Trans. Nonferrous Met. Soc. China 23(2013) 2759-2765
Taxonomy characterization and cadmium biosorption of fungus strain
Xiao-xi ZENG1,2,3, Li-yuan CHAI1, Jian-xin TANG2, Xue-duan LIU3, Zhi-hui YANG1
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Hunan Key Laboratory of Green Packaging and Biological Nanotechnology, Hunan University of Technology, Zhuzhou 412007, China;
3. School of Mineral Processing and Bioengineering, Central South University, Changsha 410083, China
Received 16 July 2012; accepted 15 November 2012
Abstract: The taxonomy characteriazation and cadmium (Cd) biosorption of the high Cd-resistant fungus M1 were investigated. The internal transcribed spacers (ITS) region and ��-tubulin genes of the strain were amplified, sequenced and analyzed by molecular biology technology. The Cd biosorption assay was performed by shaking flask. Fourier transform infrared spectroscopy was used to analyze the mycelium. The similarity of gene sequences and phylogenetic trees show the very close relation between the strain and Paecilomyces lilacinus, and the fungus M1 was identified as P. Lilacinus. The initial pH 6 and Cd concentration about 100 mg/L are optimum. Zn and Mn have a little effect on the Cd biosorption of the strain, while Cu and Pb present obvious effects. FTIR analysis shows that the fungus adsorbs Cd by esters, anhydride, and amide. With the preferable absorption capacity, fungus M1 is considered to have good prospects in bioremediation.
Key words: taxonomy characterization; cadmium biosorption; Paecilomyces lilacinu; Fourier transform infrared spectroscopy
1 Introduction
Hazardous levels of heavy metals have increased in environment by industrial activities and agricultural practices. In pollution sites, heavy metals have dispersed into subsurface sediment and groundwater, and the possibility of these elements to damage the ecosystem and human health by the food chain is increasing [1,2]. Cd is a highly toxic heavy metal and unnecessary element for organism cell. It has been widely exploited and applied in industry, agriculture and military field, which caused a serious Cd-pollution. Cd can be transported into the cytoplasm through nontoxic metals transport systems due to the structural and valence similarities. It damages cells by strong affinity to sulfhydryl groups of proteins and displacement of Zn and Fe ions from proteins [3]. The complexation of sulfhydryl groups with heavy metal cation decreased the activity of sensitive enzymes. It has been speculated that Cd can generate reactive oxygen species (ROS) to cause single-strand DNA damage. Moreover, Cd inhibits the DNA mismatch repair system, which may lead to mutations and cancer [4].
With the increase of concern on heavy metal pollution, various strategies to restore environment have been studied by researchers. Some methods have been used in treatment of wastewater, such as precipitation, adsorption on activated carbon, membrane separation, ionic exchange and flotation. Recently, removal of heavy metals from wastewaters has focused on the development of safer, more efficient, and less expensive materials [5,6]. Compared with the other methods, biosorption has the merits of low energy consumption, no second pollution and simply operation. As a way of cleaning up toxic-metal contamination, the biosorption of microbial cells for heavy metals is a promising technology [7,8]. Fungi possess good metal uptake systems with metabolism-independent biosorption. The hyphal wall was found to be a primary site of metal ion accumulation. Rhizopus and Aspergillus sp. were thought as promising biosorbers of Cd and Cr [9]. Phanerochaete chryso-sporium mycelium was used as a biosorbent for heavy metals Cd (II), Pb (II) and Cu (II) from solution [10]. The above researches showed fungus was a good material to adsorb heavy metals. There are some reports about fungi used on metals biosorption, but few reports have been published about biosorption by Paecilomyces lilacinu.
Xiangjiang River (located in Hunan province) valley is the typical heavy metals pollution. Cd is the main toxic metal and has threatened human health. In previous work, an indigenous fungus with high resistance to Cd was isolated. In order to know the possibility in bioremediation, the fungus was investigated to understand its taxonomy and biosorption capacity.
