered steels. The microstructure and scratch morphologies were studied using FEI Quanta 250 FEG SEM, while the phase compositions of materials were quantitatively analyzed by D/max 2550 (40 keV, 18 kW, Cu Kα) X-ray diffractometer and electron probe micro-analyzer (EPMA, JXA-8530F).
3 Results and discussion
3.1 Densification behavior of niobium-alloying steels
Figure 1 shows the density curves of CPM121 steels with different vanadium /niobium contents. With the increase of niobium content, both density and relative density gradually increase. More niobium additions further improve the relative density to almost 99.8% after sintered at 1165 °C for 2 h.
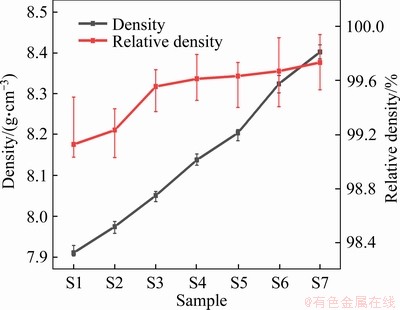
Figure 1 Variations of density and relative density of CPM 121 with different vanadium/niobium content
The DSC curves of as-annealed S1, S3, S5, and S7 steels are shown in Figure 2. The peak at (850±2) °C corresponds to the phase transformation point of α-Fe→γ-Fe, it means that niobium does not affect the nucleation and growth of γ-Fe during heating. At 1000-1050 °C, S3, S5 and S7 steels show obvious endothermic peaks, namely 1053.3, 1007.3 and 1008.3 °C. At higher temperature, the partial liquid phase appears in steels at 1204.7, 1115, 1071.3 and 1070.8 °C. For S1 steel, combined with the C-Fe-V phase diagram [29], it can be seen that the phase γ+MC enters the phase region of L+γ+MC after 1204.7 °C. For niobium-alloyed steels, the appearance of an endothermic peak at 1000-1100 °C may be caused by the dissolution of M6C/M2C from γ+MC+M6C/M2C into L+γ+MC region [30], which means that niobium addition may promote the phase transformation process. The possible appearance of partial liquid among particles helps the densification process of PM steels [28], as shown in Figure 1.
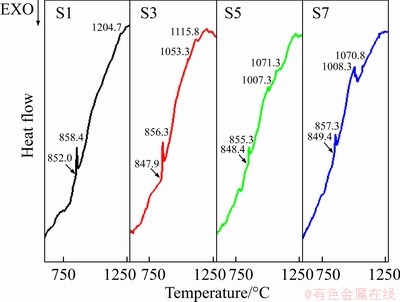
Figure 2 DSC curves of annealed CPM121 with different vanadium/niobium contents measured from RT to 1300 °C (The onset temperature and peak temperature of each peak were marked)
3.2 Microstructure and phase analysis
Phases of PM steels, which are composed of α-Fe, MC(V8C7) [7], NbC, M6C, and WC, do not change obviously before and after heat treatment according to the XRD patterns of as-sintered and as-tempered steels in Figure 3. But with increasing niobium content, peaks of V8C7 in Figure 3 shift towards lower angles signi?cantly. According to the Bragg’s law, nλ=2dsinθ, (n is the diffraction order, d is the interplanar spacing, λ is the wavelength of X-ray, and θ is the angle), lower θ values indicate bigger interplanar spacing values of V8C7 structure. This may be explained by the bigger atom radius of element niobium than vanadium, considering the solution of niobium in MC [24], which will be characterized below.
Figure 4 shows SEM images of as-tempered HSSs with different vanadium/niobium contents.