2 Experimental
2.1 Strain and medium
The fungus M1 was isolated from Zhuzhou smelter near by Xiangjiang River, which possesses high Cd-resistant capacity. Czapek medium (sucrose 30 g/L, NaNO3 2.0 g/L, K2HPO4 1.0 g/L, KCl 0.5 g/L, MgSO4 0.5 g/L, FeSO4 0.01 g/L) was used.
2.2 ITS and ��-tubulin genes amplification and sequence analysis
In order to extract the genomic DNA, the fresh mycelia were harvested and rapidly ground in a mortar with liquid N2 for 5-10 min. The pretreated mycelia were performed according to the instructor of commercial kit. The fungal universal primers ITS-1 (5��- TCCGTAGGTGAACCTGCGG-3��) and ITS-4 (5��- CCTCCGCTTATTGATATGC-3��) were used to amplify the internal transcribed spacers (ITS) region sequence [11]. Based on the lots of ��-tubulin sequences in GenBank database, a pair primers were designed as follows: f (5��-CATGCGTGAGCTTGTATGC-3��) and r (5��-TAGCCGAAAGCGAAGTTG-3��). The PCR conditions were as follows: 95 ��C for 5 min; 32 cycles: 94 ��C for 30 s, 56 ��C for 30 s and 72 ��C for 1 min; 72 ��C for 7 min and 4 ��C pause. The above amplification products were purified and sent to Shanghai Sangon Biotechnologies Co. Ltd. to sequence. The ITS and ��-tubulin gene sequences of the fungus were submitted to GenBank and analyzed with the BLAST search tool.
2.3 Cd biosorption assay
To obtain mycelia biomass, M1 was inoculated in Czapek liquid medium. The cultures were incubated on a rotary shaker at 30 ��C and 180 r/min for 3 d. The biomasses were harvested by centrifugation at 5000 r/min for 10 min, washed with sterile water three times. Mycelia were sterilized at 121 ��C for 20 min to get no-living biomass. Both of living and no-living biomass was used in biosorption. The biosorption experiments were performed for 24 h. After adsorption the Cd adsorption solutions were centrifuged at 5000 r/min for 10 min. The supernatant was filtered by 0.45 ��m micro filtration membrane. The concentration of Cd was determined by flame atomic absorption.
In the adsorption experiments, the wet mycelia were weighed to keep equal in each flask. The adsorption amount was calculated as
Q=(��i-��f)��V/md (1)
where Q is the Cd adsorption amount; ��i and ��f mean the initial and final Cd concentration, V is the Cd solution volume and md is the dry mass of biomass. The below experiments were repeated three times and average values were gained.
The influence of initial pH on Cd adsorption was studied. The Cd solution pH was adjusted from 3 to 7 by 1 mol/L HNO3 and 1 mol/L NaOH. The initial Cd concentration is 100 mg/L. Adsorption was performed in a rotary shaker with 100 r/min at 30 ��C. Shaking for 30 min, the Cd concentrations were determined.
At varied initial Cd concentration, the Cd adsorption capacity of fungus M1 was studied. The initial Cd concentrations were ranged from 20 to 200 mg/L. Other adsorption conditions were same as the above experiments. The Cd removal rate (R) was obtained by the following equation:
R=(��i-��f)/��i��100% (2)
The effects of Zn (II), Mn (II), Cu (II), and Pb (II) on Cd adsorption were alone investigated. In Cd solutions, Cd concentration was kept 100 mg/L and the other metal ion was 50 mg/L. Other adsorption conditions were same as the above experiments. Shaking at 100 r/min for 30 min, the Cd concentrations were measured.
2.4 FTIR analysis
The cultures were incubated on a rotary shaker at 30 ��C and 180 r/min for 7 d, then performed cadmium adsoption assay as 2.3. The biomasses were harvested by centrifugation at 8000 r/min for 10 min, followed freeze-drying to constant mass. Using a potassium bromide (KBr) pellet technique (1 mg sample/100 mg KBr), the above biomasses were analyzed by Fourier transform infrared spectroscopy.