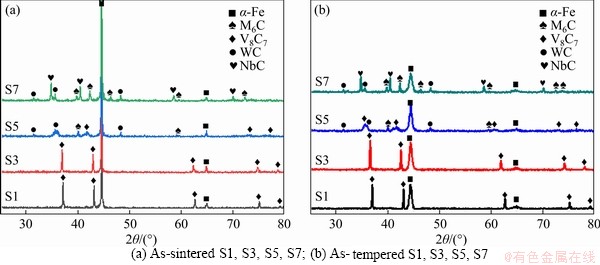
Figure 3 XRD patterns of CPM121 with different vanadium/niobium contents:

Figure 4 SEM images of CPM121 with different vanadium/niobium content after heat treatment:
When niobium content reaches 3% or more, almost no voids can be observed. It is in agreement with the density curves in Figure 1. In detail, it is found in SEM images that the material without niobium is mainly composed of the dark α-Fe matrix, gray MC carbide phase, and dark areas inside the carbides. With the increase of niobium content, the dark area in MC carbide gradually disappears, and the bright-white carbides with different shapes can be observed. From the EPMA analysis, the dark areas contain 18.35W, 16.9Mo, 51.71V, 6.08Cr, 6.15Fe and 0.81Co (wt.%), compared with 27.47W, 11.7Mo, 36.7V, 5.9Cr, 16.23Fe and 2.0Co (wt.%) in bright areas. In other words, the dark area is tungsten-poor in gray MC carbide as reported [27]. As demonstrated by GODEC et al [31], the inhomogeneous distribution of tungsten and vanadium also occurs in conventional HIP HSS and the element inhomogeneity does not affect the crystal structure of MC carbides, as observed in XRD patterns (Figure 3). Further, S3 and S5 were typically researched using EPMA dot and surface analysis (Figure 5).
In Figure 5, elements W, Mo, C, V, and Co

Figure 5 EPMA mapping results of tempered steels of S3 (a) and S5 (b)
present uniform distribution in the iron matrix. Gradient distribution of the above elements can be observed along with the interface between carbides and matrix, which verifies the vigorous mutual diffusion of atoms during sintering. The white point-like carbide is proved to be the WC-based particle. Further, with the increasing of niobium addition, the content of element W in MC carbide linearly decreases from 33.4 wt.% (S1) to 5.4 wt.% (S7), as shown in Figure 6. The lower solubility of W in gray phase and the precipitation of W-rich carbides (WC and M6C) in the matrix are related to the lower solubility of W in NbC than that in VC at high temperature, concluded from the isothermal section of C-V-W [32] and C-Nb-W [33] ternary phase diagrams above 1100 °C.
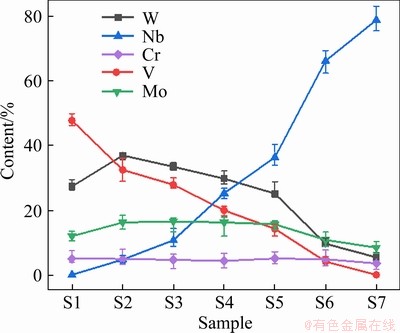
Figure 6 Variations (EPMA) of alloying elements in MC carbide in as-tempered steels as a function of niobium content
As shown in Figures 4 and 5, white rod-shaped carbide gradually appeared along with more niobium additions. Based on Figure 5, element Cr gradually performs segregation in white carbides and leanness in the matrix in S5 compared with that in S3. Moreover, EPMA results show that the white rod-shaped carbide is mainly composed of Mo, W, Cr, and Fe. In detail, compositions of white carbides are 49.2W, 28.8Mo, 6.7Fe, 9.2Cr, 4.6V, 0.9Nb, 0.6Co (wt.%) in S5, and 45.1W, 12.96Mo, 31.59Fe, 4.3Cr, 4.4Co, 0.6Nb (wt.%) in S7. Generally, M6C carbide in HSS belongs to Fe3W3C or Fe4W2C structure and M2C [34] carbide is mainly composed of Mo2C or W2C. Then when niobium acts alone (S7 steel), white carbides are concluded to be M6C-type. Though no M2C peaks can be observed in S5 in XRD curves in Figure 3 considering the low content of strip carbides, the strip carbides in S5 and S6 are still speculated to belong to M2C structure [35] considering their compositions above.
Finally, it should also be noted that more niobium addition deteriorates the microstructure and uniformity of composition of steels, manifesting as the bridging and coarsening of unevenly-distributed carbides, which will no doubt pose distinctly impacts on the mechanical properties.
3.3 Mechanical properties
3.3.1 Effect of niobium-alloying on mechanical properties
Figure 7 shows the mechanical properties of the vanadium/niobium alloyed CPM121 after heat treatment. It can be seen that the hardness values of the steels are generally at a high level (HRC 69.3-72.6) and increase linearly with niobium content. Generally, hardness of tool steel is in contradiction to strength and toughness, which are key indicators for their service performance, while PM HSSs show superior comprehensive

Figure 7 Variations of hardness, impact toughness (a), bend strength, and fracture toughness (b) of the vanadium/niobium alloyed CPM121 after heat treatment
performance. As displayed in Figure 7, with the increase of niobium content, the impact toughness, bending strength, and fracture toughness of the materials increase first and then decrease after peak values of S3 steel. Enhanced densification by niobium addition as proved by Figures 1 and 2, together with the finer grain size of S3 and S2 steel (it is calculated with Image-Pro Plus that the average grain sizes of S1, S2, and S3 are 2.944, 2.142 and 1.962 μm, respectively) in Figure 3, are responsible for the improved strength and toughness compared with S1 steel. However, more niobium content leads to the appearance of the massive strip and block M6C, M2C, and WC carbides, thus improving the hardness but seriously deteriorating the strength and toughness.