3 Results and discussion
3.1 Taxonomy characterization
ITS region and �¨Ctubulin gene sequences amplified from the genomic DNA are submitted to GenBank and the accession number are FJ461773 and FJ502248 separately. The sequences are aligned with the previously published sequences by BLAST tool. The result showed that they have more than 99% similarity to Paecilomyces lilacinus. As shown in Fig. 1 and Fig. 2, the phylogenetic trees based on ITS region and �¨Ctubulin gene are constructed by the software Clust.X. Both the phylogenetic trees also suggest the very close relationship between the fungus M1 and P. Lilacinus, and the strain is identified as P. Lilacinus.
The ITS regions are located between the 18S and 28S rRNA genes, including the ITS1 nucleotide sequence, 5.8S ribosomal RNA, and ITS2 nucleotide sequence. Due to the high variable domains ITS regions has been used extensively to detect closely related fungal isolates. HENRY et al [11] developed a method to identify Aspergillus at the species level and differentiate it from other true pathogenic and opportunistic molds, and found that both ITS 1 and ITS 2 regions were needed for accurate identification of Aspergillus at the species level. Based on the ITS region sequences, the specific primers were designed to separate the taxonomy which were hardly distinguishable on the basis of their morphology [12].
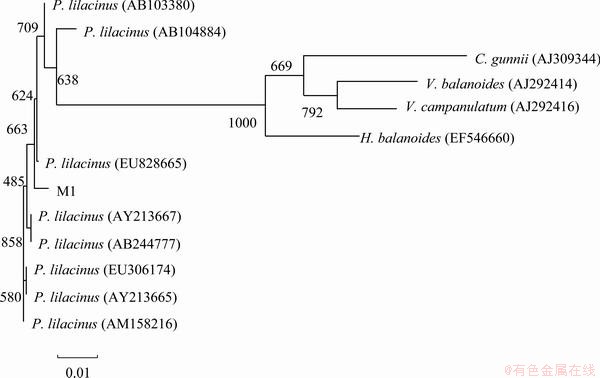
Fig. 1 Phylogenetic tree based on ITS regions sequence of fungus M1
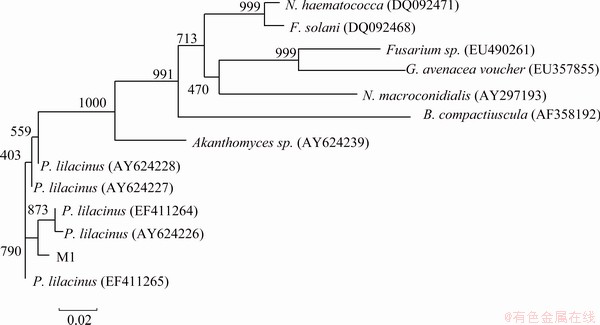
Fig. 2 Phylogenetic tree based on �¨Ctubulin sequence of fungus M1
Tubulins are soluble protein in all eukaryotic cells cytoplasm. They mainly include two types tubulin molecules called �� and ��-ubulin. The genes encode ��-tubulins are extremely conserved and have been studied in many fungi [13,14]. The ��-tubulin gene of Aspergillus flavus was found to be highly conserved between the two Aspergillus species and used as a selectable marker for transformation. Nowaday ��-tubulin gene is thought as important on taxonomy identification [15].
Based on the ITS regions and �¨Ctubulin gene analysis characteristics, the fungus M1 can be identified as P. lilacinus. P. lilacinus has the greatest potential for use in combination with selected fungicides and nematicides for the control of potato cyst nematodes [16]. Paecilomyces lilacinus strain 251 proved significant control of the root-knot nematode M incognita on tomato [17]. In the field P. lilacinus has been developed into commercial products and used to control nematodes for years [18]. Moreover, it has been studied in environment treatment. ODA et al [19] found that P. lilacinus D218 excreted PHB (3-hydroxybutyrate) and PCL (polycaprolactone) depolymerases to degraded PHB and PCL. P. lilacinus as biphenyl oxidizing organisms can oxidize chlorinated biphenyl derivatives [20]. But there are few reports about P. lilacinus used in heavy metals removal. It is maybe the first report about heavy metal removal by P. lilacinus.