Empirically, fracture morphology can more effectively reflect the density of the PM materials with high density (>98%) than the morphology of polished sections such as Figure 3, considering the filling and plastic deformation of micro-pores by abrasive diamond or SiC particles. In Figure 8, the fracture morphologies of as-tempered HSSs with different compositions are displayed. A small number of spherical pores can be seen in S1 and S2, while the pores disappear with higher niobium content. This is in agreement with Figure 1. During the final sintering stage, with the growth of sintering necks, the voids are isolated into closed pores and spheroidized [27, 35]. By bulk and grain boundary diffusion, the number and size of pores decrease simultaneously [36]. Therefore, the segregation of niobium at the defects [20] accelerates the grain boundary diffusion and promotes the densification of the material. At the same time, the increase in the solid solubility of vanadium in γ-Fe can release more carbon atoms, which also promotes the densification of materials [37]. However, as shown in Figure 3, more strip and coarsened carbides appear due to the activated diffusion induced by niobium. In Figures 8(c)-(g), HSSs show the characteristics of quasi-cleavage fracture. With the increase of niobium content, the tearing edge increases first and then decreases significantly. In Figures 8(d)-(g), the fracture surface shows a smoother cleavage plane, meaning that the strength and toughness of the

Figure 8 Fracture morphology of vanadium/niobium alloyed CPM121 steels after heat treatment:
material decrease, which is manifested as the impact toughness of the material decreases gradually when the niobium content is higher than 3%, in Figure 6.
3.3.2 Effect of niobium-alloying on secondary hardening and temper resistance
Figure 9 shows the hardness variations of different compositions of vanadium/niobium alloyed CPM121 after tempered at 580 °C for different time. It can be seen that with the increase of niobium content, the quenching hardness of the material decreases remarkably, and the hardness of as-quenched S7 even decreases to HRC58.5. According to the Nb Cottrell effect [20], niobium atoms segregated along the grain boundary (such as the grain boundary of γ-Fe/γ-Fe and residual carbide of γ-Fe) and formed high temperature pinning during quenching. Together with enhanced solubility of vanadium in γ-Fe by niobium, they will severely inhibit the nucleation and growth of ferrite, manifesting as shifting the continuous cooling transformation curve (C curve) to the right and increased hardenability. However, the pining effect and more dissolved vanadium atoms caused by excessive addition of niobium also improve the stability of the undercooled austenites and lower the Ms point. Then, a large number of undercooled austenites remain as residual and oversaturated γ-Fe, appearing as low hardness values of as-quenched steels. However, after tempering, the hardness values of S5-S7 steels increase rapidly to HRC71-73, namely obvious secondary hardening [38]. During tempering and cooling, retained austenites transform into metastable bainite or tempered martensites and precipitated carbides [37]. The hardness of the materials alloyed by less than 5%Nb varied almost

Figure 9 Hardness variations of CPM121 with different vanadium/niobium contents after tempered for different time
placidly. No obvious secondary hardening can be observed in S1-S3 steels. Thus, it can be concluded that excessive niobium additions (>5%) will markedly affect the hardenability of CPM121, and sufficient tempering is essential, for secondary hardening.
For studying the temper resistance of niobium alloyed steels, the variations of red hardness are shown in Figure 10. It is found that the values of the red hardness of niobium alloyed steels decrease slightly after reheated at 600 °C, but they still maintain at a fairly high level (HRC67.2 of S7 after kept at 600 °C for 4 h). At the same time, it can be observed that the red hardness tends to increase with the addition of niobium. For example, the hardness values of S1 steel decrease by HRC2.2, HRC4.3 and HRC5.9 after reheated at 600 °C for 2, 4 and 8 h; and the decrease of S3 steel is HRC1.6, HRC3.4 and HRC4.9, respectively. However, the hardness decrease tends to be larger with more niobium additions. For S7 steel, the values of hardness decrease are HRC4.6, HRC5.4 and HRC7.6. Therefore, the addition of appropriate amount of niobium can improve the red hardness of CPM121 steel, but the tempering resistance of CPM121 steel is deteriorated when the niobium content exceeds 5% (S5).

Figure 10 Hardness variations of tempered niobium alloyed CPM121 steels after reheated at 600 °C for 2, 4 and 8 h
Generally, the serving temperature of HSS is always below the tempering temperature of 540-600 °C, but occasional overheating during being grinded by wheels or cutting may distinctly affect the performance of HSS. Figure 11 displays the hardness curves of S1-S7 steels after reheated at different temperatures for 4 h. As a whole, the hardness values of each steel decrease by about HRC3-5 after reheated at 600 °C, HRC5-8 at 625 °C and HRC7-10 at 650 °C. Especially, the hardness of niobium-alloyed steels remains above HRC62 even after over-tempered at 650 °C for 4 h,
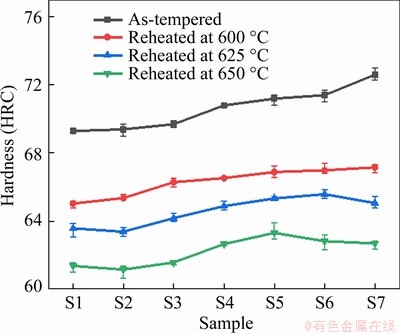
Figure 11 Hardness variations of tempered niobium alloyed CPM121 steels after reheated at 600, 625 and 650 °C for 4 h
and the hardness overall increases with increased niobium content, except that of S6 at 650 °C and S7 steels at 625 °C.
3.3.3 Effect of vanadium/niobium alloying on wear resistance
Figure 12 shows the micro-morphology of wear scratch of vanadium/niobium alloyed CPM121 steels after wear test against with YG6 hard metal balls (HRA90). It can be found that the high-speed steels with different compositions show relatively flat friction surface in the SEM images, though different wear depth of 2-4 μm can be detected by the contour graph, as presented in Figure 13. During abrasion, a large number of carbides acting as wear resistant members and strong supporting points, impede the cut of grinding ball, resulting in the flat friction surface. However, numerous microcracks can be observed on the steels alloying by more than 7% niobium. The three-dimensional contour graph shows that tough and undulating pits appear on the surface of the grinding marks in S5 and S7. Besides, the rod-like carbides tend to be broken during abrasion.

Figure 12 Scratch microstructure of CPM121 with different vanadium/niobium contents after heat treatment:
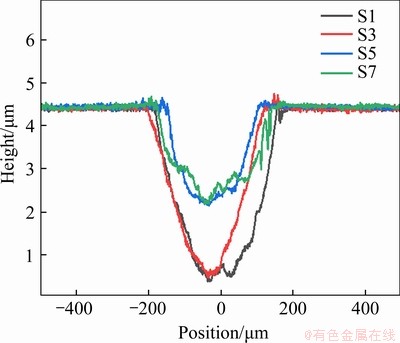
Figure 13 Section profile of wear scratch of CPM121 with different vanadium/niobium content after heat treatment
The wear scratches of niobium-alloyed steels after wear test are shown in Figure 13. With the increase of niobium content, the abrasion volume of materials decreased first and then increased. The wear volumes of S1, S3, S5, and S7 are 942.7, 743.6, 411.9 and 488.0 μm3, respectively. Therefore, proper niobium content can enhance the wear resistance of material significantly [39], but when the niobium content exceeds 7%, the wear resistance of the material decreases slowly due to abrasive wear. Besides, the big-sized carbides as wear-resisting supports are responsible for the lower friction volumes of S5-S7, which show higher hardness than the steels alloyed by less than 7%Nb.
4.1 Effect of rotary forging on properties of S3 steel
As an economic processing method combining forging with rolling, rotary forging possesses the advantages of high production efficiency, good product performance, and dramatically improve performance [40]. In this paper, the S3 steel alloyed with 3%Nb showing better comprehensive mechanical properties was used for the rotary forging test at 1100 °C.
The SEM images of S3 steel after rotary forging, quenching and tempering are shown in Figure 14. It can be seen that the rotary forging deformation does not change the type of carbides. With the increase of deformation, coarsened and slightly bridged carbides were crushed into uniform and fine carbide particles [41]. Combined with the statistics of Image-Pro Plus, it is found that the volume fraction of gray MC carbide is (24.63±0.31)% and the average size is 1.887 μm without deformation, while the volume of carbide increases to (25.70±0.17)% and (27.52±0.26)% after forging with 20% and 40% deformation, and the average size of MC carbides decreases to 1.725 and 1.386 μm, respectively. Namely, the carbide distribution and grain refinement of S3 steel can be greatly improved by rotary forging.