3.2 Effect of initial pH
The result of the effect of pH on the biosorption capacity is shown in Fig. 3. Both living biomass and no-living biomass present the similar trend. In the pH range of 3-6, the adsorption amount increases with initial pH increasing, and the Cd adsorption amount enhances from 16.77 to 36.51 mg/L, and 17.86 to 41.13 mg/L for living and no-living biomass, respectively. When pH value reaches 7, the adsorption capacity is slightly decreased. The well biosorption occurs at pH 6-7 and 5-7 by living and no-living biomass. Since alkaline solution contributes Cd ion to form hydroxide precipitation, the pH exceeding 7 is not considered to further investigate. The pH 6 is thought as the optimum initial value in this experiment.
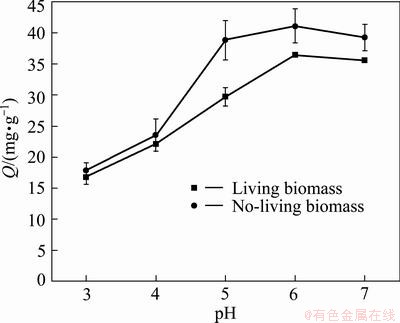
Fig. 3 Effect of pH on Cd biosorption by living and no-living biomass
It has been reported that the ability of Cd biosorption was related to initial pH. This result was similar to the previous reports on the biosorption of Cd. RANGSAYATORN et al [21] reported Cd adsorption by Spirulina platensis TISTR 8217 silica immobilized cells remained constant for a wide pH range from 4 to 7, while the maximum Cd adsorption capacity of alginate immobilized cells occurred at pH 6 [21]. The optimum pH for adsorption of Cd by Pantoea sp. TEM18 was 6.0 [22]. The pH 5 was found to be optimum for Cd biosorption by Hydrilla verticillata biomass [23]. The effect of pH on metal biosorption is related to the active sites on the cell walls. The cell wall surface contains a large number of active sites such as carboxyl, hydroxyl, amino groups, and phosphate groups. The solution pH can affect the interaction between metal ions and the active sites. At low pH, the amount of proton far exceeds that of metal ion and the active sites mainly bind proton, resulting Cd ion free in solution [24]. With the initial pH increasing, the proton decreases and the Cd ions bound to the active sites increase. Thus the Cd biosorption increased at higher initial pH. However, some heavy metal ions can form precipitation in alkaline environment and the alkaline solution was excluded in biosorption.
3.3 Effect of initial Cd concentration
The effect of initial Cd concentration on biosorption capacity is shown in Fig. 4. It is observed that with the Cd concentration increasing from 20 to100 mg/L, the adsorption amount rapid enhances from 9.45 mg/g to 36.46 mg/g and 9.85 mg/ g to 41.99 mg/ g for living and no-living biomass. When the Cd concentration exceeded 100 mg/L, the adsorption amount increased slightly. Meanwhile, the biosorption rate exhibited drop tendency. With the Cd concentration increasing from 20 mg/L to 150 mg/L, it decreases from 78.75% to 38.98% and 82.05% to 46.05 % for living and no-living biomass.
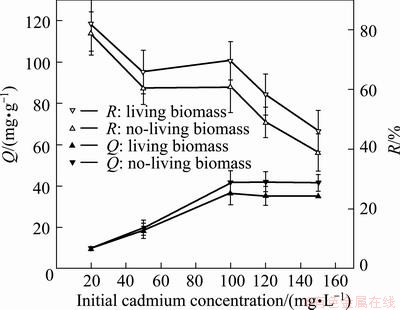
Fig. 4 Effect of initial Cd concentration on adsorption amount (Q) and Cd removal (R)
The general accepted biosorption mechanism is that metal ions are adsorbed by active sites of cell wall. Regardless of other conditions, the biosorption capacity is proportional to the amount of metal ions and active sites. So, the adsorption amount can increase with Cd concentration. In this experiment the biomass amount was constant and the active sites were limited. When the maximum active sites were occupied with metal ions, it presented no significant increase with the higher Cd concentration. At the same time, at higher Cd concentration, the increase amount of Cd free in solution was more than that of bound on sites, resulted in the adsorption rate decreasing. Some researchers have studied the influence of initial metal ions concentration on biosorption by different biomass [25]. The capacity of the biosorbents of Pantoea sp. TEM18 increased initially on increasing the initial concentrations of Cd ions and reached saturation at 150 mg/L [22]. The Cd uptake by S.platensis reached a saturation value around 150 mg/L [26]. In this experiment the Cd biosorptions of the living and no-living biomass both reached saturation at 100 mg/L. It suggests the strain is suitable to treat wastewater at about 100 mg/L.
3.4 Effect of other metals
In heavy metal polluted environment, there generally exist different metals. So, studying the effect of other metal ions on biosorption is necessary in bioremediation. Zn, Mn, Cu and Pb are the usual heavy metals in heavy metals pollution sites. The effect of them on the biosorption was investigated. From Fig. 5, it can be found that all the metals can inhibit the biosorption both by living and no-living biomass, but the inhibitions of Cu and Pb were more than Zn and Mn. In the presence of Cu and Pb, the adsorption amount lowered to 12.93 and 14.5 mg/g, respectively. It is implied that Cu and Pb would decrease the Cd biosorption of the strain.
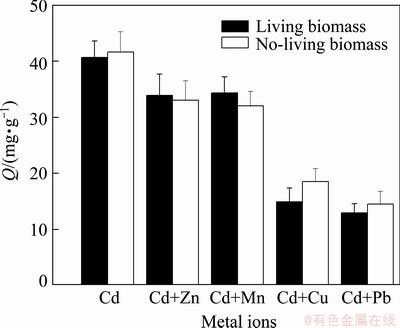
Fig. 5 Influence of other metals on Cd biosorption
Since the biosorption mechanism is related metal ion bound the active sites of cell wall, there would exist in competition of varied metal ions for active sites. Some scholars have studied the competition. AKAR et al [27] found that Cu, Cd and Ni can inhibit the Pb uptake capacity of Botrytis cinerea and the highest inhibition effect was observed in the presence of Cu followed by Ni and Cd. In the biosorption of heavy metals with Phanerochaete chrysosporium the order of affinity for competitive conditions was Cu (II)>Pb (II)>Cd (II) [28]. Competitive biosorption experiments were performed with Cd and Pb together with Cu and the biosorption capacities of the yeast biomass for all metal ions were found to be lower than in non-competitive conditions [29]. In this result, Cu and Pb also present high inhibitions on Cd biosorption, but the inhibition of Zn and Mn were weak. It may imply that there were more common affinity sites for Cu (II) and Pb (II) with Cd (II) on cell wall than the other two metal ions.
3.5 FTIR analyses
The FTIR analysis of biomass is shown in Fig. 6. The biosorption peak at 3430 cm-1 could result from the stretching vibration of N��H groups of the primary and secondary amides and O��H groups. The band at 3160 cm-1 is indicative of N��H group. The peaks at 2930 and 2850 cm-1 indicate C��H group stretching vibration. The bands at 1640 and 1750 cm-1are the results of C=O stretching. The peak at 1400 cm-1 is indicative of N��H stretching vibration from the primary amides. The biosorption peak at 1090 cm-1 is indicative of phosphate functional groups such as P=O, P��OH,  . The FT-IR spectra show the difference between the components of biomasses with varied Cd concentration (Fig. 6). Compared with the biomass which has not contacted Cd (Fig. 6(a)), the FTIR spectrum corresponding to 2 and 6 mM Cd shows (Fig. 6(b), (c)) the following difference: the peaks at 3430 and 3160 cm-1 attached to O��H and N��H groups are weak, the peaks at 2930 and 2850 cm-1 related to C��H group obviously weaken; the peaks at 1750 cm-1 related to C=O group of from esters, anhydride, and amide almost disappeared. The changes of biosorption bands mean the decrease of amino and amide. The result suggests that the Cd biosorption mechanism of the strain is related to carboxyl, hydroxyl, amino, and so on groups, which is similar to other reports [7].