During rotary forging, dynamic recovery and recrystallization constantly occurred, which may make the undissolved carbides surrounded by

Figure 14 Microstructure and fracture surface morphology of S3 steel with different swaging deformation:
dislocation with high density due to the pinning effect. Numerous defects around carbides not only promote the dissolution of alloy elements (W, Mo, V, Nb, etc.) in the matrix, but also accelerate the precipitation process on carbides. Namely, the enhanced dissolution-precipitation process of alloy elements can facilitate both refining and spheroidizing of carbides, as shown in Figure 14(e).
In Figure 14, the S3 steel without deformation shows quasi-cleavage fracture with tearing edges and fan-shaped river patterns. With the increase of deformation degree, the small cleavage surface of river pattern produced by local transgranular fracture disappears, the amounts of fine and uniformly distributed dimples and tear edges gradually increase. The phenomenon that the material exhibits a large number of plastic fractures, means that the bonding strength of the interfacial increases with the increase of deformation. Besides, refined carbides and ferrous grains modify the way of crack propagations, it will probably improve the mechanical properties of steels.
After forging, the hardness of S3 maintains at HRC 69.7-70.0. And as shown in Figure 15, it can be seen that with the increase of forging deformation, both of the toughness and strength of S3 increase linearly. When the deformation is 40%, the impact toughness and bending strength increase to 10.43 J/cm2 and 3731.9 MPa, compared with 7.25 J/cm2 and 3090.8 MPa of the material without forging. But higher deformation leads to a higher risk of cracking, especially for the ultra-high alloyed steels [41]. Thus further forging was not included for comparison. Combined with the microstructure images in Figure 14, the dispersion of micron-sized carbides, the improvement of carbide morphology

Figure 15 Mechanical properties of S3 steel with different forging deformations
and refinement are the key to improve the strength and toughness of materials, which is consistent with the variation trend of toughness and bending strength of steels. In conclusion, rotary forging can improve the mechanical properties of materials effectively, further expand their application field as high strength and super hard PM high-speed steels.
4 Conclusions
Niobium addition was introduced into CPM121 (10W-5Mo-4Cr-10V-9Co) steel by partially taking place of costly vanadium. Optimized mechanical properties and temper resistance of niobium-alloyed steels, as well as the strengthening mechanism, were investigated.
1) With the increase of niobium content, niobium atoms partially participate into the VC lattice and form MC carbides of (Nb, V)C and NbC structures. Proper niobium additions enhance the densification and lead to the refinement of MC carbides, but excessive additions lead to the formation of strip-like carbides and bulk M6C carbides, as well as WC, partially due to the less solubility of W in NbC than in VC.
2) Niobium alloyed CPM121 steels generally have high hardness (HRC69.3-72.6), temper resistance, and wear resistance. In a whole, the wear resistance and hardness increase with niobium addition. A small amount of niobium can improve the strength and toughness (peak values of 3090.8 MPa, 7.25 J/cm2 of the steel with 3%Nb) and temper resistance of CPM121 steels, but when niobium content is more than 5%, the strength, toughness, and temper resistance decrease.
3) Hot deformation of rotary forging greatly improves the toughness and strength of steels. Meanwhile, the fracture of steels after rotary forging changes from quasi-cleavage fracture to massive plastic fracture.
Contributors
The overarching research goals and the direct fabrication technology(DFT)were developed by HE Yue-hui, ZHANG Qian-kun, XIAO Yi-feng, WU Liang, QIAN Jin-wen, and LIN Nan. LI Su-wang, SHEN Wei-jun and CHEN Ze-min analyzed the measured data, and wrote the first draft of the manuscript. ZHANG Qian-kun and LI Su-wang edited the draft of manuscript. All authors replied to reviewers’ comments and revised the final version.
Conflict of interest
ZHANG Qian-kun, LI Su-wang, XIAO Yi-feng, WU Liang, QIAN Jin-wen, CHEN Ze-min, SHEN Wei-jun, LIN Nan and HE Yue-hui declare that they have no conflict of interest.
References
[1] FOLZER A, TORNBERG C. Advances in processing technology for powder-metallurgical tool steels and high speed steels giving excellent cleanliness and homogeneity [J]. Materials Science Forum, 2003, 426-432: 4167-4172. DOI: 10.4028/www.scientific.net/msf.426-432.4167.
[2] SOARES E P, VATAVUK J, PANELLI R, PILLIS M F. Evaluation of mechanical properties and microstructure of a high carbon-vanadium tool steel produced by powder metallurgy [J]. Materials Science Forum, 2006, 530-531: 140-144. DOI: 10.4028/www.scientific.net/msf.530-531.140.
[3] WANG Rong, ANDREN H O, WISELL H, DUNLOP G L. The role of alloy composition in the precipitation behaviour of high speed steels [J]. Acta Metallurgica Et Materialia, 1992, 40: 1727-1738. DOI: https://doi.org/10.1016/0956-7151(92) 90116-V.
[4] KREMNEV L S. From high-speed tungsten steel to high-temperature molybdenum steel: A century of high-speed steel [J]. Steel in Translation, 2009, 39(12): 1111-1118. DOI: 10.3103/S0967091209120195.
[5] DOBRZA?SKI L A, KASPRZAK W. The influence of 5% cobalt addition on structure and working properties of the 9-2-2-5, 11-2-2-5 and 11-0-2-5 high-speed steels [J]. Journal of Materials Processing Technology, 2001, 109(1, 2): 52-64. DOI: 10.1016/S0924-0136(00)00775-5.
[6] XU Liu-jie, WEI Shi-zhong, XING Jian-dong, LI Yan, LONG Rui. Phase structure and fine microstructure of in-situ vanadium carbides in cast high-vanadium high-speed steel [J]. Metals & Materials International, 2006, 12: 371-375. DOI: https://doi.org/10.1007/BF03027702.
[7] SHTREMEL’ M A, KARABASOVA L V, CHIZHIKOV V I, VODENIKTOV S I. On optimum alloying of high-vanadium, high-speed steel [J]. Metal Science and Heat Treatment, 1999, 41(4): 146-150. DOI:10.1007/BF02465798.
[8] WEI Shi-zhong, ZHU Jin-hua, XU Liu-jie, LONG Rui. Effects of carbon on microstructures and properties of high vanadium high-speed steel [J]. Materials & Design, 2006, 27(1): 58-63. DOI: 10.1016/j.matdes.2004.09.027.
[9] VANCURA F, MA?EK B, AI?MAN D, JIRKOV? H, WAGNER M F X, B?HME M. Modification of metastable microstructure of CPM15V steel by heat exposure after treatment in semi-solid state [J]. Journal of Alloys and Compounds, 2014, 586: S159-S164. DOI: 10.1016/j.jallcom. 2013.04.064.
[10] WU Li-zhi. Developments and challenges of China high-speed steel industry over last decade [C]// Advanced Steels. 2011: 453-461. DOI:10.1007/978-3-642-17665-4_45.
[11] YAN X G, LI D Y. Effects of the sub-zero treatment condition on microstructure, mechanical behavior and wear resistance of W9Mo3Cr4V high speed steel [J]. Wear, 2013, 302(1, 2): 854-862. DOI: 10.1016/j.wear.2012.12.037.
[12] DO K R, CHOI S H, KWON Y S, PARK D W, CHO K K, AHN I S. Study on the sintering behavior and microstructure development of the powder injection molded T42 high-speed steel [J]. Metals and Materials International, 2011, 17(6): 937-942. DOI: 10.1007/s12540-011-6011-y.
[13] AI?MAN D, RUBE?OV? K, MIKMEKOV? ?. Mini-thixoforming of high-alloyed CPM REX 121 steel [J]. Advanced Materials Research, 2014, 887-888: 1156-1160. DOI: 10.4028/www.scientific.net/amr.887-888.1156.
[14] TAKIGAWA H, MANTO H, KAWAI N, HOMMA K. Properties of high-speed steels produced by powder metallurgy [J]. Powder Metallurgy, 1981, 24(4): 196-202. DOI: 10.1179/pom.1981.24.4.196.
[15] LU L, HOU L G, ZHANG J X, WANG H B, CUI H, HUANG J F, ZHANG Y A, ZHANG J S. Improved the microstructures and properties of M3: 2 high-speed steel by spray forming and niobium alloying [J]. Materials Characterization, 2016, 117: 1-8. DOI: 10.1016/j.matchar.2016.04.010.