. The FT-IR spectra show the difference between the components of biomasses with varied Cd concentration (Fig. 6). Compared with the biomass which has not contacted Cd (Fig. 6(a)), the FTIR spectrum corresponding to 2 and 6 mM Cd shows (Fig. 6(b), (c)) the following difference: the peaks at 3430 and 3160 cm-1 attached to O��H and N��H groups are weak, the peaks at 2930 and 2850 cm-1 related to C��H group obviously weaken; the peaks at 1750 cm-1 related to C=O group of from esters, anhydride, and amide almost disappeared. The changes of biosorption bands mean the decrease of amino and amide. The result suggests that the Cd biosorption mechanism of the strain is related to carboxyl, hydroxyl, amino, and so on groups, which is similar to other reports [7].
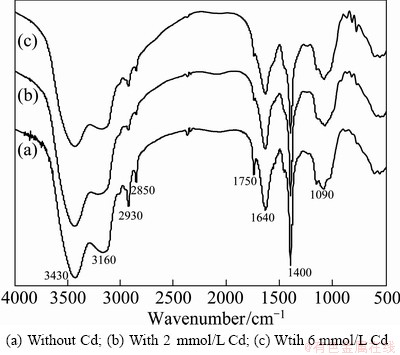
Fig. 6 FTIR spectra of fungus M1
4 Conclusions
1) ITS region and �¨Ctubulin gene sequences are aligned with the previously published sequences by BLAST tool and all have more than 99% similarity to P. lilacinus. The phylogenetic trees also suggest the very close relationship between the fungus M1 and P. Lilacinus, and the strain is identified as P. Lilacinus.
2) The initial pH has an obvious effect on Cd biosorption of the strain. The suitable initial pH values are 6-7 and 5-7 for living and no-living biomass, respectively. At initial Cd concentration of 100 mg/L, both the adsorption amount and removal rate are efficiency. Zn and Mn have a little effect on the the Cd biosorption of the strain, while Cu and Pb present obvious effects. The strain is suitable to adsorb Cd ions of solution and has potential in bioremediation of Cd pollution. FTIR suggests that the Cd biosorption mechanism of the strain is related to carboxyl, hydroxyl, amino, and so on groups.
References
[1] ZENG Xiao-xi, TAGN Jian-xin, JIANG Pei, LIU Hong-wei, DAI Zhi-min, LIU Xue-duan. Isolation, characterization, and extraction of mer gene of Hg2+ resisting strain D2 [J]. Transactions of Nonferrous Metals Society of China, 2010, 20 (3): 507-512.
[2] YANG Miao, XIAO Xi-yuan, MIAO Xu-feng, GUO Zhao-hui, WANG Feng-yong. Effect of amendments on growth and metal uptake of giant reed (Arundo donax L.) grown on soil contaminated by arsenic, cadmium and lead [J]. Transactions of Nonferrous Metals Society of China, 2012, 22 (5): 1462-1469.
[3] BANJERDKIJ P, VATTANAVIBOON P, MONGKOLSUK S. Exposure to cadmium elevates expression of genes in the OxyR and OhrR regulons and induces cross-resistance to peroxide killing treatment in xanthomonas campestri [J]. Applied and Environmental Microbiology, 2005, 71(4): 1843-1849.
[4] WANG A, CROWLEY D E. Global gene expression responses to cadmium toxicity in Escherichia coli [J]. Journal of Bacteriology, 2005, 187(9): 3259-3266.
[5] KANG S H, SINGH S, KIM J, LEE W, MULCHANDANI A, CHEN W. Bacteria metabolically engineered for enhanced phytochelatin production and cadmium accumulation [J]. Applied and Environmental Microbiology, 2007, 73(19): 6317�C6320.