[16] KIRK F A. Problems in high-speed steel manufacture and use: A challenge for economic powder metallurgy processing [J]. Powder Metallurgy, 1981, 24(2): 70-74. DOI: 10.1179/pom. 1981.24.2.70.
[17] XU Liu-jie, XING Jian-dong, WEI Shi-zhong, PENG Tao, ZHANG Yong-zhen, LONG Rui. Artificial neural network prediction of heat-treatment hardness and abrasive wear resistance of high-vanadium high-speed steel (HVHSS) [J]. Journal of Materials Science, 2007, 42(8): 2565-2573. DOI:10.1007/s10853-006-1278-y.
[18] WIEΒNER M, LEISCH M, EMMINGER H, KULMBURG A. Phase transformation study of a high speed steel powder by high temperature X-ray diffraction [J]. Materials Characterization, 2008, 59(7): 937-943. DOI: 10.1016/ j.matchar.2007.08.002.
[19] GRINDER O. The HIP way to make cleaner, better steels [J]. Metal Powder Report, 2007, 62(9): 16-22. DOI: 10.1016/ S0026-0657(07)70190-X.
[20] TAKAHASHI J, KAWAKAMI K, HAMADA J I, KIMURA K. Direct observation of niobium segregation to dislocations in steel [J]. Acta Materialia, 2016, 107: 415-422. DOI: 10.1016/j.actamat.2016.01.070.
[21] WOYDT M, HUANG S, VLEUGELS J, MOHRBACHER H, CANNIZZA E. Potentials of niobium carbide (NbC) as cutting tools and for wear protection [J]. International Journal of Refractory Metals and Hard Materials, 2018, 72: 380-387. DOI: 10.1016/j.ijrmhm.2018.01.009.
[22] WANG He-bin, HOU Long-gang, ZHANG Jin-xiang, LU Lin, CUI Hua, ZHANG Ji-shan. The secondary precipitates of niobium-alloyed M3: 2 high speed steel prepared by spray deposition [J]. Materials Characterization, 2015, 106: 245-254. DOI: 10.1016/j.matchar.2015.06.006.
[23] CHEN M Y, GOUN? M, VERDIER M, BR?CHET Y, YANG J R. Interphase precipitation in vanadium-alloyed steels: Strengthening contribution and morphological variability with austenite to ferrite transformation [J]. Acta Materialia, 2014, 64: 78-92. DOI: 10.1016/j.actamat.2013.11.025.
[24] YILMAZ A. Microstructural analysis of a new cast high-speed niobium-alloyed tool steel [J]. Metal Science and Heat Treatment, 2012, 54(7, 8): 349-354. DOI: 10.1007/s11041-012-9511-6.
[25] OKAMOTO R, BORGENSTAM A, ?GREN J. Interphase precipitation in niobium-microalloyed steels [J]. Acta Materialia, 2010, 58(14): 4783-4790. DOI: 10.1016/j.actamat. 2010.05.014.
[26] WOYDT M, MOHRBACHER H. The use of niobium carbide (NbC) as cutting tools and for wear resistant tribosystems [J]. International Journal of Refractory Metals and Hard Materials, 2015, 49: 212-218. DOI: 10.1016/j.ijrmhm.2014.07.002.
[27] ZHANG Qian-kun, JIANG Yao, SHEN Wei-jun, ZHANG Hui-bin, HE Yue-hui, LIN Nan, LIU C T, HUANG Han, HUANG Xiao-lin. Direct fabrication of high-performance high speed steel products enhanced by LaB6 [J]. Materials & Design, 2016, 112: 469-478. DOI: 10.1016/j.matdes.2016.09. 044.
[28] TORRES Y, CASELLAS D, ANGLADA M, LLANES L. Fracture toughness evaluation of hardmetals: Influence of testing procedure [J]. International Journal of Refractory Metals and Hard Materials, 2001, 19(1): 27-34. DOI: 10.1016/S0263-4368(00)00044-5.
[29] RAGHAVAN V. C-Fe-V (carbon-iron-vanadium) [J]. Journal of Phase Equilibria, 1993, 14(5): 622-623. DOI: 10.1007/BF02669147.
[30] PAN Fu-sheng, WANG Wei-qing, TANG Ai-tao, WU Li-zhi, LIU Ting-ting, CHENG Ren-ju. Phase transformation refinement of coarse primary carbides in M2 high speed steel [J]. Progress in Natural Science: Materials International, 2011, 21(2): 180-186. DOI: 10.1016/S1002-0071(12)60053-7.
[31] GODEC M, BATI? B ?, MANDRINO D, NAGODE A, LESKOV?EK V, ?KAPIN S D, JENKO M. Characterization of the carbides and the martensite phase in powder-metallurgy high-speed steel [J]. Materials Characterization, 2010, 61(4): 452-458. DOI: 10.1016/j.matchar.2010.02.003.
[32] HUANG Shui-gen, VLEUGELS J, LI Lin, van der BIEST O. Experimental investigation and thermodynamic assessment of the V―W―C system [J]. ChemInform, 2005, 36(33): 68-74. DOI:10.1002/chin.200533015.
[33] KUBLII V Z, VELIKANOVA T Y. Ordering in the carbide W2C and phase equilibria in the tungsten-carbon system in the region of its existence [J]. Powder Metallurgy and Metal Ceramics, 2004, 43(11, 12): 630-644. DOI:10.1007/s11106-005-0032-3.
[34] LEE H M, ALLEN S M, GRUJICIC M. Coarsening resistance of M_2C carbides in secondary hardening steels: Part II. Alloy design aided by a thermochemical database [J]. Metallurgical Transactions A, 1991, 22(12): 2869-2876. DOI: 10.1007/ BF02650248.
[35] AKASH A, MAYO M J. Pore growth during initial-stage sintering [J]. Journal of the American Ceramic Society, 1999, 82(11): 2948-2952. DOI:10.1111/j.1151-2916.1999.tb02186. x.
[36] KANG S J L, JUNG Y I. Sintering kinetics at final stage sintering: Model calculation and map construction [J]. Acta Materialia, 2004, 52(15): 4573-4578. DOI: 10.1016/j.actamat. 2004.06.015.
[37] KARAG?Z S, FISCHMEISTER H F. Niobium-alloyed high speed steel by powder metallurgy [J]. Metallurgical Transactions A, 1988, 19(6): 1395-1401. DOI: 10.1007/BF0 2674013.
[38] PAVL???KOV? M, VOJT?CH D, NOV?K P, GEMPERLOV? J, GEMPERLE A, Z?RUBOV? N, LEJ?EK P, JUR?I P, STOLA? P. Thermal treatment of PM-tool steel alloyed with niobium [J]. Materials Science and Engineering A, 2003, 356(1, 2): 200-207. DOI: 10.1016/S0921-5093(03)00120-5.
[39] ZAPATA W C, DA COSTA C E, TORRALBA J M. Wear and thermal behaviour of M2 high-speed steel reinforced with NbC composite [J]. Journal of Materials Science, 1998, 33(12): 3219-3225. DOI: 10.1023/A: 1004324729342.
[40] ZHOU C, MOON J R, PEACOCK S. Rotary forging of sintered iron based composites [J]. Powder Metallurgy, 1991, 34(1): 33-38. DOI: 10.1179/pom.1991.34.1.33.
[41] ZHOU Bin, SHEN Yu, CHEN Jun, CUI Zhen-shan. Breakdown behavior of eutectic carbide in high speed steel during hot compression [J]. Journal of Iron and Steel Research, International, 2011, 18(1): 41-48. DOI: 10.1016/S1006-706X(11)60009-5.
(Edited by HE Yun-bin)
中文导读
铌的添加对CPM121粉末冶金高速钢组织和性能的影响
摘要:超高钒粉末冶金高速钢通常存在加工困难、生产成本高的问题。研究以超高合金粉末高速钢CPM121 (10W-5Mo-4Cr-10V-9Co, wt.%)作为基础成分,采用元素和碳化物的预氧化球磨混合粉为原料,经压制、活化烧结制得致密化(>99.0%)的样品,研究铌的添加(w(V)+w(Nb)=10 wt.%)对CPM121粉末冶金高速钢组织和性能的影响。结果表明:铌强化高速钢表现出较高的硬度及耐磨性、优异的二次硬化性能和抗回火性。但当铌含量超过5%时则会导致组织中碳化物的粗化和偏聚现象,进而导致材料的强韧性降低。EPMA结果表明铌是MC碳化物的形成元素,促使大量W在M6C及WC碳化物中聚集。其次,旋锻变形可以大幅度提高材料的强韧性,成分为S3(3%Nb+7%V)的CPM121铌高速钢在40%的变形量下,材料的抗弯强度和冲击韧性相较于未旋锻态分别提高了20.74%和43.86%。
关键词:CPM121;铌合金化;显微组织;力学性能;抗回火性;耐磨性;旋锻;粉末冶金