[6] EI-SAYED M. EI-MORSY. Cunninghamella echinulata a new biosorbent of metal ions from polluted water in Egypt [J]. Mycologia, 2004, 96(6): 1183�C1189.
[7] GADD G M. Biosorption: critical review of scientific rationale, environmental importance and significance for pollution treatment [J]. Journal of Chemical Technology and Biotechnology, 2009, 84(1): 13-28.
[8] CHOJNACKA K. Biosorption and bioaccumulation�CThe prospects for practical applications [J]. Environment International, 2010, 36(3): 299-307.
[9] ZAFAR S, AQIL F, AHMAD I. Metal tolerance and biosorption potential of filamentous fungi isolated from metal contaminated agricultural soil [J]. Bioresource Technology, 2007, 98(9): 2557-2561.
[10] LI Q, WU S, LIU G, LIAO X, DENG X. Simultaneous biosorption of cadmium (II) and lead (II) ions by pretreated biomass of Phanerochaete chrysosporium [J]. Sep Purif Technol, 2004, 34(1-3): 135-142.
[11] HENRY T, IWEN P C, HINRICHS S H. Identification of Aspergillus species using internal transcribed spacer regions 1 and 2[J]. Journal of Clinical Microbiology, 2000, 38(4): 1510-1515.
[12] MELLO A, GHIGNONE S, VIZZINI A, SECHI C, RUIU P, BONFANTE P. ITS primers for the identification of marketable boletes [J]. Jorunal of Biotechnology, 2006, 121(3): 318-329.
[13] KEELING P J, LUKER M A, PALMER J D. Evidence from beta-tubulin phylogeny that microsporidia evolved from within the fungi [J]. Molecular Biology and Evolution, 2000, 17(1): 23-31.
[14] ZUCCARO A, SCHOCH C L, SPATAFORA J W, KOHLMEYER J, DRAEGER S, MITCHELL J I. Detection and identification of fungi intimately associated with the brown seaweed fucus serratus [J]. Applied and Environmental Microbiology, 2008, 74(4): 931-941.
[15] SEIP E R, WOLOSHUK C P, PAYNE G A, CURTIS S E. Isolation and sequence analysis of a 3-Tubulin gene from Aspergillus flavus and its use as a selectable marker [J]. Applied and Environmental Microbiology, 1990, 56(12): 3686-3692.
[16] JACOBS H, GRAY S N, CRUMP D H. Interactions between nematophagous fungi and consequences for their potential as biological agents for the control of potato cystnematodes [J]. Mycological Research, 2003, 107(1): 47�C56.
[17] KIEWNICK S, SIKORA R A. Biological control of the root-knot nematode meloidogyne incognita by Paecilomyces lilacinus strain 251 [J]. Biological Control, 2006, 38(2): 179-187.
[18] ZOU C S, MO M H, GU Y Q, ZHOU J P, ZHANG K Q. Possible contributions of volatile-producing bacteria to soil fungistasis [J]. Soil Biology & Biochemistry, 2007, 39(9): 2371-2379.
[19] ODA Y, ASARI H Y, URAKAMI T I, TONOMURA K. Microbial degradation of poly (3-Hydroxybutyrate) and polycaprolactone by filamentous fungi [J]. Journal of Fermentation and Bioengineering, 1995, 80(3): 265-269.
[20] SIETMANN R, GESELL M, HAMMER E, SCHAUER F. Oxidative ring cleavage of low chlorinated biphenyl derivatives by fungi leads to the formation of chlorinated lactone derivatives [J]. Chemosphere, 2006, 64(4): 672-685.
[21] RANGSAYATORN N, POKETHITIYOOK P, UPATHAM E S, LANZA G R.Cadmium biosorption by cells of Spirulina platensis TISTR 8217 immobilized in alginate and silica gel [J]. Environment International, 2004, 30(1): 57-63.
[22] OZDEMIR G, CEYHAN N, OZTURK T, AKIRMAK F, COSART. Biosorption of chromium (VI), cadmium (II) and copper (II) by Pantoea sp. TEM18 [J]. Chemical Engineer Journal, 2004, 102(3): 249-253.
[23] BUNLUESIN S, KRUATRACHUE M, POKETHITIYOOK P, UPATHAM S, LANZA G R. Batch and continuous packed column studies of cadmium biosorption by Hydrilla verticillata biomass [J]. Journal of bioscience and Bioengineer, 2007,103 (6): 509-513.
[24] KAEWSARN P, YU Q. Cadmium (II) removal from aqueous solutions by pre-treated biomass of marine alga Padina sp [J]. Environmental pollution, 2001, 112(2): 209-213.
[25] BAYRAMOGLU G, TUZUN I, CELIK G, YILMAZ M, ARICA M Y. Biosorption of mercury (II), cadmium (II) and lead (II) ions from aqueous system by microalgae Chlamydomonas reinhardtii immobilized in alginate beads [J]. International Journal of Mineral Processing, 2006, 81(1): 35-43.
[26] RANGSAYATORN N, UPATHAM E S, KRUATRACHUE M. POKETHITIYOOK P, LANZA G R. Phytoremediation potential of spirulina (arthrospira) platensis: biosorption and toxicity studies of cadmium [J]. Environmental Pollution, 2002, 119(1): 45-53.
[27] AKAR T, TUNALI S, KIRAN I. Botrytis cinerea as a new fungal biosorbent for removal of Pb (II) from aqueous solutions [J]. Biochemical Engineering Journal, 2005, 25(3): 227-235.
[28] SAY R, DENIZLI A, ARICA M Y. Biosorption of cadmium (II), lead (II) and copper (II) with the filamentous fungus Phanerochaete chrysosporium [J]. Bioresource Technology, 2001, 76(1): 67-70.
[29]  U. Biosorption of cadmium and lead ions by ethanol treated waste baker's yeast biomass [J]. Bioresource Technology, 2005, 96(1): 103-109.
U. Biosorption of cadmium and lead ions by ethanol treated waste baker's yeast biomass [J]. Bioresource Technology, 2005, 96(1): 103-109.
һ�꿹������ķ���ѧ������������
����ϣ1,2,3������Ԫ1��������2����ѧ��3����־��1
1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. ���Ϲ�ҵ��ѧ ��ɫ��װ������������ʡ�ص�ʵ���ң����� 412007��
3. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083
ժ Ҫ���о�һ��߿������M1�ķ���ѧ������������Ӱ�����ء�ͨ����������ѧ�������ITS �ͦ�-�ܵ��Ļ������У������в���ͷ���������ҡƿ�����������о�Ӱ���������������������أ����ø���Ҷ������Ծ�˿���������������г����������������������ԱȽϺ�ϵͳ����������������������뵭������ù�Ĺ�ϵ������������M1�ڷ���ѧ�����ڵ�������ù������ĺ�ʧ���˿��ֱ�����ʼpHֵΪ6~7��5~7�ķ�Χ�ڱ��ֳ��Ϻõ���������������ҺŨ��Ϊ100 mg/Lʱ����Ч����á�п���̶Ծ������������Щ��Ӱ�죬��ͭ��Ǧ������������Ӱ�����ԡ����������������������������Ҫ���ʻ����ǻ��Ͱ����Ȼ����йء�ʵ�������������M1���ؽ������������о��нϺõ�Ӧ��ǰ����
�ؼ��ʣ�����ѧ����������������������ù������Ҷ���������
(Edited by Chao WANG)
Foundation item: Project (50925417) supported by the National Funds for Distinguished Young Scientist, China; Project (2012BAC09B04) supported by the National Key Technology Research and Development Program, China; Project supported by the Post-doctoral Program of Central South University, China; Projects (31100082, 61171061) supported by the National Natural Science Foundation of China; Project (2012SK4028) supported by the Science and Technology Program of Hunan Province, China
Corresponding author: Zhi-hui YANG; Tel: +86-731-88830875; E-mail: yangzhihui65@126.com
DOI: 10.1016/S1003-6326(13)62794-6